Abstract
Study Design
Delayed diagnosis of degenerative cervical myelopathy (DCM) is likely due to a combination of its subtle symptoms, incomplete neurological assessments by clinicians and a lack of public and professional awareness. Diagnostic criteria for DCM will likely facilitate earlier referral for definitive management.
Objectives
This systematic review aims to determine (i) the diagnostic accuracy of various clinical signs and (ii) the association between clinical signs and disease severity in DCM?
Methods
A search was performed to identify studies on adult patients that evaluated the diagnostic accuracy of a clinical sign used for diagnosing DCM. Studies were also included if they assessed the association between the presence of a clinical sign and disease severity. The QUADAS-2 tool was used to evaluate the risk of bias of individual studies.
Results
This review identified eleven studies that used a control group to evaluate the diagnostic accuracy of various signs. An additional 61 articles reported on the frequency of clinical signs in a cohort of DCM patients. The most sensitive clinical tests for diagnosing DCM were the Tromner and hyperreflexia, whereas the most specific tests were the Babinski, Tromner, clonus and inverted supinator sign. Five studies evaluated the association between the presence of various clinical signs and disease severity. There was no definite association between Hoffmann sign, Babinski sign or hyperreflexia and disease severity.
Conclusion
The presence of clinical signs suggesting spinal cord compression should encourage health care professionals to pursue further investigation, such as neuroimaging to either confirm or refute a diagnosis of DCM.
Introduction
Degenerative cervical myelopathy (DCM) is a progressive spine condition and the most common cause of spinal cord dysfunction worldwide.1,2 Patients with DCM can present with subtle, non-specific symptoms in their upper and lower extremities, making it difficult to initially diagnose this condition.3,4 Behrbalk et al (2013) determined that the average time to diagnosis of DCM was 2.2 ± 2.3 years and that patients attended an average of 5.2 ± 3.6 physician visits before obtaining a correct diagnosis. 5 The first line physician was a primary care practitioner in 69% of cases and an orthopedic surgeon in 21% of cases. 5 DCM was most commonly mistaken for carpal tunnel syndrome or cervical disc radiculopathy without neurological deficit. Similarly, others have reported that the time between symptom onset and surgical evaluation was 17.7 ± 16.0 months. 6 This delay in diagnosis and assessment by a spinal surgeon has detrimental consequences and can result in incomplete postoperative recovery, impaired quality of life and significant disability including inability to work.7,8 DCM is often managed surgically with response to treatment dependent on degree of preoperative functional impairment and duration of symptoms.9,10 As such, timely diagnosis and management is critical in order to optimize outcomes.
Patients with DCM complain of symptoms in their upper and lower extremities, including bilateral arm paresthesia, reduced manual dexterity, gait instability, and weakness. Other symptoms include neck pain or stiffness, Lhermitte’s phenomena and urinary or fecal urgency or incontinence. On examination, patients with DCM typically present with bilateral motor and/or sensory deficits of the upper and lower extremities without facial involvement, although clinical variations can occur. 11 Furthermore, patients with DCM exhibit a combination of upper and lower motor neuron signs (manifestation of the disease that is identified during an examination) as well as abnormalities in the sensation of pain, temperature, proprioception and vibration. Upper motor neuron signs include hyperreflexia below the level of the lesion, Hoffmann sign, upgoing plantar responses, lower limb spasticity, and corticospinal distribution motor deficits. 11 Lower motor neuron signs result from compression of the nerve roots as they exit the spinal canal and commonly include muscle atrophy especially in the hands, muscle fasciculations and weakness. However, patients do not always present with clear signs of DCM, but rather demonstrate nonspecific dissociated sensorimotor deficits and subtle gait disturbances. Furthermore, common comorbidities in patients of typical DCM age, such as carpal tunnel syndrome, radiculopathy and arthritis, may further impede the diagnosis. Therefore, clinical signs of cervical myelopathy (ie spinal cord involvement) are of greatest diagnostic value.
Misdiagnosis or delayed diagnosis of DCM is likely associated with its subtle, non-specific symptoms, incomplete neurological assessments by clinicians and a lack of public and professional awareness.12,13 Developing diagnostic criteria for DCM will likely improve diagnosis and was determined as a research priority as part of the AO Spine RECODE-DCM (Research Objectives and Common Data Elements for Degenerative Cervical Myelopathy) project.14-16 The first step in this process is to identify candidate variables for inclusion in diagnostic criteria. Signs that exhibit high sensitivity and specificity, and those that are correlated with disease severity are likely the most relevant.
A previous systematic review by Cook et al (2011) aimed to evaluate the diagnostic accuracy of various clinical tests by summarizing studies that reported on sensitivity, specificity, and other metrics. 17 This review used the QUADAS (Quality Assessment of Diagnostic Accuracy Studies) tool to assess the external and internal validity of a diagnostic study and evaluate its risk of bias. Based on this scoring system, only a single study by Cook et al (2009) was rated as high quality. 18 This review concluded that the test with the highest sensitivity was the inverted supinator sign (61%), followed by the suprapatellar tendon reflex (56%) and the Hoffmann sign (44%). 17 Although the presence of clonus and the Babinski sign were not sensitive findings, they were the most specific tests (92% and 96%, respectively) for confirming a diagnosis of DCM. This review must be expanded on for the following reasons: (i) the term DCM has been introduced since its publication; (ii) several relevant studies have been conducted since 2011, including some that assess the diagnostic value of novel signs; and (iii) this review did not assess the relationship between the presence of clinical signs and disease severity.
The objective of this study was to conduct a systematic review of the literature to address the following key questions (KQ):
KQ1: What is the diagnostic accuracy (ie sensitivity, specificity, positive or negative predictive value, positive or negative likelihood ratio) of clinical signs in patients with DCM?
KQ2: What is the association between clinical signs and disease severity in patients with DCM?
Materials and Methods
The systematic review was formatted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses checklist. Neither informed consent nor Institutional Review Board approval were required due to the nature of the study. The review was not registered in PROSPERO or any other similar site.
Eligibility Criteria
Inclusion and Exclusion Criteria.
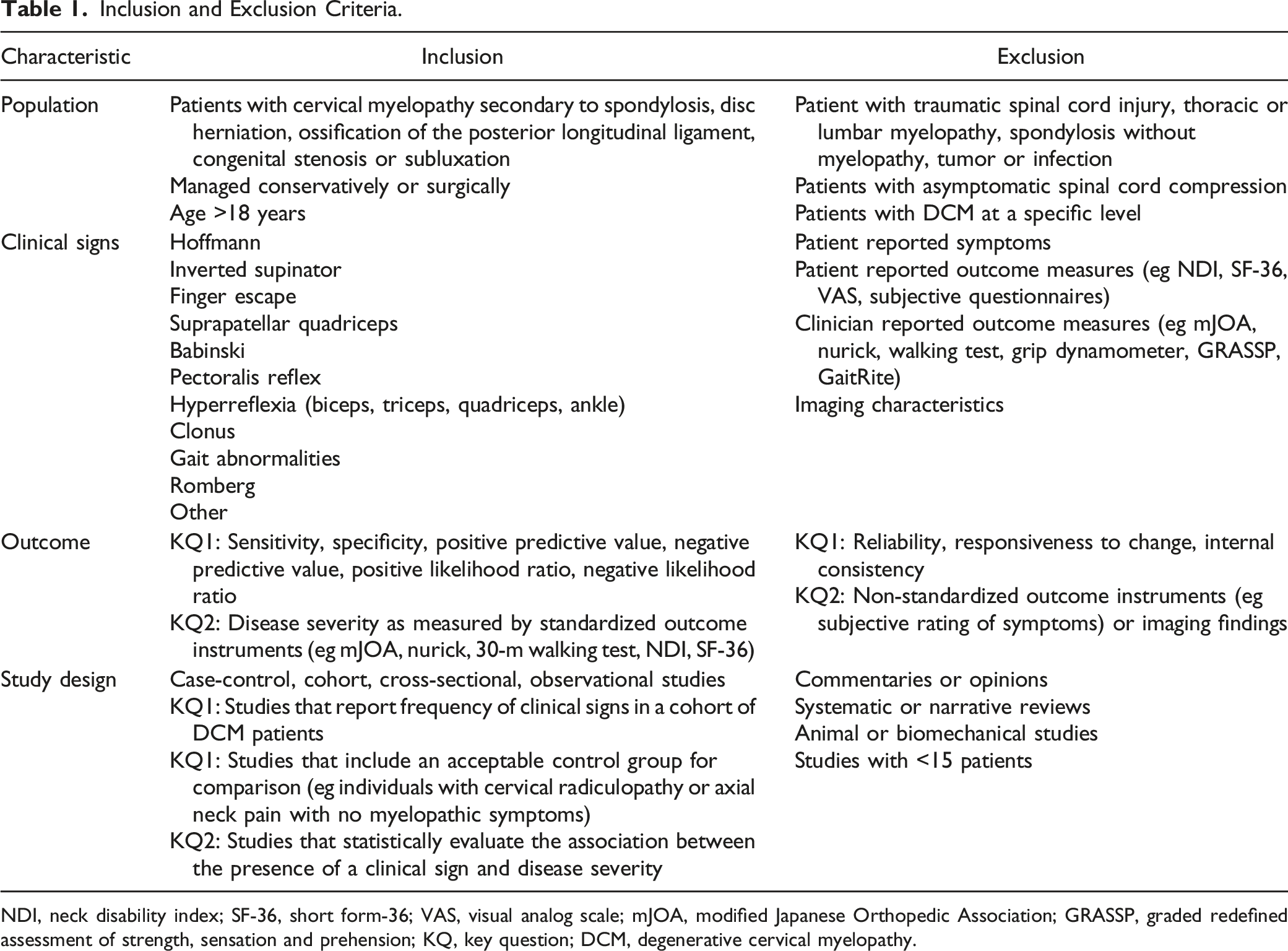
NDI, neck disability index; SF-36, short form-36; VAS, visual analog scale; mJOA, modified Japanese Orthopedic Association; GRASSP, graded redefined assessment of strength, sensation and prehension; KQ, key question; DCM, degenerative cervical myelopathy.
