Abstract
Study Design:
Classification development.
Objectives:
The aim of our study was to develop a 3-tier classification for the levels of evidence for osteobiologics and provide a description of the principles by which osteobiologics can be evaluated. BOnE (Bone Osteobiologics and Evidence) classification evaluates each osteobiologic based on the available evidence, and if the published evidence is based on clinical, in vivo or in vitro studies.
Methods:
The process of establishing the BOnE classification included 5 face-to-face meetings and 2 web calls among members of the AOSpine Knowledge Forum Degenerative.
Results:
The 3 levels of evidence were determined based on the type of data on osteobiologics: level A for human studies, level B for animal studies, and level C for in vitro studies, with level A being the highest level of evidence. Each level was organized into 4 subgroups (eg, A1, A2, A3, and A4).
Conclusions:
The use and the variety of osteobiologics for spine fusion has dramatically increased over the past few decades; however, literature on their effectiveness is inconclusive. Several prior systematic reviews developed by AOSpine Knowledge Forum Degenerative reported low level of evidence primarily due to the high risk of bias, small sample size, lack of control groups, and limited patient-reported outcomes. BOnE classification will provide a universal platform for research studies and journal publications to classify a new or an existing product and will allow for creating decision-making algorithms for surgical planning.
Introduction
The use of osteobiologics for spine fusion has increased significantly over the years with the expansion of available graft materials as well as the incidence of patients with spine pathologies. 1 -4 The ideal bone graft has osteoconductive, osteoinductive, and osteogenic properties. Osteoconductive property of bone graft refers to the 3-dimensional matrix that forms a scaffold for the eventual formation of bone. This osteoinductive matrix provides a porous environment for ingrowth of blood vessels and a surface friendly for cell growth and differentiation into bone lineage. Osteoinductive property refers to growth factors within the graft that recruit cells into the matrix and drive cell differentiation into bone forming lineage. Osteogenic property refers to the presence of cells that are crucial steps of bone formation and remodeling such as mature osteoblast and stem cells. Osteobiologics can have varying degrees of autograft, allograft, or synthetic components. Non-autograft osteobiologics can be further divided into bone graft extenders (reducing the amount of autologous bone needed while achieving similar fusion rates), enhancers (combined with autologous bone to enhance the fusion), and substitutes (have same efficacy or better than autograft). The spine biologics domain expanded from being primarily iliac bone autograft to demineralized bone materials (DBMs), bone morphogenetic protein-2 (BMP-2), synthetics, and bone marrow aspirates. Although those materials have been used in the past 2 decades, the literature on their effectiveness is inconclusive. Several systematic reviews developed by AOSpine Knowledge Forum Degenerative reported low level of evidence primarily due to the high risk of bias, small sample size, lack of control groups, and limited patient-reported outcomes. 5 -8 In addition, the majority of studies are often rated level III evidence or below. Furthermore, the vast majority of the currently available osteobiologics products, have little or, in some cases, no clinical evidence for efficacy. The choice of the osteobiologic product can greatly affect the cost and outcome of surgery. Given the very limited evidence of most osteobiologics, it is hard to predict effectiveness of the osteobiologics and how it affects patient outcomes.
A systematic method of judging the effectiveness of osteobiologics does not exist. Currently, the level of evidence that supports the use of different osteobiologics varies greatly. In fact, many products that are marketed are only supported by minimal evidence. Some materials without any osteoinductive properties are tested as bone void filler and brought to market with that minimal level of data. Other materials such as DBM may have actual osteoinductive molecules, but DBMs vary in osteoinductive activity between brands and from lot to lot within brands. Other materials may have cellular components and may even be marketed as products containing “stem cells”; however, the scientific data that the “stem cells” actually have any effect on fusion is limited to a few peer-reviewed studies with small sample sizes and often without a comparator. 9 -14 In evaluating the strength of evidence that an osteobiologic has any of the 3 main characteristics, in particular, osteogenic and osteoinductive activity, it is important to have a hierarchy of in vivo over in vitro data. Within in vivo animal models, there is a hierarchy of spine fusion models determined by the species and the time to fusion. 15,16 An example is BMP-2 and the concentration required to achieve fusion in different species along with the length of time. In general, the larger the animal the higher is the required concentration of BMP-2. However, this is not always true as the sheep interbody fusion model results in a very high fusion rate even when osteobiologics with relatively little osteoinductivity are used.
We believe that there is a need for a systematic method of rating the evidence to support the effectiveness of an osteobiologic. This would be a tremendous aid for stakeholders such as surgeons, patients, insurance companies, hospitals, and governmental agencies to quickly evaluate osteobiologics. The purpose of this article is to introduce a classification for the levels of evidence for osteobiologics and to provide a description of the principles by which osteobiologics can be evaluated.
Development Process
During the course of conducting systematic reviews on osteobiologics, AOSpine Knowledge Forum Degenerative was trying to synthesize conclusions based on the available evidence. Clinicians and researchers in our team were not able to provide any recommendation based on the results of various systematic reviews due to the lack of evidence and high risk of bias.
