Abstract
Study Design
Systematic review.
Objective
To assess the available evidence related to dose-dependent effectiveness (i.e., bone fusion) and morbidity of osteobiologics used in anterior cervical discectomy and fusion (ACDF).
Methods
Studies with more than 9 adult patients with degenerated/herniated cervical discs operated for one-to four-levels ACDF reporting used osteobiologics doses, fusion rates at six months or later, and related comorbidities were included. PubMed, EMBASE, ClinicalTrials, and Cochrane were searched through September 2021. Data extracted in spread sheet and risk of bias assessed using MINORS and Rob-2.
Results
Sixteen studies were selected and sub-grouped into BMP and non-BMP osteobiologics. For the 10 BMP studies, doses varied from 0.26 to 2.1 mg in 649 patients with fusion rates of 95.3 to 100% at 12 months. For other osteobiologics, each of six studies reported one type of osteobiologic in certain dose/concentration/volume in a total of 580 patients with fusion rates of 6.8 to 96.9% at 12 months. Risk of bias was low in three of the 13 non-randomized (18.75%) and in all the three randomized studies (100%).
Conclusions
Taking into account the inconsistent reporting within available literature, for BMP usage in ACDF, doses lower than 0.7 mg per level can achieve equal successful fusion rates as higher doses, and there is no complication-free dose proved yet. It seems that the lower the dose the lower the incidence of serious complications. As for non-BMP osteobiologics the studies are very limited for each osteobiologic and thus conclusions must be drawn individually and with caution.
Introduction
Osteobiologics are a group of synthetic and naturally derived substances aimed to facilitate bone fusion by mimicking the properties of autologous bone, with autologous iliac bone crest grafts being historically the gold standard in spine surgery. 1 These properties might enclose being osteoconductive, osteoinductive, osteogenic, or a combination.
Achieving successful arthrodesis is regarded as one of the most pursued goals in degenerative spine surgery when instability is of major concern. By 2008, more than 400 000 spine fusion surgeries were performed annually in the United States, of which 157 000 were performed on the cervical spine showing a 2.1-fold (114%) increase since the end of the previous century. 2 This coupled with the expansion of available graft materials options as well as the increasing prevalence of spine disorders lead to a forecasted growth in the global osteobiologics market size from an estimated USD 17.94 Billion in 2020 to USD 21.67 Billion by 2026 with an impressive compound annual growth rate (CAGR) of 3.19%. 3
However, with the aim of achieving successful arthrodesis, surgeons might tend to increase the volume/concentration (i.e., dose) of the inserted osteobiologics with the perception that this can escalade its efficacy in forming a faster and more solid fusion mass. Nevertheless, some used osteobiologics have their own specifically related complications, 4 and whether upgrading the doses would improve the fusion success rate or aggravate these complications is still a research point lacking enough cumulative evidence. Not to mention that with the high cost of some of these osteobiologics, raising the doses might inadvertently pile up the costs of the provided health care. 5
Thus, a careful balance should be exercised by selecting the minimum dose capable of achieving a successful fusion while reducing any possible product-related morbidities and any unnecessary inflated financial cost. The objective of this systematic review was to assess available evidence related to dose-dependent effectiveness (i.e., bone fusion) and morbidity (i.e., complications) of osteobiologics used in anterior cervical discectomy and fusion (ACDF) procedures for management of degenerative disc conditions.
Methods
Protocol and Registration
Methods of the analysis, search strategy, and eligibility criteria were specified in advance, documented in a protocol, and submitted to the International Prospective Register of Systematic Reviews (PROSPERO), submission ID: CRD42021254596.
The formulated study question(s) and defined Participants, Intervention, Comparator(s), and Outcome(s) of interest (PICO) along with inclusion and exclusion criteria as well as the reviewed studies design, publication types, and timing are detailed in Supplemental material 1.
Briefly, we sought to identify studies including adult patients older than 18 years with degenerated or herniated cervical spine discs who underwent from one-to four-level ACDF procedures where the different doses/volumes/concentrations of a used osteobiologic were reported as well as the achieved fusion rates at least 6 months postoperatively along with the comorbidities & complications which might be corelated to the used osteobiologic dose. We excluded cases with non-degenerative cervical spine pathologies as well as cases where the surgical intervention included anterior corpectomy or a posterior approach. Also, we excluded case series, case reports, studies with fewer than 10 patients in the treatment group, invitro studies, animal studies, as well as studies for which radiographic fusion rates or clinical outcomes were not reported.
Study Design
This systematic review was conducted according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.6-8 Checklist is provided in Supplemental material 2.
Electronic Literature Search
PubMed/MEDLINE, EMBASE, ClinicalTrials.gov, and the Cochrane Collaboration Library were searched through September 15, 2020, for studies published in 2000 or later in English language. Another search was conducted one year later on September 15, 2021, using the same protocol looking for any new recent literature.
The proposed strategy for MEDLINE search terms used in September 2020 and 2021 is provided in Supplemental material 3. After adaptation, similar structured search strategies were used with syntax and subject headings appropriate for each of the other databases.
