Abstract
Study Design
Methodological study for guideline development.
Objective
AO Spine Guideline for Using Osteobiologics (AO-GO) project for spine degenerative pathologies was an international, multidisciplinary collaborative initiative to identify and evaluate evidence on existing use of osteobiologics in Anterior Cervical Fusion and Decompression (ACDF). The aim was to formulate precisely defined, clinically relevant and internationally applicable guidelines ensuring evidence-based, safe and effective use of osteobiologics, considering regional preferences and cost-effectiveness.
Methods
Guideline was completed in two phases: Phase 1- evidence synthesis; Phase 2- recommendation development based on the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach. In Phase 1, key questions identified by a panel of experts were addressed in a series of systematic reviews of randomized and non-randomized studies. In Phase 2, the GRADE approach was used to formulate a series of recommendations, including expert panel discussions via web calls and face-to-face meetings.
Discussion
AO-GO aims to bridge an important gap between evidence and use of osteobiologics in spine fusion surgeries. Owing to differences in osteobiologics preparation and functional characteristics, regulatory requirements for approval may vary, therefore it is highly likely that these products enter market without quality clinical trials. With a holistic approach the guideline aims to facilitate evidence-based, patient-oriented decision-making processes in clinical practice, thus stimulating further evidence-based studies regarding osteobiologics usage in spine surgeries. In Phase 3, the guideline will be disseminated and validated using prospectively collected clinical data in a separate effort of the AO Spine Knowledge Forum Degenerative in a global multicenter clinical study.
Introduction
The current focus of AO-GO is the use of osteobiologics in Anterior Cervical Fusion and Decompression (ACDF) procedures for cervical degenerative conditions. There are three phases to the AO-GO work: Phase 1- evidence synthesis; Phase 2- recommendation development based on the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach; Phase 3- recommendation dissemination and ongoing evidence collection based on DegenPRO, a prospective data collection of spine degenerative surgery hosted by the AO Spine Knowledge Forum Degenerative (KF Degen) and the Bone Osteobiologics and Evidence classification (BOnE Classification) developed by the KF Degen,
