Abstract
Introduction
Anterior cervical discectomy and fusion (ACDF) is a standard procedure to address common cervical pathologies such as disc herniation, spondylosis, degenerative cervical myelopathy, and others. Reported pseudarthrosis rate with this procedure ranges between 10 and 15% 1 and while evidence suggests that many of the radiologic non-unions are asymptomatic, reoperation rates at the index level are higher in non-fused than in patients who achieved fusion. 2
While the gold standard bone graft material is autologous iliac crest graft, it is associated with high rates of donor site morbidity thereby resulting in increased use of alternative graft materials. Allograft is an alternative to autograft and is widely used in clinical practice, mostly for its osteoconductive properties. However, its safety and efficacy remain a concern. Allograft use as a graft material has been suggested to induce a human leukocyte antigen-antibody response and it has been suggested that allograft may delay or reduce fusion rates, increasing the risk for graft collapse and loss of normal spinal alignment; furthermore, the main concern with allograft use is the possible transmission of infections.3,4 To aid in selecting an appropriate graft source and to produce recommendations on its safety, it is important to critically analyze the complications upon their use.
Recently, the AO Spine initiated a project entitled AO Spine Guidelines for Using Osteobiologics in Spinal Degeneration (AO-GO) to guide the use of osteobiologics in ACDF. 5 The aim of this study, which was developed under the umbrella of the AO-GO project was to systematically review the literature to analyze and identify the complications associated with the use of allograft in ACDF performed at 1 or 2 levels.
Material and Methods
Eletronic Literature Search
A systematic search of PubMed/MEDLINE, EMBASE, Cochrane Library, and ClinicalTrials.gov databases was conducted for literature published between January 2000 and August 2020. Only studies reporting human clinical data with abstracts written in English were considered for inclusion. A search strategy was defined according to PICOS (participants, interventions, comparators, outcomes, and study design) approach (Supplementary Table 1). Briefly, studies including adult patients with degenerative cervical diseases undergoing 1- 2 levels of ACDF using allograft were screened. Comparative studies or case series with > 10 patients per group were included. The search strategy included MeSH terms and keywords. References of the included studies were also screened to identify additional studies.
The systematic review was planned following the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA). 6 In brief, a systematic and eliminatory selection was performed, starting with the title, then the abstract, and ending with a full-text review for final eligibility. A full-text review was performed by 2 independent reviewers and disagreement between authors was solved by a third reviewer.
Data Extraction
All studies were analyzed and characteristics such as study design, level of evidence, and funder/author’s conflict of interest were collected. Surgical details such as the number of included patients, the existence of a comparative group, number of fused levels, use of plate/fixation method, patient age, gender, body mass index (BMI), and smoking habits were also collected whenever available. Data on the following surgical complications were collected: post-operative dysphagia/dysphonia/hoarseness of voice, post-operative seroma/hematoma, wound infection/complications, post-operative radiculopathy, new neurologic deficit (defined as a 1-point decrease in motor grade), post-operative airway compromise requiring surgical intervention, epidural seroma/hematoma, cerebral spinal fluid leak/meningocele, vertebral artery injury, esophageal perforation, Horner syndrome, construct failure, the occurrence of revision surgery, and post-operative death. Fusion rate and time for fusion were also collected when reported in the study.
Medical complications such as cardiac arrest/failure/arrhythmia, deep vein thrombosis, delirium, gastrointestinal bleeding, myocardial infarction, pneumonia, pressure sores, pulmonary embolism, systemic infection, and urinary tract infection were also recorded.
Risk of Bias and Quality Assessment
Included studies were assessed for methodological quality and risk of bias by 2 reviewers using the guidance provided by the Cochrane Handbook for Systematic Reviews of Interventions. The risk of bias in the randomized controlled trials was determined using the RoB 2 tool of Cochrane Collaboration with 5 domains of risk assessment. 7 We used MINORS criteria to assess the methodological robustness of the non-randomized studies. 8 It has 12 domains of risk assessment based on which studies were graded for their methodological quality and their eligibility for inclusion in the study. Each domain was scored 0 if not reported and 1 if found to be inadequate and 2 if adequate reporting is noted for the domain concerned. The ideal score for non-comparative studies is 16 while that for a comparative study is 24.