Screening Process
After the search was run, the references were exported to EndNote. Duplicates were removed. Title and abstract screening of all records identified by the search were conducted independently by two reviewers. To ensure consistency, we conducted calibration exercises before starting the screening process. Full text of all potentially relevant articles was retrieved and screened for final inclusion independently by the two reviewers. Any disagreement was resolved by discussion with a third reviewer functioning as referee (Figure 1). PRISMA diagram.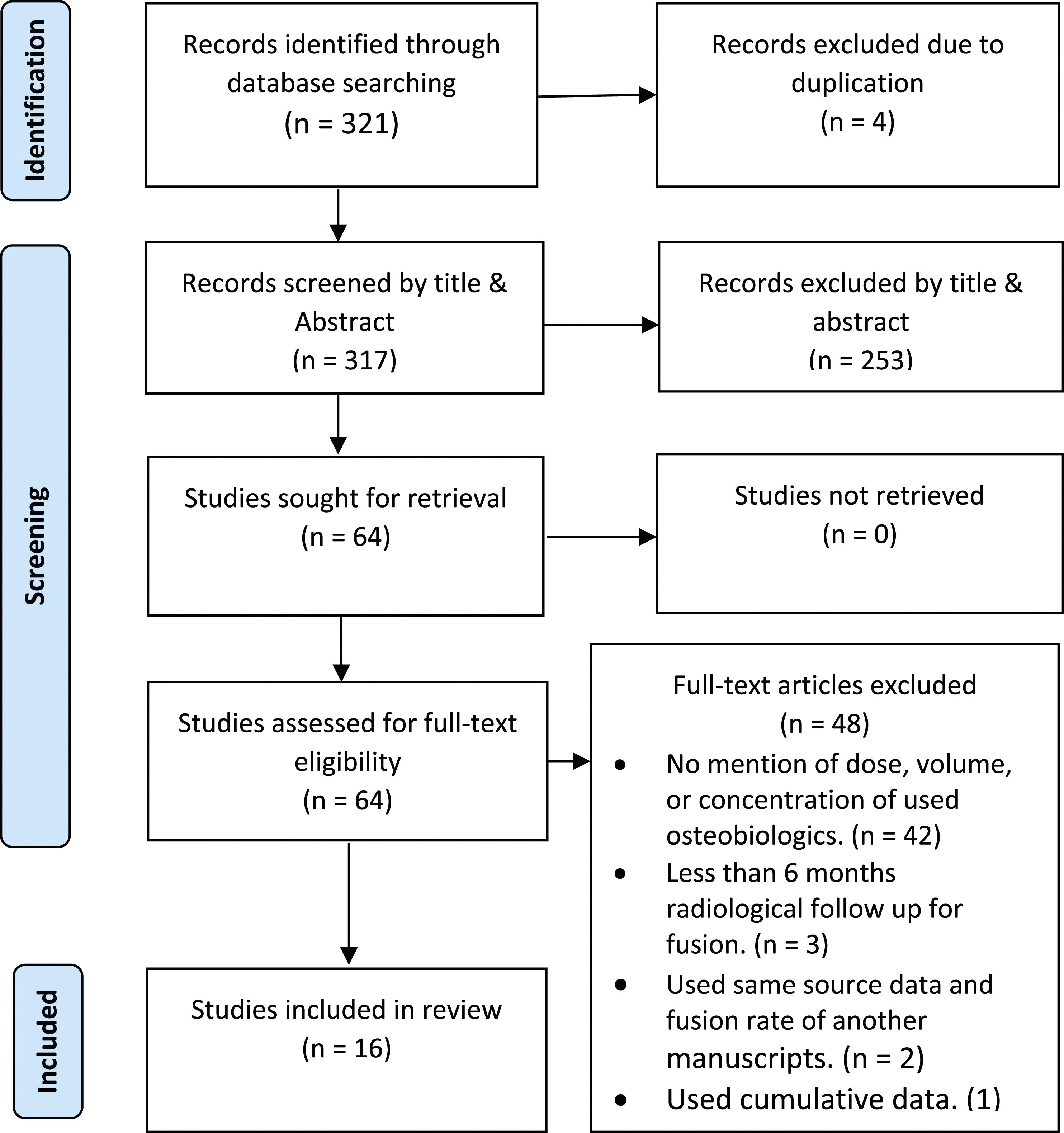
Data Extraction
One reviewer extracted data, and the process was repeated for verification. Any confusion was resolved by discussion with a second reviewer. From the included articles, the following were extracted: year of publication, first author, country where study was conducted, study design, type of osteobiologics used, commercial name, dose or concentration of used osteobiologics per disc space level, type of surgical intervention (levels operated/using of anterior plate), type of the interbody spacer used if any, sample size, gender (males %), age in years (mean ± standard deviation SD), inclusion criteria, exclusion criteria, definition of fusion used by the authors, modality of fusion assessment (Computed Tomography CT/X-ray/others), fusion at 6/9/12/24 months postoperatively, reported complications related to osteobiologics, funding if any, and any declared conflict of interest.
Osteobiologics type, dose and related fusion rates reported in the included studies.
