Abstract
Study Design
A prospective observational study.
Objectives
To investigate preoperative prevalence of neurogenic bowel dysfunction and neurogenic bladder in patients with degenerative cervical myelopathy (DCM) and examine the degree and timing of symptom improvement after cervical decompression surgery. Factors contributing to symptom improvement were also analyzed.
Methods
Among 75 patients with DCM who underwent cervical decompression, Constipation Scoring System (CSS) score, International Prostate Symptoms Score (IPSS), and Japanese Orthopaedic Association (JOA) score were assessed before surgery and 1, 3, 6, and 12 months after. Prevalence rates were calculated. Data regarding patient age, sex, disease status, disease duration, lesion level, and score changes was prospectively recorded and analyzed.
Results
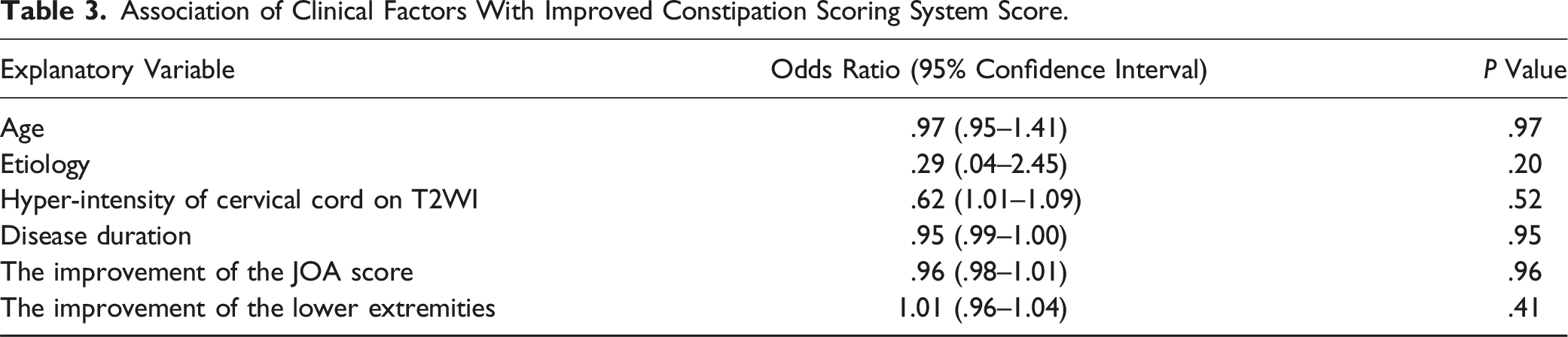
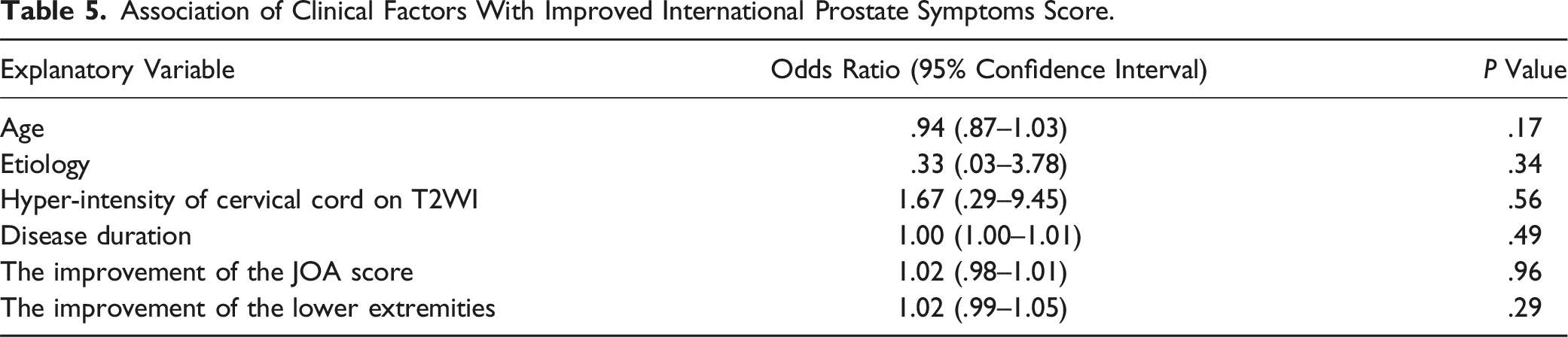
The prevalence rates of defecation and urinary dysfunction before surgery were 41.3% and 34.7%, respectively. Among the patients with defecation dysfunction, the number of patients who improved 1, 3, 6, and 12 months after surgery was 10, 9, 9, and 6, respectively. Among the patients with urinary dysfunction, the corresponding number of patients was 12, 10, 11, and 11, respectively. None of the factors we examined were significantly associated with improvement in either CSS or IPSS score; however, improvement of lower extremity JOA score tended to be associated with improvement in both.