1
and development of a clinical decision support tool. This Special Issue addresses the following objectives for Phase 1 and Phase 2: 1. To identify and evaluate the existing osteobiologics and their use in ACDF procedures. 2. To identify patient characteristics/risk factors guiding the decision on use of osteobiologics and evaluate clinical outcomes in these patient subgroups. 3. To develop evidence-based recommendations for the use of osteobiologics in ACDF procedure based on Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach.
2
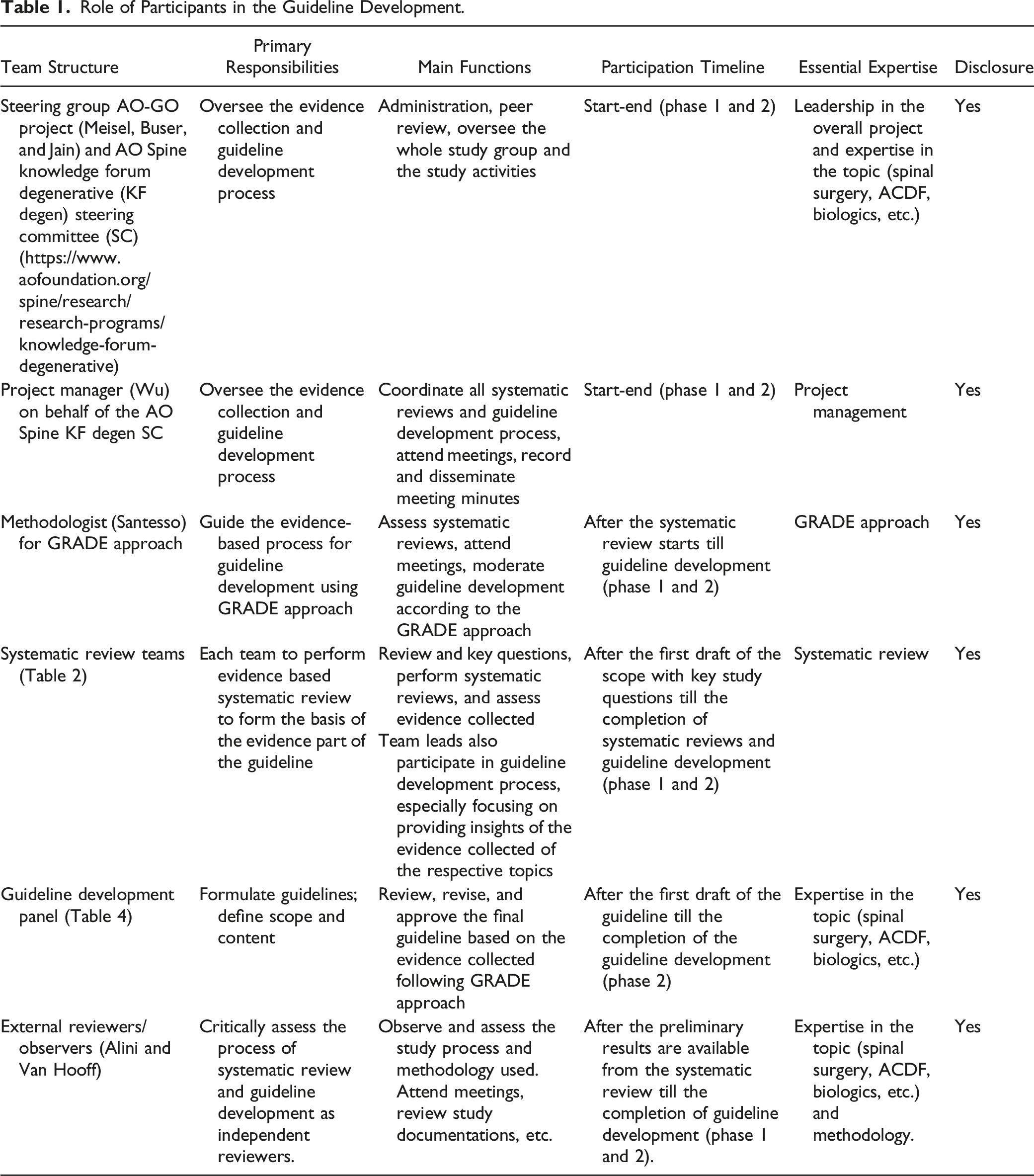
Role of Participants in the Guideline Development.
Phase 1- Evidence Synthesis
AOGO Research Topics/Questions and Team Leaders (list of team members can be found as the co-authors of the respective systematic review manuscripts of the AOGO Focus Issue).

AOGO PICO Table.

Subsequently, a series of systematic reviews synthesizing and appraising the evidence for osteobiologics were conducted. When possible study protocols for the systemetic reviews were registered in PROSPERO, the international prospective register for systematic reviews – link: https://www.crd.york.ac.uk/prospero/. For many PICOs, randomized controlled trials were not available that directly compared autograft, allograft, or a cage with osteobiologic to each other. Instead, indirect comparisons using case series of each were pooled and then compared to each other. Meta-analysis was also not possible in most comparisons and results were summarized by range of effects. Unfortunately, there was also very little data about outcomes in specific populations based on comorbidities and these analyses were not conducted. The overall strength of evidence for the clinical outcomes was assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. 5 As per GRADE, the body of evidence was specified in four categories (high, moderate, low and very low) and reflect the level of certainty in the effect estimates to support a recommendation. Several factors can increase or reduce the confidence in the effect, such as the risk of bias of the studies, the precision and consistency of the effect, the directness of the evidence and the risk of publication bias.6–11 For most outcomes, the certainty in the evidence was rated down for risk of bias because prognostic balance was likely not possible when comparing results from case series. There was often very few people across studies resulting in imprecise results.
