Abstract
Study design
Systematic literature review
Objective
To analyze the literature and describe the evidence supporting osteobiologic use in revision anterior cervical discectomy and fusion (ACDF) surgery.
Methods
A systematic search of PubMed/MEDLINE, EMBASE, Cochrane library, and ClinicalTrials.gov databases was conducted for literature reporting the use of osteobiologics in revision ACDF. We searched for studies reporting outcomes of using any osteobiologic use in revision ACDF surgeries (independently of the number of levels) in the above databases.
Results
There are currently no studies in the literature describing the outcome and comparative efficacy of diverse osteobiologic agents in the context of revision ACDF surgery. A majority of the current evidence is based only upon studies involving primary ACDF surgery.
Conclusion
The current study highlights the paucity of literature evidence on the role of diverse osteobiologics in revision ACDF, and foregrounds the need for high-quality evidence on this subject.
Introduction
Anterior cervical discectomy and fusion (ACDF) is a highly effective surgery employed in a wide variety of cervical pathologies. The management of empty disc space after discectomy and nerve root decompression in ACDF has remained an issue of controversy. 1 Diverse reconstructive options ranging from keeping the disc space empty to packing the disc space with bone grafts (autografts or allografts), or cages with or without additional reinforcement with anterior plates or inbuilt screws have been described in the literature. 2 More recently, newer osteo-biologic agents like demineralized bone matrix (DBM), synthetic bone graft substitutes, bone morphogenic proteins (rhBMP-2), and cellular bone matrix (CBM) have been employed either alone or in combination with bone grafts to enhance fusion rates through their osteogenic and osteoinductive properties.3,4
