Abstract
Study Design
Retrospective Case-Series.
Objectives
Due to heterogeneity in previous studies, the effect of MI-TLIF on postoperative segmental lordosis (SL) and lumbar lordosis (LL) remains unclear. Therefore, we aim to identify radiographic factors associated with lordosis after surgery in a homogenous series of MI-TLIF patients.
Methods
A single-center retrospective review identified consecutive patients who underwent single-level MI-TLIF for grade 1 degenerative spondylolisthesis from 2015-2020. All surgeries underwent unilateral facetectomies and a contralateral facet release with expandable interbody cages. PROs included the ODI and NRS-BP for low-back pain. Radiographic measures included SL, disc height, percent spondylolisthesis, cage positioning, LL, PI-LL mismatch, sacral-slope, and pelvic-tilt. Surgeries were considered “lordosing” if the change in postoperative SL was ≥ +4° and “kyphosing” if ≤ -4°. Predictors of change in SL/LL were evaluated using Pearson’s correlation and multivariable regression.
Results
A total of 73 patients with an average follow-up of 22.5 (range 12-61) months were included. Patients experienced significant improvements in ODI (29% ± 22% improvement, P < .001) and NRS-BP (3.3 ± 3 point improvement, P < .001). There was a significant increase in mean SL (Δ3.43° ± 4.37°, P < .001) while LL (Δ0.17° ± 6.98°, P > .05) remained stable. Thirty-eight (52%) patients experienced lordosing MI-TLIFs, compared to 4 (5%) kyphosing and 31 (43%) neutral MI-TLIFs. A lower preoperative SL and more anterior cage placement were associated with the greatest improvement in SL (β = -.45° P = .001, β = 15.06° P < .001, respectively).
Conclusions
In our series, the majority of patients experienced lordosing or neutral MI-TLIFs (n = 69, 95%). Preoperative radiographic alignment and anterior cage placement were significantly associated with target SL following MI-TLIF.
Keywords
Introduction
The malaligned spine in short-segment, degenerative disease has shown to be associated with worse health-related quality-of-life assessments, 1 increased incidence of adjacent level spondylolisthesis, 2 elevated rates of construct failure, and adjacent segment disease.2-5 Described by Foley and Lefkowtiz in 2002, minimally invasive transforaminal lumbar interbody fusion (MI-TLIF) has become a well-established procedure to address degenerative lumbar pathologies. 6
In recent decades, the maintenance and correction of sagittal alignment has shown to be significantly associated with clinical outcomes and quality-of-life. 1 Consequently, there has been a growing body of literature discussing the effect of lumbar interbody fusion on radiographic outcomes. 7 In the MI-TLIF literature, the extent of sagittal correction has come into question, with some studies suggesting a MI-TLIF is a kyphosing operation.6,8-14 However, previous studies often included heterogenous patient populations without controlling for multiple confounders (e.g., cage type), limiting the interpretability of radiographic outcomes analyses following MI-TLIF.
With the increasing utilization of MI-TLIF, identifying predictors of sagittal alignment could help guide surgical decision making. Given conflicting reports in existing literature, we focused on a homogenous series of MI-TLIF patients, with indication, clinical, and operative details remaining constant. To this end, we investigated radiographic factors that determine MI-TLIF kyphosis or lordosis in patients undergoing single-level fusion for grade 1 spondylolisthesis with minimum 1-year follow up.
Methods
Study Design and Patient Population
We conducted a single-center, retrospective case series of consecutive patients who underwent MI-TLIF by a single spine surgeon from 2015-2020. Inclusion criteria included patients undergoing single-level surgery for grade 1 degenerative spondylolisthesis. Patients receiving expandable interbody devices undergoing unilateral facetectomies with a contralateral facet release were included. Multiple-level TLIF, revision TLIF, surgeries utilizing osteotomies, prior lumbar fusion at adjacent levels, or cases involving infection, tumor, or osteoporosis were excluded. This case series has been reported in line with the PROCESS Guideline 15 and was approved by our university’s IRB (IRB# 202004195). A waiver of consent was obtained.
Surgical Technique
All patients underwent unilateral facetectomies with a contralateral facet release using an osteotome and high-speed drill, along with bilateral pedicle screw fixation. Expandable interbody devices were placed, with the size, type, and lordotic angle chosen based on the surgeon’s preference and patient anatomy. MI-TLIF surgeries were performed by a standard operative technique that has been previously described.16-21 Surgeries were performed on a modular ProAxis Jackson table (Mizuho), which would be oriented at the start of the case in a flexed position to facilitate the approach. Tiger Jamshidi needles (Stryker) and fluoroscopy were used for percutaneous pedicle screw insertion, and the MetRx sequential dilating tubular retractor system (Medtronic) was used to dilate down to the bilateral facet complex. Though there is mixed evidence regarding its impact on LL, bilateral facetectomies are a well-established and effective technique in MI-TLIF.1,9,17,22 At our institution, we typically perform a unilateral facetectomy with a contralateral facet release. This consisted of using electrocautery to disrupt the facet capsule, then placing a half inch straight osteotome on the facet and malleting until the joint could be felt to move. This joint was then packed with bone auto and allograft at the end of the case. When more substantial lordosis is needed, we generally pursued alternative interbody techniques (e.g., ALIF) or performed open TLIFs with extensive bilateral facetectomies. On the facetectomy side, the craniocaudal extent of the superior articulating process was exposed and drilled, with special attention to leave a portion of the medial inferior articulating process and pars intact to protect the thecal sac, and the traversing and exiting nerve roots. Bone shavings were collected for use as the autograft. Curettes and endplate shavers were used for the discectomy, with care to directly visualize the anterior annulus. In this study, either a straight or “bullet” cage or an articulating or “banana” cage was used. Our straight cages expanded after bone graft was packed whereas our articulating cages allowed for packing of bone graft after cage expansion. After endplate decortication, bone aspirate concentrate and allograft were packed into the anterior disc space. Then, an inserter was used to place a packed expandable interbody device into the disc space, with care to place the cage in the anterior annulus. Depending on the device type, allograft was also packed into the cage after expansion. After final rod placement using pre-contoured lordotic rods, both reduction of spondylolisthesis and posterior compression of the screw-rod construct were performed. In this process, we used reduction screws and “broke” the ProAxis bed from flexion to extension. The wound was then closed in a standard fashion.
Outcome Measures
Patient reported outcome (PRO) measures included the Oswestry Disability Index (ODI– normalized to 100%) and the Numeric Rating Scale (NRS-BP/10) for low back pain. PROs and serial radiographs were obtained preoperatively, and at approximately postoperative day 1, 6 weeks, 3 months, 6 months, 1 and 2-year routine follow-ups. Two independent reviewers blinded to patients’ outcomes assessed radiographic images.