Population
This review targeted adult patients (>18 years) with cervical myelopathy secondary to spondylosis, disc herniation, ossification of the posterior longitudinal ligament (OPLL), congenital stenosis or subluxation. Eligible studies consisted of patients treated surgically or managed conservatively. Studies were excluded if they included patients with traumatic spinal cord injury, thoracic or lumbar myelopathy, asymptomatic spinal cord compression, tumor or infection or if they focused only on patients with DCM at a specific level.
Clinical Signs
Description of Clinical Tests.

Outcome
Definitions and Equations for Metrics of Diagnostic Accuracy.

TP, true positive; FN, false negative; FP, false positive; PPV, positive predictive value; NPV, negative predictive value; TN, true negative; LR+, positive likelihood ratio; LR-, negative likelihood ratio.
Study Design
For KQ1, this review targeted studies that (i) summarized the frequency of clinical signs in a cohort of DCM patients or (ii) compared the frequency of clinical signs between DCM patients and a control group. An example of an appropriate control group is a group of individuals with cervical radiculopathy or axial neck pain with no myelopathic symptoms. For KQ2, this review aimed to identify studies that statistically evaluated the association between a clinical sign and a measure of disease severity. Studies were excluded if they were commentaries, opinions, animal or biomechanical studies, review articles or surveys or if they consisted of less than 15 patients.
Search, Study Selection and Data Collection Process
Studies were identified using 2 electronic databases: MEDLINE and Cumulative Index to Nursing and Allied Health Literature. The last search was performed on June 29, 2021. A detailed search strategy was developed. The strategy was first developed in MEDLINE and then appropriately modified for the other database. Only studies involving humans and written in English were considered for inclusion.
All titles and abstracts were evaluated in a standardized manner by 2 independent reviewers. The abstracts were sorted based on predefined inclusion criteria. Full text investigation was completed for potentially relevant studies. Disagreement between reviewers was resolved through discussion. The search strategy is illustrated in Appendix 1.
The following data were collected from each included article: inclusion and exclusion criteria; demographics of both patients and controls; clinical signs and relevant definitions; results on diagnostic accuracy; and results on the association between clinical signs and DCM severity. For each clinical sign, true and false positives and negatives were extracted, allowing for the calculation of sensitivity, specificity, positive and negative predictive values and likelihood ratios. In some cases, our calculations for these metrics of diagnostic accuracy differed from those reported in the primary study. Data were not extracted on potential clinical signs that could not be distinguished from symptoms based on full text analysis (eg “sensory abnormalities,” “gait difficulties,” “upper or lower extremity weakness.”)
Risk of Bias in Individual Studies
The upgraded QUADAS-2 tool is a 11-question instrument used to evaluate the quality of diagnostic accuracy studies. 19 This tool was used in this review in order to assess the risk of bias of individual studies. To apply the QUADAS-2 tool, the evaluator must answer “yes,” “no” or unclear” to 11 questions within 4 domains. In this review, the studies were rated as having “low” risk of bias if less than 4 questions were answered “no” or” unclear,” “moderate” risk if 4 to 8 questions were answered “no” or “unclear” and “high” risk if more than 8 questions were answered “no” or “unclear.”
Data Analysis
Forest plots were created using RevMan. From each article, we extracted the number of patients who had the disease and tested positive (true positive), did not have the disease and tested positive (false positive), had the disease and tested negative (false negative), and did not have the disease and tested negative (true negative). From these values, sensitivity and specificity were computed and plotted. In some studies, we estimated each value using prevalence data in combination with reported sensitivity and specificity. In other studies, only true positives were reported. The 95% confidence intervals for sensitivity and specificity were automatically generated by RevMan using standard error.
Results
Study Selection
The search yielded a total of 1709 citations. An additional 151 studies were identified from a scoping review and by searching reference lists. Eighteen duplicate studies were removed. After abstract and title review, 1589 records were excluded. Two hundred and fifty-three studies were retrieved for full text investigation. Of these, eleven satisfied inclusion and exclusion criteria and had an appropriate control group. An additional 61 studies reported on frequency of clinical signs in a cohort of DCM patients and were also included. Commonly, studies were excluded if they (i) discussed cervical spine pathology in asymptomatic individuals; (ii) were systematic or narrative reviews; (iii) evaluated the association between clinical signs and specific levels of spinal cord compression; and (iv) assessed the diagnostic accuracy of patient- or clinician-reported outcome measures (Figure 1). An overview of the search process.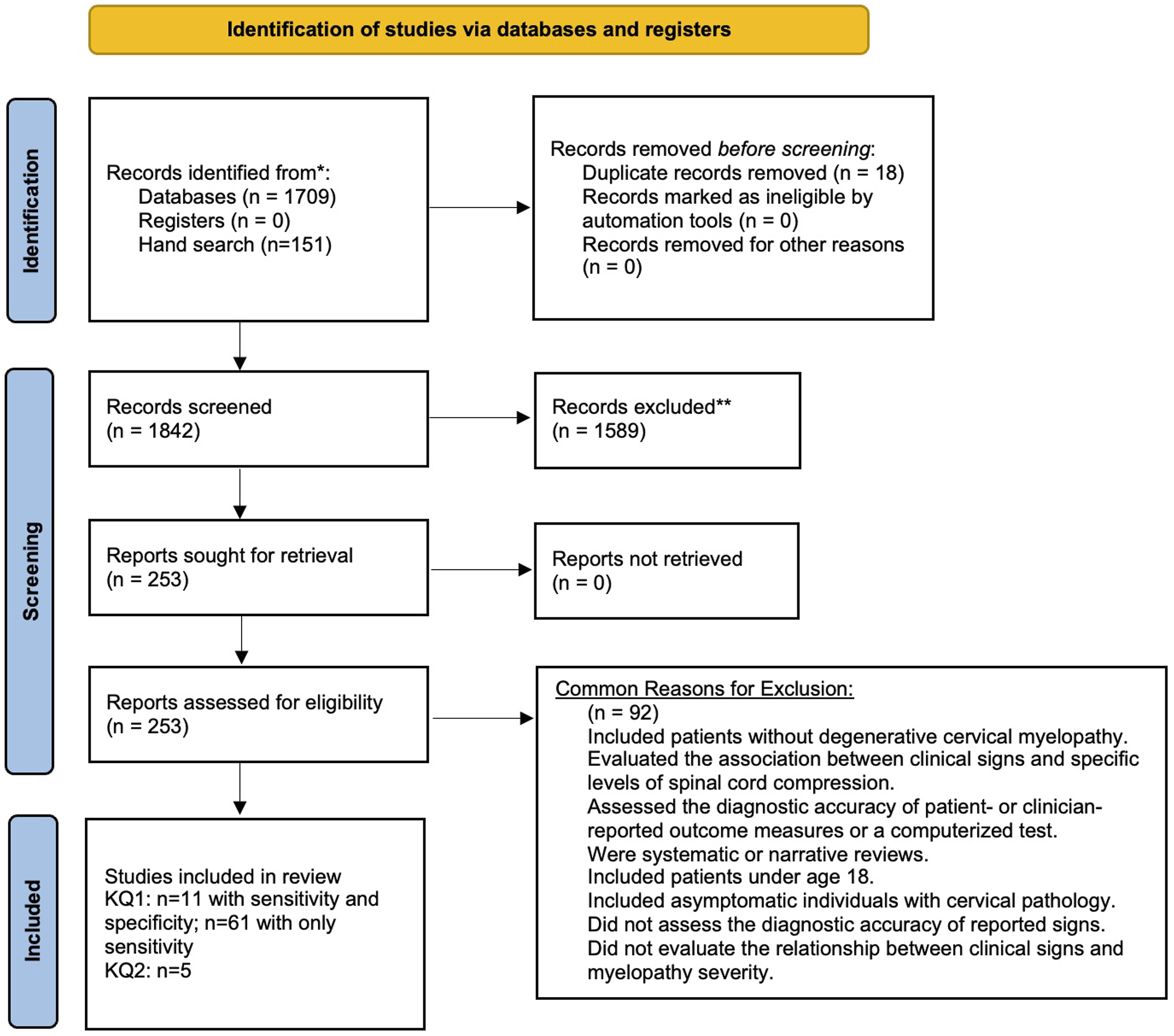
Study Characteristics
Summary of Studies That Included a Control Group and Assessed the Diagnostic Accuracy of Various Clinical Signs.