Since the success of ACDF procedures relies upon the solidity of the final fusion achieved, there has been a huge interest in the development of diverse synthetic or biological agents, which can meliorate the osseous healing across the disc space.5,6 A majority of such research on the role of osteo-biological and bone-substitute agents has focused on primary ACDF surgeries.1–6 As a direct impact of population aging, the rate of revision ACDF has been on the rise globally. In a recent study, the 2-year revision rates have been reported to range between 2.1% and 9.13% for single-level ACDF; and between 4.4% and 10.7% for multi-level ACDF. 7 The need for achieving fusion in such revision scenarios is of even greater significance, as the overall outcome tends to progressively deteriorate with every additional procedure undergone. 8 In this context, the influence of different bone graft materials and other osteo-biological alternatives on the fusion, overall outcome, and complication rates in patients undergoing revision ACDF needs to be evaluated. The objective of the current study was to systematically review the literature, and describe the evidence supporting the use of osteo-biologics in the revision ACDF surgery.
Material and Methods
Electronic Literature Search
PICOS Framework used for the Search Strategy.
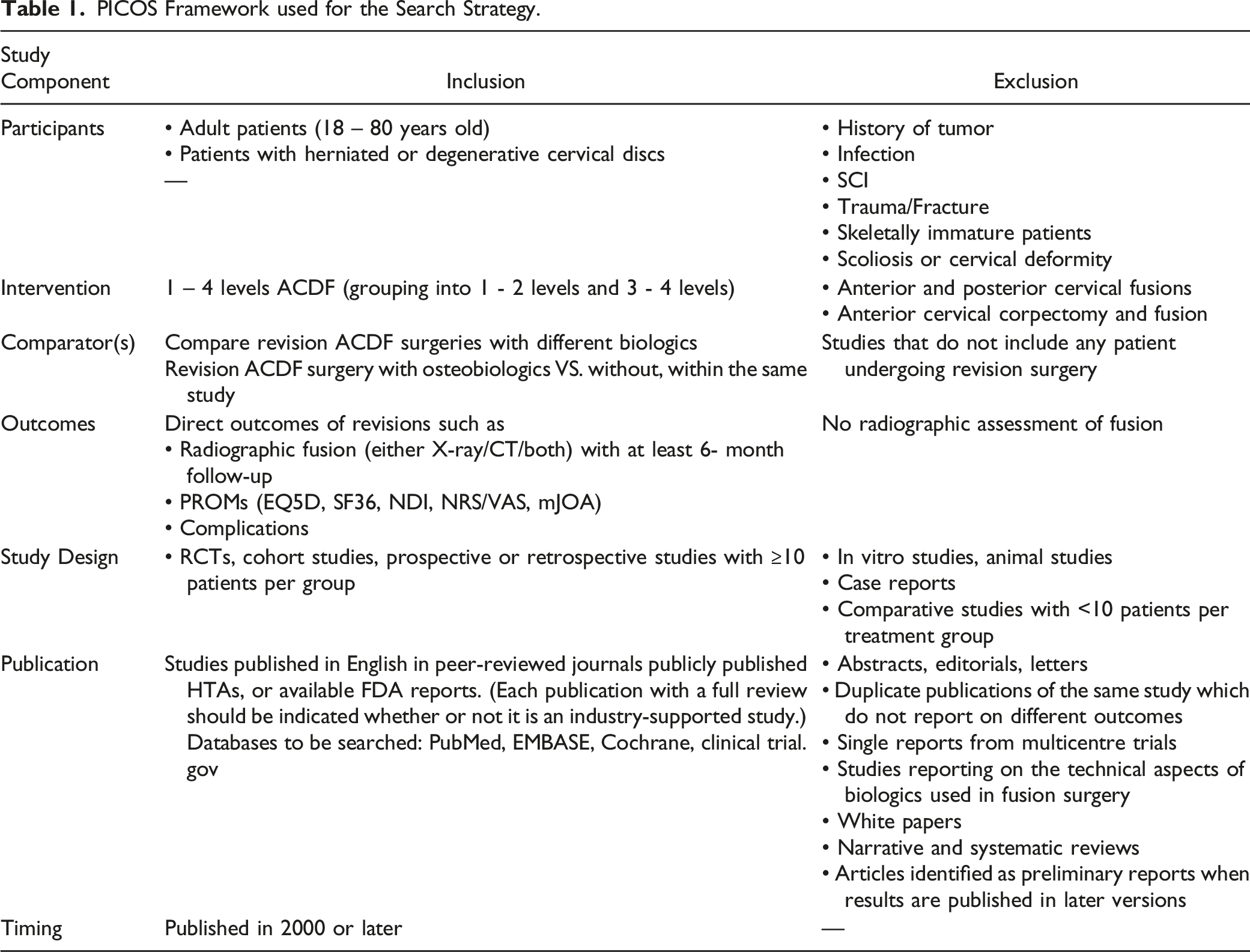
Eligibility Criteria and Screening
To be included in the final review, studies had to report on revision ACDF surgeries (independently of the number of levels) using any osteobiologic. Titles and abstracts were screened by 2 independent reviewers and disagreements were discussed among reviewers and solved through consensus.
Quality Assessment and Synthesis
Risk of bias assessment was planned to be performed using the Methodological Index for Non-Randomized Studies (MINORS) and the Cochrane Risk of Bias tool version 2.0 (RoB 2) for non-randomized and randomized clinical studies respectively.10,11 Certainty of evidence was planned to be assessed using the GRADE approach.