Conclusions
The prevalence of symptoms of defecation and urinary dysfunction in patients with DCM was 41.3% and 34.7%, respectively. Decompression surgery improved symptoms in 20% to 46% of patients.
Keywords
Introduction
Degenerative cervical myelopathy (DCM) encompasses a wide range of motor, sensory, and autonomic symptoms and signs that result from spinal cord compression and dysfunction. In severe cases, the compression can lead to problems with bowel and bladder function, including loss of control. Neurogenic bowel dysfunction and neurogenic bladder are 2 major complications of spinal cord injury. However, the Japanese Orthopedic Association (JOA) score and the Japanese Orthopaedic Association Cervical Myelopathy Evaluation Questionnaire (JOACMEQ) only evaluate urinary symptoms. The relationship between DCM and neurogenic bowel dysfunction has not been well investigated. Neurogenic bowel dysfunction is classified into 2 types according to neurologic level and lesion severity: reflex bowel dysfunction and areflexic/flaccid bowel dysfunction. 1 In patients with spinal cord dysfunction, both neurogenic bowel dysfunction and neurogenic bladder can affect lifestyle and quality of life. A web-based survey regarding neurogenic bowel dysfunction in Japan showed that 55% of spinal cord injury patients and 51% of spina bifida patients report severe neurogenic bowel dysfunction. 2
This study aimed to investigate the following in DCM patients undergoing cervical spine decompression surgery with or without fusion: (1) preoperative prevalence of neurogenic bowel dysfunction and neurogenic bladder; (2) degree and timing of postoperative improvement of neurogenic bowel dysfunction and neurogenic bladder; and (3) factors contributing to improvement of bowel and bladder symptoms.
Methods
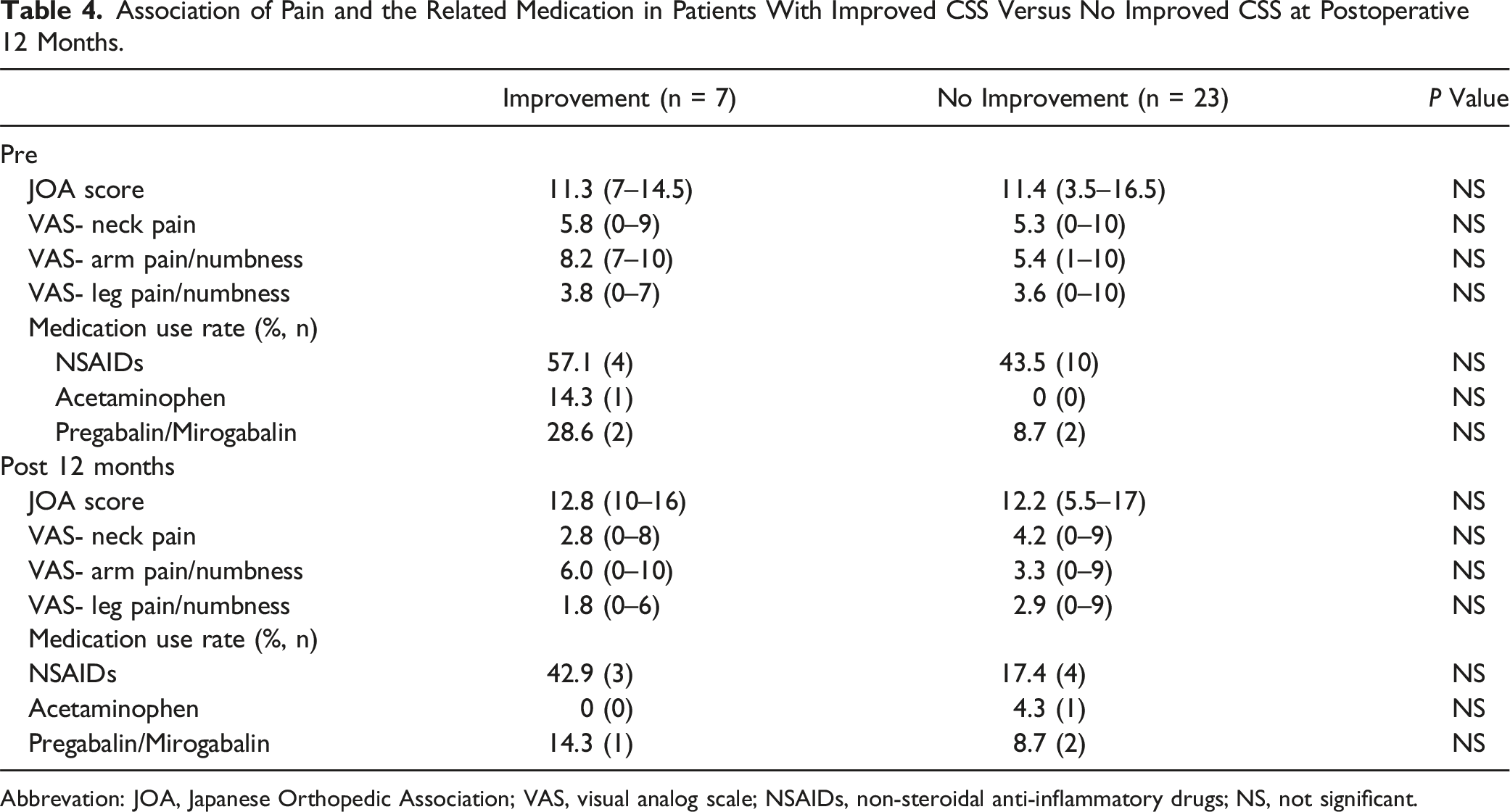
In total, 184 patients with surgically treated DCM were included from February 2018 to December 2020. Inclusion criteria were: (1) neurological symptoms and signs compatible with DCM, such as increased deep tendon reflexes and bilateral extremity pain or numbness; and (2) cervical spinal cord compression demonstrated on magnetic resonance imaging or