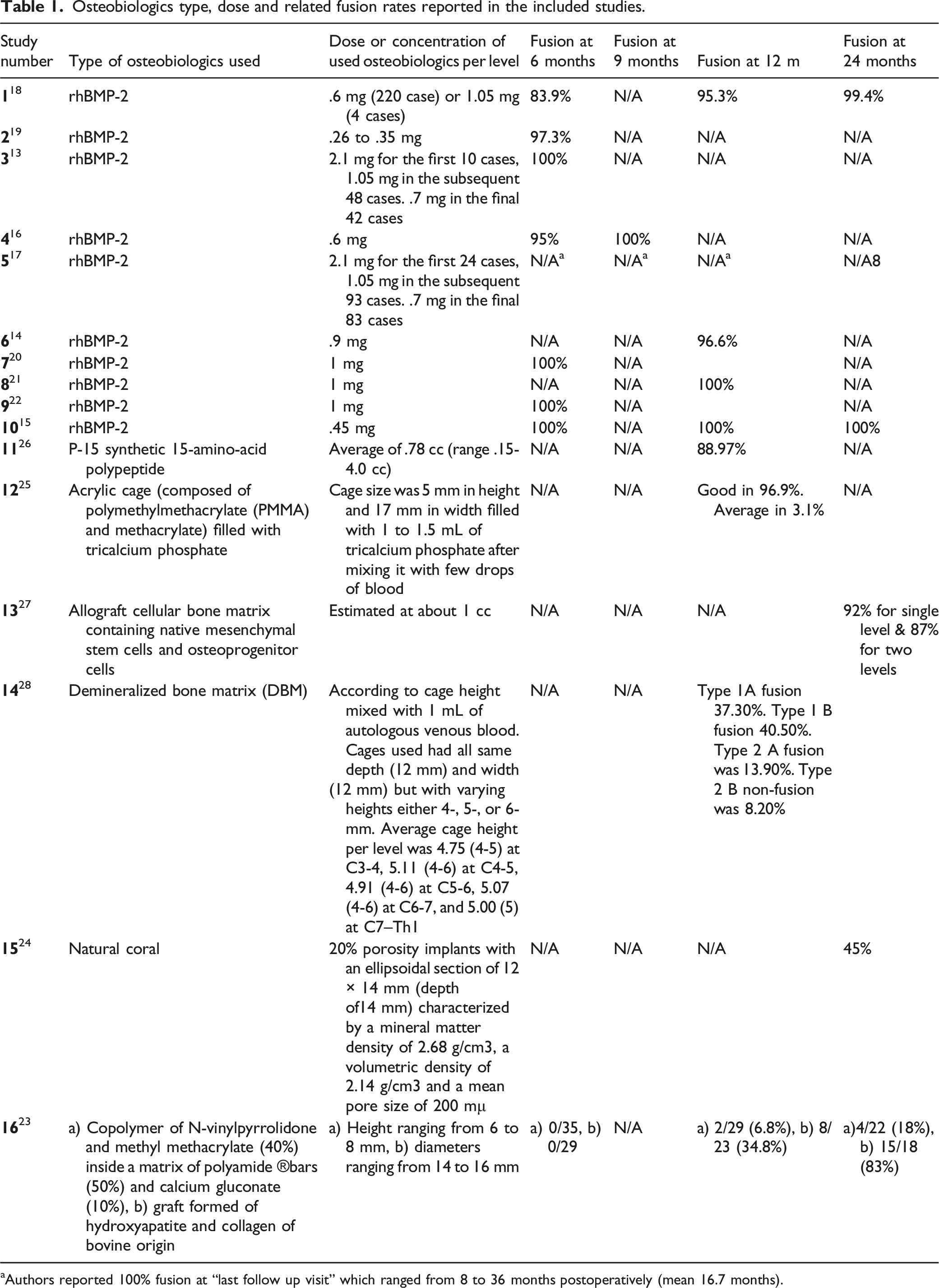
aAuthors reported 100% fusion at “last follow up visit” which ranged from 8 to 36 months postoperatively (mean 16.7 months).
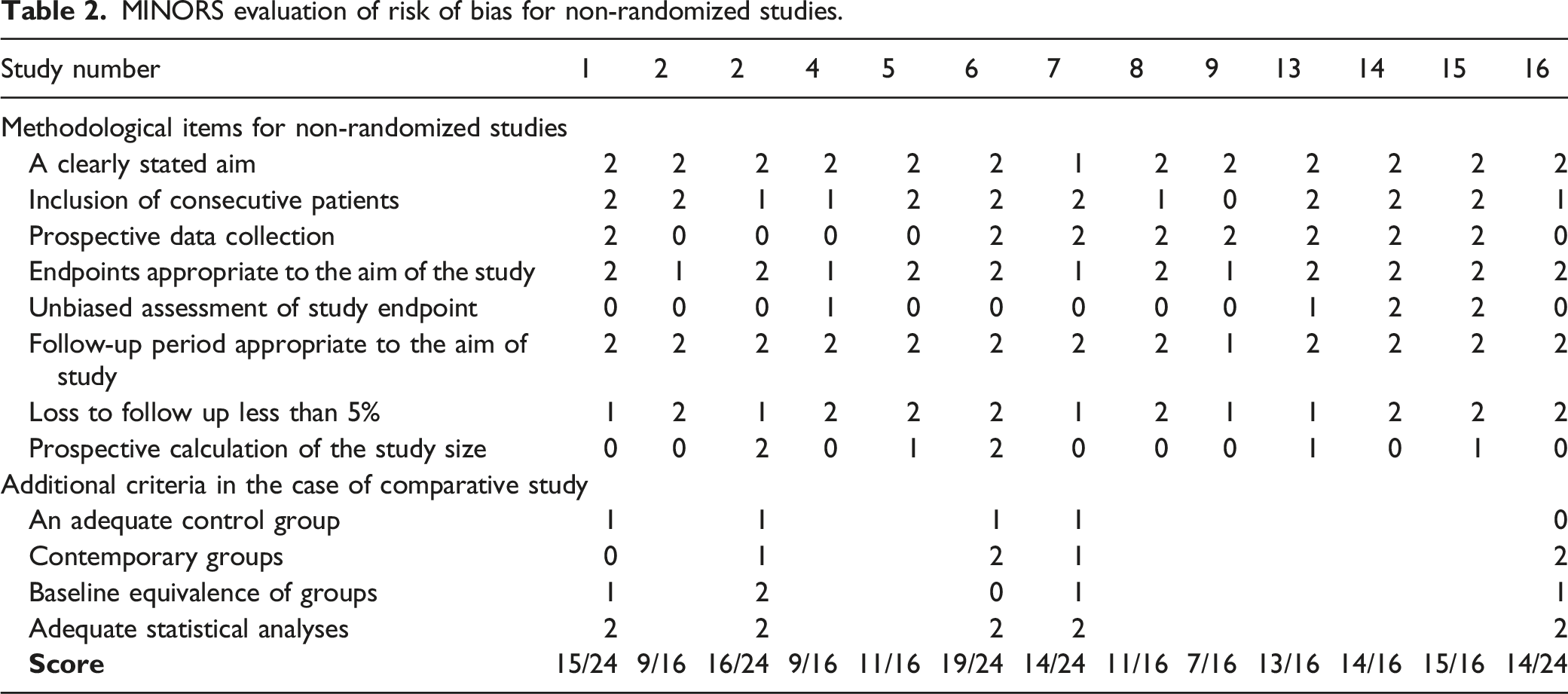
MINORS evaluation of risk of bias for non-randomized studies.