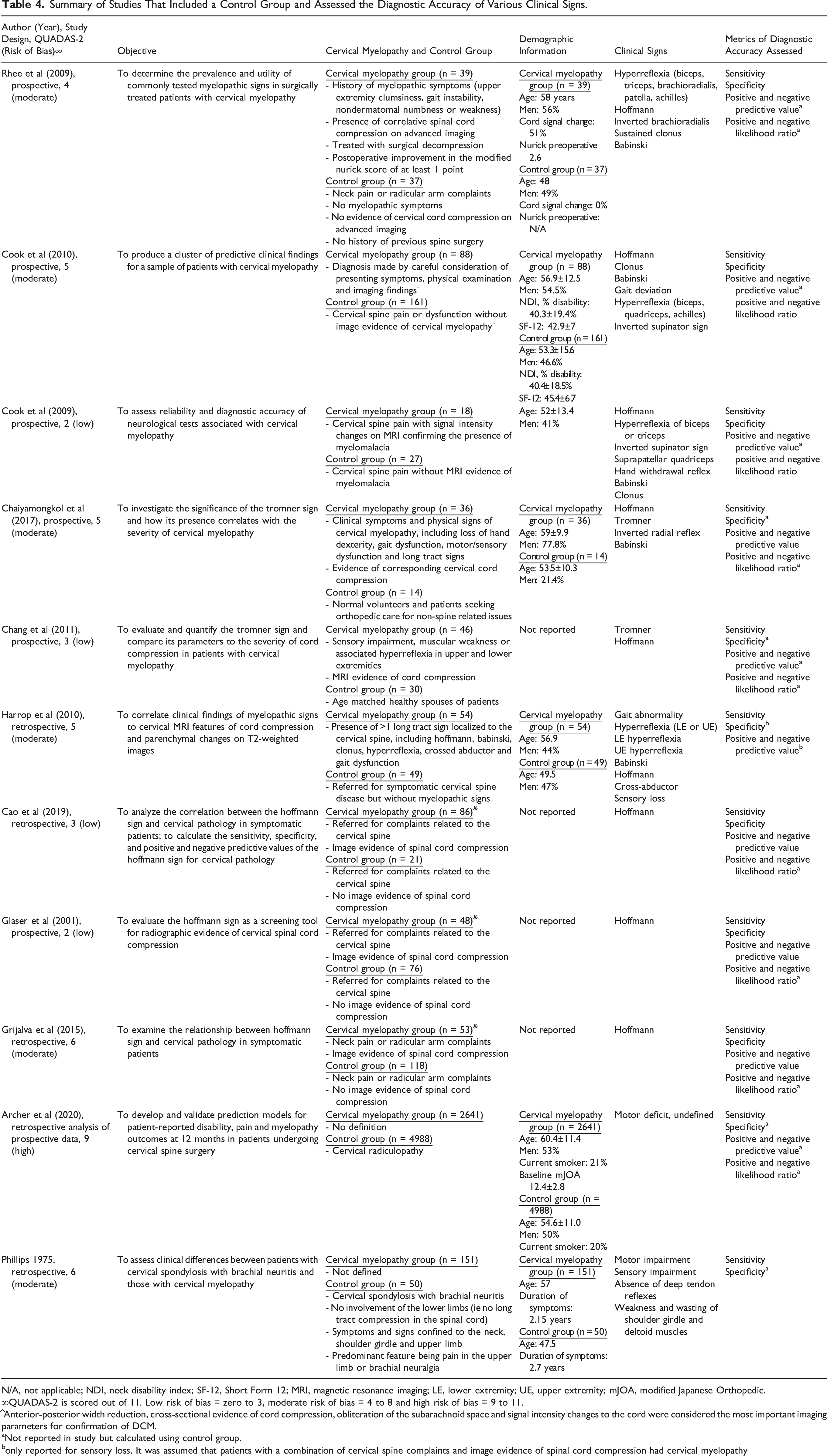
N/A, not applicable; NDI, neck disability index; SF-12, Short Form 12; MRI, magnetic resonance imaging; LE, lower extremity; UE, upper extremity; mJOA, modified Japanese Orthopedic.
∞QUADAS-2 is scored out of 11. Low risk of bias = zero to 3, moderate risk of bias = 4 to 8 and high risk of bias = 9 to 11.
^Anterior-posterior width reduction, cross-sectional evidence of cord compression, obliteration of the subarachnoid space and signal intensity changes to the cord were considered the most important imaging parameters for confirmation of DCM.
aNot reported in study but calculated using control group.
bonly reported for sensory loss. It was assumed that patients with a combination of cervical spine complaints and image evidence of spinal cord compression had cervical myelopathy
Sensitivity and specificity of pathological reflexes in degenerative cervical myelopathy: Results of eleven studies that included a control group.
