Abstract
Study design
Systematic review.
Objectives
The aim of this study is to analyze the complications related to multilevel anterior cervical discectomy and fusion (ACDF) using osteobiologics other than bone morphogenetic protein (BMP).
Methods
A systematic review of the literature was conducted using PubMed, EMBASE, Cochrane Library, and ClinicalTrials.gov databases. The search to identify studies reporting complications in multilevel ACDF surgery using osteobiologics other than bone morphogenetic protein was performed in August 2020. The study was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA).
Results
A total of 584 articles were found after searching the databases and removing duplicates. Next, screening was performed in a double reviewer process, and 153 eligible articles—with 4 retrospective studies—in full-text were selected; these met all inclusion criteria. A total of 197 patients received 3-level ACDF, while 72 patients received 4-level ACDF. Osteobiologics were used in all selected articles, allograft was used in 4 studies, autologous bone graft was utilized in 3 articles, and hydroxyapatite was used in 1 article. The main complications reported were dysphagia, adjacent segment disease, and pseudarthrosis.
Conclusions
Given the limited evidence, no conclusions on complications in multilevel ACDF using osteobiologics other than BMP could be made. However, descriptively, the most common complications found were dysphagia, adjacent segment disease, and pseudoarthrosis. Further prospective studies separately analyzing complications in multilevel ACDF by osteobiologics and a number of treated levels are needed.
Introduction
Anterior cervical discectomy and fusion (ACDF) has gained popularity in spine surgery as a validated, simple, and useful technique for a wide variety of cervical spine disorders. 1 When the anterior approach is adopted during direct decompression and fusion for degenerative spine conditions, it is associated with a lower rate of wound complications, readmission, and revisions as compared to posterior approach. 2 Degenerative disc disease can affect various levels of the cervical spine; therefore, multilevel ACDF surgery is indicated.
There is increasing knowledge as well as great variability in the graft used by spine surgeons for ACDF surgery across the world. 3 However, the use of osteobiologics in multilevel ACDF is much less understood and studied; hence, information regarding adverse events related to the use of osteobiologics in context of a more complex procedure with higher expected complication rates is necessary. 4 This is also important because of the increased cost of the procedure and consequent impairment of the patient’s quality of life. 5
The knowledge and evidence available to understand and treat degenerative disc conditions by ACDF using bone morphogenetic protein (BMP) as graft is growing. 6 However, multilevel ACDF with osteobiologics other than BMP in context of degenerative disc disease is remarkably scarce 7 ; therefore, the decision-making with respect to related-cost and morbidity is more challenging and poorly understood.
The aim of the present systematic review was to analyze the spectrum and frequency of complications associated with multilevel ACDF surgery when osteobiologics other than BMP were used. This systematic review is part of a large project of multiple systematic reviews focusing on osteobiologics in spine surgery led by AO Spine Knowledge Forum Degenerative. This systematic review focuses on multilevel ACDF with osteobiologics other than BMP, while other studies investigate BMP and other spine surgeries.
Methods
We performed a systematic review of the literature using PubMed, EMBASE, Cochrane Library, and ClinicalTrials.gov databases. The search was performed in August 2020 to identify studies reporting complications in multilevel ACDF surgery using osteobiologics other than BMP that were published in English. The restriction of the time period from 2000 to 2020 aimed to include only recent spine surgery and patient care techniques, which are updated frequently. No grey literature search outside these databases was conducted. The systematic review was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA). 8
Search Strategy, Inclusion and Exclusion Criteria
PICO search strategy Table for the systematic review.

Abbreviations: BMP, Bone morphogenetic protein; ACDF, Anterior cervical discectomy and fusion; RCT, Randomized controlled trial; SCI, Spinal cord injury.
Complications Included
We defined the reported complications in 4 main groups as follows: peri-operative surgical complications, mid or long-term outcomes, medical complications, or other.
Peri-Operative Surgical Complications
Neurologic deterioration ≥1 motor grade as per the ASIA motor scale, new postoperative radiculitis or neuropathic pain, wound dehiscence, superficial wound infection, deep wound infection, airway compromise requiring surgical intervention, dysphagia, dysphonia/hoarseness of voice, anterior cervical hematoma/seroma, epidural hematoma/seroma, cerebrospinal fluid leak/meningocele, vertebral artery Injury, esophageal perforation, Horner´s Syndrome, peri-operative death.
