Abstract
Study Design:
Cost-effectiveness analysis.
Objectives:
To determine the 7-year cost-effectiveness of cervical disc replacement (CDR) and anterior cervical discectomy and fusion (ACDF).
Methods:
We analyzed 7-year Short Form-36 Health Survey data collected from the Prestige Cervical Disc investigational device exemption study (IDE). The SF-6D algorithm was used to convert this data into health state utilities. Costs were calculated from the payer perspective, and quality adjusted life years (QALYs) were used to represent effectiveness. A Markov transition-state model was used to evaluate the cost-effectiveness of single-level CDR versus ACDF, and a Monte Carlo simulation was performed to assess the probabilistic sensitivity of the model.
Results:
CDR generated a 7-year cost of $172 989 compared to a 7-year cost of $143 714 for ACDF. CDR generated 4.53 QALYs compared to 3.85 QALYs generated by ACDF. The cost-effectiveness ratio of CDR was $38 247/QALY, while the cost-effectiveness ratio of ACDF was $37 325/QALY. The incremental cost-effectiveness ratio of CDR was $43 522/QALY, under the willingness to pay threshold of $50 000/QALY. Our probabilistic sensitivity analysis demonstrated CDR would be chosen 56% of the time based on 10 000 simulations.
Conclusions:
Single-level CDR and ACDF were both cost-effective strategies at 7 years for treating degenerative conditions of the cervical spine. Both the Markov simulation and the Monte Carlo simulation demonstrate CDR to be the more cost-effective strategy at 7 years. Continued analysis of IDE data should be performed to validate long-term cost-effectiveness of these treatment strategies.
Introduction
Anterior cervical discectomy and fusion (ACDF) has long been considered the “gold standard” for treating degenerative conditions of the cervical spine that are refractory to conservative management. 1 –5 While outcomes following ACDF are generally favorable, the technique restricts cervical range of motion and may accelerate adjacent segment degeneration. 6 –8
Cervical disc replacement (CDR) was developed to mitigate some of the negative aspects of ACDF, and as a motion-preserving treatment modality it may be an effective alternative. 9,10 The increasing popularity of CDR has been complemented by a number of Food and Drug Administration (FDA) investigational device exemption (IDE) studies that have demonstrated similar clinical outcomes between ACDF and CDR for both 1- and 2-level procedures. 11 –18
In the case of 2 competing treatment modalities, cost-effectiveness analysis (CEA) can be an important tool for providing additional economic insight into the benefits of one procedure over another. 19 Current studies demonstrate CDR to have lower costs than ACDF for 1- and 2-level procedures in the short term, but few studies have established long-term cost-effectiveness of CDR. 20 –22
The Prestige LP Cervical Disc (Medtronic Inc, Minneapolis, MN) has superior clinical outcomes at 7-year follow-up compared to ACDF for single-level procedures, but the cost-effectiveness of this device at long-term follow-up (7 years) for single-level procedures has yet to be determined. 11,12 Our objective with this study was to use 7-year follow-up data from the single-level Prestige CDR IDE to conduct a cost-effectiveness analysis comparing CDR to ACDF. Our results will provide an economic perspective for making an informed decision about which treatment modality to use for treating degenerative conditions of the cervical spine.
Materials and Methods
Study Design
This CEA model followed the guidelines of the Panel on Cost-Effectiveness in Health and Medicine. 23 This panel established a set of methodological guidelines that when followed provide standardization and allow for the establishment of consistent quality and comparable results. This study did not require institutional review board approval as all data obtained from the original FDA IDE study was completely de-identified.
A Markov state-transition model was constructed using a common decision-analysis software package (TreeAge Pro 2015; TreeAge Software, Williamstown, MA). A Markov state-transition model allows for the direct comparison of the relative costs and effectiveness of different treatment options. The model was constructed from the payer perspective, in which only the health care described in the catalog of reimbursed items is relevant and the reimbursement rates directly reflect the costs of the model.
The primary effectiveness in this study was presented as quality-adjusted life years (QALYs). Costs were expressed in 2014 dollars. All input variables in this model were discounted at the standard rate of 3% to reflect present-day value. The outcomes of the model were expressed as incremental cost-effectiveness ratios (ICERs)($/QALY), which allow for the comparison of the relative cost-effectiveness of 2 procedures. Outcomes and transition-state probabilities were determined from the data presented in the Medtronic Prestige Single-Level CDR IDE study.
Model Structure
The conceptual structure of the Markov transition-state model is demonstrated in Figure 1. The model represents the 2 treatment strategies, CDR and ACDF, that can be used to treat single-level cervical degenerative disc disease. Each surgical strategy was represented by 5 discrete transition states: (1) Well State, (2) Index Revision State, (3) Adjacent Segment Revision State, (4) Complication State, and (5) Death State.