Overall Strength of Evidence
The overall strength of evidence across all the included studies was assessed for the main outcomes using the precepts outlined by the Grading of Recommendations Assessment, Development and Evaluation (GRADE) working group9-11 and recommendations made by the Agency for Healthcare Research and Quality (AHRQ). 12 In determining the quality (strength) of evidence regarding a given outcome, the overall quality may be downgraded 1 or 2 levels based on the following domains: (1) risk of bias due to study limitations; (2) inconsistency (heterogeneity) of results; (3) indirectness of evidence (eg, hard clinical outcomes); (4) imprecision of effect size estimates (eg, width of confidence intervals); and (5) publication or reporting bias. Publication and reporting bias are difficult to assess, particularly with fewer than 10 RCTs. 13 Publication bias was unknown in all studies and thus this domain was eliminated from the strength of evidence table. The initial quality of the overall body of evidence begins as high for RCTs and low for observational studies. The body of evidence for methodologically strong observational studies may be upgraded 1 or 2 levels if there are no downgrades in the primary domains listed above and 1 or more of the following are met: (1) large magnitude of effect; (2) dose-response gradient; and (3) all plausible biases would decrease the magnitude of an apparent effect. The final overall quality (strength) of the body of literature expresses the validity of the conclusions derived from them.
Results
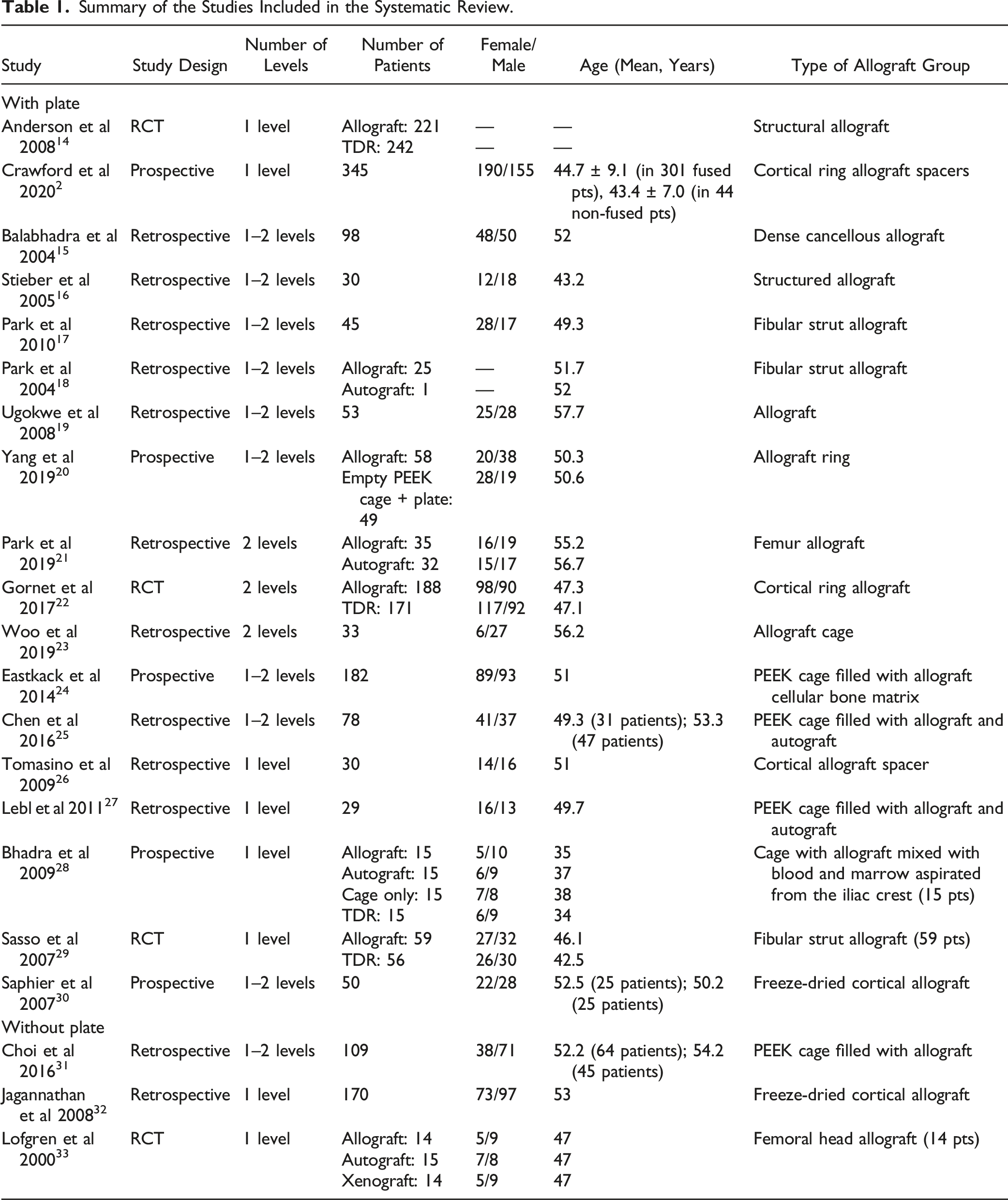
A search for studies with patients undergoing 1- or 2- level ACDF using allograft was performed to identify complications associated with allograft usage. From 584 potentially relevant citations, 21 studies met the inclusion criteria and were included in the final analysis (Figure 1). Of the 21 included studies, 4 were randomized controlled trials (RCTs), 4 were prospective, and 13 were retrospective studies respectively. These