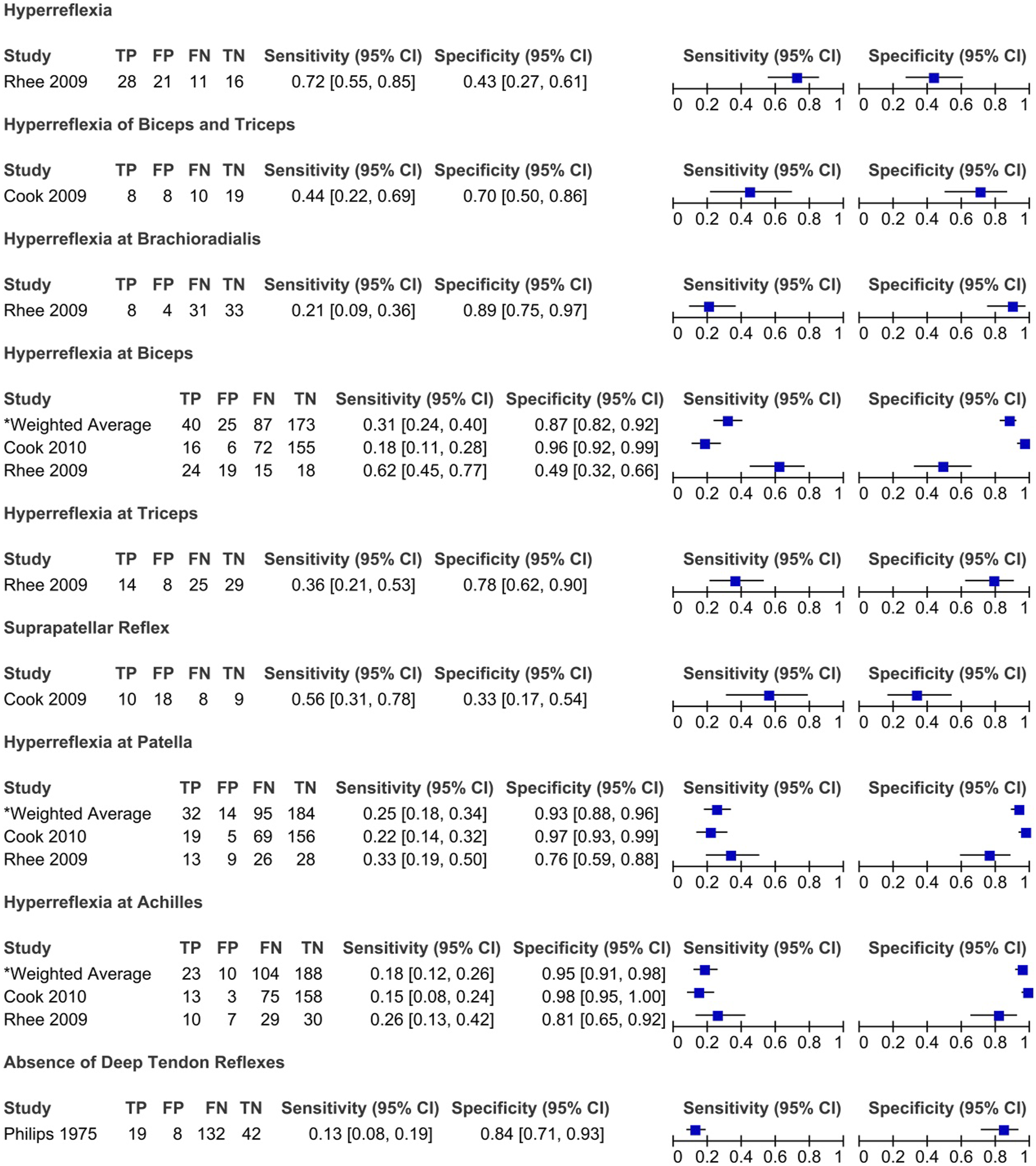
Sensitivity and specificity of hyperreflexia and hyporeflexia in degenerative cervical myelopathy: Results of eleven studies that included a control group.
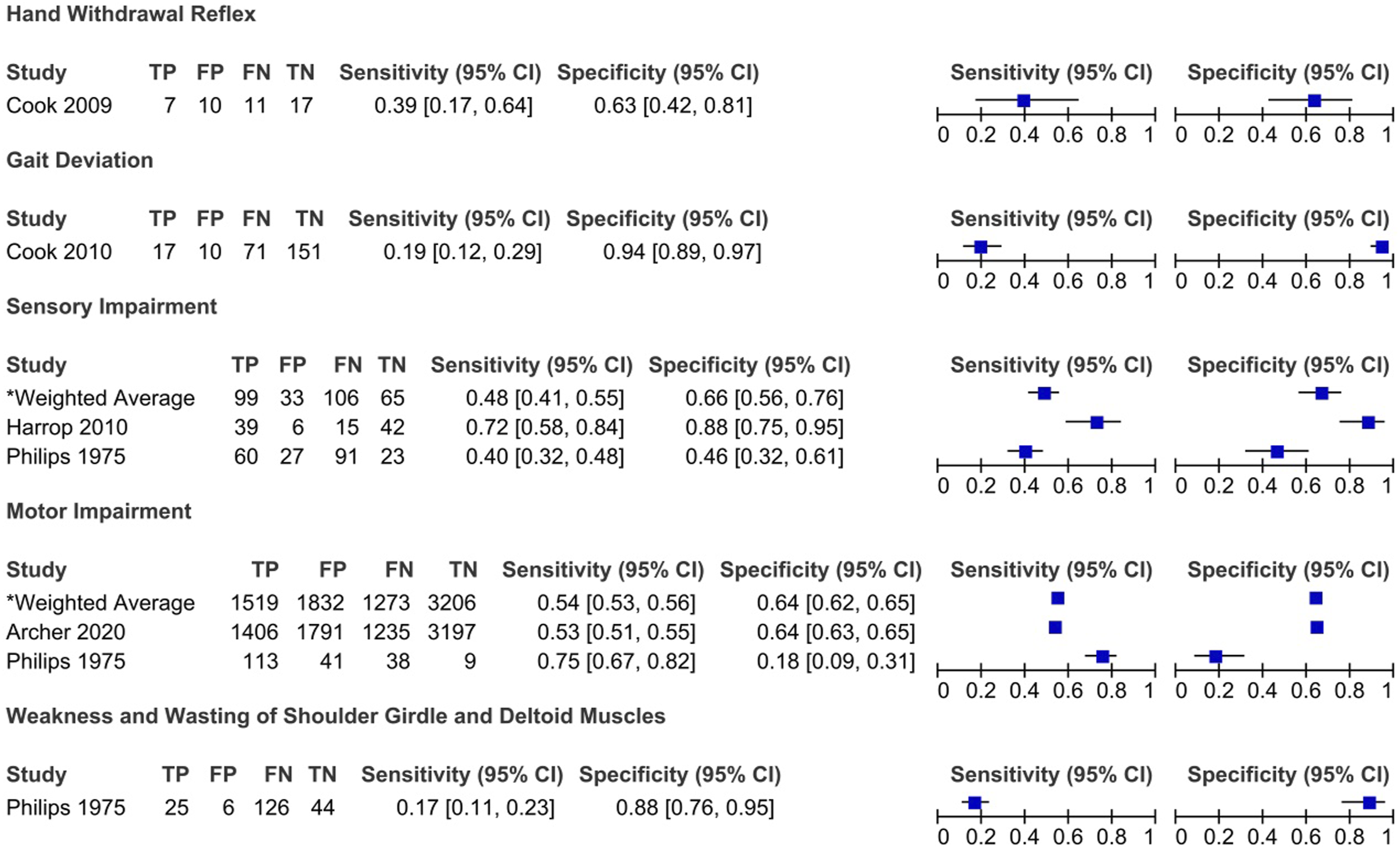
Sensitivity and specificity of sensory and motor impairment and gait dysfunction in degenerative cervical myelopathy: Results of eleven studies that included a control group.
For KQ2, the search identified 5 studies that evaluated the association between various clinical signs and disease severity as measured by the JOA (n = 3),32,35,88 Nurick (n = 2),20,88 mJOA (n = 1), 36 European Myelopathy Score (EMS) (n = 1), 88 Prolo Score (n = 1) 88 and Cooper Myelopathy Scale (CMS) (n = 1). 88 The clinical signs that were discussed in these studies were Hoffmann (n = 4), Babinski (n = 4), hyperreflexia (n = 3), clonus (n = 2), inverted supinator sign (n = 1), dysdiadochokinesia (n = 1), pathological reflexes (n = 1), hypertonia (n = 1), paresis of the upper and lower extremity (n = 1) and great toe proprioception (n = 1).
Risk of Bias of Individual Studies
The QUADAS-2 scoring system was applied to the eleven studies that discussed diagnostic accuracy of various signs and included a control group. The risk of bias was deemed to be low in 4 studies,18,23,25,26 moderate in 620-22,24,27,29 and high in 1. 28
Results of Individual Studies
What is the diagnostic accuracy (i.e. sensitivity, specificity, positive or negative predictive value, positive or negative likelihood ratio) of clinical signs in patients with DCM?