Sagittal segmental parameters included segmental lordosis (SL), disc angle (DA), 23 anterior disc height (ADH), posterior disc height (PDH), and percent spondylolisthesis. Cage position was determined by the center point ratio (CPR), 24 and cage subsidence was defined as migration greater than 2-mm (Supplemental Figure 1). 25 Sagittal spinopelvic parameters were overall lumbar lordosis (LL), PI-LL mismatch, sacral slope (SS), and pelvic tilt (PT). Change in radiographic alignment was calculated by subtracting the postoperative from the preoperative measurement. Based on the minimal detectable change (MDC), 26 surgeries were considered to be “lordosing” if postoperative induction of segmental or overall lordosis was ≥ +4° and “kyphosing” if ≤ -4°. 26
Statistical Analysis
Descriptive data were analyzed via univariate analyses. In our series, missing radiographic data (<5%) were imputed using observed radiographic measurements based on predictive mean matching.27-30 We used 5 imputed datasets and pooled results according to Rubin’s rules.31,32 Pearson correlations were used to evaluate the association of preoperative radiographic measures with ΔSL or ΔLL. Variables that had a P < .1 were entered into multivariable linear regression models, with P < .05 used as the threshold for retention. The intraclass correlation coefficient (ICC) was calculated for a subset of 10% of the radiographs to assess the inter-rater reliability. All statistical analyses were performed in R, version 4.1.4.
Results
A total of 73 patients were included in this study with a mean follow-up of 22.5 months (range 12-61 months). The male:female ratio was 40:32 (55/44%) and 44 (60%) patients had an ASA score ≤2. The majority of patients underwent L4-5 level surgeries (n = 57, 68%). All patients experienced significant improvements in ODI (29% ± 23% improvement, P < .001) and NRS-BP (3.3 ± 3 point improvement, P < .001) at latest follow-up (Figure 1). One patient experienced a postoperative epidural hematoma that was evacuated and was subsequently discharged home in good condition. Eight (12%) patients underwent reoperation for pseudarthrosis (n = 1), adjacent segment disease (n = 5), or hardware failure (n = 2). There was no difference in pseudarthrosis rates requiring revision surgery between straight and articulating cages (P = 1). Cage subsidence was observed in 22 (30%) cases, two of which were grade 1 (25-50% loss of postoperative disc height).
