Abstract
Study design
This study constitutes a systematic review of the literature.
Objective
The aim of this study was to identify and present all available studies that report on the costs of osteobiologics used in anterior cervical discectomy and fusion (ACDF).
Methods
The literature was systematically reviewed to identify studies with specific inclusion criteria: (1) randomized controlled trials and observational studies, (2) in adult patients, (3) with herniated disc(s) or degenerative cervical spine disease, (4) reporting on either direct or indirect costs of using specific osteobiologics in an ACDF operation. (5) Only studies in English were included. The quality of the included studies was assessed using the MINORS and RoB 2.0 tools.
Results
Overall, 14 articles were included; one randomized controlled trial and 13 observational studies. The most commonly used osteobiologics other than autograft/iliac crest bone graft (ICBG) were allograft and bone morphogenetic protein (BMP). None of the studies was reported to be industry-supported. There was considerable heterogeneity on the reported costs. Overall, most studies reported on surgery-related costs, such as anesthesia, operating room, surgical materials and surgeon’s fee. Only two studies, both using allograft, reported the exact cost of the osteobiologic used (450 GBP, $700). Some of the studies reported on the cost of care during hospitalization for the surgical operation, such as radiology studies, emergency room costs, cardiologic evaluation, laboratory studies, pharmacy costs, and room costs. Only a few studies reported on the cost of follow-up, reoperation, and physical therapy and rehabilitation.
Conclusion
Based on the data of this current systematic review, no recommendations can be made regarding the cost-effectiveness of using osteobiologics in ACDF. Given the high costs of osteobiologics, this remains a topic of importance. The design of future studies on the subject should include cost effectiveness.
Introduction
Anterior cervical discectomy and fusion (ACDF) has long been considered the gold-standard operation for the treatment of symptomatic cervical disc degeneration non-responsive to conservative treatment. 1 The original technique described in 1958 used iliac crest bone graft (ICBG) obtained from the iliac crest of patients during the fusion operation.2,3 Since then, many alternatives have emerged with varying cost, availability, efficacy, and safety. 4 However, autograft remains the most commonly implemented graft method to this day. 5 Each available option comes with its advantages and drawbacks, but an important consideration when treating patients is the cost of care.
During the decade between 2001 and 2010, more than 3.5 million fusion operations were performed in the USA, culminating in more than 285 billion dollars in charges. 6 The overall mean cost of a single-level ACDF operation in the USA has been reported to be around $13900, while the cost increases with each additional level. 7 Needless to say, the cost of an ACDF operation, and any surgical operation for that matter, can be significantly affected by medical and non-medical factors.7-9 Some examples for the latter include geography (different continents, countries, and even cities), type of practice (private versus public), and insurance coverage and policies. Medical factors can be defined as those that are directly linked to the operation itself and the subsequent medical care. Those can be, amongst others, related to the surgeon, the surgical instrumentation, the surgical approach, the length of hospitalization, the patients’ comorbidities, the peri- and post-operative complications etc.
Although many studies have reported on the efficiency of alternative osteobiologics and their adverse events profile, not many studies have compared them from a cost-effectiveness standpoint. As a result, there is no consensus on whether using those alternative osteobiologics in place of ICBG is cost-effective for patients and healthcare systems alike.
The current systematic review aimed at identifying and presenting all studies available in the literature reporting on the costs of the various osteobiologics available for ACDF.
Materials and Methods
The review protocol was not registered on PROSPERO. The extracted data for all studies are available upon reasonable request.
Literature Search
Two investigators (GM and MCD) performed an individual electronic database search of the PubMed (MEDLINE), EMBASE, clinicaltrials.gov, and Cochrane Library databases. The exact search algorithms for all databases are available in Appendix A. The last literature search was performed on October 25th, 2020. The PRISMA guidelines for reporting reviews and meta-analyses were followed (Appendix B). 10
Inclusion and Exclusion Criteria
Our inclusion criteria were:
(1) adult patients (18-80 years old), (2) with herniated disc(s) or degenerative cervical spine disease that underwent an ACDF operation. (3) We included randomized controlled trials (RCTs), retrospective or prospective cohort studies, and case-control studies with at least ten patients in each study arm, that (4) reported on the direct and/or indirect costs (5) for at least one arm of their study or compared the costs of different osteobiologics or the costs of osteobiologic use versus no osteobiologic use between the study arms. In addition, we only included studies that (6) specified precisely which type of osteobiologic they used. Finally, only studies (7) in the English language were considered eligible for inclusion.