We decided to generate a universal classification for all osteobiologics by applying general concepts used to classify clinical publications based on the level of evidence. We believe that Bone Osteobiologics and Evidence (BOnE) classification will enhance preoperative treatment planning, will provide sound basis for research studies and outcomes evaluation, and simplify physicians’ and hospitals’ decision making. In addition, this classification could be used by industry when designing the next generation of their graft material.
The process of establishing the BOnE classification included 5 face-to-face meetings and 2 web calls among members of the AOSpine Knowledge Forum Degenerative. Initial draft of the BOnE classification was based on a notion that it should be possible to divide osteobiologics into a few categories that are easily recognizable by clinicians, researchers, patients, and industry. The 3 levels of evidence were determined based on the type of data on osteobiologics. Initial classification included level A for human studies, level B for animal studies, and level C for in vitro studies, with level A being the highest level of evidence. Each level was organized into 3 subgroups (eg, A1, A2, and A3). During the initial testing of the BOnE classification, it was realized that each category required a level 4 subgroup for evidence of very low quality. Once the BOnE classification was revised, AOSpine Knowledge Forum Degenerative members had a final round of discussions where consensus was reached and the BOnE classification was formed as shown in Figure 1.
BOnE classification.
The grading principle is very straightforward: evaluation of each osteobiologic would start with the highest level of evidence and it would be downgraded based on the available evidence primarily focusing on peer review publications. White papers would not be considered. If an osteobiologic has both human and animal level 1 evidence, the osteobiologic would be classified as A1, which is the highest level of evidence.
To demonstrate the impact of BOnE classification, preliminary grading was done for several common and heavily used biologics, including rh-BMP2, Grafton DBM, Osteofil DBM, and OsteoCel. Based on a preliminary analysis, the findings are presented in Table 1. During the preliminary grading each biologic was initially given the highest grade (A1) as described above and was then downgraded, if applicable, based on the available evidence. For example, for OsteoCel biologic final grade was A4 due to the presence of uncontrolled case series (4 studies) or lesser quality retrospective cohort studies (1 study). Our follow-up studies will systematically examine each biologic by analysis of available evidence and will grade each biologic using BOnE classification. In addition, our follow-up studies will consider both approved and off-label uses for each biologic when applicable and incorporate into grading.
Examples of BOnE Classification Grading.
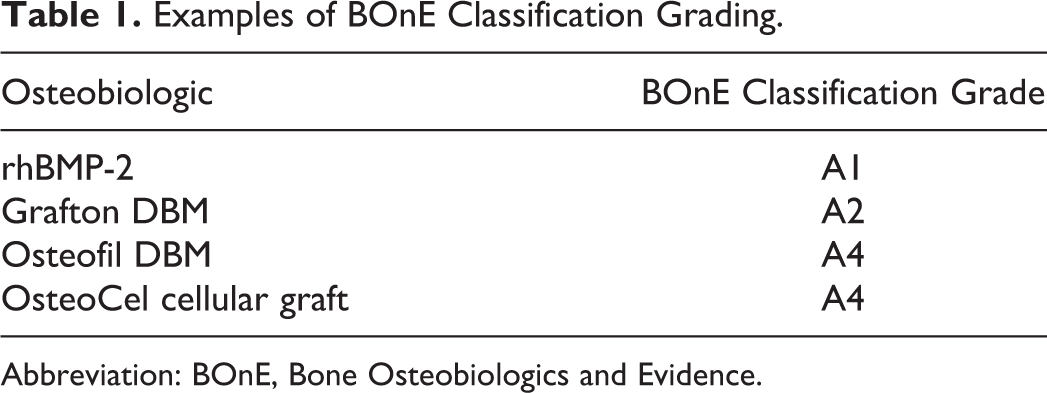
Abbreviation: BOnE, Bone Osteobiologics and Evidence.