myelography. We excluded patients with cervical spondylotic radiculopathy alone; lower urinary tract pathology such prostatic hypertrophy; concomitant thoracic or lumbar lesions; and history of spinal, urological, digestive or gynecological surgery. Patients taking medication that can affect bowel or bladder function or who were managing urinary dysfunction using self-catheterization were excluded from the improvement analyses. Of these 184 patients, 75 patients met these criterion and data was prospectively collected. All patients underwent a urologic evaluation by a urologist to rule out lower urinary tract lesions. Detailed micturition and defecation histories were obtained from each patient using the Constipation Scoring System (CSS) and International Prostate Symptoms Score (IPSS). Clinical symptoms and CSS, IPSS, and JOA scores were assessed immediately before and 1, 3, 6, and 12 months after surgery. Additionally, the JOA recovery rate regarding lower motor/sensory function was calculated using the following formula: recovery rate = (JOA score regarding lower motor/sensory function 1 year after surgery − preoperative JOA score regarding lower motor/sensory function)/(6 − preoperative JOA score regarding lower motor/sensory function) × 100. Factors including patient age, sex, disease status, disease duration, lesion level, and changes in CSS, IPSS, and JOA scores were recorded. Pain due to sympathetic hyperactivity can cause defecation difficulties. 3 Visual analog scale (VAS) for neck pain, arm pain/numbness and leg pain/numbness and medication for pain and numbness use rate was investigated, comparing CSS improved group with no improved group at postoperative 12 months. Furthermore, correlations between the improvement of IPSS/CSS and clinical factors including age, disease duration, preoperative upper extremity/trunk, lower extremity and bladder score in JOA scoring system and the JOA recovery rate regarding lower motor/sensory function were calculated by Spearman rank correlation coefficient and visualized by heatmap. The study was approved by the institutional review board of our hospital (FHR 2022-4) and written informed consent was obtained from all patients.