33
Complete descriptive statistics and surgical details for the overall series are shown in Table 1. The ICC revealed “excellent” agreement in measuring radiographic parameters (all >.90). Longitudinal data describing patient-reported outcome (PRO) measures obtained preoperatively and at latest follow-up after MI-TLIF surgery. The smooth lines represent mean PRO scores with error bars representing SD in both directions. All MI-TLIF patients experienced significant improvements in ODI (A) and NRS-BP (B) at >1-year follow up compared to baseline status (***P < .001). ODI: Oswestry Disability Index for physical disability (/100%); NRS: Numeric Rating Scale for low back pain (/10). Patient Demographics, clinical, and surgical Characteristics. *ODI = Oswestry disability index. defined as migration greater than 2-mm; ASD: Adjacent segment disease.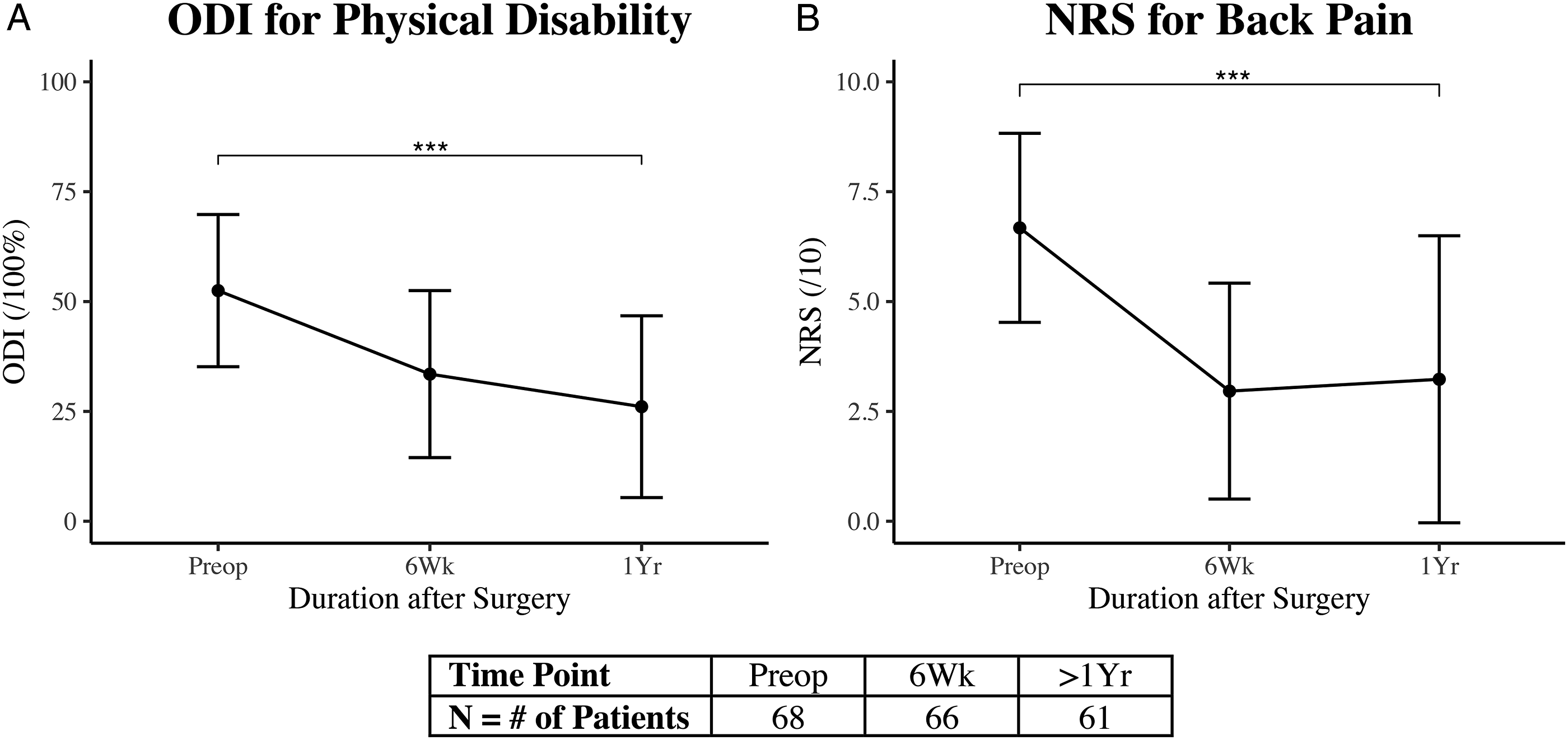

Index-Level Segmental Parameters
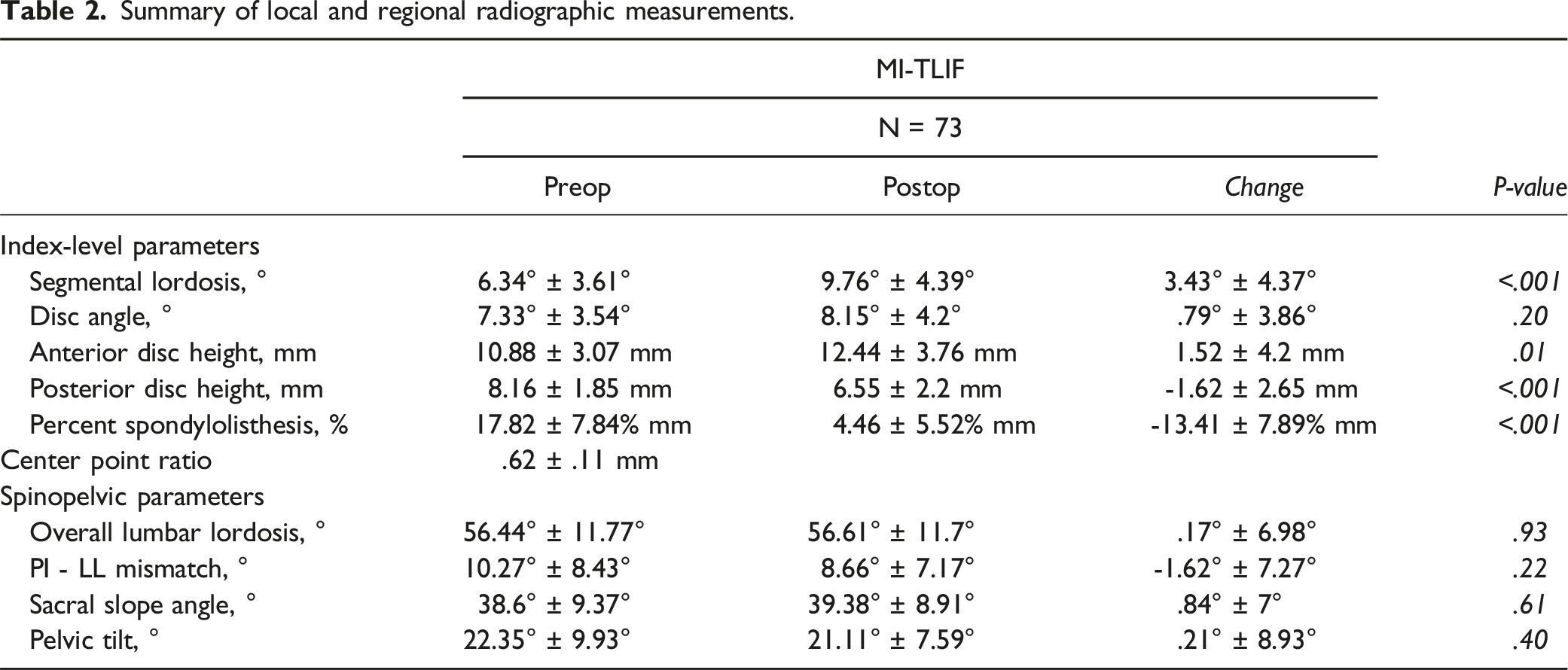
Summary of local and regional radiographic measurements.
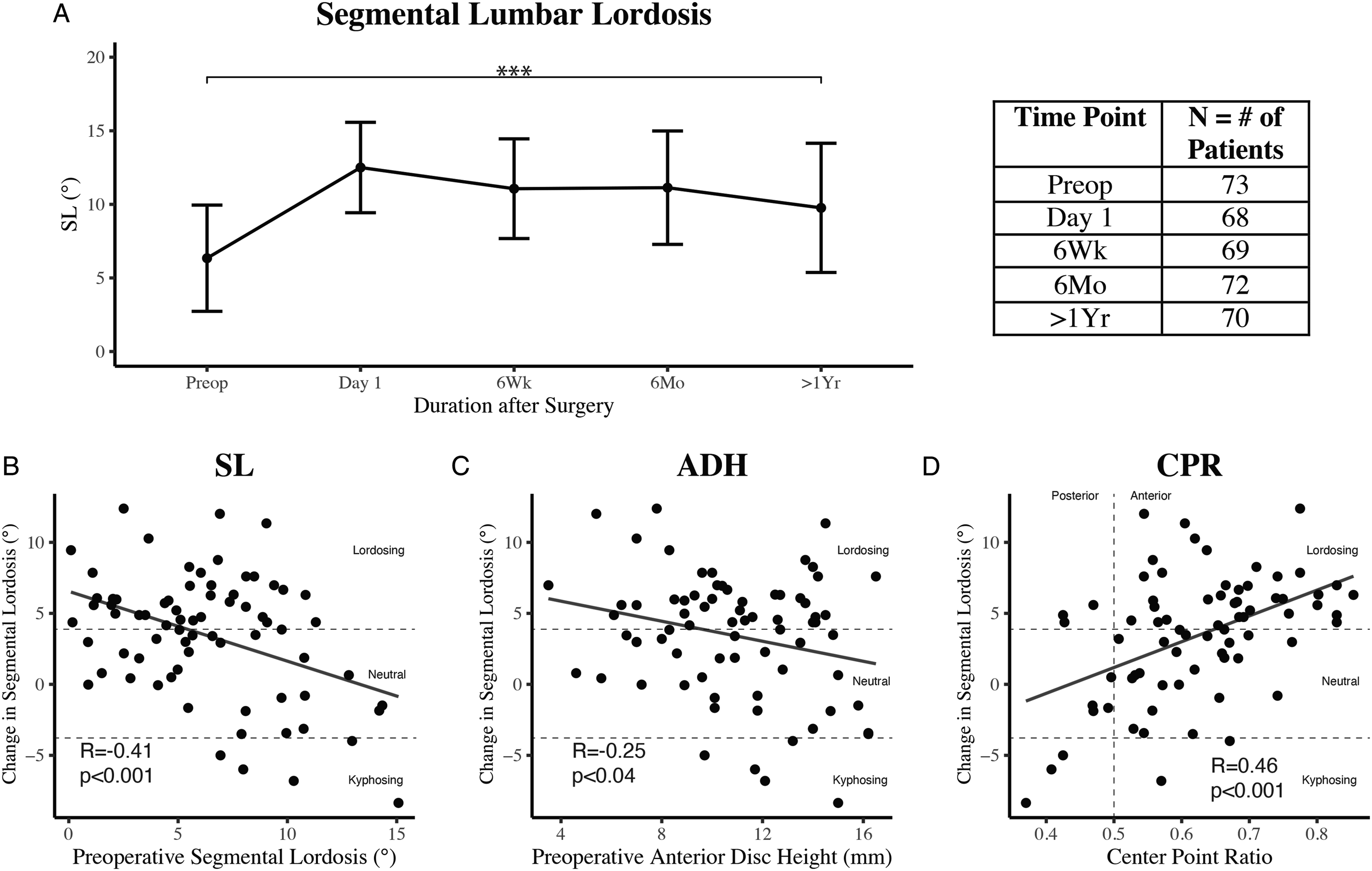
(A) Longitudinal data describing segmental lordosis obtained preoperatively and during routine postoperative clinic visits following MI-TLIF. Smooth lines represent the mean lordotic angle (°) with error bars representing SD in both directions. On average, MI-TLIF patients experienced significant improvements in segmental lordosis (***P < .001) at >1-year follow up compared to baseline status. Preoperative segmental lordosis (B), anterior disc height (C), and center point ratio (D) were significantly correlated with change in segmental lordosis at last follow up, with ordinary least squares (OLS) regression lines shown. Horizontal dashed lines delineate lordosing, neutral, and kyphosing MI-TLIF according to the minimal detectable change (MDC) associated with Cobb’s lumbar lordosis (4°). The vertical dashed line (D) refers to a center point ratio of .5, indicating midline cage placement.