Phase 2- Recommendation Development based on GRADE
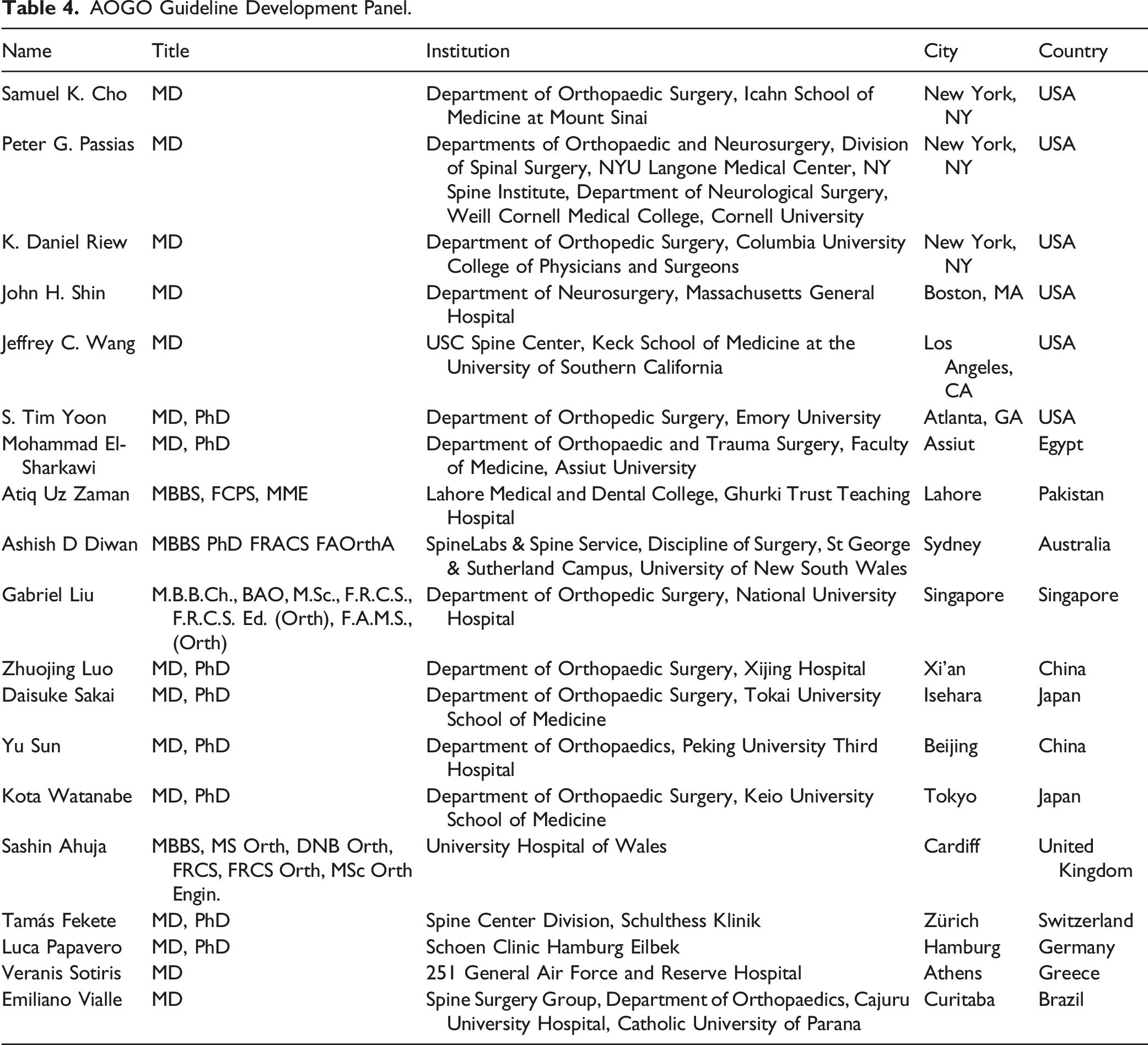
AOGO Guideline Development Panel.
Discussion
AO-GO is aiming to help various stakeholders, including surgeons, to choose and use appropriate osteobiologics, companies to develop and advance osteobiologics products, hospitals to use suitable osteobiologics products, and regulatory authorities to oversee osteobiologics development and usage. By developing a guideline for osteobiologics use, we also aim to encourage evidence-based research on osteobiologics and related products.