studies included a total of 2463 patients, of which 1867 were treated with allograft and 596 were controls. In 6 of the studies, ACDF with allograft was compared with one or more groups, such as ACDF with autograft in 4 studies, total disc replacement (TDR) in 4 studies, ACDF with an empty PEEK cage in 2 studies, and ACDF with bovine xenograft in 1 study. In 5 of the studies, the allograft was within a PEEK cage, whereas in the other 16 studies structural allografts were used. Plate and screws were used for fixation in 18 of the included studies. (Table 1) PRISMA flowchart of the included studies. Summary of the Studies Included in the Systematic Review.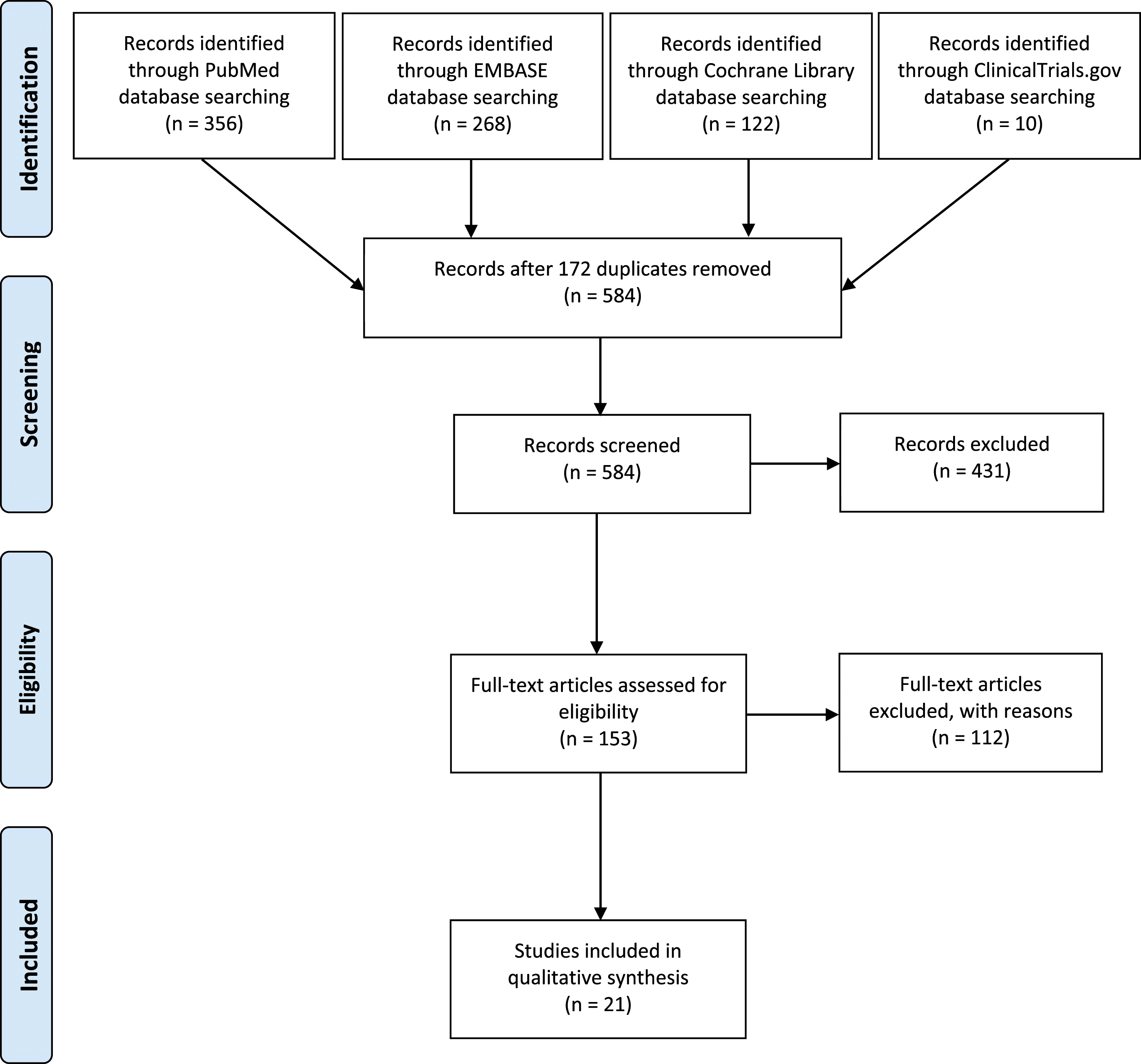
Quality Assessment
The methodological quality of the included RCTs was presented as a traffic-light plot and bar plot as in Supplementary Figure 1. The included RCTs had a low risk of bias. The methodological quality of the non-randomized studies assessed with 12 domains of MINORS criteria is presented in Supplementary Table 2. The range of total MINORS score achieved by the prospective studies was from 11 to 21 which is acceptable for analysis. The total MINORS score of the retrospective studies ranged from 5 to 11 since only 7 domains of MINORS criteria apply to them.
Fusion Rate
Fusion was reported in 14 of the 21 included studies (Supplementary Table 3). All studies assessed fusion with neutral anteroposterior and lateral radiographs, while 8 of the included studies used flexion/extension films. In 2 studies20,24 CT scans were used to assess fusion in all cases, whereas in 1 study, 17 a CT scan was used in cases where fusion was doubtful after radiographic analysis.
Reported fusion rate at 12 or more months varied between 68.5% 21 and 100%.17,20,28 In 3 of the studies, no plate was used for fixation, while in 11 studies a plate was used for fixation. Fusion in studies where no plate was used for fixation ranged between 92% and 96% and in those where a plate was used varied between 68.5% and 100%.