Mid- or Long-Term Complications
Non-union, construct failure with loss of correction (screw pull out, plate dislodgement); construct failure without loss of correction (cage subsidence, screw pull out, plate dislodgement); revision surgery for incomplete neurologic decompression and persistent symptoms.
Medical Complications
Cardiac arrest/failure/arrhythmia, deep vein thrombosis, delirium, gastrointestinal bleeding, myocardial infarction, pneumonia, pressure sores, pulmonary embolism, systemic infection, urinary tract infection.
Other
Complications not included in the previous lists.
List of Osteobiologics Included
Each included study was categorized according to the number of treated levels and the osteobiologics used. The osteobiologics included were: Autologous bone graft, such as autologous iliac crest bone (AICB); Allogenic bone graft; Bone marrow aspirate (BMA); Demineralized bone matrix (DBM); Hydroxyapatite (HA); Platelet-rich plasma (PRP); Ceramic-based bone graft; Mesenchymal stem cells (MSC); Other synthetic materials (Synthetic grafts-ß-TCP, bioglass, etc.).
Assessment of Risk of Bias
Non-randomized studies for comparative and non-comparative studies were assessed with the Methodological Index for Non-randomized Studies (MINORS). 9 The maximum score was 16 for non-comparative studies and 24 for comparative studies.
Results
Screening and Selection Processes
A total of 756 articles were found in the initial search of all 4 employed databases. After the duplicates were removed, 584 articles were screened on the title and abstract level. A total of 153 full-text articles were analyzed with 4 studies meeting the inclusion and exclusion criteria (Figure 1). A detailed search query of PubMed and Embase databases is shown in Supplement 1. PRISMA (preferred reporting items for systematic reviews and meta-analyses) flow diagram for identification and selection of studies.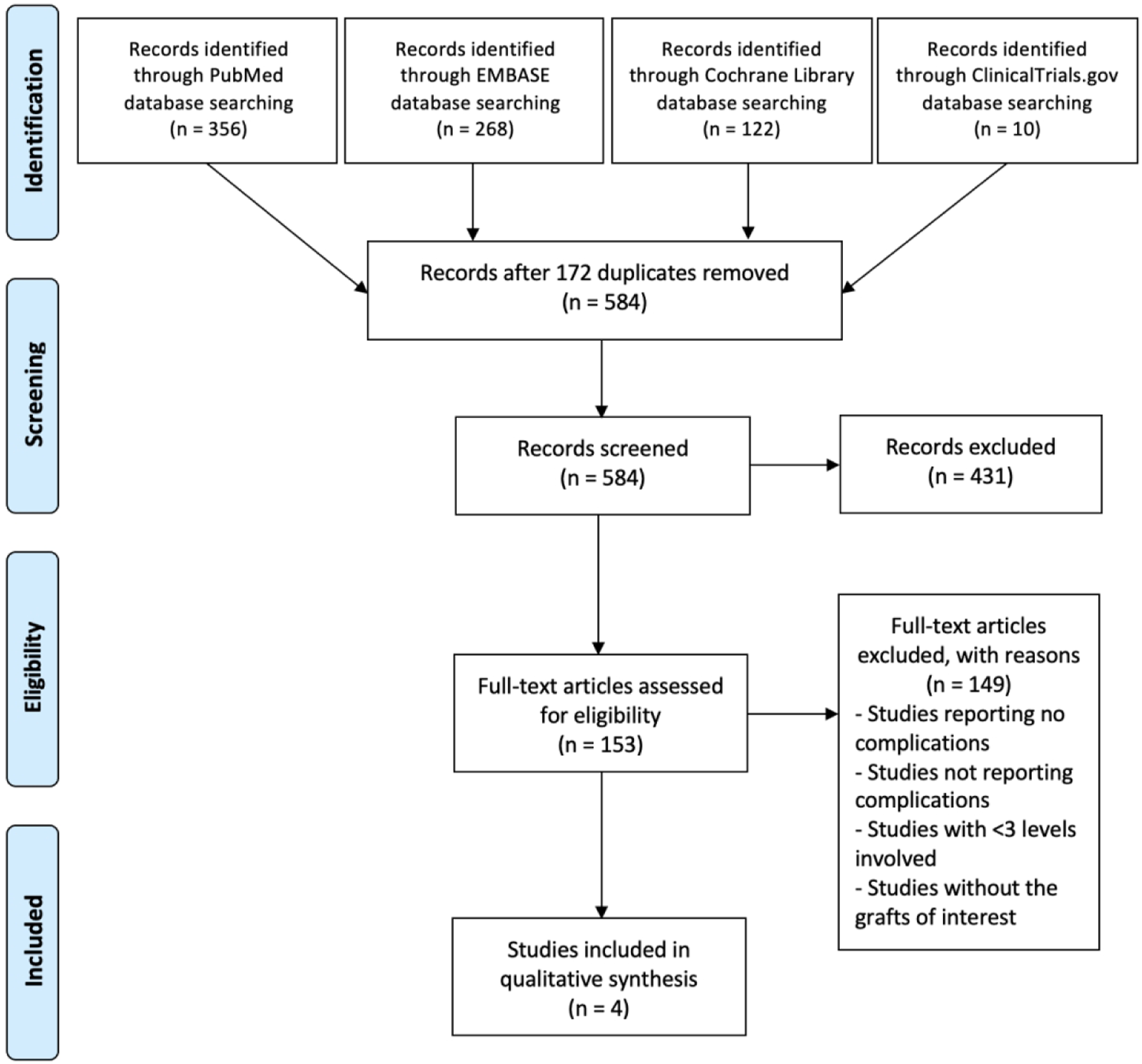
Risk of Bias
Summary of the 4 studies included in our systematic review.
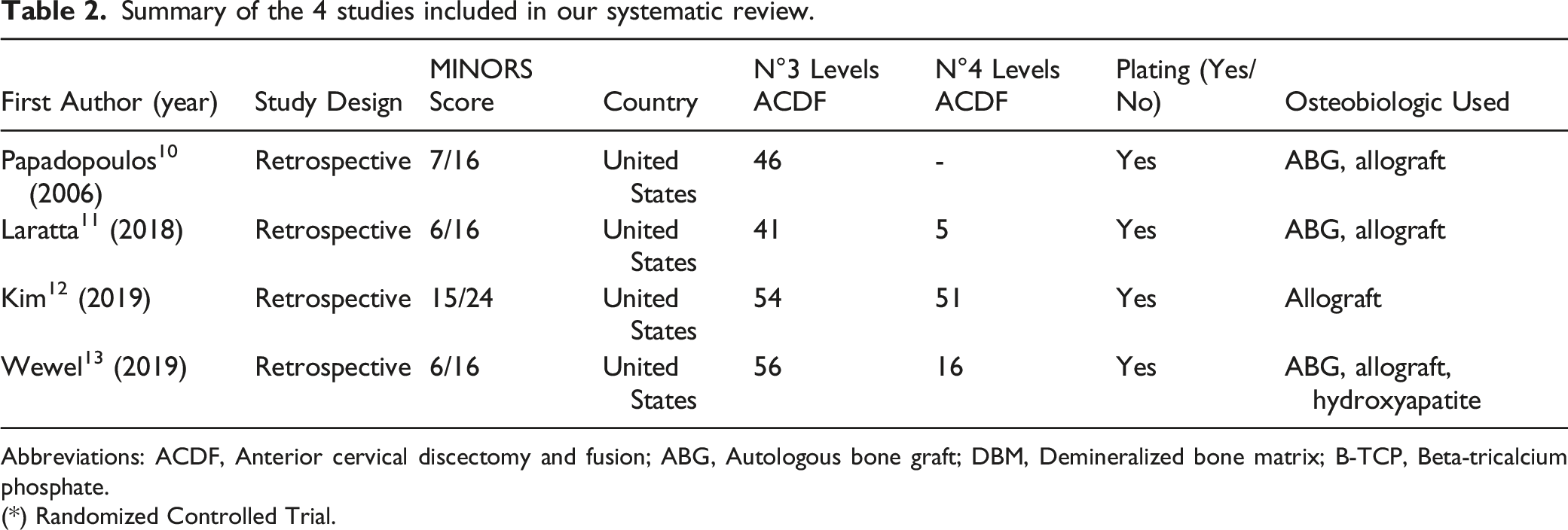
Abbreviations: ACDF, Anterior cervical discectomy and fusion; ABG, Autologous bone graft; DBM, Demineralized bone matrix; B-TCP, Beta-tricalcium phosphate.
(*) Randomized Controlled Trial.
The financial disclosures from all authors of the 4 included articles are shown in Supplement 2.