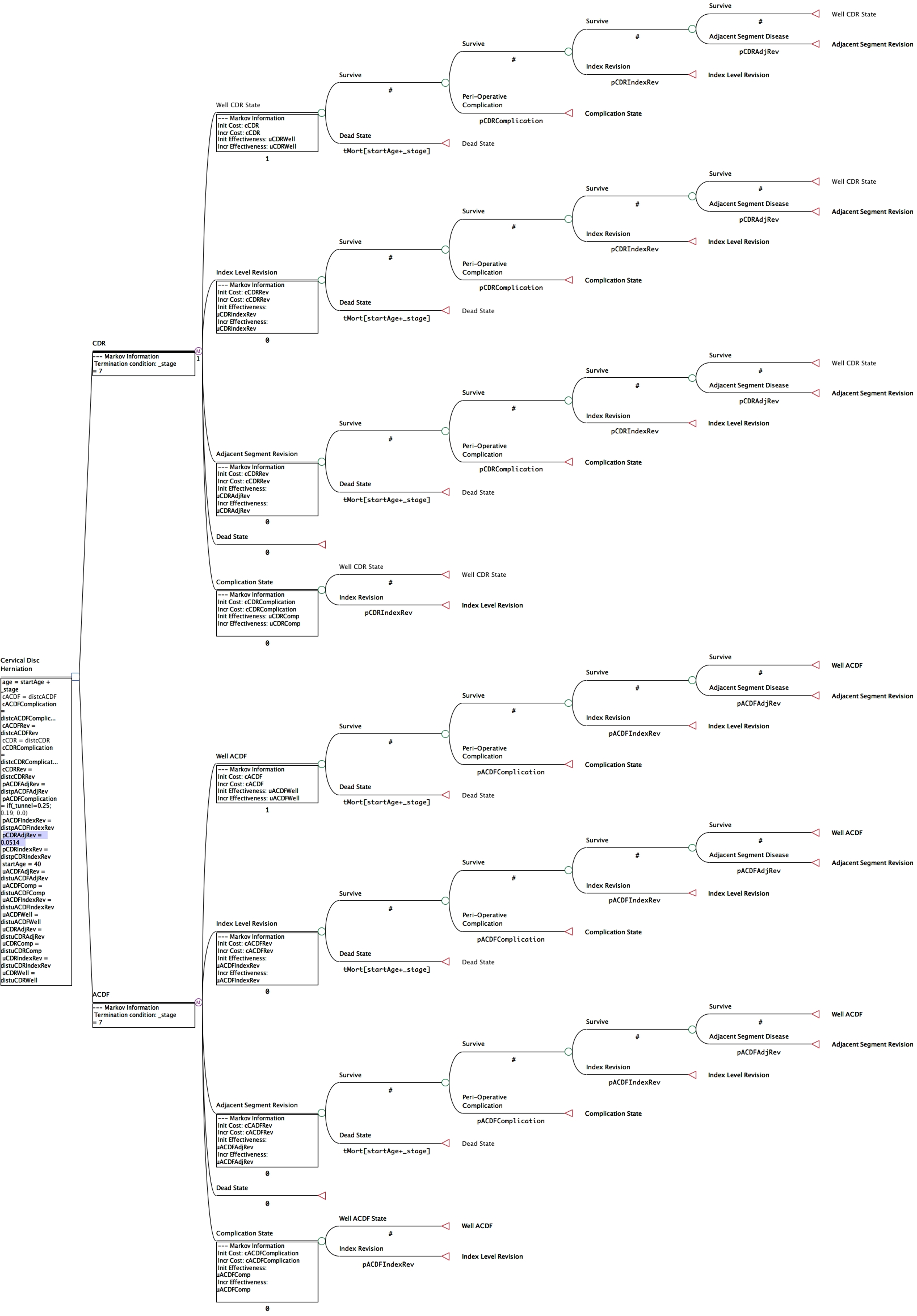
Visual representation of the Markov transition-state model. Each surgical treatment was associated with 5 distinct transition-states: (1) Well State, (2) Index Revision State, (3) Adjacent Segment Revision State, (4) Complication State, and (5) Death State.
Transition probabilities between the various transition-states were determined from the Medtronic Prestige Single-Level IDE study (Table 1). These probabilities were expressed as probability of occurrence/annum. Perioperative death was assigned a value of zero as there were no perioperative mortalities in the IDE study and the risk of death from anesthesia remains extremely low. Patients could only enter into the complication for the first 90 days of the first cycle to represent any perioperative complication risk. Similarly, a 90-day complication window could also be entered after index or adjacent segment revision.
Transition Probabilities Used in the Model.

Abbreviations: ACDF, anterior cervical discectomy and fusion; CDR, cervical disc replacement; CDC, Centers for Disease Control and Prevention.
Health state utility values were assigned to each of the Markov transition states. The utility value represents the value obtained by the patient for remaining in that health state for one cycle.
A willingness-to-pay threshold of $50 000/QALY was used. A probabilistic sensitivity analysis was performed using a Monte Carlo simulation of 10 000 cycles to validate the input variables in the model. Confidence intervals were reported at 95%.