Fusion was reported in 4 studies with a comparative control group, although in 1 of these studies the control group had only one patient. 18 Yang et al 20 and Bhadra et al 28 reported a 12-month fusion rate of 100% in the allograft and the control group where controls were empty PEEK cages and autograft and plate, respectively. However, radiographic fusion occurred sooner in the autograft than in the empty PEEK cage group. 20 Park et al 21 reported the lowest 12-month fusion rate (68.5%) compared with the autograft group (93.8%).
Construct Failure With Loss of Correction
Construct failure with loss of correction (subsidence, screw pull out, plate or cage dislodgement) was reported in 7 studies (Supplementary Table 3). Most studies defined subsidence as a > 2 mm decrease in the anterior or posterior functional spine unit. The most frequent construct failure was cage subsidence, followed by 3 cases of graft extrusion, 31 2 cases of screw backout, 30 and 1 case of cage migration. 27 From these cases, 3 cases required revision surgery, which is further detailed below.
Revision Surgery
Revision surgery at the index level was reported in 14 studies (Supplementary Table 3) and ranged between 0%15,33 and 10.3%. 27 Most studies did not report the reason for revision surgery, and the most frequently reported reason was non-union, in 8 cases.
Post-Operative Dysphagia or Dysphonia
Post-operative dysphagia or dysphonia was reported in 12 studies (10 with plate and 2 without plate fixation; 4 comparative and 8 non-comparative) and ranged between .5% 24 and 14.4% 22 (Supplementary Table 4). In studies where a plate was used, this complication ranged between .5% 24 and 14.4%, while in the 2 studies where no plate was used, the rate of dysphagia or dysphonia was 6.4% 31 and 8.8%. 32
Six studies,20,25-27,31,32 which included 42 cases of dysphagia or dysphonia from a total of 483 allograft ACDF patients operated, reported the evolution of dysphagia. There was a complete resolution within the first 3 months in 33/42 cases and persistence in 9 patients.