Data Synthesis
A total of 197 patients received 3-level ACDF while 72 received 4-level ACDF. The osteobiologics used in the selected articles were autologous bone graft in 3 studies, allograft in 4 articles, and hydroxyapatite in 1 article (Table 2). A meta-analysis was not possible due to the heterogeneity in study design, quality of reporting and assessment; therefore, it cannot be pooled.
Postoperative Complications
Analysis of the primary outcomes reported in the 4 studies included.
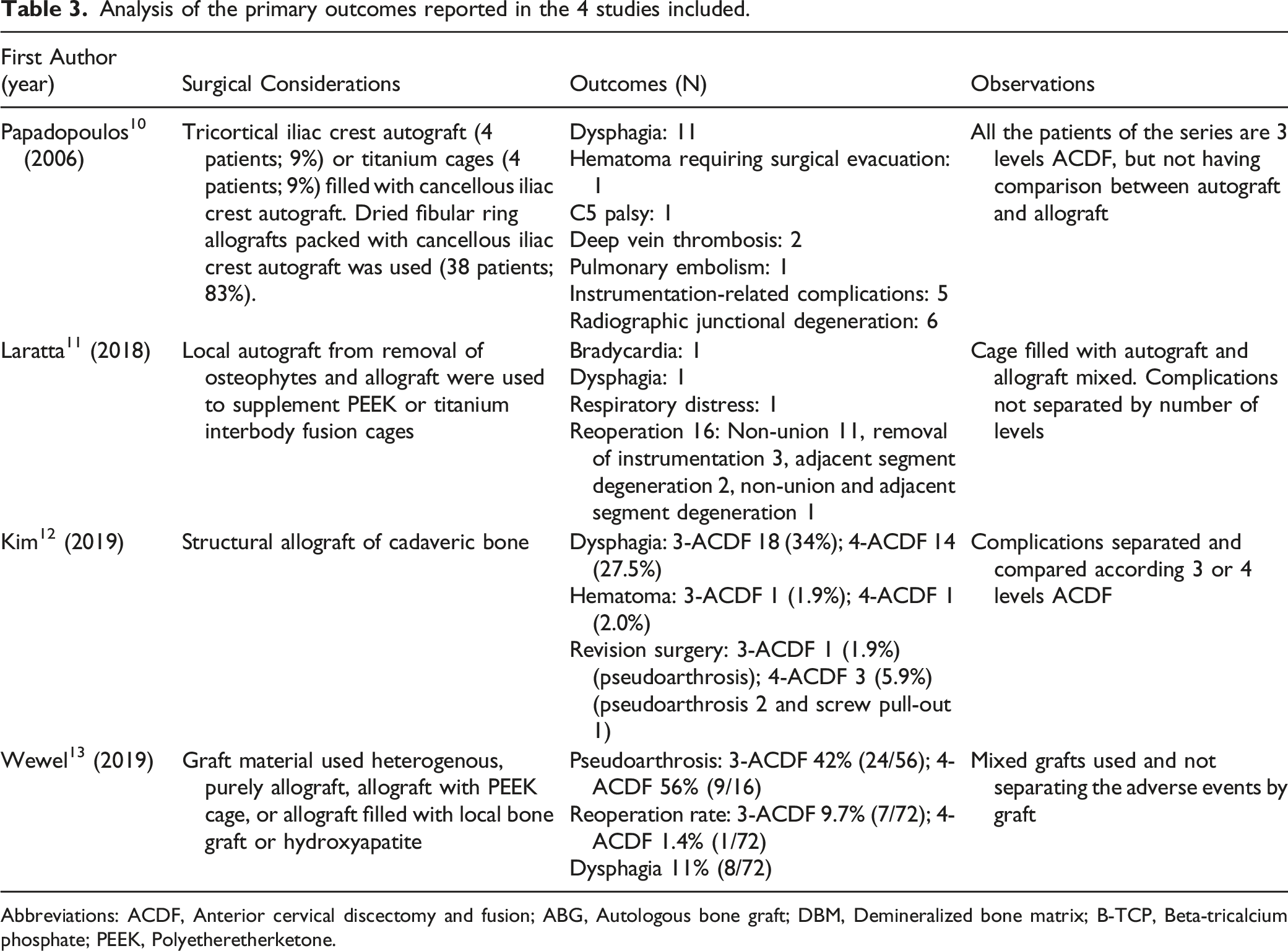
Abbreviations: ACDF, Anterior cervical discectomy and fusion; ABG, Autologous bone graft; DBM, Demineralized bone matrix; B-TCP, Beta-tricalcium phosphate; PEEK, Polyetheretherketone.