Constipation Scoring System
The CSS is based on 8 questions concerning bowel evacuation. 4 For each question, the patient chooses one of 5 options indicating severity of a particular symptom. The answers are assigned points ranging from zero to 4. The total score can therefore range from zero to 30. The 8 questions refer to the following evacuation symptoms: (1) frequency of bowel movements, (2) difficult/painful evacuation efforts, (3) subjective feeling of evacuation completeness, (4) abdominal pain with evacuation, (5) time in lavatory per evacuation attempt, (6) assistance needed for evacuation, (7) number of unsuccessful evacuation attempts per 24 hours, and (8) duration of constipation. Symptomatic neurogenic bowel dysfunction was defined as total CSS score ≥4. 5 “Significant improvement” was defined as improvement from CSS score ≥4 to ≤3 after surgery.
International Prostate Symptoms Score
The IPSS is based on 7 questions concerning urinary symptoms and one question concerning quality of life. 6 For each question, the patient is asked to choose one of 6 answers that indicate severity of a particular symptom. The answers are assigned points from zero to 5. The total score can therefore range from zero to 35 (asymptomatic to very symptomatic). The 7 questions refer to the following urinary symptoms: (1) incomplete emptying, (2) frequency, (3) intermittency, (4) urgency, (5) weak stream, (6) straining, and (7) nocturia. “Symptomatic” neurogenic bladder was defined as total IPSS score ≥8. “Significant improvement” was defined as improvement from IPSS score ≥8 to ≤7 after surgery.