Studies that included a control group
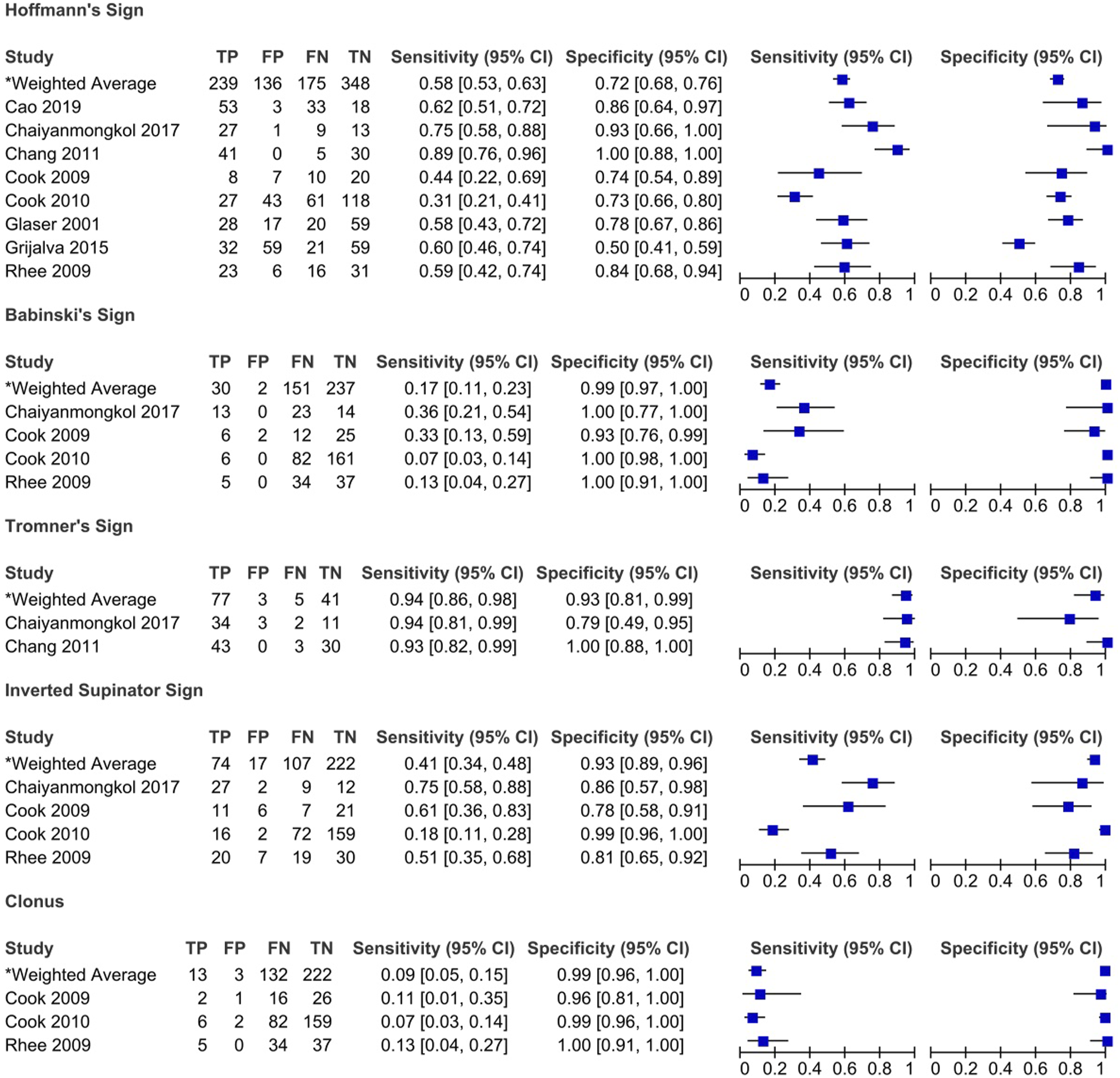
Rhee et al (2009) compared the prevalence of various myelopathic signs between a cervical myelopathy group and a control group (patients with neck pain or radicular arm complaints with no image evidence of spinal cord compression or history of previous cervical surgeries). 20 Based on their results, patients with cervical myelopathy were more likely to exhibit any myelopathic (79% vs 57% in control group) or provocative sign (Hoffmann 59% vs 16%; inverted supinator sign 51% vs 19%; Babinski 13% vs 0%; and clonus 13% vs 0%) than individuals in the control group. In contrast, there were no significant differences in the frequency of hyperreflexia between the myelopathy and control groups. 20 Interestingly, 21% of patients diagnosed with cervical myelopathy, and ultimately treated surgically, demonstrated no myelopathic signs on examination. Biceps hyperreflexia had the highest sensitivity (62%), followed by Hoffmann sign (59%) and inverted brachioradialis (51%). In contrast, the sensitivities of Babinski (13%), clonus (13%) and hyperreflexia of the brachioradialis (21%) were low. The majority of myelopathic signs were specific, especially Babinski (100%) and clonus (100%).
Cook et al published 2 studies that evaluated the diagnostic accuracy of various clinical signs.18,21 In the 2009 study, patients were recruited if their primary complaint was cervical pain and were included in the cervical myelopathy group if they had signal intensity changes on MRI. 18 Based on their results, the inverted supinator sign had the highest sensitivity (61%), followed by the suprapatellar tendon reflex (56%), whereas Babinski (93%) and clonus (96%) demonstrated the highest specificity. 18 Based on likelihood ratios, only Babinski and inverted supinator sign exhibited significant diagnostic accuracy. Specifically, the positive likelihood ratio for Babinski was 4.5 with a post-test probability of 73%. This indicates that the probability of the disease increases by 73% if this test is positive. The negative likelihood ratio for the inverted supinator sign was .5 with a post-test probability of 25%. Cook et al (2009) also evaluated whether combining clinical signs affected diagnostic accuracy. 18 Unfortunately, the post-test probabilities of identifying DCM were not improved by combining clinical signs. In a second study, Cook et al (2010) aimed to develop a prediction model for the diagnosis of DCM. 21 Clinical signs that had a positive likelihood ratio greater than 2 or a negative likelihood ratio less than .5 were initially entered into a regression model and were retained if the P-value was less than .1. 21 The 4 clinical signs that were deemed the most important for the diagnosis of DCM were gait deviation, Hoffmann, inverted supinator sign and Babinski. Age greater than 45 was also included in the model. It was found with a sensitivity of 94% that a patient was unlikely to have DCM if they exhibited none or only a single 1 of these 5 variables. In contrast, the presence of any 3 of the 5 variables being positive yielded a specificity of 99% and a positive likelihood ratio of 30.9, with a post-test probability of 94%; implying a high probability of the patient having DCM. 21
Harrop et al (2010) evaluated the frequency of myelopathic signs in a cohort of patients with evidence of cervical spondylosis and symptoms of cervical stenosis. 24 Patients who exhibited greater than 1 long-tract sign localized to the cervical spinal cord comprised the myelopathy group, while those who did not were considered controls. 24 The sensitivity of signs ranged from 44% (Babinski) to 91% (gait abnormality), with hyperreflexia (85%), Hoffmann (83%) and lower extremity hyperreflexia (81%) demonstrating values above 80%. The presence of sensory loss was 72% sensitive and 88% specific for identifying cervical myelopathy and exhibited positive and negative predictive values of 87% and 74%, respectively.
Diagnostic Accuracy of Clinical Signs Used to Diagnose Degenerative Cervical Myelopathy: Results of Twelve Studies That Included a Control Group.