Utilities
Utility values can range from 0 to 1, with 0 representing death and 1 representing perfect health. Overall, health state utilities are a required part of any CEA analysis as they represent a reliable measure of the health-related quality of life. When a CEA model is run over a number of cycles, health state utilities are combined with survival estimates and are aggregated across the model to generate QALYs.
Short Form 36 (SF-36) data from the Single-Level Prestige LP CDR IDE study for 1212 CDR patients and 183 ACDF patients was used. The raw SF-36 data was collected at baseline, 12 months, 24 months, 36 months, 60 months, and 84 months postoperatively. SF-36 data was converted into health state utilities using the SF-6D algorithm, which is a preference-based index obtained from a sample of the general population using the recognized valuation technique of standard gamble (Table 2).
Utility Scores at Each Time Point as Derived From the SF-6D.
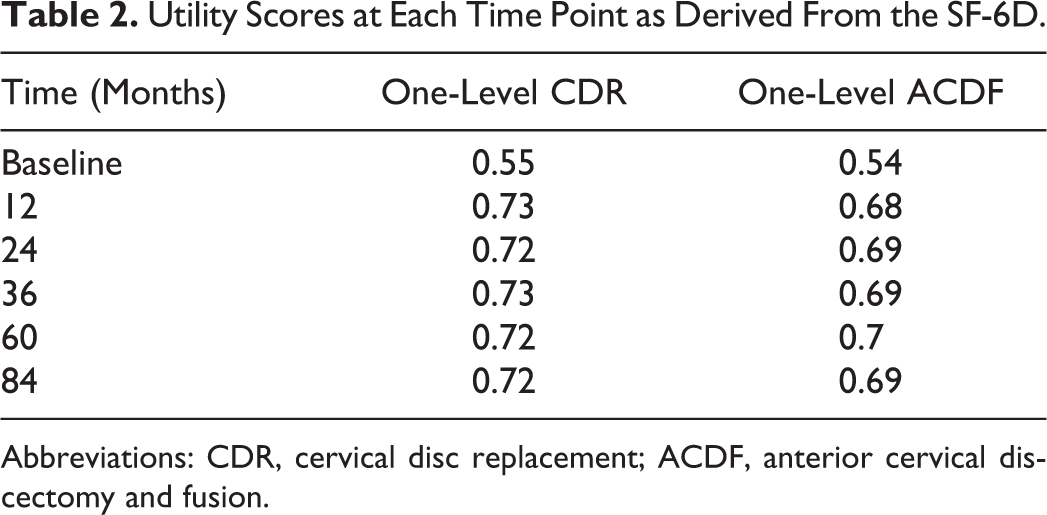
Abbreviations: CDR, cervical disc replacement; ACDF, anterior cervical discectomy and fusion.
Revision surgery at the index segment or adjacent segment were assumed to have a utility similar to that of the base-case state (preoperative state).
Costs
Gross cost methodology was used to estimate the direct costs of both CDR and ACDF. International Classification of Diseases, Ninth Revision, Diagnosis Related Group (DRG) and Current Procedural Terminology (CPT) codes specific for each surgical procedure were used. Combining the reimbursement rates for each DRG and CPT using 2014 Medicare date, an accurate representation of the direct costs could be obtained (Table 3). To reflect the values of a private payer system, Medicare reimbursement rates were multiplied by 140%. All costs in this study were denominated in 2014 dollars.
Costs in 2014 Dollars for Selected CPT and DRG Codes.
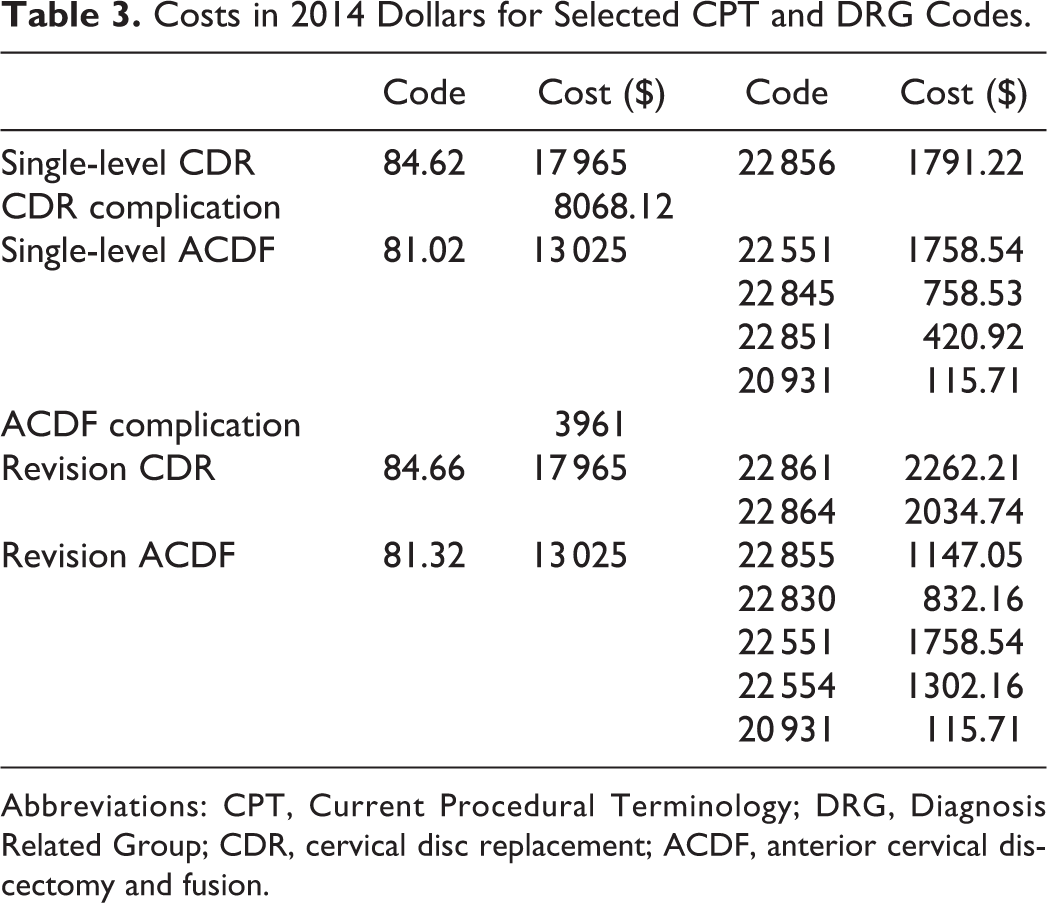
Abbreviations: CPT, Current Procedural Terminology; DRG, Diagnosis Related Group; CDR, cervical disc replacement; ACDF, anterior cervical discectomy and fusion.