Statistical Analysis
Statistical analyses were performed using JMP statistical software version 13 (SAS Institute Inc., Cary, NC, USA). Comparisons were performed using the chi-square test, Mann–Whitney U test, and Fisher exact probability test as appropriate. Binomial logistic regression was performed to calculate odds ratios (ORs) with 95% confidence interval (CI). P < .05 was defined as significant.
Results
Patient Characteristics.

Values shown are numbers or means with range.
Abbrevation: JOA, Japanese Orthopaedic Association; IPSS, International Prostate Symptoms Score; CSS, Constipation Scoring System.
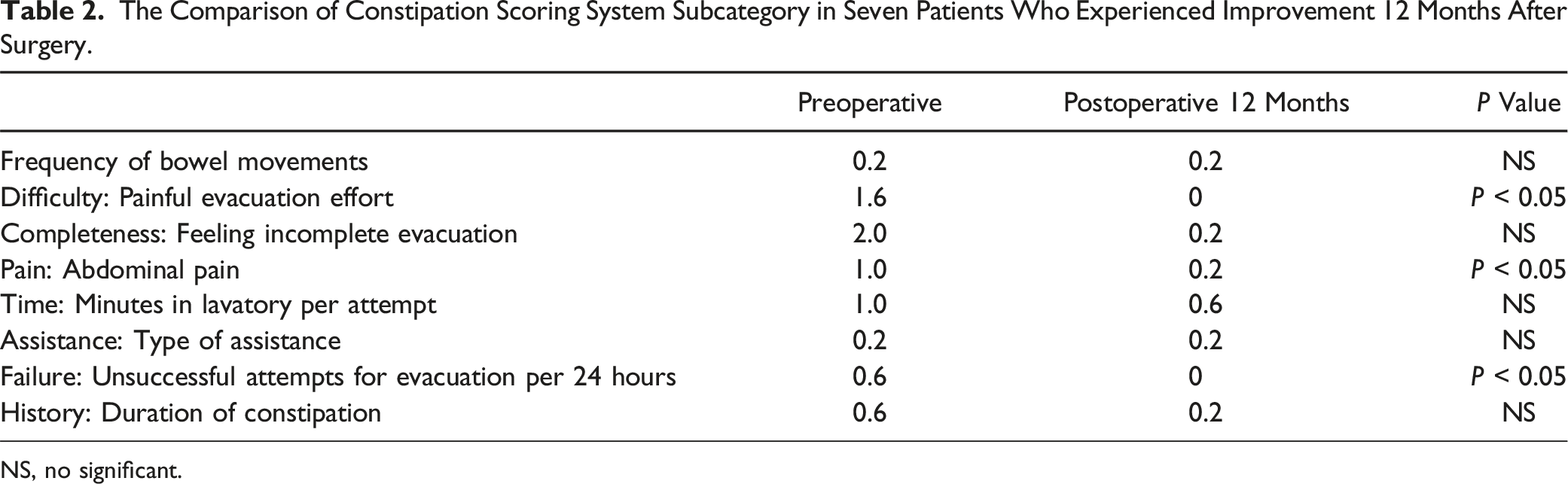
Among the 31 patients with defecation symptoms, the number of patients who improved at 1, 3, 6, and 12 months after surgery was 11, 10, 10, and 7, respectively (Figure 1). In the 7 patients who showed improvement in defecation symptoms 12 months after surgery, the subcategory scores for difficult/painful evacuation, success of evacuation, and abdominal pain with evacuation had especially improved (Table 2). Among the 7 patients with improved defecation symptoms 1 year after surgery, two had preoperative urinary symptoms. Only one of these 2 experienced improved urinary symptoms after surgery. The group with improved CSS had a higher mean improvement in lower extremity JOA than the group without improvement (OR 1.02; 