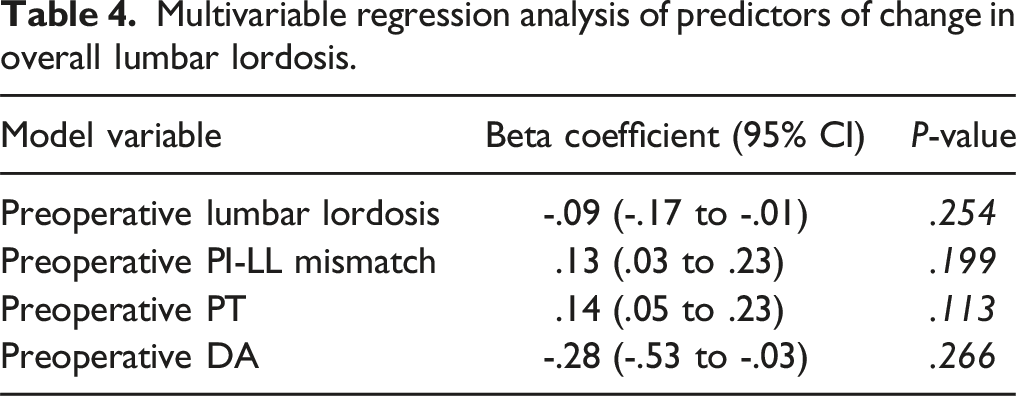
Multivariable regression analysis of predictors of change in segmental lordosis.

Overall Lumbar Lordosis and Pelvic Parameters
When assessing overall lumbar lordosis and pelvic parameters, sixty-nine (95%) patients were included at minimum 1-year follow-up. Mean baseline LL, PI-LL mismatch, SS, and PT were 56.44° ± 11.77°, 10.27° ± 8.43°, 38.6° ± 9.37°, and 22.35° ± 9.93° respectively, and were not different at latest follow-up (Figure 3(A), Table 2, P > .05). When assessing change in LL, 20 (27%) patients experienced lordosing, 22 (30%) kyphosing, and 31 (43%) neutral TLIFs (Supplemental Table 1). On bivariate analyses, preoperative LL (r = -.31, P = .008), PI-LL mismatch (r = .32, P = .005), PT (r = .30, P = .009), and DA (r = -.25, P = .03), were significantly correlated with ΔLL (Figure 3(B)–(E)). Level treated, cage type, and cage subsidence were not associated with ΔLL (P > .05). However, on multivariable regression analyses, no variables were significantly associated with ΔLL (Table 