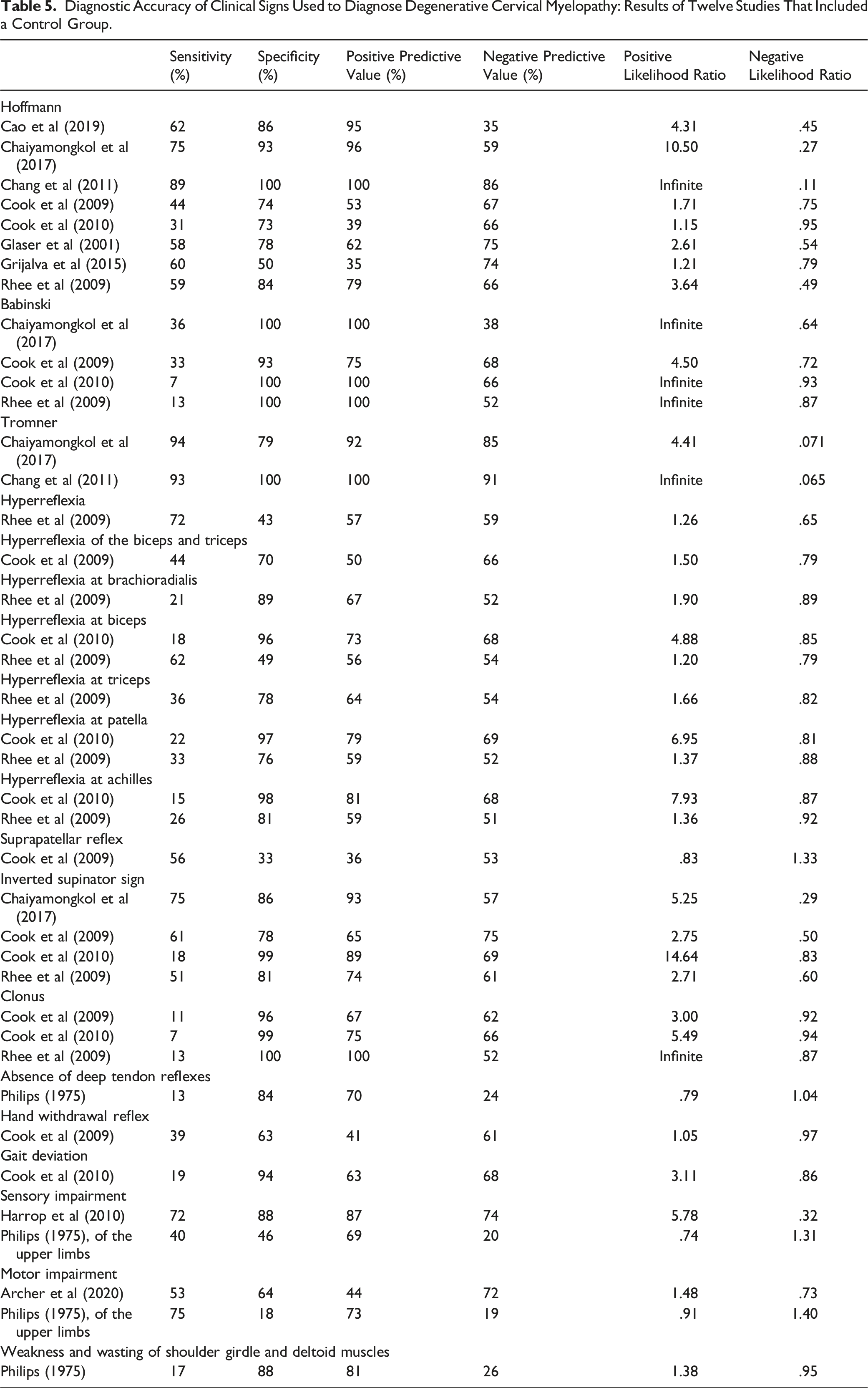
Three studies evaluated the relationship between the presence of the Hoffmann sign and cervical pathology in symptomatic patients.25-27 In these studies, patients were considered to have cervical myelopathy if they were referred for complaints related to the cervical spine and had image evidence of cord compression. The control group consisted of individuals who were referred for similar symptoms but had no evidence of cord compression. Based on their results, the Hoffmann sign was 58% to 62% sensitive and 50% to 86% specific for identifying individuals with DCM.
Finally, 2 studies compared the rates of motor and sensory impairment between patient with cervical myelopathy and those with cervical radiculopathy or cervical spondylosis with brachial neuritis.28,29 The sensitivity of motor impairment ranged from 53% to 75% and specificity from 18% to 64%. Further results are provided in Table 5 for the diagnostic accuracy of sensory impairment, absence of deep tendon reflexes and weakness and wasting of shoulder girdle and deltoid muscles.
Studies that reported on frequency
Sensitivity of Clinical Signs Used to Diagnose Degenerative Cervical Myelopathy.
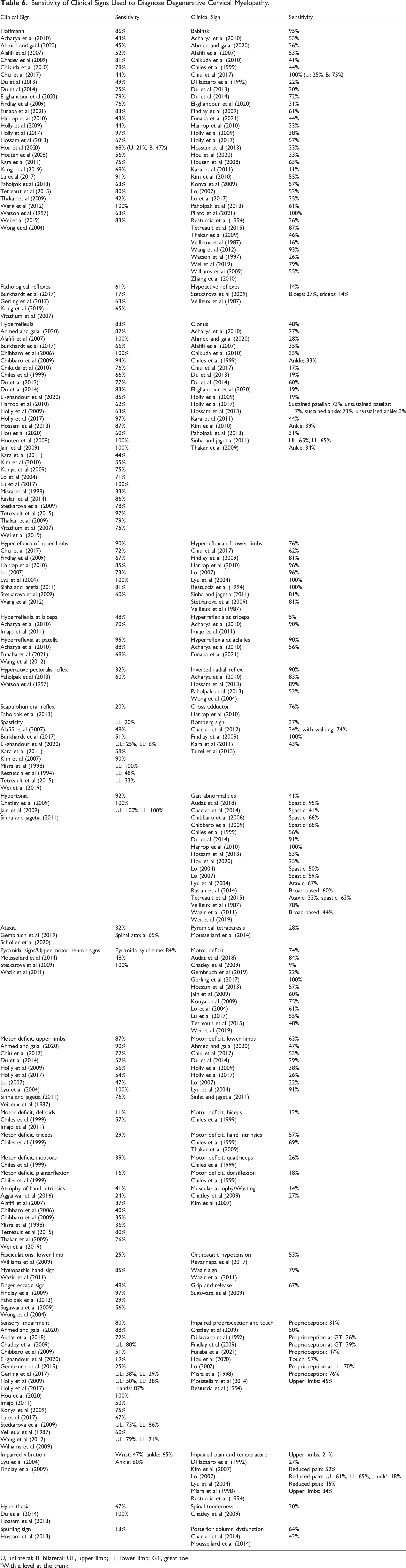
U, unilateral; B, bilateral; UL, upper limb; LL, lower limb; GT, great toe.
aWith a level at the trunk.
What is the association between clinical signs and disease severity in patients with DCM? Five studies evaluated the association between the presence of various clinical signs and disease severity as measured by the Nurick, JOA or mJOA, EMS, CMS and Prolo scores (Table 7).20,32,35,36,88 Patients with a Hoffmann sign had more severe total JOA scores than those without this sign.
35
In single studies, a Hoffmann sign was also associated with worse upper and lower limb motor scores on the JOA.32,35 However, Funaba et al (2021) failed to detect a relationship between lower extremity JOA motor scores and the Hoffmann sign,
35
while Chikuda et al (2010) noted no correlation between a Hoffmann sign and upper extremity JOA motor scores.
32
Of note, patients with a unilateral Hoffmann sign were less severe than those with bilateral Hoffmann sign.
36
Compared to patients with a Babinski, those with a Hoffmann sign had higher (less severe) mJOA scores.
36
Finally, there were no associations between the Nurick score and the presence of a Hoffmann sign.
20
Two studies identified that patients with a Babinski had more severe lower extremity JOA motor scores than those without this clinical finding.32,35 In contrast, there was no association between a Babinski and total JOA, upper extremity JOA motor or Nurick scores.20,32,35 Single studies demonstrated that clonus was associated with more severe lower extremity JOA motor scores
32
but not with upper extremity JOA motor or Nurick scores.20,32 Furthermore, Vitzthum and Dalitz (2007) identified an association between (i) the presence of dysdiadochokinesia and worse JOA, CMS upper extremity and EMS scores; (ii) upper extremity weakness and worse JOA, CMS upper extremity and EMS scores; and (iii) lower extremity weakness and worse Nurick scores. Finally, this review determined no association between (i) greater toe proprioception and total or subscores of the JOA;
35
(ii) hyperreflexia and JOA, Nurick, CMS, EMS or Prolo scores;
32
and (iii) Nurick scores and hyperreflexia of the triceps, brachioradialis, patella and achilles or the inverted brachioradialis reflex.
20
The Association Between Various Clinical Signs and Disease Severity.