95% CI, .96-1.04; P = .41; Table 3); however, this difference was not statistically significant. The improvement of CSS was not significantly correlated with pain and pain/numbness medication (Table 4). Constipation Scoring System status (improved vs. unimproved) in 30 patients with defecation symptoms immediately before surgery and 1, 3, 6, and 12 months after surgery. The Comparison of Constipation Scoring System Subcategory in Seven Patients Who Experienced Improvement 12 Months After Surgery. NS, no significant. Association of Clinical Factors With Improved Constipation Scoring System Score. Association of Pain and the Related Medication in Patients With Improved CSS Versus No Improved CSS at Postoperative 12 Months. Abbrevation: JOA, Japanese Orthopedic Association; VAS, visual analog scale; NSAIDs, non-steroidal anti-inflammatory drugs; NS, not significant.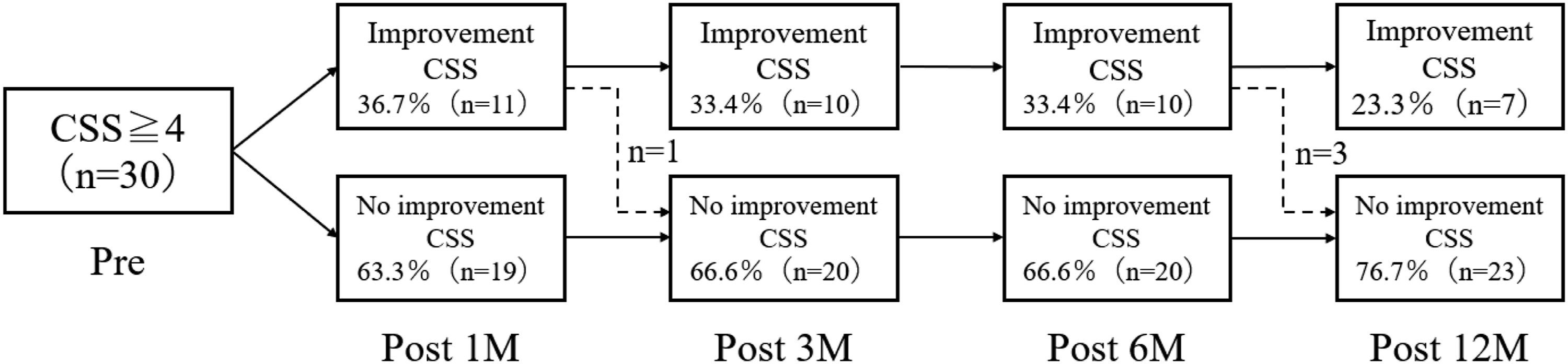
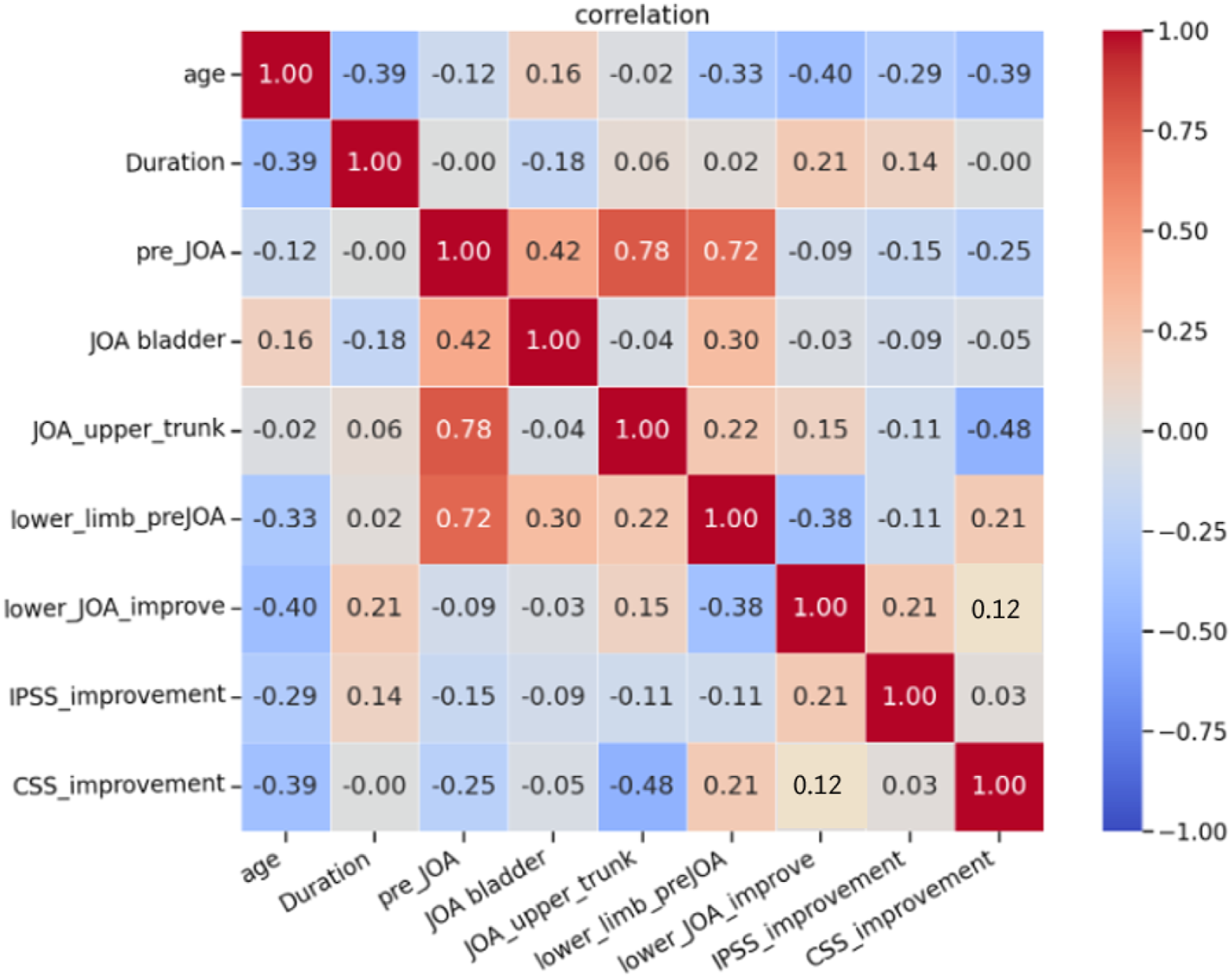
Among the 26 patients with urinary symptoms before surgery, one patient was excluded from the improvement analysis because they began performing self-catheterization for urinary symptoms after surgery. Among the remaining 25, the number of patients who improved at 1, 3, 6, and 12 months after surgery was 13, 11, 12, and 12, respectively (Figure 2). Among the 12 patients whose urinary symptoms improved 1 year after surgery, eight had defecation symptoms. Among these, 7 patients did not experience improvement of defecation symptoms. The