Our exclusion criteria were: (1) studies with patients deemed as skeletally immature, diagnosed with infections, scoliosis/cervical deformities, spinal cord injury, spinal fracture, or patients with a history of tumor. Moreover, (2) studies that reported on combined anterior and posterior cervical discectomy and fusion and those on anterior cervical corpectomy and fusion were excluded. (3) Studies that did not have any cost-related information and those (4) which did not specify the exact osteobiologic used were also excluded. Finally, we excluded (5) in vitro/animal studies, case reports, studies with less than ten patients in each arm, abstracts, letters to editors, white papers, and narrative or systematic reviews.
Definitions
Direct costs were defined as the costs of osteobiologic, surgery, hospitalization, follow-ups, complications, and revisions. Indirect costs were defined as the costs for rehabilitation, physiotherapy, and medications, as well as taking into account the time to return to work.
Osteobiologics were grouped into the following nine categories: autologous bone graft, allogeneic bone graft/bone marrow aspirate, demineralized bone matrix, hydroxyapatite, bone morphogenetic protein (BMP), platelet-rich plasma, ceramic-based bone graft, mesenchymal cells, and other synthetic materials (eg, bioglass).
Data Extraction
The data were extracted using a predesigned standardized data form by two independent investigators (GM and MCD). The following data were extracted, when available: First author’s name, year of publication, study design, country, setting (inpatient, outpatient), osteobiologic used in each study arm, number of levels fused, age, male/female ratio, number of participants in each study arm, the total cost for each study arm, direct and indirect cost breakdown, smoker status of the patients, length of follow-up and patient-reported outcomes at the longest follow-up available. Finally, we noted whether the studies were industry-supported or not. Discussion resolved any discrepancy between the reviewers, and if consensus could not be reached, a third senior investigator was consulted (AKD).