Laratta et al 11 reported 41 patients with 3-level ACDF and 5 patients with 4-level ACDF using cages filled with autograft and allograft mixed. Complications occurred in 3 (6.5%) patients: 1 patient had symptomatic bradycardia, 1 patient had postoperative throat swelling, and 1 patient had respiratory distress. Sixteen patients (35%) had a revision surgery within 2 years with 11 of these patients (24%) returning for non-union. Three (7%) patients returned for hardware removal, 2 (4%) patients returned for adjacent segment disease (ASD), and 1 (2%) patient returned for non-union plus ASD.
Kim et al 12 reported separately and compared their experience according to 3- or 4-level ACDF using structural allograft (cadaveric bone) with plating and screws. One patient in each group experienced postoperative hematoma which did not require surgical intervention. Transient dysphagia was the most common peri-operative complication in both groups and affected 34.0% of patients undergoing 3-level ACDF and 27.5% of patients undergoing 4-level ACDF (P = .61). One patient in the 3-level ACDF group and 3 patients in the 4-level ACDF group underwent revision surgery (1.9% for 3-level vs 5.9% for 4-level, P > .05). Revision surgery in 3-level ACDF was performed in 1 patient for pseudoarthrosis, while revision surgery for pseudoarthrosis was done 2 patients and screw pull-out in 1 patient in 4-level ACDF.
Wewel et al, 13 in their experience, included 56 patients with 3-level ACDF and 16 patients with 4-level ACDF using allograft consisting purely of allograft, allograft with PEEK cage, or allograft filled with local bone graft or hydroxyapatite. Pseudoarthrosis was seen in 56% (9/16) in 3-level ACDF and 42% (24/56) in 4-level ACDF. Eight patients were symptomatic and required posterior spinal instrumentation for a total reoperation rate of 11.1% (8/72). Seven patients initially had a 3-level anterior fusion resulting in a total re-operation rate of 9.7% (7/72), 1 patient in 4-level fusion resulted in a total reoperation rate of 1.4% (1/72), and transient dysphagia occurred in 8/72.
Discussion
In single-level ACDF surgery, an inherent lower-risk is assumed, but it is not a risk-free surgical procedure, and an increment in the rate of complications is expected according to the number of treated levels, particularly dysphagia. 14 ACDF is 1 of the most frequently performed spine surgical procedures, and the gold standard material employed for achieving bone fusion is autologous bone graft, which carries donor site-related complications. 15 Therefore, other osteobiologics have been developed to avoid this associated morbidity with BMP being 1 of the most widely used; they are associated with a significant increment in cost and other adverse events.6,16 The present systematic review aimed to synthetize the best evidence available regarding the related-complications of multilevel—3- or 4-level—ACDF using osteobiologics other than BMP.
Three- or 4-level ACDF is considerably less reported in the literature than 1- or 2-level. Therefore, the complications reported are scarce and the adverse events related to osteobiologics in this scenario are remarkably missing. Nonetheless, the literature available regarding anterior cervical corpectomy and fusion 17 or posterior approach18,19 for the surgical treatment of multilevel degenerative disease is expanding. Independently of the osteobiologics used and considering different number of treated levels, the most frequently reported complication are transient dysphagia and pseudoarthrosis requiring reoperation, as observed by the present systematic review.10,12,13 A recent comprehensive qualitative description of pooled incidence including different number of treated levels showed adjacent segment disease as the main complication reported (8.1%), followed by dysphagia (5.3%), C5 palsy (3.0%) and pseudoarthrosis (2.0%). 20
Notably, when reporting dysphagia, formal and validated measurement tools to diagnose, quantify severity and impact on quality of life, and follow-up were not used. This fact can lead to an inconsistency and discrepancy in incidence of dysphagia when it is analyzed. 21 In the same line of thought, we considered pseudoarthrosis when it was textually reported, having variability in clinical and radiological criteria. A recent well-performed study comparing 3- and 4-level ACDF was not included in our systematic review due to the search data, which showed a greater rate of revision surgery of 3-level (9.3%) over 4-level (0%) ACDF caused by dysphagia, pseudarthrosis, and adjacent segment disease. 22
The available literature did not allow to separate complications according to the different osteobiologics in multilevel ACDF surgery. Cho et al 23 reported complications non-separately by number of treated levels in 86 cases of 3-level ACDF, and they found graft complications—radiological findings of collapse, non-union, and dislodged graft—in 4% of patients using iliac crest with plate and 50% in iliac crest alone. In their study, Moreland et al 24 included 13 patients with 3-level ACDF and 2 patients with 4-level ACDF using titanium cages filled with DBM or ß-TCP and compared them with allograft without instrumentation and which was non-differentiated by number of treated levels; there were 2 postoperative complications in the titanium cage group—postoperative hemorrhage requiring reoperation, and reoperation of an early adjacent segment disease. Chang et al, 25 in their clinical series including 16 patients with 3-level ACDF and 5 patients with 4-level ACDF, analyzed the clinical and radiographic outcomes comparing ACDF using cages filled with autogenous cancellous bone marrow vs hydroxyapatite graft, and there were no graft complications including collapse, non-union, or dislodgement. One patient had transient postoperative dysphagia.