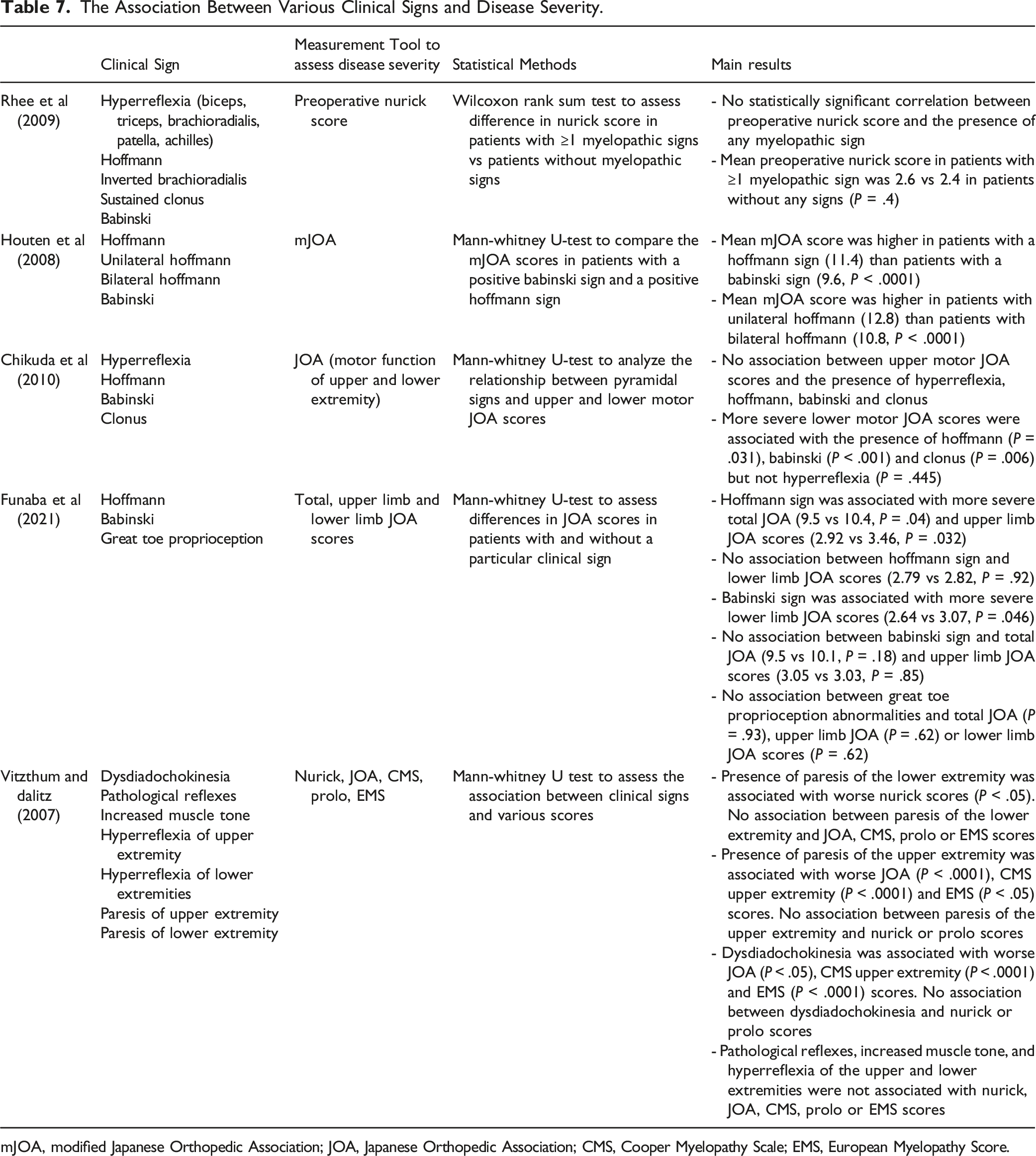
mJOA, modified Japanese Orthopedic Association; JOA, Japanese Orthopedic Association; CMS, Cooper Myelopathy Scale; EMS, European Myelopathy Score.
Discussion
This systematic review aims to summarize the diagnostic accuracy of various clinical tests used to evaluate patients with DCM. Based on the results of the eleven studies, the most sensitive clinical tests were the Tromner sign and hyperreflexia, while the most specific tests were Babinski, the Tromner sign, clonus and the inverted supinator sign. Signs that may be eligible for inclusion in diagnostic criteria should demonstrate both moderate to high sensitivity and specificity. Of note, the majority of clinical signs identified in this review were either sensitive or specific, but not both. Furthermore, based on 1 series, examination findings may be absent entirely and a diagnosis of DCM can be made using a combination of patient-reported symptoms and imaging findings. As a result, it is important for clinicians to have a high index of suspicion for the diagnosis of DCM and to use a variety of clinical examination maneuvers to assess each patient. Furthermore, physicians should feel empowered to order imaging of the cervical spine (or even advanced imaging) if a patient demonstrates any of these signs of DCM or symptoms referable to the neck (please see separate study on symptoms of DCM). Given the impact of early surgery on neurological recovery and other patient outcomes, we must get to the point where subtle signs and symptoms of myelopathy trigger early referral for neuroimaging and assessment by experts. Timely diagnosis of DCM is a top priority to ensure these individuals either get the surgical treatment they need, or are at the very least, monitored for disease progression by physicians with significant expertise in DCM. The results from this systematic review and a scoping review on important DCM symptoms will be used in combination with expert opinion to generate diagnostic criteria that can guide a variety of health care professionals towards a diagnosis of DCM.
Upper Motor Neuron Signs
The diagnostic value of the Hoffmann sign has been extensively studied in individuals with cervical spine complaints. Based on the results of this review, the clinical utility of this sign is controversial given the wide range of sensitivities (25% to 100%), specificities (50% to 100%) and positive (1.15 to infinite) and negative (.11 to .95) likelihood ratios. Similar to other pathological reflexes, the Hoffmann sign results from decreased inhibitory input from the descending fibers of the corticospinal tract; this decreased inhibition causes exaggerated activation of motor neurons from connections with both sensory neurons and interneurons. 36 It is postulated that patients with peripheral neuropathy or radiculopathy may have a falsely negative Hoffmann sign due to interference with the normal reflex arc. 26 However, in a study by Houten and Noce (2008), the Hoffmann sign was evident in patients with known diabetic neuropathy despite normal to diminished deep tendon reflexes. 36 The technique of eliciting this reflex is challenging to master and interrater variability may contribute to the range of results across studies. Cook et al (2009), however, demonstrated substantial agreement (89%) between a neurosurgeon and a physical therapist in identifying a Hoffmann sign in a population of patients with cervical pain seeking a surgical consult. 18 Inter-rater reliability is likely to differ across specialists and may be lower when assessed by physicians who encounter these patients less frequently. Finally, a positive Hoffmann sign may be present in .7% to 3% of the population as well as in patients with upper motor neuron dysfunction from an intracranial process. 91 Although an asymmetric Hoffmann reflex may be more pathological, Houten and Noce (2008) demonstrated that a bilateral Hoffmann sign is associated with more definitive cord compression and more severe DCM. 36 This clinical sign should be used in combination with other signs and symptoms the support a diagnosis of myelopathy. Compared to the Hoffmann sign, the Tromner sign demonstrated superior sensitivity (94%) and specificity (93%) supporting its utility in both ruling in and out DCM.22,23
An upgoing plantar response, known as the Babinski sign, is a primitive reflex that disappears at 1 to 2 years of age but can reappear in the setting of upper motor neuron dysfunction. In the studies included in this review, the Babinski sign was highly specific for diagnosing cervical myelopathy. The control groups, however, were either healthy individuals or patients with cervical spine complaints without image evidence of spinal cord compression or myelomalacia. As such, the values for specificity may be falsely elevated as a Babinski sign may also be present in patients with noncervical or nonspondylotic disorders that result in upper motor neuron dysfunction. The reliability of the Babinski sign also ranges from poor to moderate in cohorts of individuals with and without a variety of neurological diseases.18,92,93 This variability in assessment stems from differences in technique as well as in interpretation. Of note, the Babinski sign requires both extension of the great toe and fanning of the other toes; withdrawal of the foot or isolated toe extension are not consistent with a positive test. Finally, patients with a Babinski sign had worse mJOA scores than those with a Hoffmann sign, indicating that release of this primitive reflex is associated with more severe corticospinal tract dysfunction. 36 The presence of clonus is also highly specific (99%) but demonstrates poor sensitivity.