Quality of Studies Assessment
The quality of the included studies was assessed using the risk of bias 2 (RoB 2) tool for RCTs and the MINORS tool for cohort studies. The RoB 2 tool considers the randomization process, the deviations from the intended interventions, the missing outcome data, the measurement of the outcome, and the selection of the reported result to estimate the overall bias. The MINORS tool assesses the studies based on the study design and execution (clearly stated aim, consecutive patients, prospective collection of data, appropriate endpoints, unbiased assessment, appropriate follow-up, minimal loss of patients to follow-up, prospective power analysis, adequate control group, proper statistical analysis and comparability of the two study groups).
Results
Selection and Characteristics of the Included Studies
The electronic database search resulted in 868 unique studies after the removal of duplicates. Of those, 677 were excluded after the title and abstract review resulting in 191 articles eligible for full-text review. After reviewing the full text of all articles, 14 studies were appropriate for inclusions in the current systematic review.11-24 The exact process of the database search is presented in the form of a PRISMA flowchart in Figure 1. PRISMA flowchart for study selection.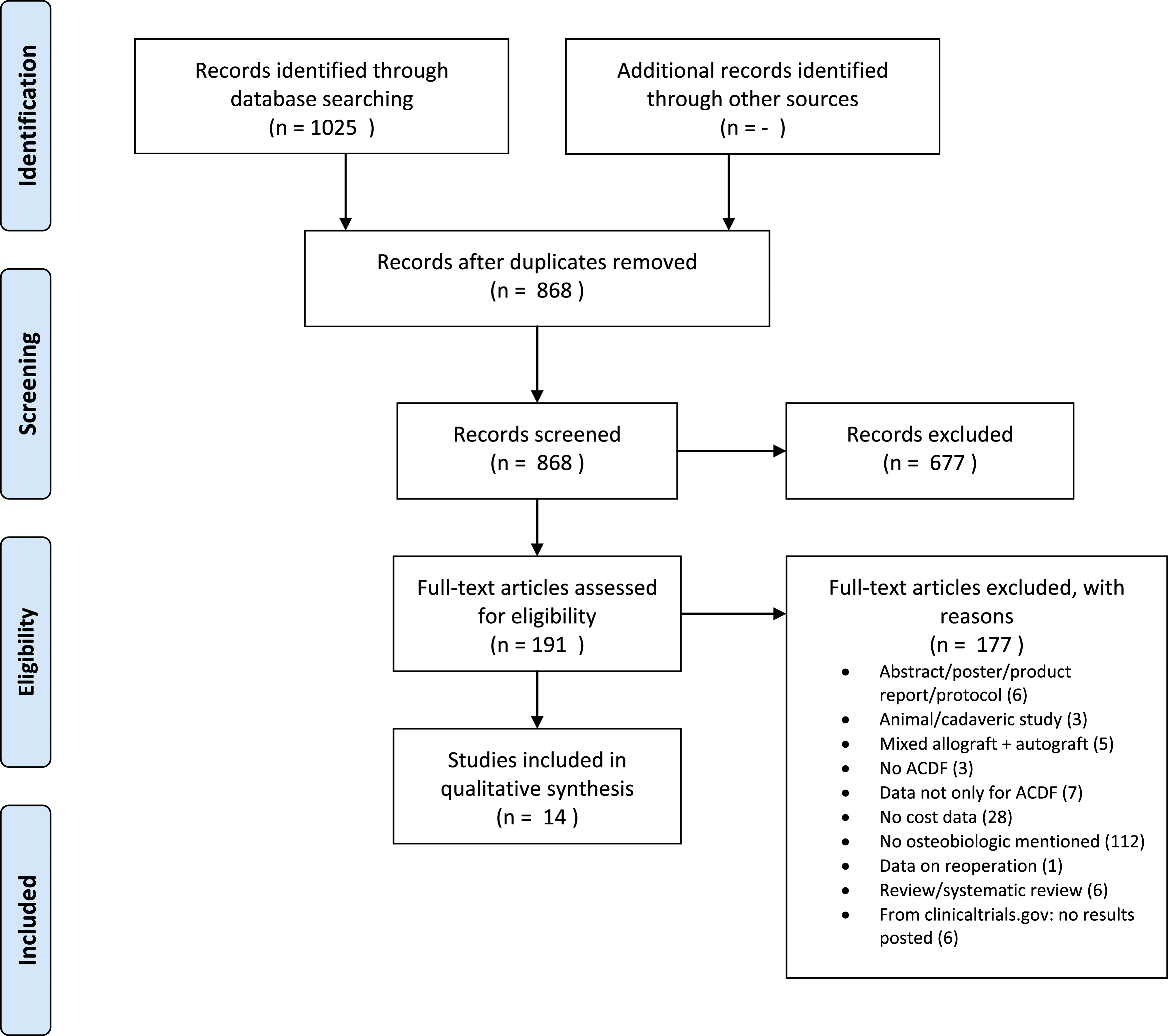
Table Presenting the Basic Characteristics of the Included Studies.
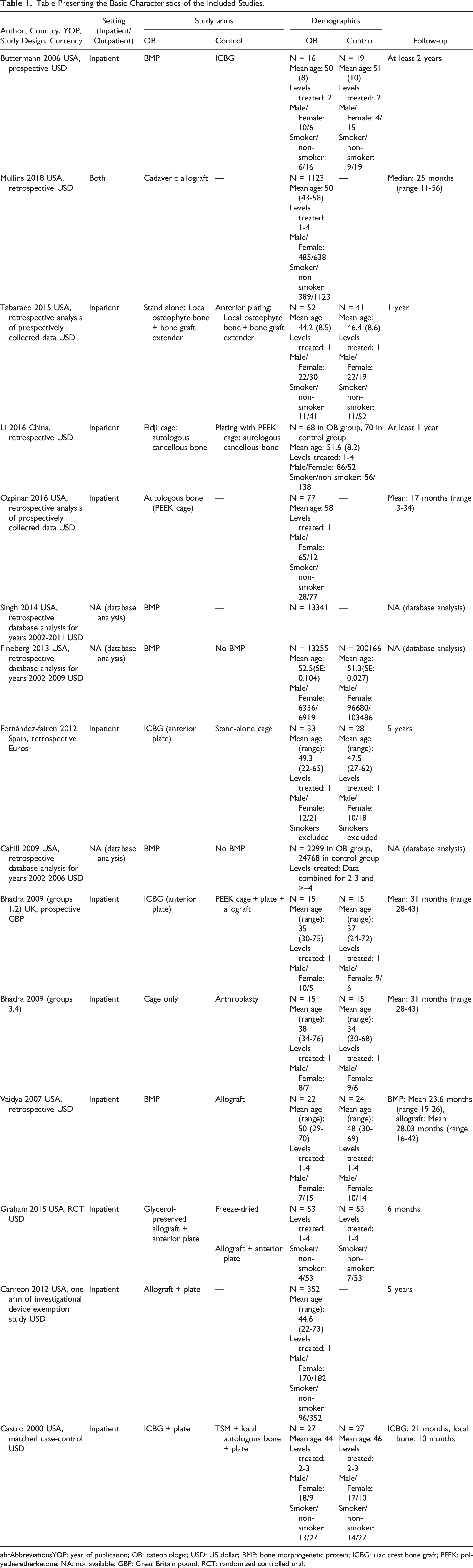
abrAbbreviationsYOP: year of publication; OB: osteobiologic; USD: US dollar; BMP: bone morphogenetic protein; ICBG: iliac crest bone graft; PEEK: polyetheretherketone; NA: not available; GBP: Great Britain pound; RCT: randomized controlled trial.