4). (A) Longitudinal data describing overall lumbar lordosis obtained preoperatively and during routine postoperative clinic visits following MI-TLIF. Smooth lines represent the mean lordotic angle (°) with error bars representing SD in both directions. Overall lumbar lordosis remained stable at >1-year follow. Preoperative overall lumbar lordosis (B), PI-LL mismatch (C), pelvic tilt (D) Multivariable regression analysis of predictors of change in overall lumbar lordosis.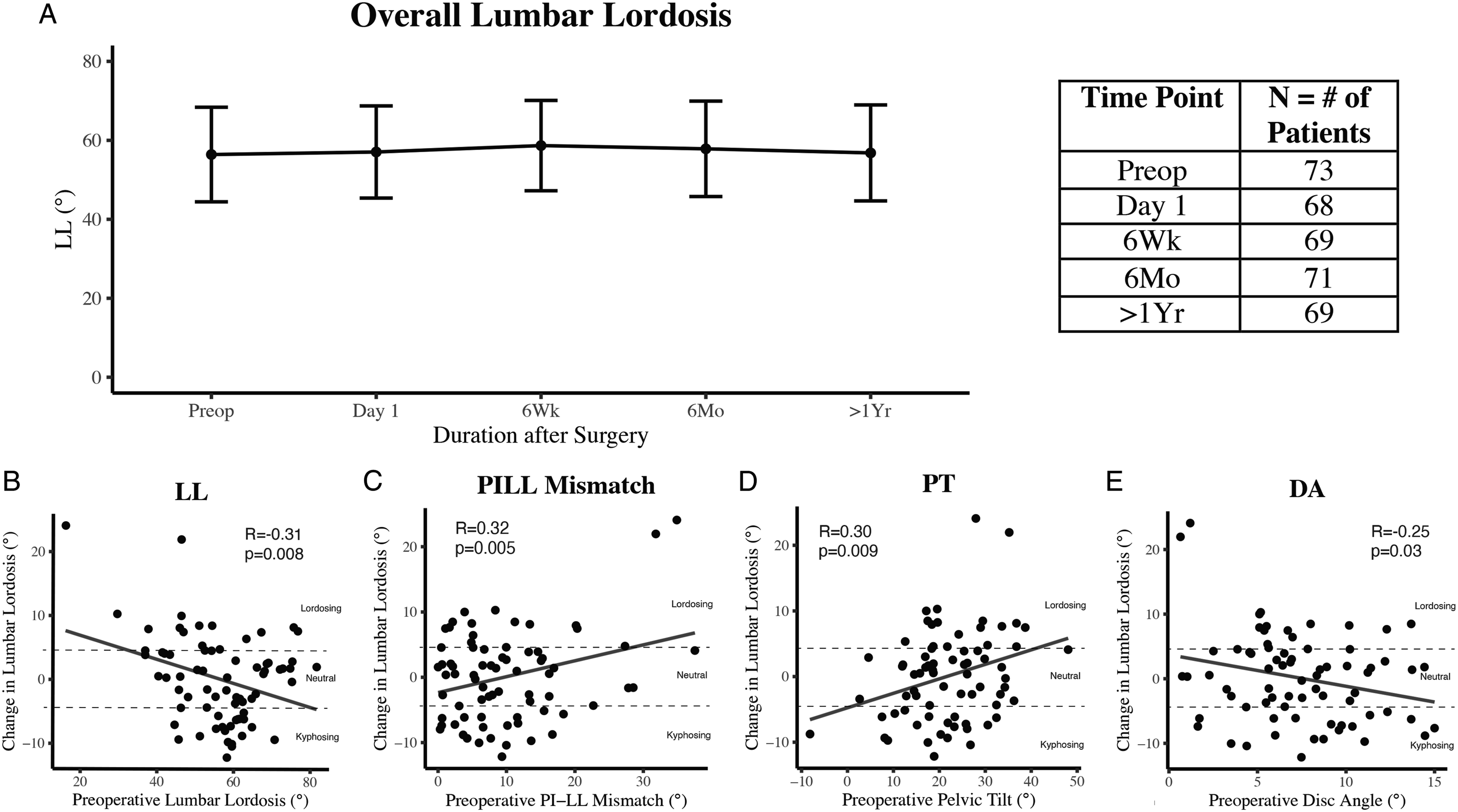
Stratification of Outcomes by Lordotic Group
Summary of clinical and radiographic outcomes stratified by ΔSL group.
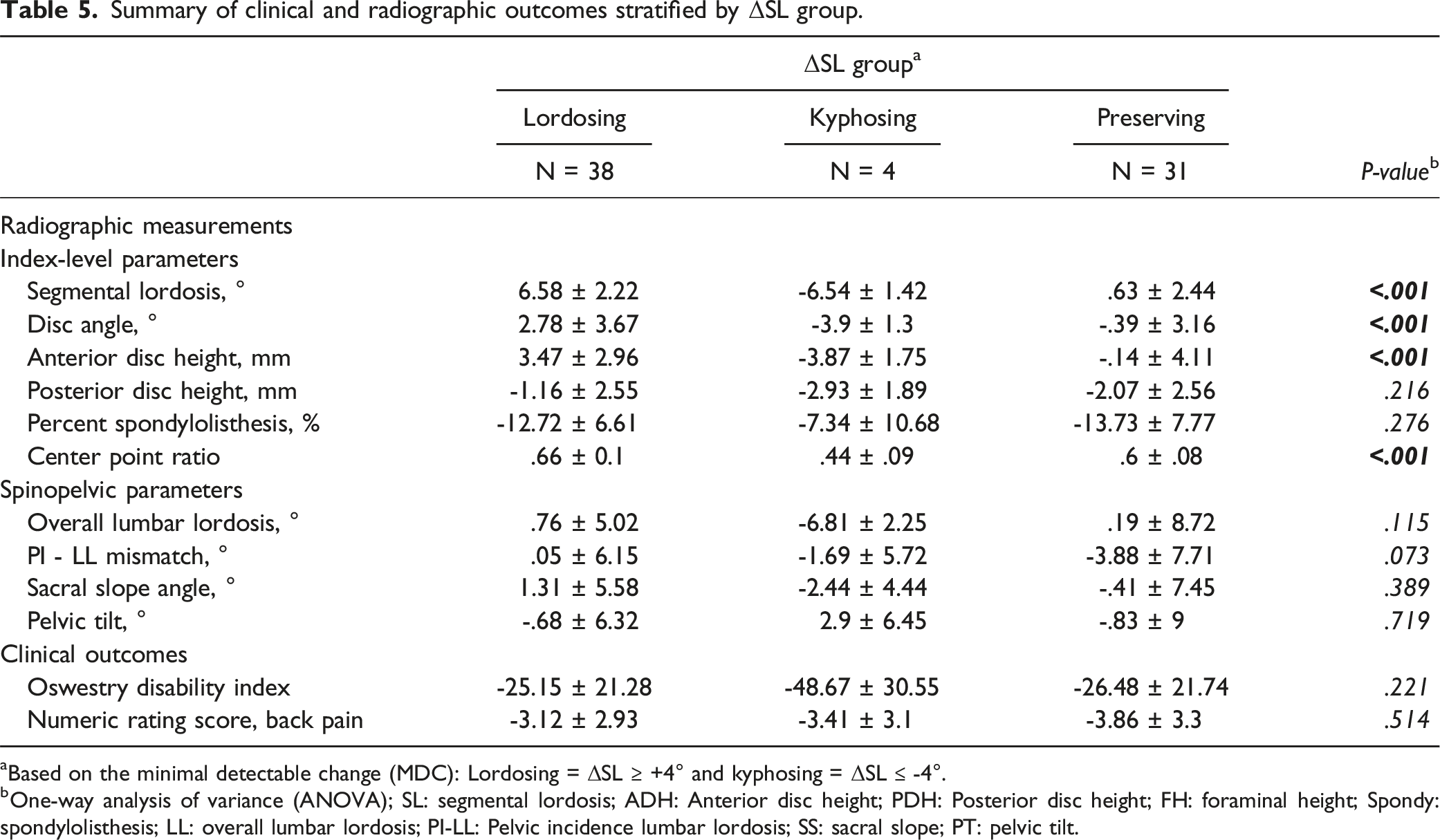
aBased on the minimal detectable change (MDC): Lordosing = ΔSL ≥ +4° and kyphosing = ΔSL ≤ -4°.
bOne-way analysis of variance (ANOVA); SL: segmental lordosis; ADH: Anterior disc height; PDH: Posterior disc height; FH: foraminal height; Spondy: spondylolisthesis; LL: overall lumbar lordosis; PI-LL: Pelvic incidence lumbar lordosis; SS: sacral slope; PT: pelvic tilt.