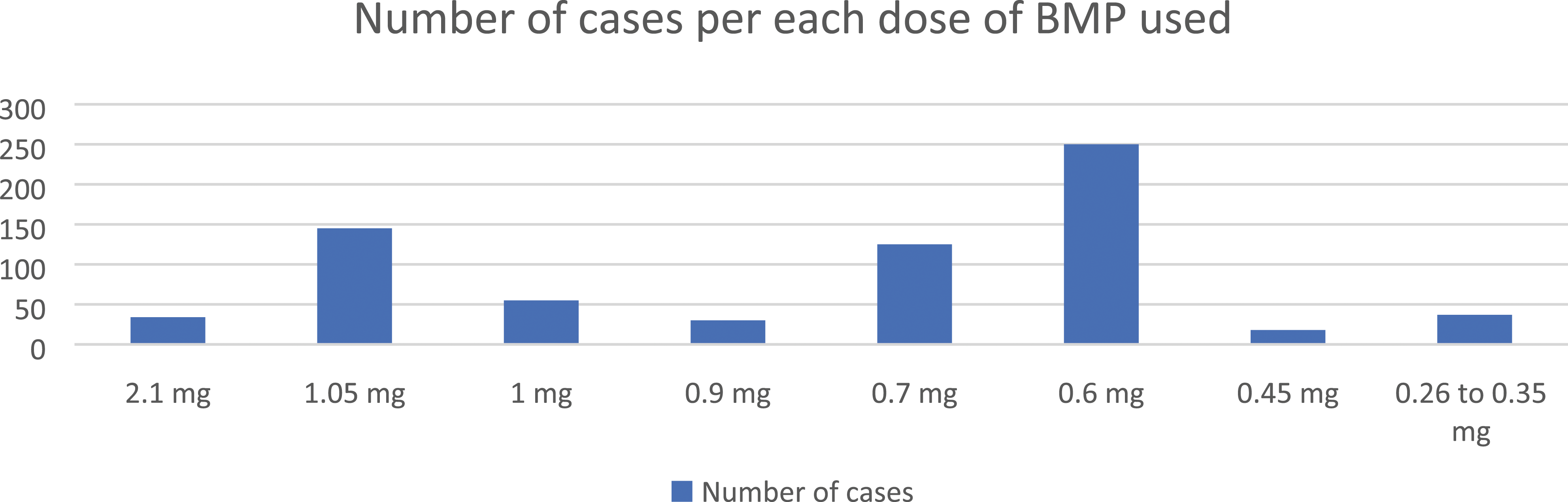
BMP doses used in the studies with its related number of cases.
Risk of Bias
Each study was evaluated for risk of bias using the preset criteria from Methodological index for non-randomized studies (MINORS) 11 for the non-randomized studies and from the Risk Of Bias tool (ROB 2.0) 12 for the randomized studies. This was conducted independently by two reviewers after an initial calibration exercise, and any disagreement was resolved by discussion with a third reviewer.
MINORS provides a longer set of questions (9 vs 12) for non-comparative studies vs comparative studies. The items were scored zero if not reported; 1 when reported but inadequate; and 2 when reported and adequate. The global ideal score was 16 for non-comparative studies and 24 for comparative studies. Low risk of bias is considered when a study total score for a certain outcome is ≥13/16 (for non-comparative studies) or ≥21/24 (for comparative studies). 11 ROB 2.0 tool assesses risk of bias arising due to faulty randomization process, due to deviations from the intended interventions, due to missing of some outcome data, due to inaccurate measurement of the outcome, and/or due to selective reporting of results. 12
Results
The search strategy done in September 2020 returned 318 potentially relevant citations leading to a final 16 studies being selected. The repeated search conducted in September 2021 returned three more relevant citations, but none fulfilled the predefined inclusion criteria. PRISMA diagram for the most updated search is provided in Figure 1. The detailed extracted data from the 16 studies is tabulated in Supplemental materials 4 to 11. As 10 of the 16 studies were reporting doses of Bone Morphogenetic Protein (BMP), the 16 studies were further sub-grouped into BMP and non-BMP osteobiologics for ease of analysis.