Risk of Bias Assessment
The risk of bias assessment for the only RCT revealed “Some Concerns” regarding the presence of risk of bias. More specifically, “some concerns” for risk of bias were present in domains 2 (“Deviations from the intended”) and 3 (“Missing outcomes”). It should be noted that these concerns were not relevant with the aim of our current systematic review.
The assessment of case series and cohort studies using the MINORS tools, revealed an overall moderate quality for the case series (scores ranged from 9/16 to 12/16) and moderate quality for the cohort studies (scores ranged from 16/24 to 18/24). The exact score for each study is presented in Appendix C.
ACDF Costs and Osteobiologics
Overall, most studies reported on the surgery-related costs, such as anesthesia costs, theatre costs, costst for surgical materials (eg, plates, cages, screws), and surgeon’s fees. Only two studies, both using allograft, reported the exact cost of the osteobiologic used.20,22 Bhadra et al reported that the cost of allograft was 450 GBP, while Graham et al reported a cost of $700.
Some of the studies reported on the cost of care during the hospitalization for the surgical operation, such as radiology studies, emergency room costs, cardiologic evaluation, laboratory studies, pharmacy costs, and room costs. Furthermore, only a few studies reported on the cost of follow-up, reoperation, and physical therapy and rehabilitation.
Of the included studies, four included only one group/arm that was relevant to our study. Two studies reported cost data in patients who underwent ACDF using allograft; the total cost reported by Mullins et al 14 was $18095 for outpatient (day case) surgery, which increased to $24492 for inpatient surgery. Carreon et al 23 reported a total cost of $15714. Another study reported some of the direct costs associated with the use of autologous bone graft but not the exact osteobiologic cost or the total cost of the operation 15 ; and finally, a study on the use of BMP reported a varying total cost ranging from $14401 in 2003 to $17271 in 2007, for the period 2002-2010. 16 None of the above studies reported the exact cost of the osteobiologic used.
The remaining ten included studies reported two or more groups relevant to our analysis. Amongst the studies that included comparative groups, two of them compared the costs of using BMP versus not using BMP. The total cost reported by Fineberg et al was $18011 with BMP vs. $12919 without BMP. 17 In comparison, the total cost reported by Cahill et al was $46112 with BMP vs. $31179 without BMP. 19 At first glance, the extra cost associated with the use of BMP might seem to range from $5092 to $14933, however, it was not clear whether this was due to the osteobiologic itself or any undefined associated healthcare costs.
Similarly, Buttermann compared BMP versus ICBG, and the total costs reported were $36155 with BMP vs. $30800 with ICBG. 11 Vaidya et al compared BMP vs. allograft but did not provide information on the total costs. 21 Tabaraee et al compared the costs of using local osteophyte bone plus bone graft extender in stand-alone cages versus a cage-plus-plate; these costs were $10475 for the former vs. $11402 for the latter. 13
Details on the individual costs reported in each study are shown in Appendix D. All appendices can be accessed through the link to the Supplemental Documents for the AOGO Publication folder.