Sensitivity Analysis
As CEA models are built on advanced statistical decision and a certain level of uncertainty exists with respect to parameters and observable variables within the model, it is necessary to perform a robust sensitivity analysis to validate the model. A probabilistic sensitivity analysis (PSA) was used in our model, which is a procedure by which all the input variables are considered as random quantities and can be associated with a probability distribution. A Monte Carlo simulation was used to perform the PSA across 10 000 discrete iterations.
Results
Base Case
The model was constructed to treat a 40-year-old patient with single-level degenerative disc disease who had failed appropriate conservative care and was an ideal surgical candidate for CDR or ACDF.
Procedure-Related Costs
Costs were generated using a 140% multiple of 2014 Medicare reimbursement for the associated DRG and CPT codes for each procedure. All costs were discounted at 3% to represent present-day value. The base case analysis generated a 7-year cost of $172 989 for CDR and $143 714 for ACDF (Table 4). ACDF resulted in a 7-year cost savings of $29 275 when compared with CDR.
Base Case Cost-Effectiveness Rankings of One-Level ACDF Versus CDR.
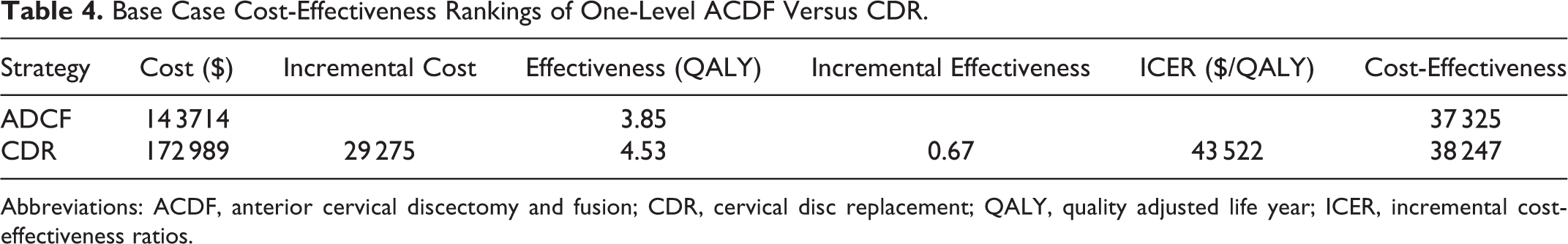
Abbreviations: ACDF, anterior cervical discectomy and fusion; CDR, cervical disc replacement; QALY, quality adjusted life year; ICER, incremental cost-effectiveness ratios.
Procedural Effectiveness
CDR resulted in a generation of 4.53 QALYs, while ACDF resulted in 3.85 QALYs (Table 4). Over the 7 years represented by the model, CDR resulted in a net gain of 0.67 QALYs when compared with ACDF.
Procedural Cost-Effectiveness
CDR resulted in an increased incremental cost of $29 275 with an incremental gain of 0.67 QALYs when compared with ACDF. The cost-effectiveness ratio of CDR was $38 247/QALY, while ACDF was $37 325/QALY. The ICER of CDR was $43 522/QALY, which reached the willingness to pay (WTP) threshold of $50 000/QALY (Table 3).