Next Steps, Challenges, and Conclusions
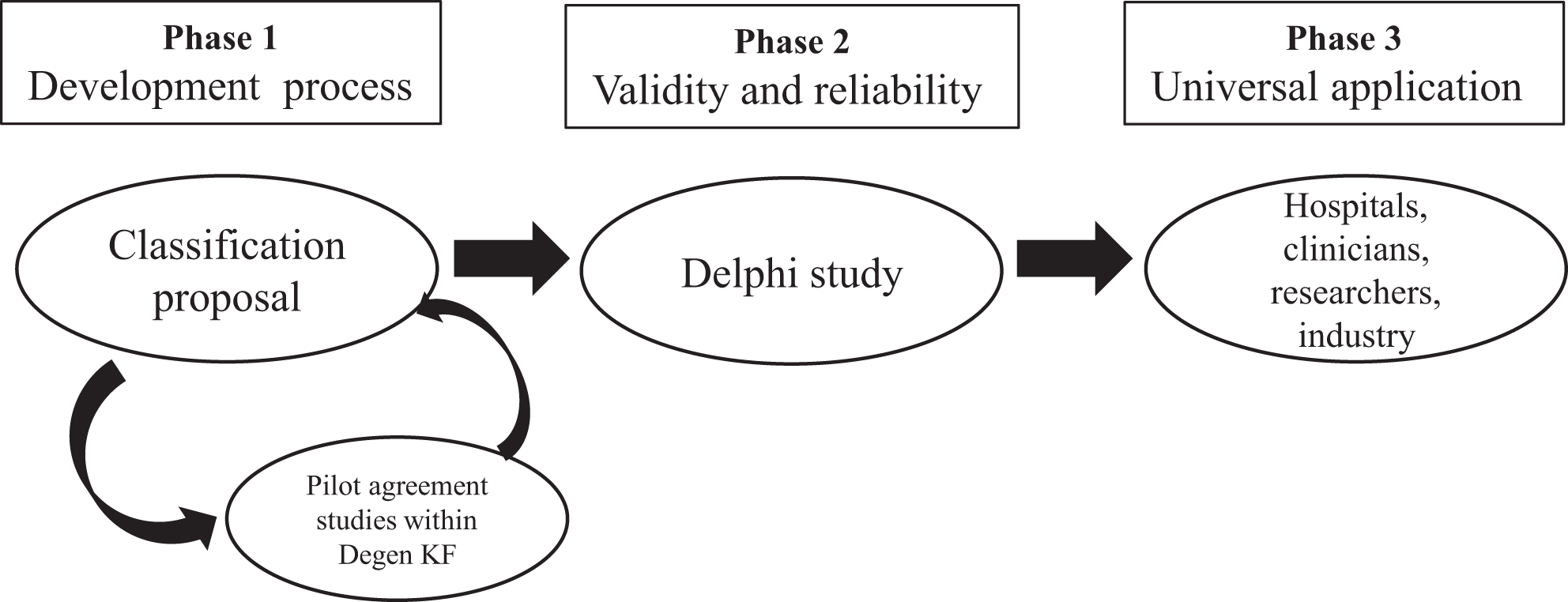
Methodological pathway for classification development was proposed by Audige et al and has been adopted by several AO Foundation classifications. 17 The original pathway suggests 3 main phases: (1) development phase, (2) reliability testing, and (3) association with patient-reported outcomes. A modified version of methodological pathway was used during the development and will be used for validation of BOnE classification, including (1) development phase, (2) validity and reliability, and (3) universal application (Figure 2).

Modified methodological pathway for establishing a classification. 17
Next steps will focus on establishing the classification’s content validity and reliability. The AOSpine Knowledge Forum Degenerative will organize a Delphi process including various stakeholders such as clinicians and researchers to test the validity of BOnE classification. Inter- and intraobserver within AOSpine Knowledge Forum Degenerative grading will be done for each current osteobiologics to test the reliability of BOnE classification. Key leaders in the spine fusion field will be invited to conduct rating of most common osteobiologics, and their scores will be used to generate the interobserver rating.
With the growing number of biologics as well as published studies, BOnE classification will have to be tested and updated for each specific biologic on a frequent basis. This can pose a challenge regarding its validity. Our team plans to develop an AOSpine platform that would have a constant feed from peer-review publications into the grading system.
In summary, BOnE classification will provide a universal platform for research studies and journal publications to classify a new or an existing product, will allow for creating decision-making algorithms for surgical planning, and help the spine community as a whole to understand pros and cons of each osteobiologic.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Disclosures outside of submitted work:ZB-consultancy: Xenco Medical, AO Spine (past); Research Support: SeaSpine (paid to the institution), Next Science (paid directly to institution); North American Spine Society: committee member; Lumbar Spine Society: Co-chair Research committee, AOSpine Knowledge Forum Degenerative: Associate member HJM – Dr Meisel is consultant (money paid to institution) - Regenerate Life Sciences GmbH for DiFusion (ongoing), Co.don (past); royalties from: Medtronic, Fehling Aesculap (past); stocks (money paid to institution) - Regenerate Life Sciences GmbH in DiFusion STY- Dr Yoon owns stock in Phygen, Alphatec, Meditech; royalties Meditech Advisors, Stryker Spine (Paid directly to institution), grant from AOSpine (Paid directly to institution/employer), research support from Biomet (Research support given to AREF), research support from Nuvasive and Medtronic DB – Consultant – Vallum, Royalties – America, Medtronic, Fellowship Support – AOSpine (paid directly to institution). JCW – Royalties – Biomet, Seaspine, Amedica, DePuy Synthes; Investments/Options – Bone Biologics, Pearldiver, Electrocore, Surgitech; Board of Directors - North American Spine Society, AO Foundation (20,000 honorariums for board position, plus travel for board meetings), Cervical Spine Research Society; Editorial Boards - Spine, The Spine Journal, Clinical Spine Surgery, Global Spine Journal; Fellowship Funding (paid directly to institution): AO Foundation PH- royalties for Medtronic, nuvasive, and summer boomer.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was organized and funded by AOSpine through the AOSpine Knowledge Forum Degenerative, a focused group of international spine experts. AOSpine is a clinical division of the AO Foundation, which is an independent medically guided not-for-profit organization. Study support was provided directly through the AOSpine Research Department.