group with improved IPSS had a higher mean improvement in lower extremity JOA than the group without improvement (OR, 1.02; 95% CI, .99-1.05; Table 5); however, this difference was not statistically significant. The improvement of IPSS was not obviously correlated with preoperative upper extremities/trunk, preoperative lower extremities and preoperative bladder JOA score (Figure 3). The population with a significantly positive relation was only the JOA recovery rate regarding lower motor/sensory function (.21). The improvement of CSS was correlated with preoperative lower extremities JOA score and the JOA recovery rate regarding lower motor/sensory function (0.21, 0.12). Constipation Scoring System status (improved vs. unimproved) in 29 patients with defecation symptoms immediately before surgery and 1, 3, 6, and 12 months after surgery. Association of Clinical Factors With Improved International Prostate Symptoms Score. Correlation matrix for CSS/IPSS. Some clinical factors were negatively related, represented in blue, and others were positively related, represented in red.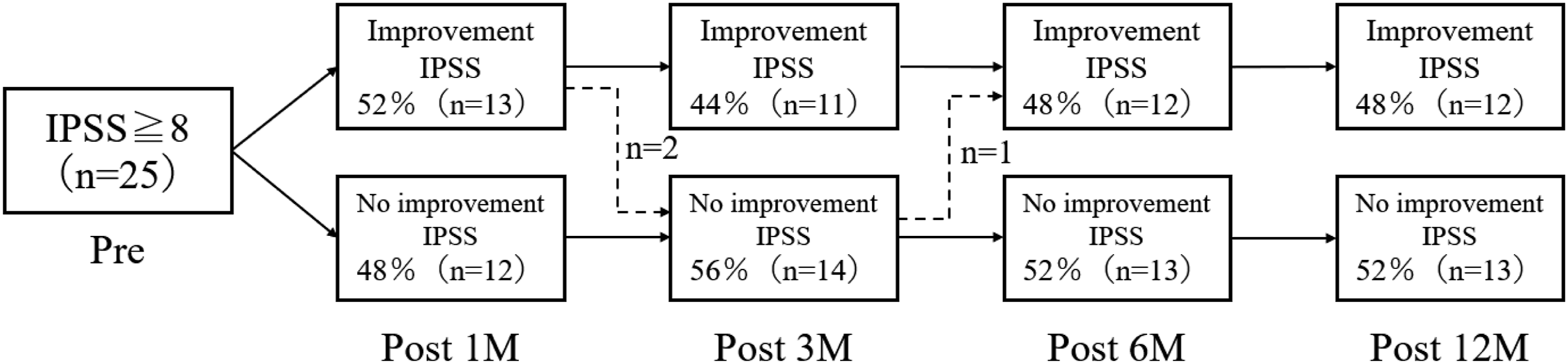
Fifteen patients had both defecation and urinary symptoms before surgery. Among these, one patient (6.7%) experienced improvement in both types of symptoms after surgery; 6 (40%) experienced improvement in only urinary symptoms and 5 (33.3%) experienced improvement in only defecation symptoms. Neither defecation nor urinary symptoms improved in 3 patients (20%).