Hyperreflexia at any deep tendon has moderate sensitivity for detecting cervical myelopathy (79%, with 47% of studies reporting a sensitivity over 80%). The sensitivity of this sign improves when assessing reflexes exclusively in the lower extremities (85%) compared with the upper extremities (78%).24,34,50 Evaluation of individual reflexes demonstrated a wide range of sensitivities: brachioradialis (21%), biceps (18% to 70%), triceps (5% to 90%), patella (22% to 95%) and achilles (15% to 90%). These results indicate that hyperreflexia, particularly in the lower extremities, may be an important screening tool for DCM. This finding is consistent with the underlying pathology of DCM as reflexes become exaggerated below the level of spinal cord compression due to reduced descending inhibition from the corticospinal tract. Furthermore, the reflexes in the upper extremities may be increased or decreased depending on the level of canal stenosis and the involvement of the nerve root as it exits the neural foramen. Presence of exaggerated reflexes in the upper extremities can further help to localize the level of spinal cord compression. For example, a hyperactive pectoralis reflex may indicate a myelopathic level above C4 whereas normal upper extremity reflexes are typically seen with a level at C6 to C7 or below.39,94 Notably, peripheral neuropathy may affect deep tendon reflexes; specifically, there was a significant difference in the frequency of achilles hyperreflexia in patients with diabetes (0%) compared to those without (35%, P = .04). 20 Caution should be taken in ruling out DCM in patients with peripheral neuropathy when reflexes are normal or diminished. Finally, the inverted supinator reflex has shown promising results with respect to specificity (93%) and therefore may be used to confirm a diagnosis of DCM in patients with cervical complaints.18,20-22,30
Sensorimotor and Gait Dysfunction
Patients with DCM will often have motor and sensory dysfunction of their upper and lower extremities. The sensitivity, however, is variable for motor deficits (9 to 100%) and low for impaired pain, proprioception, temperature and vibration sensation. Weakness in patients with DCM often begins in the intrinsic hand muscles and is likely due to anterior horn cell damage as opposed to nerve root compression. 33 As a result, hand weakness is more sensitive (60%) than motor deficits in the iliopsoas (39%), triceps (29%), quadriceps (26%), dorsiflexors (18%), plantarflexors (16%), biceps (12%) and deltoids (18%).33,70,89
Gait impairment may be 1 of the earliest manifestations of DCM. However, subtle changes in a patient’s stance and stability when walking may be difficult to appreciate early in the disease course. A myelopathic gait is often described as unsteady, broad-based or spastic and is a result of impaired proprioception in addition to upper motor neuron dysfunction. Furthermore, patients with DCM exhibit reduced gait velocity, a shortened stride length, increased double support time, a wide step width and slower cadence.95-99 DCM should be considered in patients with recurrent falls and gait deterioration. However, this clinical sign did not demonstrate high sensitivity given the wide variety of conditions that can impair gait, including osteoarthritis, peripheral neuropathy, obesity and vascular disease. Romberg sign can also be used to detect proprioception dysfunction but demonstrates poor sensitivity in DCM.30,34,37,51,84 When walking, however, the sensitivity of the Romberg sign increases from 34% to 74%. 34 This finding is clinically relevant as patients with DCM may sustain recurrent falls, especially in the dark. Overall, advanced gait assessments are essential to quantify subtle gait disturbances.
Based on the results of this systematic review, a combination of clinical signs is likely required to support a diagnosis of DCM. According to Cook et al (2010), the presence of 1 of the following has a high sensitivity (94%) for identifying individuals with DCM: gait deviation, Hoffmann sign, inverted supinator sign, Babinski sign and age over 45. 21 Given these results, DCM could be safely excluded in patients who did not have any of these 5 criteria. 21 Similarly, Rhee et al (2009) demonstrated that the presence of greater than or equal to 1 provocative sign (Hoffmann, inverted supinator sign, Babinski and clonus) was approximately 70% sensitive for diagnosing DCM. 20 However this study also identified cases of DCM with no positive examination findings. Furthermore, while the presence of an increasing number of clinical signs increases specificity and helps to confirm the diagnosis of DCM, it also appears to correlate to disease severity. Consequently, examination findings alone are not sufficient to make or exclude a diagnosis of DCM, especially early in the disease course.
Limitations
While this is the first review to summarize the sensitivity and specificity of clinical signs in DCM, there are limitations that should be mentioned. First, there were only eleven studies that included a control group in their analysis; as a result, there is limited information on the specificity of various DCM signs. Furthermore, of these eleven studies, 7 were rated as either moderate or high risk of bias on the QUADAS-2, questioning the validity of these results. In some of the studies, individuals were considered controls if they had neck, radicular or cervical spine pain or other symptoms but did not have myelomalacia or spinal cord compression on neuroimaging. It is increasingly recognized that patients can still be diagnosed with DCM in the absence of signal change or even spinal cord compression on static MRI. Moreover, several patients will have a combination of symptoms and signs of myelopathy and radiculopathy given that degenerative changes can simultaneous compress the spinal cord and nerve root. As such, these control groups may be suboptimal for assessing the accuracy of various symptoms. Other studies used patients referred to the orthopedic clinic for other complaints or healthy controls such as the spouses of patients. These reflect better control groups for the question of interest. Second, values for sensitivity and specificity are extracted from studies that are screening a particular population and not just a random group of individuals. Nonetheless, this review provides invaluable information on some of the most common signs of DCM and will undoubtedly improve understanding of this condition.
Conclusion and Future Directions
Patients with DCM can present with subtle, non-specific signs in their upper and lower extremities, making it difficult to initially diagnose this condition. Based on the results, the most sensitive clinical tests for diagnosing DCM were the Tromner sign and hyperreflexia, while the most specific tests were the Babinski, Tromner sign, clonus and inverted supinator sign. Signs that may be eligible for inclusion in diagnostic criteria should demonstrate both moderate to high sensitivity and specificity. While the presence of these clinical signs is helpful, examination findings may be absent entirely and a diagnosis of DCM can still be made using a combination of patient-reported symptoms and imaging findings. Furthermore, the absence of upper motor neuron signs does not rule out a diagnosis of DCM. In patients with clear symptoms of myelopathy, standard or advanced imaging, formal gait assessment and neurophysiology may be required to confirm the diagnosis. The current systematic review provides a framework to create a diagnostic toolkit for specialists, primary care physicians, and allied health professionals. • Patients with DCM can present with subtle, non-specific signs in their upper and lower extremities, making it difficult to initially diagnose this condition. • The most sensitive clinical tests for diagnosing DCM are the Tromner sign and hyperreflexia • The most specific tests are the Babinski, Tromner sign, clonus and inverted supinator sign. • Examination findings may be absent entirely and a diagnosis of DCM can still be made using a combination of patient-reported symptoms and imaging findings. • Absence of upper motor neuron signs does not rule out a diagnosis of DCM.
Supplemental Material
Supplemental Material - The Value of Clinical Signs in the Diagnosis of Degenerative Cervical Myelopathy: Results of a Systematic Review
Supplemental Material for The Value of Clinical Signs in the Diagnosis of Degenerative Cervical Myelopathy: Results of a Systematic Review by Zhilin Jiang, Benjamin Davies, Carl Zipser, Konstantinos Margetis, Allan Martin, Stavros Matsoukas, Freschta Zipser-Mohammadzada, Najmeh Kheram, Andrea Boraschi, Elina Zakin, Oke Righteous Obadaseraye, Michael G. Fehlings, Jamie Wilson, Ratko Yurac, Chad E Cook, Jamie Milligan, Julia Tabrah, Shirley Widdop, Lianne Wood, Elizabeth A. Roberts, Tanzil Rujeedawa, Lindsay Tetreault on behalf of the AO Spine RECODE-DCM Diagnostic Criteria Incubator in Global Spine Journal
Footnotes
Acknowledgments
This research aligns with the AO Spine RECODE-DCM top research priority ‘Diagnostic Criteria’ selected by people living and working with DCM. For further information on how this process was conducted, why this question was prioritized, and global updates on currently aligned research, please visit aospine.org/recode/diagnostic-criteria. This article, including the broader efforts to establish diagnostic criteria for DCM, is led by the RECODE-DCM Diagnostic Criteria Incubator Group. This was initially launched, with support from AO Spine through the AO Spine Knowledge Forum Spinal Cord Injury, a focused group of international Spinal Cord Injury experts. The oversight and support of the incubator has now transitioned to ![]() , a global charity focused on DCM.
, a global charity focused on DCM.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Correction (September,2024):
In Table 2 the positive Babinski’s sign is a great toe extension. This previously stated it was a great toe flexion.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