In their prospective randomized controlled trial, Graham et al 26 included 19 patients with 3-level ACDF and 6 patients with 4-level ACDF. They performed a non-separated evaluation by number of treated levels comparing glycerol-preserved and freeze dried Cloward dowels in ACDF surgery. Fifty-nine patients (55.7% of the 106 study subjects) experienced at least 1 of the 108 adverse events reported. Typical reported events included dysphagia and pain in the neck area or along the arms, shoulders, and back. Heidt et al 27 published their experience in which 79 patients received 3-level ACDF and 3 patients receiving 4-level ACDF with autograft from iliac crest or allograft for fusion with a rigid or semi-rigid plate. No significant differences was observed between groups in radiographic adjacent segment disease (allograft 19.6%, autograft 29.4%, P = .101) or reoperation rates (allograft 4.3% vs autograft 7.8%; P = .250) at final follow-up.
Limitations of Present Systematic Review
This systematic review has some limitations that need to be recognized. First, it does not include comparative studies against corpectomy as they were excluded in the PICO Table according to the established exclusion criteria for all AOGO Project systematic reviews. Second, the scarce evidence available includes a list of osteobiologics other than BMP; therefore, the conclusions should be taken with caution and it must be kept in mind that knowing complications related specifically to each osteobiologic is not possible. Other limitations arise due to the limited data the current study was able to perform an analysis of from the total pooled data. Finally, well-designs tools for registering complications and grading their severity were not used, such as Spinal Adverse Events Severity System, version 2 (SAVES-2) 28 or the classification system proposed by Dindo-Clavien. 29
Conclusions
This systematic review attempted to summarize the best evidence available in multilevel ACDF using osteobiologics other than BMP. Consequent to the substantial heterogeneity of the included studies, making a conclusion with recommendations to the spine community is not possible. However, descriptively, the most common complications were dysphagia and pseudoarthrosis. Further prospective studies analyzing osteobiologics in multilevel ACDF separately to improve the strength of evidence are needed.
Supplemental Material
Supplemental material - Analysis of Complications in Multilevel Anterior Cervical Discectomy and Fusion Using Osteobiologics Other than Bone Morphogenetic Protein: A Systematic Review
Supplemental material for Analysis of Complications in Multilevel Anterior Cervical Discectomy and Fusion Using Osteobiologics Other than Bone Morphogenetic Protein: A Systematic Review by Juan P Cabrera, Neha Agarwal, Mohamed Kamal Mesregah, Ricardo Rodrigues-Pinto, Yabin Wu, Christopher Martin, Zorica Buser, Jeffrey C Wang, Hans-Jörg Meisel and the AO Spine Knowledge Forum Degenerative in Global Spine Journal
Supplemental Material
Supplemental material - Analysis of Complications in Multilevel Anterior Cervical Discectomy and Fusion Using Osteobiologics Other than Bone Morphogenetic Protein: A Systematic Review
Supplemental material for Analysis of Complications in Multilevel Anterior Cervical Discectomy and Fusion Using Osteobiologics Other than Bone Morphogenetic Protein: A Systematic Review by Juan P Cabrera, Neha Agarwal, Mohamed Kamal Mesregah, Ricardo Rodrigues-Pinto, Yabin Wu, Christopher Martin, Zorica Buser, Jeffrey C Wang, Hans-Jörg Meisel and the AO Spine Knowledge Forum Degenerative in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