Wound Related Complications
Six studies reported wound-related complications, mainly infections (Supplementary Table 4). Forty-three of 51 wound complications, however, were derived from a single study, 22 which reported a 22.8% infection rate in 188 patients. Resolution of wound infection was reported in 4 studies (5 infections in the allograft group and 2 in the control group) and all were treated with oral antibiotics except for 1 case of bovine xenograft infection which required xenograft removal at 6 months. 33
Other Reported Complications
Two studies reported on the occurrence of post-operative radiculopathy, with an incidence of 1.1% 24 and 1.8% 14 but none of these studies reported how the complication was managed. New neurologic deficit (motor grade > 1) after surgery was reported in 2 studies with incidence of 1.4% 14 and 1.9%. 19 The post-operative cerebrospinal fluid leak was reported in 2 studies with incidence of 1.4% 14 and 1.7%. 20 Post-operative airway compromise requiring re-intervention was reported in 2 studies and occurred in 1.4% 14 and 3.3% 26 of included patients. Post-operative subcutaneous seroma/hematoma was reported in 3 studies with the incidence of 1.4%, 14 6.7%, 26 and 6.9%, 27 of which only 1 patient required re-intervention. One case of epidural hematoma, which caused spinal cord compression was reported 26 and the patient was submitted for revision surgery 3 weeks after the initial procedure. Jagannathan et al 32 reported a case of Horner syndrome in a cohort of 170 patients submitted to ACDF using an allograft (Supplementary Table 5).
Medical complications were inconsistently reported in analyzed studies. Only one study reported a death in the control (TDR) group. 22
Discussion
The ideal graft material to use in ACDF is a matter of controversy. Despite knowing the advantages and disadvantages of mostly commonly used grafts, it is still unclear to recommend an appropriate choice. In this study the most frequently reported complication was post-operative dysphagia or dysphonia, followed by revision surgery and wound-related complications, particularly infection.
Only 14 out of the 21 included studies reported fusion rate. Concerning the assessment of fusion rate the criteria largely varied between studies, with 2 studies routinely used CT scans, one used CT scans in doubtful cases and 8 studies used flexion/extension films but rarely specified which radiologic factors were analyzed in each case. While few studies have suggested that non-union can be accurately identified on both plain radiographs and CT images34,35 a recent systematic review recommended using < 1 mm of motion between spinous processes on extension and flexion views to confirm fusion in ACDF. 36 The inconsistency in reporting fusion and the non-uniform way in which it was reported in the 14 studies may be the reason for the large variability in the rate of fusion among them (68.5–100%). Three studies reported a 100% fusion rate. Interestingly, in all these studies, a plate was used for allograft fixation. The role of plate fixation in reducing allograft subsidence and in increasing fusion rates to values similar to those found with autograft has previously been reported.37,38 However, not all studies where plates were used for fixation had higher fusion rates and in the study reporting the lowest fusion rate (68.5%) plates were used. 21
Post-operative dysphagia or dysphonia was reported in twelve out of twenty-one studies. However, their reporting was not precise, with some studies not distinguishing between dysphagia and dysphonia. These, however, are completely 2 different complications. Dysphagia is a subjective symptom related to difficulty in swallowing after surgery. Its causes may be due to neuronal, mucosal or muscular structures and the most frequent cause may be swelling of the surrounding soft tissue structures, alterations in the normal esophageal motility, and altered sensation resulting from traction during surgery.39,40 Other nonsurgical factors such as position of the endotracheal tube cuff have also been implicated in post-operative dysphagia. 39 Dysphonia after anterior cervical surgery is often attributed to vocal cord paralysis due to laryngeal nerve injury 41 and its incidence has been reported between .06 and 11%, with persistence occurring in 0–3.5% cases. 42 It was not possible to understand if this complication occurred more frequently with allograft than with other graft materials, due to the limited comparative data in the included studies. Dysphagia or dysphonia was reported in studies with or without plate fixation but its incidence was higher in studies where plates were used for fixation. Furthermore, of the 3 studies reporting incidence of dysphagia or dysphonia above 10%, all used plated constructs.22,25,30 Of them, there was only 1 comparative study that reported 14.4% dysphagia/dysphonia in plated ACDF. 22 Other studies have suggested that the more extensive retraction and dissection required to implant a plate and triangulate the screws may be responsible for the higher incidence of these complications in the plated. 43 In the present review only 6 of the identified studies reported the evolution of the dysphagia or dysphonia with time, They reported resolution of the dysphagia or dysphonia mostly in the first 3 months.