BMP Osteobiologics
There were 10 studies that focused on BMP.13-22 Regarding the studies design, four were retrospective cohort studies,13,16,17,19 four prospective cohort studies,14,20-22 one prospective historically controlled study, 18 and one randomized Controlled study (RCT). 15 All studies have been conducted in the United States, using the same BMP product under the commercial name “INFUSE” (Medtronic, MN, USA). The BMP doses used per level varied between 0.26 mg to 2.1 mg, for the number of cases used per each dose please refer to Figure 2, Table 1, and supplemental material 5. The total number of enrolled cases was 694 cases, their mean age was 45.75 years with males representing 47% (only one study with total of 11 cases did neither report the gender nor the mean age 20 ). Regarding type of surgical intervention, all studies reported ACDF with anterior plating, “one level” in 351 cases, “two levels” in 90, “three levels” in 88, “four levels” in 6, “one or two levels” in 18, “one or two or three levels” in 41, and “two or more levels” in 100. Polyetheretherketone (PEEK) cages were used as an interbody spacer in half the studies,13,16-18,21 while a cortical ring allograft was used in three studies.14,15,20 The remaining two studies used a cortico-cancellous allograft 19 and a bioabsorbable cage 22 respectively (Supplemental material 6).
For the fusion radiological assessment method implemented, X-ray alone was used in one study, 18 CT alone in one study, 16 both Xray & CT together in two studies,15,17 while performing Xray only initially followed by CT as a back-up method when the X-ray images were indeterminate or when pseudoarthrosis suspected was used in three studies.13,14,19 The radiological method was not mentioned in the remaining three studies.20-22 Only five studies (50%)13-17 followed the fusion assessment criteria recommended by Oshina and Rhee,9,10 where ISM less than or equal to 1 mm on dynamic lateral X-rays was used in two studies and CT scan was used for all patients in another three studies with or without X-ray (Supplemental material 8). The reported fusion rates as well as their reporting postoperative intervals are detailed in Table 1 and supplemental material 9. Complications were reported in 8 of the 10 studies,13,14,16-21 as expected the most common complications related to BMP were dysphagia, local soft tissue edema, surgical site collection (hematoma/seroma), dyspnea, and heterotopic calcification. The incidence of these complications in each study is detailed in Supplemental material 10. Two studies received funding from the studied BMP product manufacturing company,15,18 authors in four studies (including the two which received the funding) declared financial ties with the studied BMP product manufacturing company.15,17,18,22 Authors of another two studies declared possible conflict of interest by being paid consultants in two other spine companies not related to the studied BMP product.13,19 (Supplemental material 11)
Non-BMP Osteobiologics
There were six studies that focused on non-BMP osteobiologics.23-28 Each reported the use of one type of osteobiologic in certain dose/concentration/volume except one study which compared two osteobiologics. 23 The compositions of the reported osteobiologics are detailed in Table 1. The data is relatively heterogenous in comparison to the BMP group studies. Regarding the studies design, two were randomized control studies (RCT),25,26 three were prospective cohort studies,24,27,28 and the sixth one was a retrospective cohort study. 23 The six studies have been conducted in six different countries i.e., USA,26,27 Canada, 26 Turkey, 28 Iran, 25 Spain 23 & Belgium. 24 The total number of enrolled cases was 580 cases. Regarding type of surgical intervention, “single level ACDF with anterior plating” was reported in a total of 98 cases, “one level without plating” in 165, “two levels with plating” in seven, “two levels without plating” in 88, and “one or two” levels with plating in the remaining 222. PEEK cages were used as an interbody spacer in two studies,27,28 while cortical ring allograft was used in one study. 26 In the remaining three studies the disc spacers were made from the osteobiologic material itself.23-25 For the standard fusion assessment protocol implemented, X-ray alone was used in one study, 28 Xray initially followed by CT as a back-up method when the X-ray images were indeterminate or when pseudoarthrosis was suspected in three studies,23,24,26 while performing both Xray & CT together was done in two studies.25,27 All six studies described their definitions for fusion but only two studies25,27 followed the fusion assessment method recommended by Oshina and Rhee.9,10 The reported fusion rates as well as their reporting postoperative intervals are detailed in Table 1 and supplemental material 9. Complications were reported in 3 of the 6 studies,23,24,26 and most encountered complications were mechanical failures related to the interbody graft especially for those interbody spacers made wholly from the osteobiologic material, where collapse, displacement, and/or local kyphosis have been reported. Dysphagia was reported only with the BOD (27%) and Surgibone (7%) grafts. 23 Three studies received funding from the studied product manufacturing company,25-27 and authors in one of these studies declared financial ties with the studied product manufacturing company. 26
Risk of Bias of Tested Outcomes
The overall risk of bias for the tested outcome was considered low in 3 from the 13 non-randomized studies (18.75%),24,27,28 and for all the 3 randomized studies (100%).15,25,26 This sums up to 37.5% (6/16) of all the included studies. Table 2 summarizes the overall scoring for the risk of bias for the primary outcome and the factors related to the rating, as described in the Methods for non-randomized studies.
Discussion
The number of spinal fusion surgeries is progressively increasing on annual basis, 2 with a parallel increase in the number of available osteobiologics being promoted for surgeons as an essential adjuncts to fusion interventions in a market expected to exceed 20 Billion US dollars in just few years. 3 The surgeon’s choice of a specific osteobiologic would logically depend on its efficacy in achieving solid fusion, possible attributed side effects and morbidities, and its cost. As with any bio substance, the presence of dose-effect and dose-side effect correlations are expected. This study aims to investigate these correlations in the osteobiologics used in ACDF surgeries based on the available literature.