Discussion
In the evaluation of DCM symptoms, neurogenic bowel dysfunction is frequently overlooked. However, DCM patients sometimes experience improvement of defecation symptoms after cervical decompression surgery. Davies et al surveyed symptoms associated with DCM from the perspective of people living with the condition, in which urinary and defecatory symptoms comprised approximately 20% to 40%. 7 Patients-reported outcome measures such as JOA scoring system and IPSS/CSS are useful tools to capture information on an individual’s perception of his or her level of impairment, disability and quality of life.8,9 Although the prevalence of symptoms related to storing and/or voiding urine in DCM patients ranges between 40% and 80%,10,11 the relationship between DCM and neurogenic bowel dysfunction has not been reported. All patients do not always achieve neurological recovery after surgery. Rehabilitation could improve disabled function. Especially, targeted rehabilitation is important as soon possible after surgery, depending on the clinical manifestations. 12 It is reported that body weight-supported treadmill excise has a benefit for improving bladder and bowel function. 12
The parasympathetic innervation of the right colon is derived from the vagus nerve. In the left colon and anorectum, parasympathetic innervation is derived from preganglionic parasympathetic neurons in the sacral spinal cord whose axons innervate postganglionic neurons in the peripheral ganglia located in the wall of the intestine. The sacral preganglionic neurons receive input from the brainstem via descending tracts that travel through the cervical and thoracic spinal cord.13,14 Sympathetic input to the gastrointestinal tract arises from the spinal cord (T5 to L3 levels).13,14 The locations of the defecation and urinary pathways within the spinal cord are different. Nathan et al reported that the afferent and efferent tracts serving defecation run within a narrow band extending from the periphery of the cord to approximately the lateral horn of the grey matter. 15 In contrast, the descending pathway responsible for coordination of the bladder and urethral sphincter is located mainly in the lateral column, while the ascending pathway is located in the dorsal column. 16 Moreover, the intestinal wall contains intrinsic pacemakers and a neural network that is part of the sympathetic and parasympathetic chain that programs smooth muscle activity that is necessary to expel waste material 17 ; the neurologic system controlling urinary function lacks such a network. Furthermore, defecation can be additionally facilitated by volitional increases in intra-abdominal pressure. 18 Valles et al reported that weak abdominal muscles contribute to constipation in patients with incomplete spinal cord injury above the T7 level, including the cervical cord. 19
The DCM patients in our study exhibited discrepancies in improvement of defecation and urinary function after surgery. Voluntary abdominal muscle control, which aids defecation, may be one explanation. Other possibilities include the difference in storage capacity for stool and urine as well as the fact that the defecation system is more complicated than the urinary system.
Approximately 30% to 40% of the DCM patients in our study had symptoms of defecation and/or urinary dysfunction. Symptom improvement was observed only 1 month after cervical decompression surgery in 20% to 50% of these patients and persisted for at least 1 year. According to the present study, bladder score in JOA scoring system could not show the subtle changes of bladder function in DCM. The JOA scoring system seems to be unsatisfactory tool to examine neurogenic bladder function. Also, JOA scoring system could not originally evaluate bowel function at all. To investigate neurogenic bladder and bowel function in DCM, IPSS/CSS scoring system is more preferable to JOA scoring system. None of the factors we examined were significantly associated with improvement in either CSS or IPSS; however, improvement of lower extremity JOA score tended to be associated with improvement in both. As in previous studies of patients with cauda equina syndrome, 5 the CSS indices for difficult/painful evacuation efforts and number of unsuccessful evacuation attempts per 24 hours significantly improved in our DCM patients after surgery. In addition, our patients also experienced improvement in abdominal pain with evacuation. We speculate that the spasticity of defecation-related muscles decreased after decompressive surgery, similar to the decreased extremity spasticity frequently observed after surgery, which resulted in improved defecation function. Also, an improvement of bowel symptoms persisted up to one year in patients with cauda equina syndrome, 5 whereas there was a gradual decrease in the number of patients that noticed an improvement in their bowel symptoms as time progressed. Compared with patients with cauda equina compression, more multi-factors as mentioned above may play an important role in the pathogenesis of bowel movement disturbance in the patients with cervical myelopathy. To the best of our knowledge, no previous study has described the relationship between constipation and DCM. Further investigation of the relationship between anorectal function and DCM using anorectal manometry is warranted.
This study has several limitations. First, the causes of constipation were varied and may have been multifactorial, as patient mental status, meal content, and activities of daily living can affect defecation. Second, the CSS cut-off value of 4 to define symptomatic neurogenic bowel dysfunction was arbitrary. Although we used a score of 4, the rate of patients with symptomatic neurogenic bowel dysfunction would have differed if a different cut-off score was used. Finally, the sample size was small and had limited statistical power. Despite these limitations, we believe that our study provides important information regarding defecation and urinary symptoms in patients with DCM.
Conclusion
The prevalence of symptoms of defecation and urinary dysfunction in patients with DCM was 41.3% and 34.7%, respectively. Decompression surgery improved symptoms in 20% to 46% of patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