Discussion
Study Overview
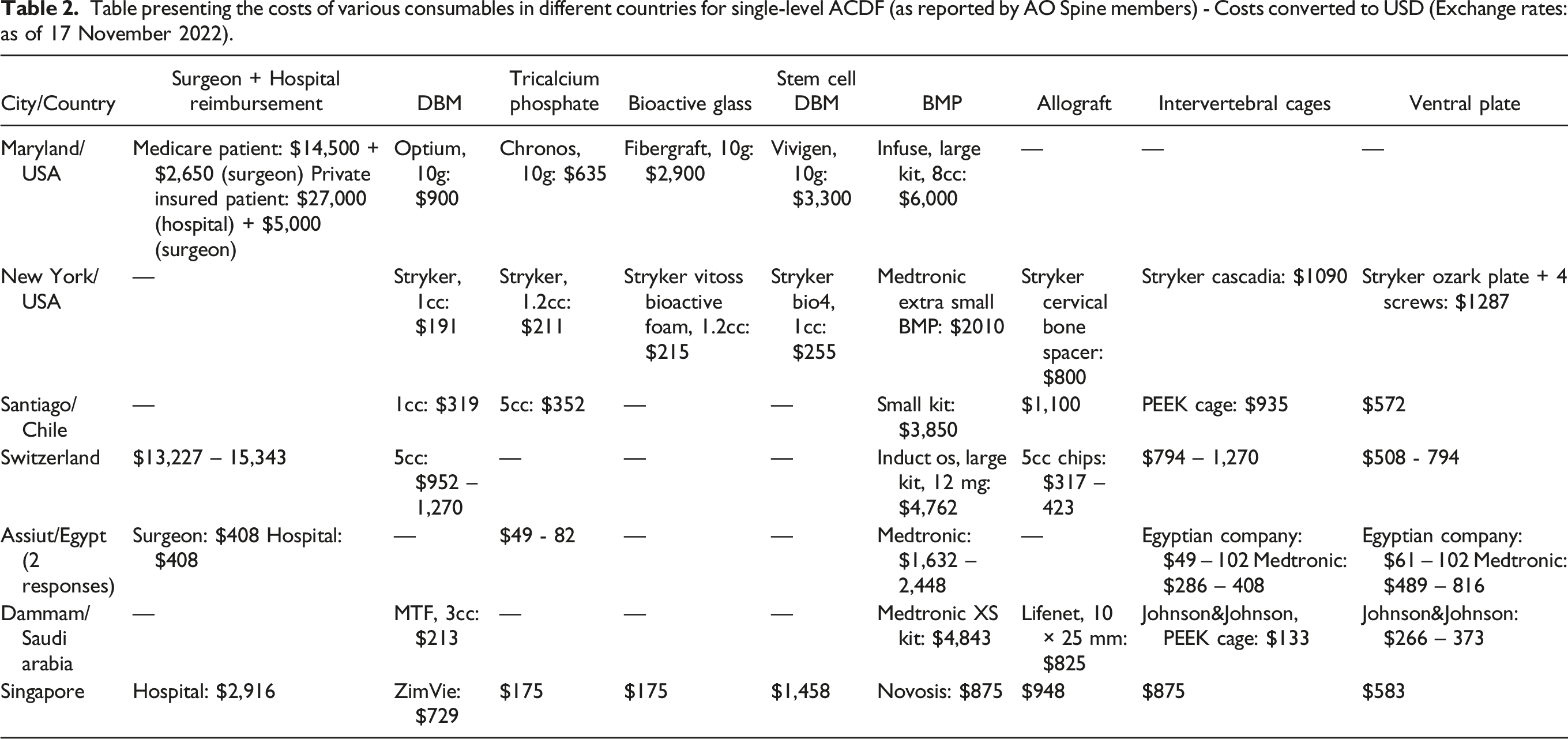
Table presenting the costs of various consumables in different countries for single-level ACDF (as reported by AO Spine members) - Costs converted to USD (Exchange rates: as of 17 November 2022).
Despite the fact that ACDF has been an extensively studied procedure, our electronic database search resulted only in 14 articles that reported on the costs for at least one relevant study group. Within this set, the most commonly reported alternative osteobiologics were allograft and BMP. Most of the available data indicated that alternative osteobiologics usually resulted in greater costs than ICBG; however, we were not able to pool data from different studies together due to the wide variability of parameters (eg, levels of fusion, variability of what contributed to the total costs reported) in each study.