BMP Osteobiologics
In 1965, Urist reported that demineralized bone matrix (DBM) implanted in muscular tissues induces ectopic formation of bone tissues which postulated the presence of certain bioactive factors responsible for inducing bone formation. 29 The bone formation characteristics of isolated BMP-2 was further highlighted when the osteogenic activities of 14 BMPs were compared using an adenoviral gene transfer technique yielding BMP-2 to be among the most potent. 30
However, BMP-2 desired positive influence on postoperative fusion does not come without drawbacks, most commonly seen are local tissue swelling causing dysphagia and/or life-threatening breathing difficulties, 31 usually attributed to BMP-2 being chemoattractant for lymphocytes, monocytes, and macrophages. 32 Besides, BMP-2 has been reported to up regulate the proliferation of endothelial cells causing increase in local micro vascularity inducing hyperemia rather than true exudate. 33 Thus in most BMP cases with postoperative neck swelling causing acute breathing difficulties or dysphagia, when an emergent re-exploration was done, no identifiable drainable fluid collections were detected but rather diffuse swelling of the anterior neck soft tissue structures. 31 Also, excessive bone growth “heterotopic ossification” might exceed the boundaries of the operated interbody space causing pressure on surrounding nearby vital structures or even fusion of non-targeted adjacent segments. 34 Experimentally in animal models, it has been shown that the rate of bone formation to be dependent on the amount of implanted BMP-2, with the period needed for bone formation reduced from 14 to 5 days using higher doses of BMP-2. 35 Again, in animal models, the associated inflammatory edema has been also proven to be dose dependent, with the magnitude of swelling increases with increasing the dose of applied BMP-2. 36
It is thus crucial while planning to use BMP-2 in ACDF to delineate the fine line between achieving desired fusion while avoiding undesired excessive ossification and/or local soft tissues edema. As it has been experimentally shown, these desired effects and undesired side effects are both dose-dependent. In this systematic review, we tried to delineate that line (if any) by answering a few questions.
Were the tested doses of BMP-2 precisely reported? And can BMP doses be precisely selected and accurately reproduced during surgeries?
As mentioned earlier, all the BMP studies included in this systematic review have been conducted in the United States, using the same BMP product which is sold under the commercial name “INFUSE” (Medtronic, MN, USA). The used BMP is recombinant human bone morphogenetic protein-2 (rhBMP-2) administered on an absorbable collagen sponge (ACS) carrier. And according to the manufacturer, neither the rhBMP-2 nor the ACS can produce new bone tissue independently and it is only when they are used together that the bone induction process could be initiated. The manufacturer supplies it in six different kit configurations with each kit containing 1 of 5 different graft “ACS” sizes (.7, 1.4, 2.8, 5.6, & 8.0 cc) along with 1 of 3 different BMP vials (1.05, 4.2, & 12 mg). The BMP vial is diluted with preset amount of saline and then evenly distributed over the ACS and left for at least 1 minutes till becoming evenly soaked. After preparation, any chosen kit will yield a unified concentration of 1.5 mg BMP/1 cc. 37 Surgeons can use the appropriate graft size in cc (or even part of a graft e.g. 1/2 or 1/3) to fill the interbody spacer used. Thus, desired doses can be accurately and easily calculated and reproduced in the daily practice settings. Interestingly enough, BMP has not been yet granted FDA approval for use in the cervical spine, 38 so all the included studies represent off-label clinical application. Also, it worth noting that the FDA released an advisory on life threatening complications associated with BMP use in cervical spine fusion in 2008. 39
Were the Postoperative Fusion Rates Reported in Accordance with the Current Literature Recommendations for Assessing Anterior Cervical Fusion?
Different methods are used to confirm successful fusion or diagnose pseudarthrosis after anterior cervical spine fusion, with Surgical re-exploration being the gold standard despite being impractical and static X-rays being the most used despite being inaccurate. 40 Accurate diagnosis may be challenging, and different surgeons may disagree. 41 These inconsistency in reporting within the concerned literature may indirectly compromise reaching helpful conclusions. 42
In the 10 BMP studies included in this review, five studies followed the current literature recommendations for anterior cervical fusion assessment by Oshina et al and Rhee et al.9,10 Two studies13,14 applied the ISM method with CT as a backup for indeterminate cases, and fusion was confirmed in 129/130 cases. The other three studies15-17 used CT for assessment of all enrolled cases, and fusion was confirmed in 247/248 cases at 6 months with the only patient who did not show fusion had undergone previous cervical fusion procedure, had a 3-level ACDF, and was a smoker who eventually showed solid fusion after repeating the CT at the 9 months follow up. 16 The cases which were assessed using “literature recommended methods and criteria” represents more than half the cases in all the BMP studies included in this review [378/694 (54%)], and are reported for doses varying from 0.45 to 2.1 mg per level.
To be noted that studies where CT assessment was used only after the initial X-ray screening deemed indeterminate but did not apply the ISM method on the initial X-ray were considered not aligning with the current literature recommendations, 19 because there is a high probability of falsely appreciating fusion in the initial x-rays for the cases that were not referred for confirmatory CT.43,44
In this systematic review, the data analysis showed that the recommended fusion assessment methods and criteria were applied on a large number and percentage of enrolled cases covering a wide range of used BMP concentration. The sensitivity and specificity of this assessment method has been confirmed by other authors 45 which would further strengthen any drawn conclusions regarding dose-efficacy correlation. 41
What is the Lowest BMP Dose that Achieved a Successful Fusion Rate?