While multivariable regression results from the multiple imputed datasets are reported, as a sensitivity test, all analyses were also performed in the complete-case dataset and documented in Supplemental Tables 2 and 3.
Discussion
MI-TLIF is a useful technique to address degenerative lumbar etiologies; however, its ability to induce lordosis remains controversial, with several studies reporting MI-TLIF to be a kyphosis-inducing operation.14,34 Multiple practices have been developed to combat these deficiencies, from lordotic operative tables 35 to advancements in medical devices36-38 and surgical techniques. 17
However, there is significant heterogeneity in studies investigating restoration of lordosis following MI-TLIF. 39 Inclusion of single-level and multi-segment procedures in the same study prevents accurate conclusions regarding SL and LL outcomes. Variation in interbody devices and surgical techniques further confound findings. 39 In addition, many studies lack long-term follow up, and therefore, fail to account for cage settling or subsidence. 9 Given this evidence gap, we aimed to evaluate the impact of MI-TLIF on segmental and overall lumbar lordosis, with all other factors, including levels treated, surgical technique, and cage type, remaining constant.
Predictors of Lordosis in MI-TLIF
Consistent with previous studies evaluating single-level MI-TLIF, we found preoperative spine habitus to be significantly associated with radiographic outcomes.16,40 In a cohort of 171 single-level MI-TLIF patients, Vaishnav et al 40 demonstrated that patients with low preoperative SL (<15°) experienced the greatest correction in SL regardless of cage type. Regarding regional alignment, Khalifeh et al 16 reported in a cohort of 68 patients undergoing transfacet MI-TLIF that patients with hypolordotic spine types (LL <40°) experienced the greatest increases in SL and LL. Specifically, in our series, baseline SL predicted ΔSL at long-term follow-up, a finding that persisted on multivariable regression analysis. This finding may be attributable to the fact that hypolordotic spine types possess a higher propensity for radiographic improvement compared to hyperlordotic patients, who are at or near maximal lordosis.5,9
In addition to patient-specific factors (e.g., radiographic habitus), interbody devices can also have significant implications on postoperative lordosis. Interbody device technology makes MI-TLIF possible, as narrowed working corridors prevent placement of large cages.36,38 In our study, we found anterior cage positioning to be significantly associated with ΔSL, which was the strongest relationship in multivariable analyses.1,41-43 Our series saw 63 (86%) devices placed anteriorly (CPR >.5), with a mean CPR of .62 ± .11. Such placement maximizes the anterior pivot effect of the cage, lengthening the anterior column (reflected by ΔADH: +1.52 ± 4.2 mm, P < .001), reducing rod strain, and providing a stiffer construct. 43 This finding may also partly explain why our series experienced a modest, yet significant decrease in PDH (Δ-1.62 ± 2.65 mm, P < .001). However, this decrease in PDH was not associated with new-onset neurological deficits in our series, a finding that is further corroborated by steady improvements in ODI and NRS (Figure 1).
In addition to its effect on lordosis, cage placement onto the cortical load-bearing anterior hypophyseal ring has shown to reduce the risk of cage subsidence. 44 In the MI-TLIF literature, cage subsidence has shown to decrease disc height and lordosis, potentially preventing fusion and worsening outcomes. 45 A variety of risk factors of cage subsidence have been identified, ranging from low bone mineral density 46 to surgical technique, 25 with mixed evidence regarding the risk of cage subsidence in expandable vs static cages.36,37,47 At our institution, recognizing the risk of expandable cages due to an increased axial compression load at the cage-endplate interface, 44 we take care in ensuring anterior placement of our interbody device onto the cortical load-bearing fibrous anterior hypophyseal ring. In our series, we suggest that anterior cage positioning may have reduced the risk of cage subsidence, thereby limiting potential loss of postoperative lordosis. Ultimately, we found that the impact of cage subsidence at long-term follow-up was not substantial enough to have a major effect on ΔSL or ΔLL.
In comparison to ΔSL and consistent with existing literature, our patients did not experience significant changes in overall lumbar lordosis at latest follow-up (ΔLL: .17° ± 6.98°, Figure 3(A)).19,21 However, the neutral effects on overall lumbar lordosis were expected, as patients in our series underwent solely single-level surgery without osteotomies. These results suggest that adjacent unfused segments compensated for the modest changes in segmental lordosis to produce a stable overall alignment.48,49 Though many preoperative radiographic parameters correlated with ΔLL (Figure 3(B)–(E)), none persisted on multivariable analysis (Table 4). Finally, we found multivariable regression results to be identical between the multiple imputation analysis (Tables 3 and 4) and complete case analysis (Supplemental Tables 2 and 3), further demonstrating the durability of these findings.
Important to note, lordosis in the lumbar spine is not homogenously distributed.50,51 In order to account for this potential confounder of postoperative lordosis, a subgroup analysis was performed comparing ΔSL and ΔLL between levels treated. Specifically, surgeries were binned into those undergoing L4-5 (n = 57, 78%) or L5-S1 (n = 1, 1%) vs L2-3 (n = 7, 10%) or L3-4 (n = 8, 11%). For ΔSL, the L4-5 or L5-S1 group improved by 3.41° ± 4.43° whereas the L2-3 or L3-4 group improved by 3.19° ± 4.5° (P = .87). For ΔLL, the L4-5 or L5-S1 group improved by .63° ± 7.08° whereas the L2-3 or L3-4 group decreased by -2.08° ± 5.98° (P = .16). In addition, the level treated was not a significant predictor of ΔSL or ΔLL on multivariable regression. Therefore, while lumbar lordosis is not evenly distributed throughout the lumbar spine, the level treated in our study did not significantly impact our results.
Findings in Context
Though existing studies have assessed the effect of MI-TLIF on sagittal alignment,9,19,39,40,52 heterogenous patient populations limit robust assessments of radiographic predictors of segmental and global lordosis. Specifically, inclusion of multi-level surgery, variation in facetectomy technique, and use of differing cage technology may confound analyses. Consistent with previous studies,40,43 we reinforce the importance of preoperative patient selection and operative technique on sagittal alignment following MI-TLIF. Our long-term follow-up (mean: 22.5 months) and multivariable analyses controlling for multiple confounders, including the effects of cage subsidence, further demonstrate the durability of our findings.
Similar to previous reports, the majority of patients in our series experienced lordosing MI-TLIFs when defined using the MDC (4°) of Cobb’s lumbar lordosis 26 (n = 38, 52%) or as ΔSL >0° (n = 54, 74%) as described in prior studies. 9 We suggest that the increases in SL seen in our case may be attributed to the combined effects of expandable cage technology, cage positioning, use of an advanced adjustable operating bed, and maintenance of the posterior tension band.