Probabilistic Sensitivity Analysis
The results of the Monte Carlo simulation are demonstrated in Table 5. Figure 2 represents the cost-effectiveness scatter plot of the 10 000 simulations comparing CDR and ACDF. Simulations to the right of the WTP line represent those values in which CDR is the more cost-effective strategy. Assuming a WTP $50 000/QALY, the cost-effectiveness acceptability curve indicated that CDR would be chosen 56% of the time based on 10 000 simulations (Figure 3).
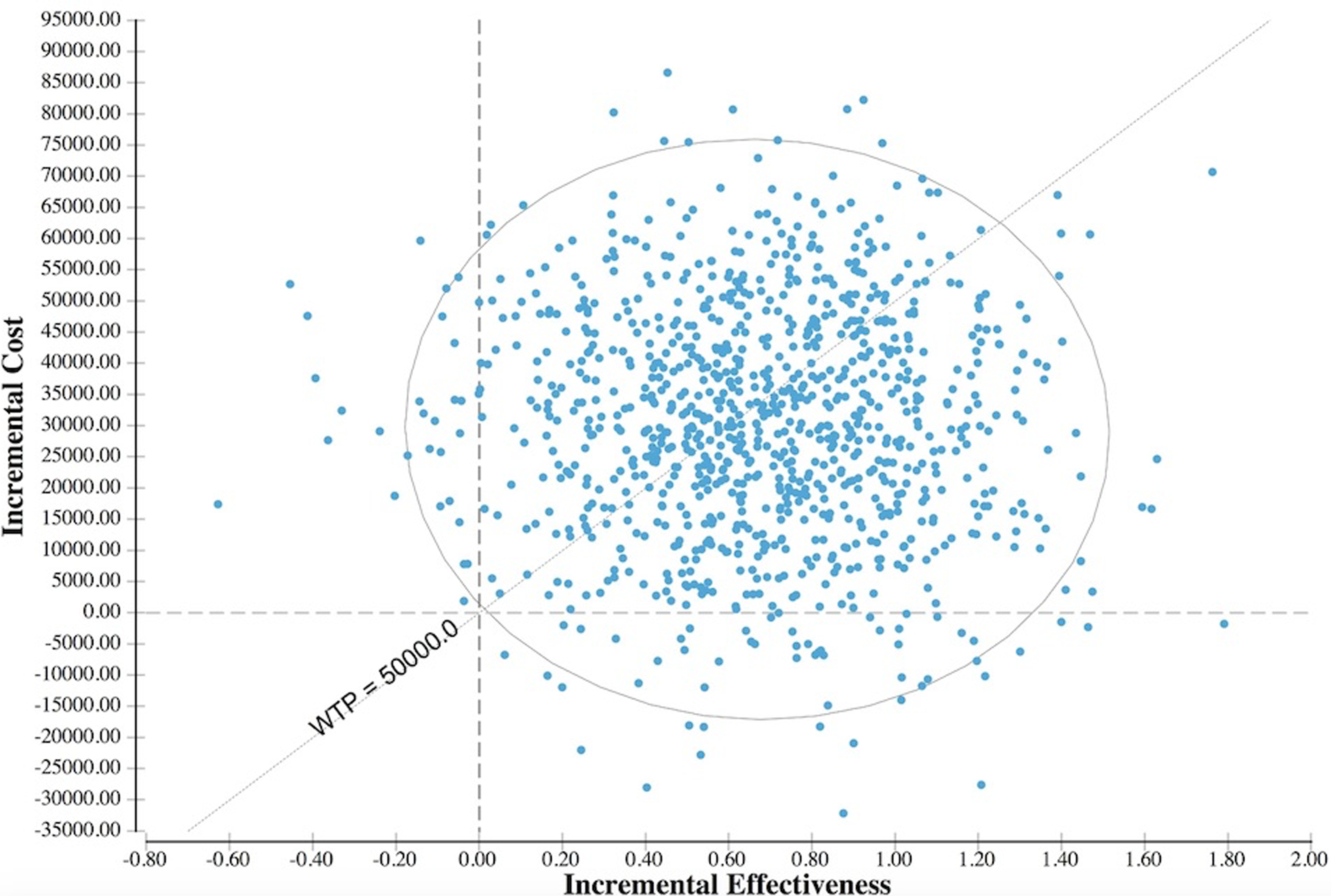
Depiction of the cost-effectiveness scatter plot of 10 000 simulations. Simulations to the right of the willingness to pay (WTP) line indicate values where CDR is the more cost-effective option.