As mentioned earlier, the BMP doses reported in the included studies varied between 0.26 mg to 2.1 mg per level. Three retrospective cohort studies directly compared the outcome of different BMP doses, all used PEEK cages as the interbody spacer, and all reported the lowest used dose to be about 0.6 to 0.7 mg. In 2008, Tumialan et al compared three different doses in a large series: 2.1 mg for the first 24 cases, 1.05 mg in the subsequent 93 cases, and 0.7 mg in the final 83 cases. The authors reported 100% fusion rate detected in all cases by the “last follow-up” visit confirmed by routine CT scans. 17 In 2013, Lu et al reported a similar 100 cases series in which the used 2.1 mg for the first 10 cases, 1.05 mg in the subsequent 48 cases, and 0.7 mg in the final 42 cases. The fusion rate was also 100% at 6 months period confirmed by ISM method. 13 In 2017, Burkus et al compared 0.6 mg dose in 220 cases to 1.05 mg in 4 cases, and they concluded that “there were no meaningful differences between the groups in clinical outcomes, rates of fusion, …”. However, the fusion assessment radiological criteria were not aligning with “literature recommended” assessment methods. 18
Regarding the studies that reported utilization of single BMP dose, the lowest doses reported were by Pourtaheri et al where a dose varying between 0.26 to 0.35 mg BMP per level (between 1/8 and 1/6 of an extra-small 1.4 cc ACS) was used in 37 cases. 19 The interbody spacer used was a cortico-cancellous allograft where a 4 mm drill holes were made in the top and bottom of the allograft spacers, and the rhBMP-2 was placed in these holes. Fusion rate was 97.3% at 6 months postoperatively (only one patient didn’t show fusion by 6 months). The fusion had been mainly assessed using static x-rays images, and the authors intraoperatively placed a fibrin sealant anteriorly over the grafts for containment of the rhBMP-2 from the anterior structures in an attempt to reduce the risk of postoperative dysphagia. 19 Whether the fibrin sealant might had a negative impact on osseous fusion process in this study remains unclear but animal studies confirmed sealant inhibition of allografts fusion.46,47 Another similarly low BMP dose of 0.45 mg placed within a cortical ring allograft has been reported by Baskin et al for 18 cases with 100% fusion rate at 6 months postoperatively. The study is a RCT and fusion has been confirmed by routine CT but the number of cases is relatively small and the study was funded by the manufacturer of the used BMP product. 15 According to a recent systematic review, it is unlikely that structural allograft had any positive impact on fusion rate of this low BMP dose vs PEEK cages used for the aforementioned higher doses. 48
In three smaller studies (not included in this current systematic review due to low number of enrolled cases) a fusion rate of 100% was reported for concentrations of 0.2-0.4, 0.7-1.1, 1.4-2.1 mg/level. 49 These three small studies along with another six studies (listed in this current systematic review) were included in a meta-analysis published in 2016, where the authors concluded that the lowest concentration analyzed (.2-.6 mg/level) had a fusion rate similar to the cohort that received the highest dose (1.1-2.1 mg/level), and thus they recommended the use of .2-.6 mg/level of BMP in ACDF. 50 In another meta-analysis published in 2020, Wen et al reported that the average fusion rate analyzed from 17 studies was 98.8% in low dose (<.7 mg/level) of rhBMP-2, 98.22% in middle dose (.7-1.1 mg/level) of rhbmp-2 and 95.29% in high dose (>1.1 mg/level) of rhBMP-2, indicating that lower doses might achieve at least similar if not marginally better fusion rates than higher doses. 51
From the reported dose-related fusion rates reviewed in this systemic review as well as in other publications, it seems that BMP doses lower than .7 mg per level or may be even as low as .2 or .45 mg per level are enough to achieve satisfactory fusion rates in ACDF as equal as higher doses.
Are BMP Related Side Effects Dose-Dependent?
Although probably side effects would escalate with increasing the BMP dose used considering the induced biological cellular interactions, it was difficult to withdraw comparisons regarding dose-dependent side effects occurrence in the included studies because of two main drawbacks. First, the lack of standardization about how to define, measure and report these side effects. Taking dysphagia as an example, as a common specific postoperative morbidity associated with BMP, there has been inconsistency in how to define or to grade, while different studies had different reporting metrics, e.g. cumulative rate, 18 mild and moderate and severe, 19 frequent and occasional, 16 significant and insignificant, 17 Dysphagia score, 13 …. etc., so actual correlation between the dose of the BMP used and the degree of dysphagia cannot be accurately assessed across different studies. Second, while fusion rate is assessed per operated level i.e., “level dose”, complications are correlated to the “total dose” of BMP, so even when a small dose per level is used (e.g., 0.5 mg) still a multilevel application could sum up to a much higher dose. This suggests that while the total dose of BMP used might impact the severity of dysphagia, it is only one of the many other possible contributors as the number of exposed levels, extent of dissection and retraction, and duration of surgery.