BMP in ACDF
To date, BMP has been officially approved for use in anterior lumbar interbody fusion, and its implementation in posterior lumbar interbody fusion and ACDF procedures is considered to be “off-label.”17,27,28 The cost-effectiveness of BMP use has been more extensively reported in studies with patients that underwent lumbar fusion procedures. In these studies, although BMP was more costly than conventional ICBG, the authors suggested that this effect will be counterbalanced by increased fusion rates and fewer complications.29,30
Similarly, the use of BMP in ACDF has been associated with higher costs than ICBG.17,19 However, in contrast with its lower complication rates in the lumbar spine, BMP has been associated with significantly higher rates of dysphagia and/or hoarseness in ACDF patients, 19 while its cost-effectiveness has not been proven in ACDF per se. Therefore, it remains unclear whether its use in ACDF can be deemed cost-effective even when considering the fusion and complication rates.
The data collected by our systematic review are not sufficient to make solid recommendations in favour of or against the use of BMP for ACDF, from a cost-effectiveness standpoint.
Allograft in ACDF
The use of allograft bone for spinal fusion operations was introduced in an effort to avoid harvesting ICBG, which is associated with post-operative pain and the risk of infection. 21 In this current systematic review, we identified two case series that reported on the costs of allograft implementation in a group of patients undergoing ACDF.14,23 Another study compared allograft costs with BMP costs and found that allograft was cheaper than BMP. 21 In addition, a RCT compared glycerol-preserved allograft costs with freeze-dried allograft costs; unfortunately, no total costs were reported. 22 Furthermore, another study from the UK compared allograft costs with ICBG, empty cage, and arthroplasty costs; allograft was cheaper than autograft, equally expensive as arthroplasty, and more expensive than cage alone. 20
Based on the data identified by this current systematic review, no recommendations can be made regarding the cost-effectiveness of allograft usage in ACDF.
Limitations
The current systematic review has some limitations that ought to be acknowledged. The studies available in the literature were not consistent on what data they included in their cost analysis. Some studies reported the total costs of an ACDF operation; however, they did not provide details on what was defined as total costs. Others provided cost data only for specific equipment/osteobiologics, but did not report the total costs. Another important limitation is that some of the studies used the same database (nationwide inpatient sample database) to identify their patient samples and, thus, patient overlap cannot be excluded. Furthermore, an important limitation found in all cost analysis studies is the wide variation between different countries and their healthcare systems on the costs of the same procedure and equipment/consumables. This became apparent in our case after circulating a simple questionnaire amongst a few AO Spine members from different countries. As illustrated in Table 2, the costs of various consumables present important variability between different healthcare systems, and sometimes even within the same country.
One of the limitations of the data available is the heterogeneity of the data reported. This relates to many parameters. For example, costs and charges were not both always reported within the same article, some studies reporting on the one and other studies reporting on the other. Equally, there is always a difference in health economics between different healthcare systems; we found that published studies provide a bias towards US based healthcare costs.
Different healthcare systems have different cost implications in the osteobiologic as well as the total cost; one observation is that while BMP raises the cost, its cost effectiveness remains unclear.
Conclusion
While cervical disc degeneration and ACDF remain some of the most extensively studied aspects of modern spine surgery, data on costs and cost-effectiveness are infrequent. Where reported, such data suffered from heterogeneity, and its translation between different healthcare economies is challenging. Based on the data identified by this current systematic review, no recommendations can be made regarding the cost-effectiveness of osteobiologics usage in ACDF. It is imperative that the design of future studies considers cost effectiveness, and that surgeons are always conscious of the implications of healthcare economics in their practice.
Supplemental Material
Supplemental Material - What Is the Evidence Surrounding the Cost-Effectiveness of Osteobiologic Use in ACDF Surgery? A Systematic Review of the Literature
Supplemental Material for What Is the Evidence Surrounding the Cost-Effectiveness of Osteobiologic Use in ACDF Surgery? A Systematic Review of the Literature by Andreas K. Demetriades, Georgios Mavrovounis, Moritz Deml, Kyaw Min Soe, Zorica Buser, Hans-Jörg Meisel, and AO Spine Knowledge Forum Degenerative in Global Spine Journal
Footnotes
Acknowledgments
We are grateful to Dr Mohamed Mesregah and Dr Amit Jain for their advice; and to Dr Yabin Wu, Senior Research Project Manager, AO Spine, for his invaluable contributions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