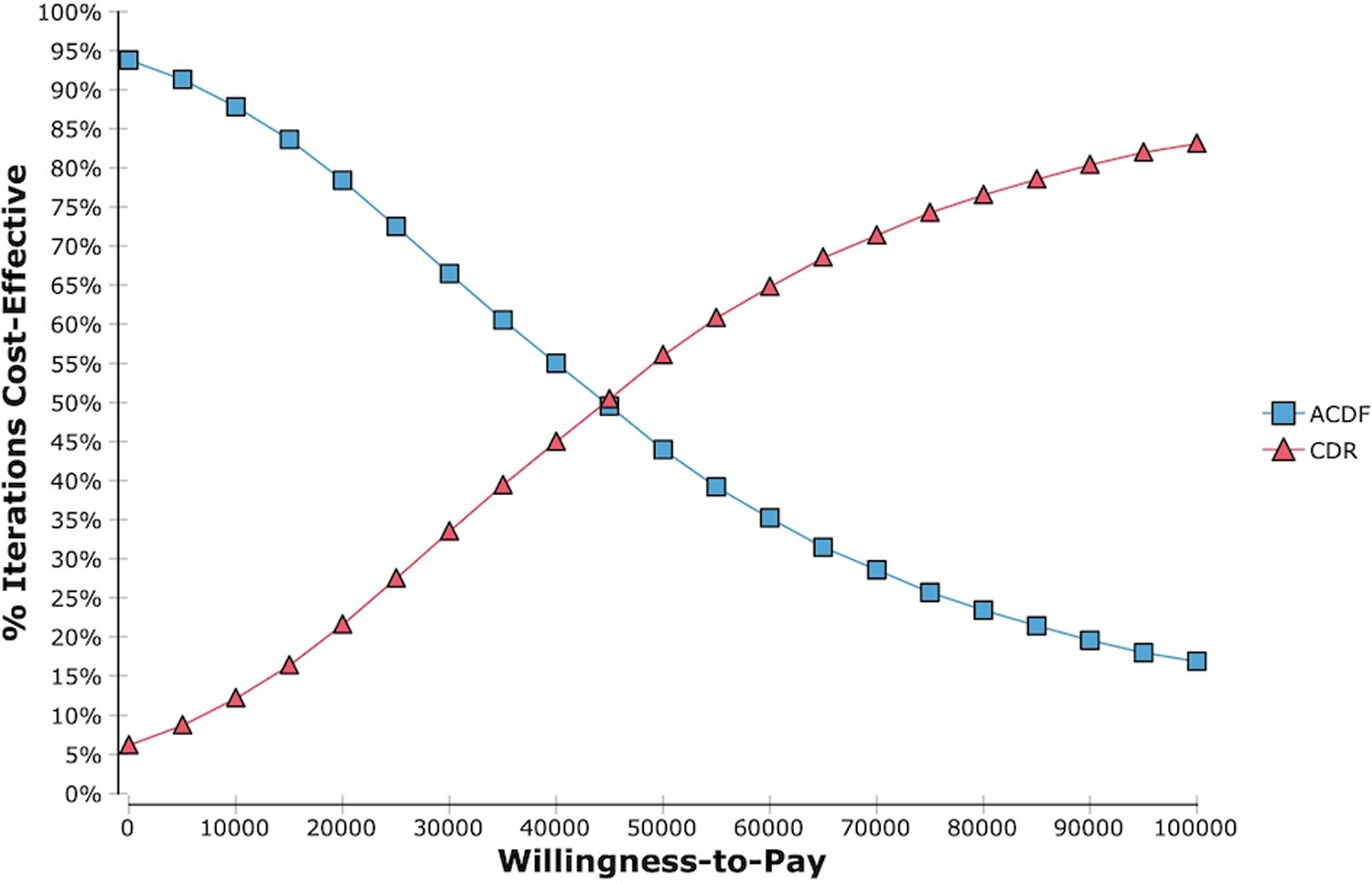
Based on 10 000 simulations, the cost-effectiveness acceptability curve indicated CDR would be chosen 56% of the time, assuming a WTP $50 000/QALY.
Results of the Monte Carlo Probabilistic Sensitivity Analysis of 10 000 Case Simulations.