The two included studies that tested different doses within its own cohort group of patients could not provide a clear conclusion. Tumialan et al, reported that they noticed no apparent reduction in the complication rate after a reduction of the dose of rhBMP-2 from 2.1 to 0.7 mg, although they noted that they have not observed any further incidents of excess interbody bone formation at the dose of 0.7 mg per level. 17 And it is to be noted also that they routinely administered 10 mg of dexamethasone at the start of surgery in their nondiabetic patients as a putative neuroprotectant, a practice they declare that it is not based on any scientific data and that their use of dexamethasone is independent of the use of rhBMP-2 and was not intended to address prevent rhBMP-2–related edema problems. 17 However, Dexamethasone has shown ability in rodent models to reduce BMP-2 induced edema52-54 which might induce similar effect in humans ameliorating the effect of high BMP doses. In the second included study, Lu et al reported that they found a weak correlation between the total dose of BMP used (also ranged from 2.1 to 0.7 mg) and dysphagia score after performing a rank correlation analysis (correlation coefficient 0.166, P = 0.046). 13
In a study not included in this systematic review due to small number of enrolled cases, Frenkel et al detected dysphagia in 4 of 5 patients (80%) who received 2.1 mg BMP per level and in 1 of 3 (33.3%) who received 1.4 mg/level. 49 Wen et al reported that the complication rate pooled from nine ACDF studies was 0% when low dose (<0.7 mg/level) rhBMP-2 was used, 15.26% for middle dose (.7-1.1 mg/level), and 24.14% for high dose (>1.1 mg/level). 51 In a similar systematic review conducted by Hofstetter et al in 2016 for seven ACDF studies using different inclusion criteria from the one used in this systemic review, it has been shown that complication rates were 7.9% with 0.7-1.05 mg BMP per level, and 10.2% with 1.1-2.1 mg BMP per level exhibiting a significant positive correlation with the dose of BMP used per level. The authors admitted the inconsistent definition of neck swelling and dysphagia in the current literature, and that their analysis included only the most severe complications requiring reoperation, readmissions, reintubations, or tracheostomies. 50
Another important point to consider is that despite the reported BMP associated morbidities, it is important to investigate whether the rate of complications after surgeries using low BMP doses (e.g., 0.45 mg per level) is different from those commonly seen after surgeries using other osteobiologics (e.g., iliac crest autograft) for single or multiple levels ACDF or not.
Non-BMP Osteobiologics
The data provided by the available literature in this group of studies is relatively heterogenous in comparison to the BMP group as each included study reported the use of one type of osteobiologic in certain dose/concentration/volume except the study by Ibanez et al 23 which compared two osteobiologics. The reported doses for each osteobiologic within its specific study was rather an average or range rather a fixed value except for Biocoral® coral elliptic cylindrical grafts 24 which was 12 × 14 × 14 mm with a fixed volumetric density of 2.14 g/cm3. So, there have not been neither fixed or directly compared doses to assess comparative dose-related fusion or complications.
All the included studies reported surgeries for one or two levels only with none reporting for three or four levels so utilization in multiple level surgeries is yet to be assessed. The earliest fusion rates in this group of osteobiologics were not reported before the 12 months follow up period, with the lowest fusion achieved by BOD (6.8%) 23 and the highest achieved with Acrylic cage filled with 1-1.5 mL of tricalcium phosphate mixed with few drops of blood (96.8%). 25 Generally, the fusion rates reported in this group were relatively lower and after longer follow-up periods than in the BMP group.
As expected, reporting of post operative dysphagia and soft tissue swelling was uncommon, but of interest, mechanical failures related to the interbody spacers made wholly from the osteobiologic material as interbody space collapse, graft fracture, graft displacement, and/or postoperative local kyphosis have been reported by Ibanez et al. 23
Limitations of this Systemic Review
The quality of the results of any systematic review depends heavily on the quality of included studies. We attempted to be entirely compliant with preset PICO and the PRISMA guidelines. The average number and quality of studies available for non-BMP osteobiologics in ACDF were insufficient, and therefore further studies are warranted. A body of quality literature has been available for BMP studies, which allows for more definitive conclusions. Some quality studies were not included because they did not provide sufficient information regarding the dose of BMP used per level in relation to outcome and complications. We tried to explore effects of reproducible BMP dose preparation and standardized assessment of post operative fusion, but multiple other factors might have influenced the efficacy of bone fusion and the complication rates including for example patient comorbidities, surgical technique, and use of supplemental instrumentation, and our results could have certainly been biased by these factors. Thus, the results of this systematic review should serve as the foundation for future well-designed prospective RCT studies to provide better guidelines on the dosing of BMP in anterior cervical fusion surgery.
Conclusions
Taking into consideration the heterogenicity of reporting in the related available literature, it seems that for rhBMP-2 usage in ACDF procedures, doses lower than 0.7 mg per level can achieve equal successful fusion rates as higher doses, and there is no complication-free dose proved yet. It seems that the lower the dose the lower the incidence of serious complications, and that multilevel applications may tend to increase the severity of complications either directly or indirectly. As for non-BMP osteobiologics the studies are almost one per each studied osteobiologic and thus conclusions must be drawn individually and with caution.
Supplemental Material
Supplemental Material - Dosing Strategy for Osteobiologics Used in Acdf Surgery, Influence on Fusion Rates and Associated Complications. A Systematic Literature Review
Supplemental Material for Dosing Strategy for Osteobiologics Used in Acdf Surgery, Influence on Fusion Rates and Associated Complications. A Systematic Literature Review by Waeel O. Hamouda, Sotiris Veranis, Oscar Krol, Navraj S. Sagoo, Peter G. Passias, Zorica Buser, Hans Joerg Meisel and Tim Yoon in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