As opposed to open TLIF, the posterior ligamentous complex
53
remains intact in MI-TLIF. Maintenance of these structures limits expansion of the posterior vertebra, which can be leveraged to maximize restoration of segmental lordosis. Under these circumstances (i.e., mobile anterior disc and relatively fixed posterior disc), cage positioning becomes paramount. Specifically, anterior placement of the interbody device can maximize the construct’s “wedge” effect, promoting segmental lordosis (Figure 4(A)). Conversely, if the cage is placed too posteriorly, the superior vertebral body may hinge over the device, potentially kyphosing the local segment (Figure 4(B)). Indeed, we found cage positioning to be the strongest predictor of ΔSL, with every .1 increase in CPR predicting a 1.5° increase in SL (Table 3). Illustration demonstrating the effect of expandable cage positioning on segmental lordosis in single-level MI-TLIF treated with unilateral facetectomy and expandable interbody devices. 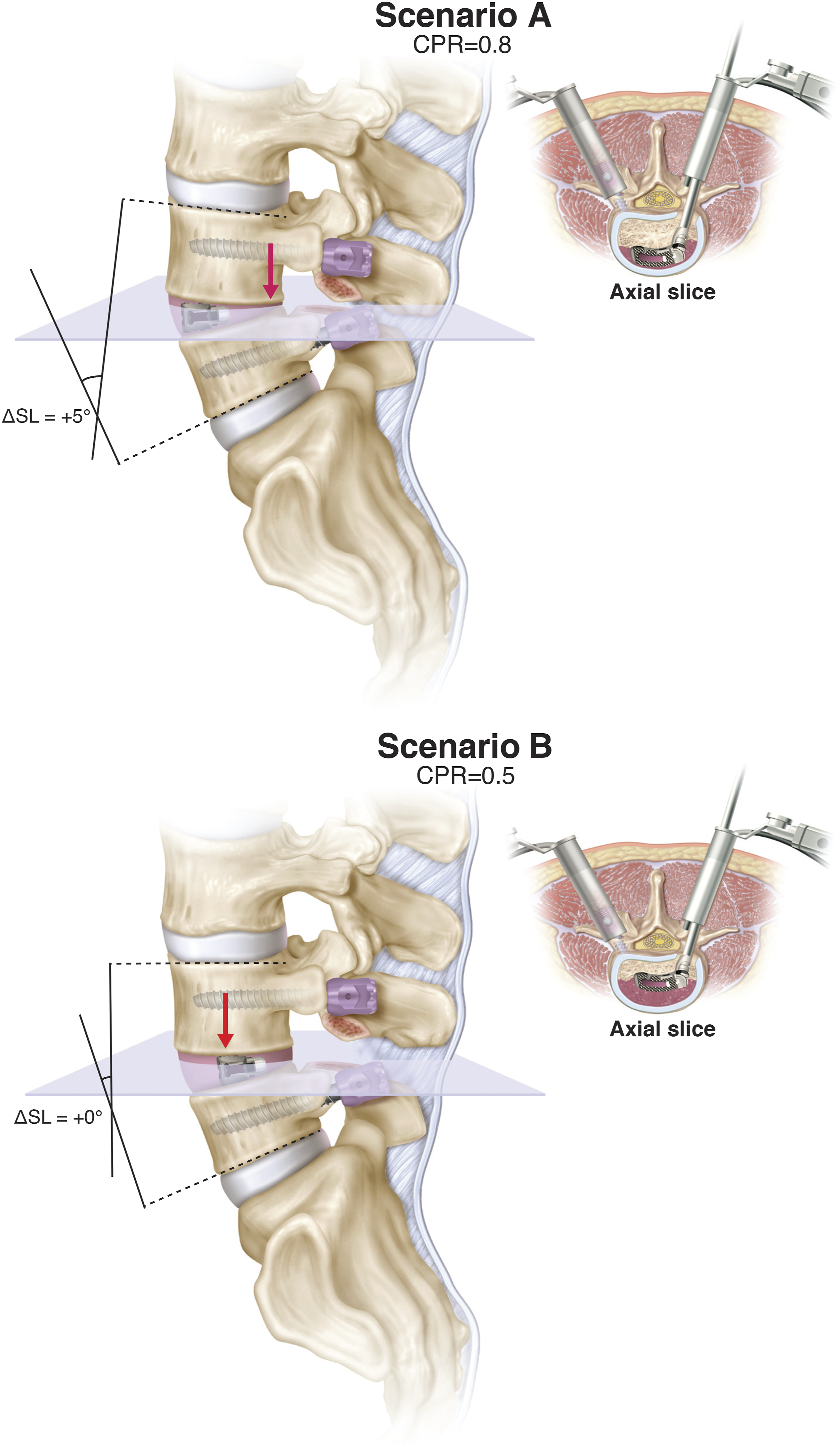
This phenomenon may be further augmented through the use of expandable interbody devices, which are increasingly utilized during MI-TLIF to address technical limitations of an minimally-invasive approach. 38 In our series, 61 (84%) of the expandable interbody devices were articulating “banana” cages and 12 (16%) were straight “bullet” devices. Developed more recently to combat the alleged kyphosing nature of TLIFs, articulating “banana” cages theoretically provide greater index-level lordosis through their improved ability to articulate along the anterior apophyseal ring. 49 As compared to a recent study by Toop et al, 49 our series found no difference between articulating (ΔSL: 3.23° ± 4.39°) and straight cages (ΔSL: 3.18° ± 4.36°) (P = .97). This may be due to our unequal distribution of cage types, smaller sample size, or other patient-specific factors, and therefore, should be further evaluated in future studies.
Though expandable cages may theoretically provide larger increases in lordosis than non-expandable cages, complications such as cage subsidence and cage migration can nullify these potential advantages. 37 Although our study was unable to compare static and expandable cages, there is a strong body of literature that suggests that static cages can likely achieve similar lordotic outcomes in single-level MI-TLIF. In comparative studies assessing lordotic outcomes between static and expandable cages, the evidence remains mixed.21,38,54 In a meta-analysis comparing static and expandable MI-TLIF patients, Ali Alvi et al 36 demonstrated the superior ability of expandable cages to promote segmental lordosis compared to static cages, with neutral effects on overall lumbar lordosis. Taken together, the literature suggests that both static and expandable cages are capable of providing similar restoration of lordosis after single-level MI-TLIF. The variation in results in existing literature implies that interbody device type alone is not the primary determinant factor of lordotic outcomes, but rather a combination of operative technique, interbody devices, and surgical adjuncts.