Results
A search for studies reporting on the use of osteo-biologics in ACDF revision surgeons was performed. From the 2231 potentially relevant citations, after title screening, 52 proceeded for abstract screening. However, only 1 relevant citation
5
was identified at this stage and after full-text assessment, the shortlisted article did not fit into the inclusion and exclusion criteria. Hence, no studies were identified to report the use of osteobiologics in revision ACDF surgery for inclusion in the systematic review as shown in Figure 1. PRISMA flow diagram for analysis of studies for inclusion in the analysis.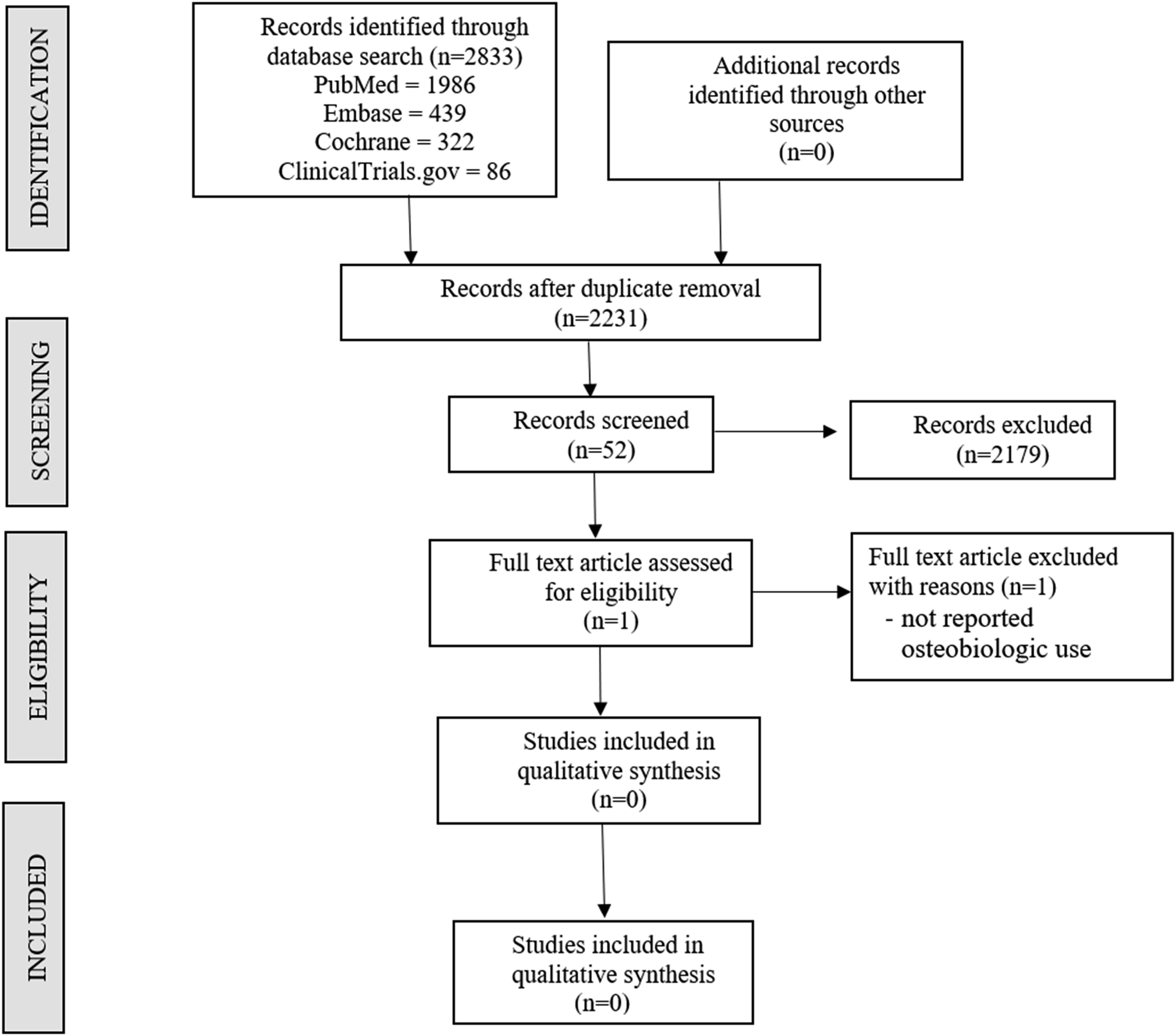
Discussion
As a direct consequence of the progressive increase in the number of cervical surgeries performed worldwide, the rates of revision cervical surgeries have also steadily escalated.8,12 The revision surgery rates have been reported to vary for different cervical spine procedures, ranging from 2.1 to 9.3% for ACDF, 1.8 to 5.4% for cervical disc arthroplasty (CDA), 2.9 to 5% following posterior cervical foraminotomy (PCF), 2.1 to 13% after laminoplasty; and 2 to 27% following laminectomy and fusion.6,13,14 The most common underlying pathologies leading to revision spinal procedures include adjacent-segment degeneration (ASD), pseudo-arthrosis, infection, same-segment disease, sagittal deformity (or kyphosis), or a combination of the aforementioned diagnoses.1–8,12 Although a carefully-planned revision surgery results in a satisfactory outcome; with every additional intervention undergone, complication rates like pseudo-arthrosis, implant failures, wound-related issues, bleeding, infections, etc. also correspondingly increase. 8 Whether the use of bone graft or other bone substitutes influences the fusion rates after ACDF, is still a debatable issue.12,15,16 Such a question carries even greater significance in the context of revision ACDF surgery. 17 The current systematic review was thus planned to evaluate the role of osteo-biologic agents on the overall outcome of patients undergoing revision ACDF. The literature search focused on articles that discussed and compared the roles of different bone grafts or other bone-substitute materials (eg autograft, allograft, DBM, CBM, or BMPs), either alone or as a supplement to cages (standalone cage or cage reinforced with in-built screws or plates) on the fusion and overall outcome following revision ACDF surgery.
Despite an extensive search of the existing literature, we could not identify any article evaluating the role of osteobiologic agents in determining the clinical, radiological, and functional outcomes of patients in revision ADCF surgeries. Despite being a very relevant subject; unfortunately, the current literature is still substantially deficient in evidence on this subject. A majority of the current evidence on osteobiologics is based on studies involving primary ACDF.1–8,12