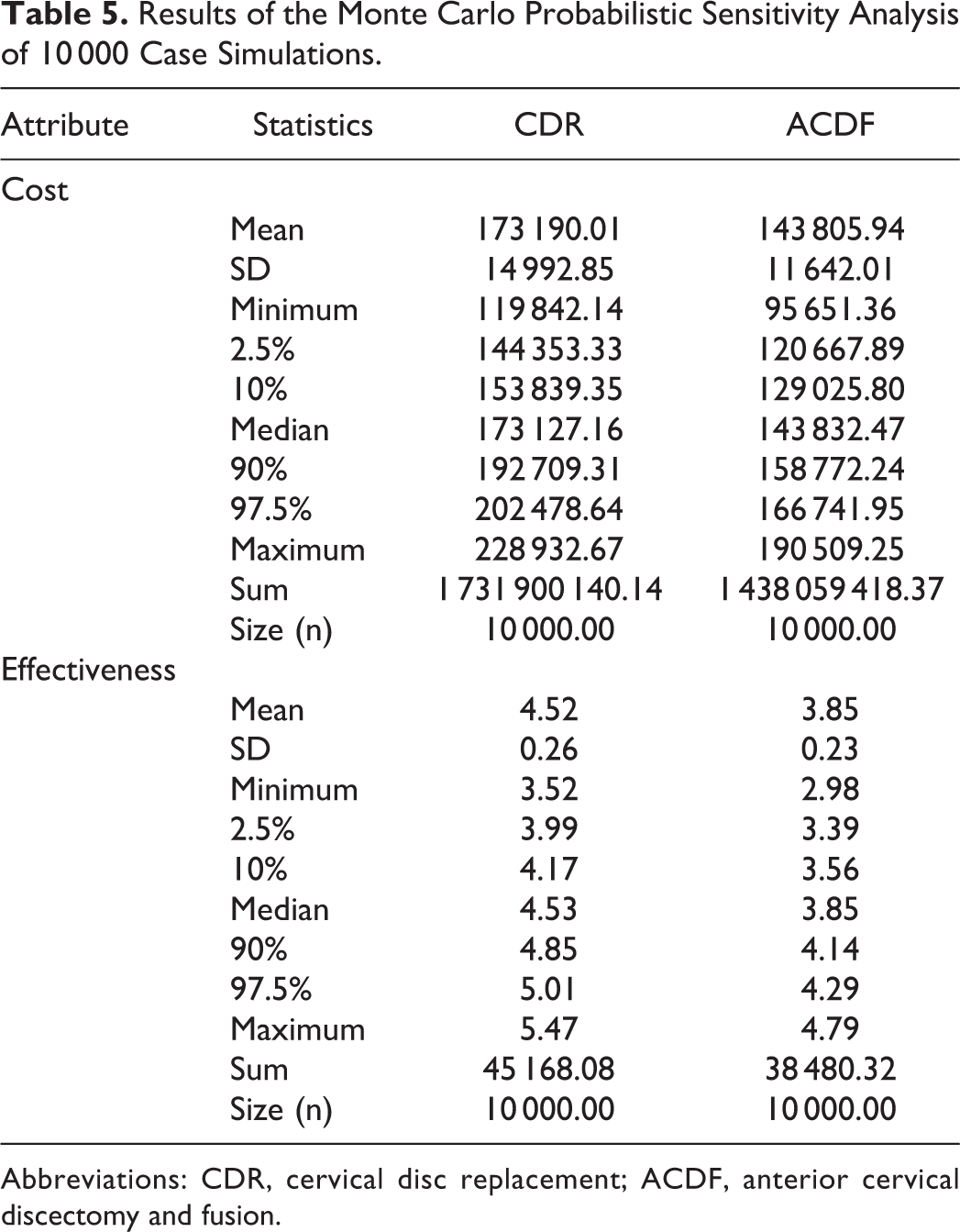
Abbreviations: CDR, cervical disc replacement; ACDF, anterior cervical discectomy and fusion.
Discussion
The objective with this study was to independently analyze prospectively collected data with 7-year clinical follow-up from the Prestige Cervical Disc Investigational Device Exemption Study. A number of FDA IDE studies comparing both 1-level and 2-level CDR to ACDF have demonstrated similar clinical outcomes between the 2 treatment modalities. 11 –17 Cost-effectiveness analyses have also suggested CDR to be a more cost-effective alternative than ACDF out to 5 years, but the cost-effectiveness has yet to be assessed at 7 years posttreatment. 20 –22
This study utilized a Markov-state transition model with a robust probabilistic sensitivity analysis to assess the relative cost-effectiveness of single-level CDR versus ACDF at 7 years. A Markov model allows for effective modeling of the decisions, outcomes, and associated cost involved in medical decision making. The transition state model allows for a patient to travel between various health states over time based on the real-world probability of doing so. Markov modeling of health care decision making is a well-validated technique and has been applied widely within orthopedics and increasingly within the realm of spine surgery. This study is one of the first to utilize the technique of Monte Carlo simulations as a means of performing a robust sensitivity analysis to validate the input variables used as part of the model.
The utility values in our model were derived from the SF-36 data from the Prestige IDE trial comparing outcomes of CDR and ACDF. SF-36 was the chosen metric as it is a validated means of translating these scores into a quantitative health utility value for use in CE modeling. 24,25 SF-36 data was converted into health state utilities using the SF-6D algorithm, which is a preference-based index obtained from a sample of the general population using the recognized valuation technique of standard gamble.
The results of this Markov simulation demonstrated that CDR was a more cost-effective option at 7 years when compared with ACDF. While both strategies can be considered cost-effective options with CE ratios less than $50 000/QALY, the ICER when comparing CDR and ACDF was found to favor CDR. Furthermore, while CDR was shown to be incrementally more expensive than ACDF, there was a significant gain in QALYs when compared with ACDF. The results of this analysis are further supported by the results of the PSA. The input variables were validated as accurate when analyzed across 10 000 simulations. Furthermore, CDR was found to be the more cost-effective option 56% of the time. The use of PSA analysis provides a more robust validation of the input variable. Historically, CEA studies have utilized 1- and 2-way sensitivity analyses to validate variables of interests. These types of analyses are considered deterministic and fail to consider the possible correlation or the underlying uncertainty about the variable of interest, focusing instead on a set of arbitrarily chosen values. PSA consider all of the input variables as random quantities with an assigned probability distribution to them.