Subsidence was the most frequently reported construct failure. Plate use has been reported to decrease the subsidence rate37,38 but, in the analyzed studies, subsidence was only reported in studies where plates were used. Overall, the revision rate was 6.4% with the most frequent reason for revision being non-union. Wound-related complications, particularly wound infections are particularly rare after ACDF. In this systematic review, however, a 5.7% wound-related complication rate was found. This surprisingly high rate of complications, however, was mostly derived from 1 study which reported a 22.8% infection rate. 15 Resolution of wound infection was scarcely reported but, required only oral antibiotics and no revision surgery was reported for this specific reason after ACDF using allograft.
Serious complications, such as post-operative new neurologic symptoms, cerebrospinal fluid leak, airway compromise, seroma and hematoma (including epidural), and Horner syndrome were reported in only one or two studies each. Due to the impreciseness of the included studies, it is not known if these complications simply did not occur or were not analyzed. However, and due to the seriousness of these complications, one may speculate that their incidence was low and that the cases in which they occurred are mirrored in the reported cases.
The literature on 1- or 2-level ACDF using allograft provides a low-quality evidence to generate recommendations and has significant limitations to analyse complications of this graft type. The results from this study are limited in their robustness due to the nature of literature available on the subject. First, there was a lack of appropriate controls in the identified studies, with few studies comparing allograft against TDR and different graft types (mostly autograft). Second, the complications were inconsistently reported among the included studies and their definition and grading were not precise and uniform. Third, the authors of most papers fail to report on the resolution of the complications. Fourth, most of the identified complications are not specific to allograft use and, therefore, the lack of comparative studies with autograft or other osteobiologics makes it difficult to assess if allograft is, in itself, responsible for any of these complications. It is most likely that these complications are related to the surgical approach, dissection, disc level operated and comorbidities of the patients included in the studies. This makes it very difficult to provide sound recommendations on the safety profile of allograft use in ACDF.
To overcome such difficulties and to aid in providing recommendations on the use of allograft and other osteobiologics, it is imperative that regulators advise healthcare management to maintain surgerical registries and report the complications in a uniform manner. Adverse event severity systems such as the SAVES-V2 system should also be used to identify and report complications in spinal surgeries. 44 Future studies should include a comparison between different graft materials for use in ACDF surgery. Ideally, randomized controlled trials are needed to aid in selecting the ideal graft type for individual clinical scenarios.
Conclusions
The overall reporting of complications was low with very few comparative studies. Reported complications with allografts are within the range of other osteobiologics and autografts and in most cases may not attributable to the use of osteobiologics and may be complications of the procedure itself. There was substantial heterogeneity between studies to allow for a sound conclusion and recommendation on complications associated with the use of allograft in 1- or 2-level ACDF. Despite a large number of osteobiologics currently available in the market, there is a lack of guidelines on their usage based on their efficacy and safety outcomes, stressing the need for well-designed studies with sufficient sample size and appropriate comparator groups.
Supplemental Material
Supplemental Material - Complications of the Use Allograft in 1-or 2-Level Anterior Cervical Discectomy and Fusion: A Systematic Review
Supplemental Material for Complications of the Use Allograft in 1-or 2-Level Anterior Cervical Discectomy and Fusion: A Systematic Review by Ricardo Rodrigues-Pinto, Sathish Muthu, Sara E. Diniz, Juan Pablo Cabrera, Chris Martin, Neha Agarwa, Hans-Jorg Meisel, Jeffrey Wang, Zorica Buser, and AO Spine Knowledge Forum Degenerative in Global Spine Journal.
Footnotes
Acknowledgments
The authors would like to thank Yabin Wu for the management of the AO Spine Knowledge Forum Degenerative and for all the planning, support and valuable contribution throughout the research conducting to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