Based on the available evidence, although the use of osteobiologic agents seems to enhance fusion rates; there is no strong evidence to suggest a significant difference in the overall outcome with different materials (autograft vs allograft vs other osteobiologics) in single-level primary ACDF. 8 In multi-level ACDF, agents with osteoinductive and osteogenic potentials are generally favored. 5 A large, retrospective study by Pirkle et al 18 demonstrated lower non-union rates (of 1.97%) with allograft, in comparison with interbody cages (5.32%). In a prospective randomized controlled trial, the use of local auto-graft from the marginal osteophytes resected at the time of discectomy, when used as fillers within the cage, had a similar outcome as compared with the allograft. 1 Wang et al 19 showed that in primary ACDF, there was no difference in the fusion rates among 3 different graft materials (local autograft vs DBM vs combination of local autograft and DBM). In another non-randomized, prospective trial, single-level ACDF with CBM-filled polyether ether ketone (PEEK) inter-body spacer resulted in 92% fusion at 2 years, with no case of pseudo-arthrosis. 15 CBMs still require stronger evidence from large-scale, prospective cohort studies before being routinely recommended. 15
In general, revision ACDFs are associated with significantly increased risks of multiple adverse events like thrombo-embolic complications, recurrent laryngeal nerve palsy, dysphagia, higher need for blood transfusion, greater rates of return to the operating room, surgical-site infections, a longer length of hospital stay, and higher re-admission rates (within 30 days).4–8 The decision-making regarding the surgical approach to revision ACDF is based on the presence of ASD, implant migration or mal-position, segmental kyphosis, and residual spinal canal stenosis. 3
We could observe that a majority of studies on revision ACDF focus on adjacent segment degeneration, and there is a paucity of studies on revision procedures required for index-level pathologies. The most common index-level pathology leading to revision surgery is pseudoarthrosis.3,20–22 Some of the risk factors for pseudoarthrosis include the number of levels of pathology (multi-level), bone quality of patients (osteopenia or osteoporosis), patient factors (smokers), type of fixation (lower rates with plating constructs in multi-level pathology), and type of implant [greater incidence with PEEK implants].1–8,12 Many pseudoarthrosis patients are asymptomatic, and the most common symptom necessitating revision surgery is intractable neck pain with radiculopathy. Radiological diagnosis of non-union is made based on dynamic lateral radiographs (Ghiselli or Cannada techniques); or CT scans.20–22 It has been reported that 70% of radiological non-unions at 1 year go onto fuse at the end of 2 years. 20–22 Recently, Alhashash et al 3 recommended a treatment algorithm for revision ACDF surgeries. In the presence of ASD, implant migration, segmental kyphosis, and residual spinal stenosis, they recommended approaching the spine anteriorly; while in the absence of any of these factors, the posterior approach is more suitable. In patients with poor bone quality and multilevel pathologies, combined approaches may be recommended. Based on our literature search, there is a tendency to prefer a posterior approach for revision surgeries following ACDF surgery.3,6–8,12,15–17
Author's Perspective
In our experience, 3 crucial factors determine the decision-making process regarding the type of osteobiologic agent used in revision ACDF surgery, which includes: 1. pathological cause for revision, 2. number of surgical levels, and 3. quality of the patient's bone.