Furthermore, all patients in our series utilized an open-frame hinged surgical table. In our institution, we utilize a flexed position to support decompression, instrumentation, and interbody device placement, followed by an extended position to fix the patient in a lordotic alignment during rod insertion.21,35 Although their true efficacy has not been established, these adjustable operative tables may have further contributed to the gains in segmental lordosis seen in our series.
Surgical technique has also been associated with corrections in lordosis.9,17 Specifically, though the evidence is mixed, bilateral facetectomies have been suggested to promote greater correction in lordosis.1,9,17,22 Biomechanically, bilateral facetectomies theoretically allows for greater compression across the instrumented vertebra to promote lordosis, with support from recent cadaveric studies. 55 However, in a study of 57 patients undergoing open TLIF with either unilateral or bilateral facetectomy, Tye et al 56 found no difference in SL correction at 1-year follow-up. In a more recent study in a cohort of 40 MI-TLIF patients, Archer et al 22 also did not find significant differences in SL when comparing unilateral and bilateral facetectomies. The conflicting evidence may suggest inconsistencies in surgeon technique, including the size and extent of the facetectomy, as well as confounding from other variables (e.g., cage type, operative bed, cage positioning, extent of posterior compression). In addition, in MI-TLIF, maintenance of the posterior ligamentous complex may interfere with posterior compression of the screw-rod construct.
Though bilateral facetectomies may have increased our lordotic potential, our study only included unilateral facetectomies with contralateral facet release, which allowed for a homogenous series of patients. Furthermore, our results suggest that advanced interbody device technology (i.e., expandable “banana” cages), anterior cage placement, ancillary surgical adjuncts (e.g., modulating ProAxis table), and careful patient selection are still contributors to improvements in lordosis, independent of facetectomy technique. Our long-term follow up (mean: 22.5 months) coupled with our low incidence of reoperations (11%) further demonstrate the stability of our results.
Our findings emphasize that predictors of alignment correction are multifactorial and cannot be predicted solely based on the type of interbody fusion procedure performed.5,49 However, they are meaningful as they may enable surgeons to have greater accuracy in their preoperative plan. In a narrative review of various types of minimally invasive interbody procedures, Uribe et al 5 makes the distinction between alignment ‘preservation’ vs ‘restoration’ following interbody fusion. Taken together, our results suggest that single-level MI-TLIF, under certain situations, is able to restore segmental lordosis. More specifically, it can maintain alignment without exacerbating underlying kyphosis. Therefore, if a surgeon aims on treating single-level degenerative lumbar disease through MI-TLIF, careful assessment of the patient’s preoperative radiographic habitus and attention to cage placement intraoperatively may maximize the ability to adequately restore SL.
In addition, our results suggest that single-level MI-TLIF, under our study conditions, has a neutral effect on overall lumbar lordosis and is generally sufficient to preserve global lordosis. As reflected through our results, in the setting of single-level surgery, global sagittal alignment (e.g., PI-LL mismatch) and clinical outcomes were not significantly different among lordosing, kyphosing, and preserving subgroups defined using ΔSL (Table 5). As expected, changes in certain index-level radiographic parameters (e.g., SL, DA, ADH) were significantly greater in lordosing patients (P < .001). This is likely in part due to significantly greater CPR values in lordosing patients (P < .001) indicating more anterior cage placement, promoting a “wedge” effect. Important to note, however, the gains in lordosis seen in our cohort were still modest. Specifically, a mean improvement of 4° may not translate into meaningful sagittal correction and lordotic gains should be interpreted in the context of the patient’s overall alignment and needs for lordosis. Consequently, if restoration of overall lumbar lordosis is the goal of surgery, surgeons may need to perform a different interbody fusion approach, such as an anterior lumbar interbody fusion (ALIF), 14 or consider use of alignment-correcting osteotomy techniques.
Limitations
Limitations of this study include its single-center design and retrospective nature. In addition, given that all patients underwent the same surgery (i.e., all single-level MI-TLIF performed on an open-frame hinged surgical table with unilateral facetectomies and a contralateral facet release with expandable cages), we were unable to assess the individual contribution of operative technique (i.e., modular operative table, cage placement, facetectomy type, instrumentation technique) or interbody device types (e.g., expandable vs static cages). Future studies comparing these domains while keeping other variables constant are critical to assessing the contributory effect of factors influencing lordosis.
Furthermore, our study includes two types of expandable interbody devices. One subtype, our straight “bullet” cage, does not allow for packing of bone graft after expansion, and therefore, has a theoretical increased risk for pseudarthrosis because pre-placed bone graft is less tightly packed. However, given that our study cohort only had one case of pseudarthrosis requiring revision surgery, we were not able to compare arthrodesis success between these cage types. Moreover, patients in our cohort experienced significant reductions in spondylolisthesis, similar to previous studies assessing radiographic outcomes following single-level MI-TLIF.57,58 Although our study focused on lordotic outcomes following MI-TLIF, future studies should examine predictors of other radiographic outcomes, such as reduction of spondylolisthesis. Last, though we included two independent reviewers, some of our findings are within the measurement error for Cobb’s lordosis 26 and may have limited clinical translations.
Conclusions
Given conflicting reports describing sagittal correction following MI-TLIF, we examine a homogenous, consecutive series of single-level MI-TLIF patients undergoing unilateral facetectomies and a contralateral facet release with expandable cages. In our series, the majority of patients experienced restoration of segmental lordosis (52%, n = 38), with neutral effects on overall alignment. At latest follow-up, preoperative segmental lordosis and cage position were significant predictors of ΔSL, which remained significant when including potential confounders on multivariable regression. Our results suggest that in order to maximize lordotic potential in single-level MI-TLIF approaches, surgeons should carefully select patients based on preoperative spine habitus and maximize anterior cage placement intraoperatively.
Supplemental Material
Supplemental Material - Predictors of Postoperative Segmental and Overall Lumbar Lordosis in Minimally Invasive Transforaminal Lumbar Interbody Fusion: A Consecutive Case Series
Supplemental Material for Predictors of Postoperative Segmental and Overall Lumbar Lordosis in Minimally Invasive Transforaminal Lumbar Interbody Fusion: A Consecutive Case Series by Justin K. Zhang, Jacob K. Greenberg, Saad Javeed, Braeden Benedict, Kathleen S. Botterbush, Christopher F. Dibble, Jawad M. Khalifeh, Samuel Brehm, Deeptee Jain, Ian Dorward, Paul Santiago, Camilo Molina, Brenton H. Pennicooke and Wilson Z. Ray in Global Spine Journal
Footnotes
Acknowledgements
The authors would like to acknowledge Dr Yikyung Park for her biostatistical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