Our results demonstrate the cost benefits of CDR, and the literature suggests clinical benefits as well. Burkus et al independently analyzed single-level 7-year clinical and radiographic outcome data from the Prestige Cervical Disc IDE. 11 The authors found that the overall success rate was 6.4% greater in the CDR group at 5 years, though this was not statistically significant. The overall success at 7 years, however, was 12.6% greater in the CDR group and was statistically significant, suggesting long-term clinical success of CDR and underscoring the importance of long-term follow-up. The authors defined overall success by 5 criteria; postoperative neck disability index score improvement of at least 15 points from preoperative, maintenance or improvement in neurologic status, functional spinal unit (FSU) disc height maintenance, no serious adverse event, and no additional surgical procedure. When examining individual clinical and radiographic outcomes, the authors observed a statistically significant lower mean neck pain score of 4.2, higher mean SF-36 score of 1.9, and greater neurologic success in the CDR group compared to the ACDF group. The authors suggest the greater neurologic success in the CDR group may have been attributed to a larger posterior decompression that occurred during CDR. There were no statistical differences between the groups with regard to arm pain, FSU failure, adjacent segment angular motion, dysphagia, or implant removal. There were higher rates of secondary surgery (4.8% vs 13.7%), revision surgery (0% vs 2.1%), supplemental fixation (0% vs 2.3%), and adjacent-level surgery in the ACDF group compared to the CDR group (4.6% vs 11.9%), all of which were statistically significant. With respect to adverse events, the CDR group had fewer spinal events (20.9% vs 38.9%), but more urogenital events (20.1% vs 12.2%), than the ACDF group, and also had a higher incidence of broken screws (5 cases vs 0 cases) than the ACDF group. All of these adverse event findings were statistically significant. These results suggest several advantages of CDR compared to ACDF with respect to clinical outcomes and additional required surgeries. 11 A second clinical and radiographic study of the 7-year Prestige cervical data by Gornet et al corroborated these findings. 26
Different types of cost-effectiveness analyses have shown CDR to be a more cost-effective option than ACDF. 20,21,27,28 Ament et al compared cost-utility of CDR and ACDF for 2-level procedures using 5-year follow-up data. 20 They used the ICER to calculate the cost-utility outcome, and a value under the commonly accepted threshold of $50 000 WTP would favor CDR. The authors used a Markov model to analyze costs and health utility outcomes. While CDR was associated with a greater cost of $1687 than ACDF over 5 years, it was also associated with less productivity loss of $34 377 over 3 years. The authors postulate the reduced productivity loss of CDR compared to ACDF was due to earlier return to work in the CDR group. The CDR cohort had 3.57 QALYs compared to 3.38 QALYs in the ACDF cohort. Incorporating the productivity loss, the authors suggest that CDR dominates ACDF from a cost utility perspective at 5 years. We found similar results in that despite initial greater cost of CDR, incremental improvement in QALYs in the CDR cohort suggest its effectiveness over ACDF. These types of cost-effectiveness analyses offer an economic perspective that may help inform decision-making when considering 2 treatment options.
This study has several limitations that are worth noting. Our model assumes a definite number of potential outcomes states, for which a patient may only enter once. For example, a patient may only enter into a revision state once, which may understate the possibility of requiring further revision surgery. Additionally, the use of a transition state model may undersimplify the real-world health transitions that a patient can make as part of their postoperative course. The cycle length was set to 1 year in this model. It is important to note that in real life a patient is not bound to cycle length and may in fact transition between health states more frequently than what is able to be accurately modeled with a decision analysis.
Another potential limitation of this study is the use of only direct costs in the model. The authors utilized gross-costing techniques based off of the payer perspective. Indirect costs including return to work data were not included in this study. Indirect costs require the use of micro-costing techniques, which have been shown to be cumbersome and difficult to accurately collect. Furthermore, the use of gross-costing has been repeatedly shown by health care economist to be a more plausible economic measure for routine use.
This study provides a validated model of the relative cost-effectiveness of single-level CDR and ACDF at 7 years. There remains uncertainty surrounding the potential long-term survivability of the CDR implant. Though this study represents one of the first to present data out to 7 years, its remains possible that CDR implant failure can occur at a later date. Continued surveillance of the IDE studies with long-term follow-up is necessary to validate the findings in this model.
Conclusion
One-level CDR and ACDF are both cost-effective strategies for the treatment of single-level degenerative disc disease at 7 years. The results of this model indicate, however, that CDR is the more cost-effective strategy with an ICER less than $50 000/QALY. The results of this Markov simulation were strongly supported with the use of a Monte Carlo simulation of 10 000 cycles. Further surveillance of the IDE studies is required to validate the long-term validity of this model.
Footnotes
Authors’ Note
The device discussed in this article is approved by the US Food and Drug Administration.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