In single-level revision ACDF surgery performed for ASD, local autograft (from osteophytes) or allograft may be the best choice. On the other hand, in revisions performed for pseudoarthrosis, agents with reliable osteogenic and osteoinductive properties are required to provide the optimal milieu for tissue healing. Although local autograft is both osteogenic and osteoinductive, its availability is substantially limited, especially in revision scenarios. We no longer use iliac crest autograft routinely in our practice, because of the concerns regarding donor-site morbidity (except in contexts of limited resource availability). In our current scenario, allografts supplemented with DBM, CBM, and BMP may offer the best outcome in such pseudoarthrosis scenarios (especially in multi-level disease), although the newer agents are still not FDA-approved for this use and can also be prohibitively expensive. Bone marrow aspirate (procured from the iliac crest or sternum) mixed with allograft substrate can be a cheaper alternative, offering the advantages of providing both osteoinductive and osteogenic environments with mitigated risks for donor-site morbidity. In patients undergoing revision ACDF for cervical deformity (involving multi-level surgeries with possibly, anteroposterior approaches) or patients with poor bone quality, allograft combined with 1 of the aforementioned agents with osteoinductive and/or osteogenic properties may be preferred. For revision ACDF performed in the setting of infection, we do not add any adjuvant graft materials until the gross infection has been eradicated. In these patients, we prefer to use only autografts. Thus, we decide upon osteobiologic use in revision scenarios, taking into consideration every patient on an individual basis in the background of all these aforementioned factors. We emphasize the clear paucity of data on this subject, and highly recommend the need for future research on this subject.
Our study has a few limitations. We have not included a grey literature search for studies not available in online databases which might have prevented us from including some potential studies on the subject. However, to be more comprehensive we search 4 databases as per the PRISMA guidelines. We have applied language restriction to the article selection to English, which might have also limited the inclusion of any potential studies in other languages.
Conclusion
Our detailed literature search failed to reveal any major studies evaluating the role of osteobiologics in the revision ACDF scenario. Considering the complexity of the situation and the utmost need to ascertain good fusion in the context of revision ACDF, the substantial need to explore the options of traditional and novel osteobiologics, and compare their relative efficacies as supplements to the conventional fusion constructs cannot be understated. Thus, the current study strongly highlights the need for large-scale prospective, randomized-controlled studies to comparatively evaluate the role of diverse osteobiologics in the context of revision ACDF surgery.
Supplemental Material
Supplemental Material - What Is the Evidence Supporting Osteobiologic Use in Revision Anterior Cervical Discectomy and Fusion?
Supplemental Material for What Is the Evidence Supporting Osteobiologic Use in Revision Anterior Cervical Discectomy and Fusion? by Sathish Muthu, Sara Elisa Diniz, Vibhu Krishnan Vishwanathan, Patrick C Hsieh, Aidin Abedi, Tim Yoon, Hans-Jorg Meisel, Zorica Buser, Ricardo Rodrigues-Pinto, and AO Spine Knowledge Forum Degenerative in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