The methods used ensured that the guideline development complied with accepted methodology and standards. Despite the paucity of studies directly comparing autograft, allograft and osteobiologics to each other, and the lack of data for the use of osteobiologics in populations with comorbidities, the guideline group balanced available evidence of benefits and harms with costs and other factors and made clinically useful recommendations that will be disseminated and validated in Phase 3. The group of experts identified for the guideline development ensured participation of clinical experts, key opinion leaders, stakeholders, and policy makers who will facilitate implementation into routine clinical practice. The systematic reviews and evidence-based guideline will also be presented in conferences and published in international peer-reviewed journals.
Compared to artificial intelligence (AI) and medical learning (ML) applications such as diagnostic imaging and classification in spine research, predictive analytics and clinical decision support tools are in their infancy. Advances in AI novel technologies and availability of data registries have enabled outcome prediction analysis and development of a clinical decision support system to some extent. Nijmegen Decision Tool for Chronic Low back pain is a recent decision support tool based on predictors such as patient’s demographics and symptoms, which is yet under development.5,12–14 In fact, to date there is no application of ML in a decision support tool for spine surgeries. Accountability, risk of bias, ethics and data protection are the major hurdles for implementing ML technology in health care. Availability of large datasets for algorithm training, validation and accuracy testing pose other major challenges.
This paper presents Phase 1 and Phase 2 for guideline development, the AOGO osteobiologics grading system is currently being developed and Phase 3 is in preparation. In future, the systematic reviews will need to be updated and the recommendations revised if needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ZB –Grants/ Research support: AO Spine (co-PI on a AOSpine Knowledge Forum grant, Paid directly to institution/employer); Nexus Spine (Paid directly to institution/employer), Medical Metrics (Paid directly to institution/employer); MiMedx (past, Paid directly to institution/employer), SBIR NIH (past, Paid directly to institution/employer); Medical Metrics (past, Paid directly to institution/employer); Next Science (past, Paid directly to institution/employer); Consultancy: Next Science (paid directly to institution/employer); NIH (advisor consultant); Scientific Advisory Board: Medtronic, Medical Metrics; Trips/Travel: AO Spine (Travel Expense Reimbursement, travel reimbursements for AO Spine Knowledge forum meetings and congresses), NASS (Travel Expense Reimbursement); Board of Directors: LSRS (Nonfinancial, Co-chair of Program Committee); Committees: AO Spine Knowledge Forum Degenerative (Steering Committee Member); North American Spine Society: Research Project Management Committee (Vice chair), Section on Biologics & Basic Research (Co-chair), The Spine Journal Peer Reviewers (Committee member). Patents (issued): Biomarkers for painful intervertebral discs and methods of use thereof; HJM – Dr Meisel is consultant (money paid to institution) - Regenerate Life Sciences GmbH for DiFusion (ongoing), Co.don (past); royalties from:Medtronic, Fehling Aesculap (past); stocks (money paid to institution) - Regenerate Life Sciences GmbH in DiFusion JCW – Royalties – Biomet, Seaspine, Amedica, DePuy Synthes; Investments/Options – Bone Biologics, Pearldiver, Electrocore, Surgitech; Board of Directors - North American Spine Society, AO Foundation (20,000 honorariums for board position, plus travel for board meetings), Cervical Spine Research Society; Editorial Boards - Spine, The Spine Journal, Clinical Spine Surgery, Global Spine Journal; Fellowship Funding (paid directly to institution): AO Foundation . AJ – Dr Jain has Consulting: DePuy Spine, Globus Spine, Stryker Spine. Yoon ST – Dr Yoon has Consulting: Alphatec; Research Support (Investigator Salary): Empiric Spine (Paid directly to institution/employer); Royalties: Meditech; Stock Ownership: Medyssey; Trips/Travel: AOSpine. Wang JC – Royalties – Biomet, Seaspine, Synthes, Novapproach; Investments/Options – Bone Biologics, Pearldiver, Electrocore, Surgitech; Consulting: Bioretec, Angitia, Epidutech, Depuy; Board of Directors - AO Foundation, National Spine Health Foundation; Editorial Boards - Global Spine Journal Editor-in-Chief; Fellowship Funding (paid to institution): AO Foundation. Wu Y: none; Agarwal N: none; Santesso N: none.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
