Abstract
Study design
Systematic review.
Objective
To investigate the efficacy of nonsurgical interventional treatments for chronic low back pain (LBP) caused by facet joint syndrome (FJS).
Methods
A systematic review of the literature was conducted to identify studies that compared interventional treatments for LBP due to FJS among them, with usual care or sham procedures. Studies were evaluated for pain, physical function, disability, quality of life and employment status. The RoB-2 and MINORS tools were utilized to assess the risk of bias in included studies.
Results
Eighteen studies published between January 2000 and December 2021 were included (1496 patients, mean age: 54.31 years old). Intraarticular (IA) facet joint (FJ) injection of hyaluronic acid (HA) did not show significant difference compared to IA corticosteroids (CCS) in terms of pain and satisfaction. FJ denervation using radiofrequency (RF) displayed slightly superior or similar outcomes compared to IA CCS, physical therapy, or sham procedure. IA CCS showed better outcomes when combined with oral diclofenac compared to IA CCS or oral diclofenac alone but was not superior to IA local anesthetic and Sarapin. IA platelet-rich plasma (PRP) led to an improvement of pain, disability and satisfaction in the long term compared to IA CCS.
Conclusion
FJS is a common cause of LBP that can be managed with several different strategies, including nonsurgical minimally invasive approaches such as IA HA, CCS, PRP and FJ denervation. However, available evidence showed mixed results, with overall little short-term or no benefits on pain, disability, and other investigated outcomes.
Introduction
Low back pain (LBP) represents a worldwide burden with significant epidemiological, social, and economic resonance. Indeed, it is considered among the most common musculoskeletal conditions globally 1 and the leading cause of years lived with disability2-4.
Spinal causes of LBP include myofascial pain, intervertebral disc degeneration, spinal stenosis, sacroiliac joint pain, and facet joint syndrome (FJS)5-9. Other causes include tumors, infections, deformities, neuropathic pain, inflammatory conditions (such as ankylosing spondylitis and rheumatoid arthritis) and non-spinal causes (including visceral causes as well as mood disorders). The literature has been widely focused on intervertebral discs as the source of LBP; however, facet joints (FJ) also play a major role in generating LBP.
FJS is characterized by wear and tear of joint surfaces due to degenerative processes, spondylolisthesis, septic arthritis, and systemic inflammatory phenomena. FJ osteoarthritis is the most common facet pathology. This syndrome appears to be the cause of 10-15% of chronic LBP cases in the young adult population and at even higher rates in the elderly. 10 The diagnosis of chronic LBP related to FJS involves several steps, from physical examination to provocative maneuvers together with imaging, 11 even though the correlation between clinical symptoms and degenerative spinal changes is still unclear. 12 Indeed, these examinations do not provide diagnostic certainty. A FJ nerve block with a local anesthetic can support the identification of FJs as the source of chronic LBP. 13
The first-line treatment comprises a multimodal approach involving medications (acetaminophen, non-steroidal anti-inflammatory drugs, muscle relaxants and antidepressants), physiotherapy and eventually psychotherapy.14,15 When conservative treatment is not sufficient, patients may benefit from local or intraarticular (IA) injections (using corticosteroids – CCS – and/or local anesthetics, hyaluronic acid – HA - or platelet-rich plasma – PRP -), prolotherapy (e.g., injection of a solution not containing biologic material with the goal of improving pain using dextrose or phenol) or physical (pulsed or conventional radiofrequency, cryoneurolysis) denervation techniques to eliminate FJ pain. 16 Surgical management including lumbar facet joint replacement is still controversial.17,18 Spinal fusion and decompression can be indicated as a last resort in patients with symptoms refractory to nonoperative modalities or in advanced stages with associated synovial facet cyst causing radiculopathy and/or spinal stenosis. 19
The aim of the present study was to systematically review the literature to investigate the efficacy of minimally invasive interventional procedures routinely used for the clinical management of LBP related to FJS. While previous studies have been mainly focused on a specific subset of nonsurgical interventions for FJS (e.g., FJ nerve block, rafiofrequency denervation, see below), this report offers a wide perspective on available therapeutic strategies to tackle LBP due to FJS, with the objective of providing evidence-based guidance to treatments that have been described in the literature so far.
Materials and Methods
The methods for this systematic review followed accepted standards for systematic review/comparative effectiveness reviews for rigor, quality, and transparency including those described by the Agency for Healthcare Research and Quality (AHRQ), 20 IOM Standards for Systematic Reviews, 21 and the PCORI Methods Guide. 22
Electronic Literature Search
A systematic search of PubMed/MEDLINE, Scopus, CINAHL, EMBASE, CENTRAL, and Cochrane databases was performed for literature published through January 2000 and December 1, 2021. Briefly, we sought to identify comparative studies investigating the effect of minimally invasive interventional procedures for treating LBP due to FJS. The research question was formulated using a PICOS-approach: Patient (P); Intervention (I); Comparison (C); Outcome (O) and Study design (S). The aim of this systematic review was to select those articles which described “if adult patients with LBP due to FJS (P) treated by nonsurgical minimally invasive interventional procedures (I) have better results in terms of clinical outcomes and complications (O) when treated with other approaches already in use (C)”. For this purpose, only randomized control trials (RCTs), cohort studies (CS), prospective (PS) or retrospective (RS) studies with ≥ 10 patients per group were included. Only studies on humans with abstracts written in English were considered, with no other limits placed on the search. The Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines were used to improve the reporting of the review as described by Moher et al. 23 For the search strategy, the following key words were used: “low back pain”, “zygapophyseal joint”, “facet joint", “syndrome”, “denervation”, “neurolysis”, “ablation”, “radiofrequency”, “nerve block”, “steroid”, “injections”. The keywords were used isolated or combined. The complete search strategy with the Medline string is reported as Supplementary File 1. Eventually, additional studies among the reference lists of the selected papers and systematic reviews were searched and included upon adherence with the search criteria.
Study Selection
The initial search of the article was performed by three reviewers (L.A., A.R. and M.D.F). In case of disagreements, the consensus of a third reviewer (G.P.) was asked. The research was conducted using the CADIMA software.
24
The following research order was used: titles were screened first, then abstracts and full papers. A paper was considered potentially relevant and its full text reviewed if, following a discussion between the two independent reviewers, it could not be unequivocally excluded on the basis of its title and abstract. The full text of all papers not excluded based on abstract or title was subsequently evaluated. The number of articles excluded or included were recorded and reported in a PRISMA flowchart (Figure 1). Search strategy flow diagram in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) protocol.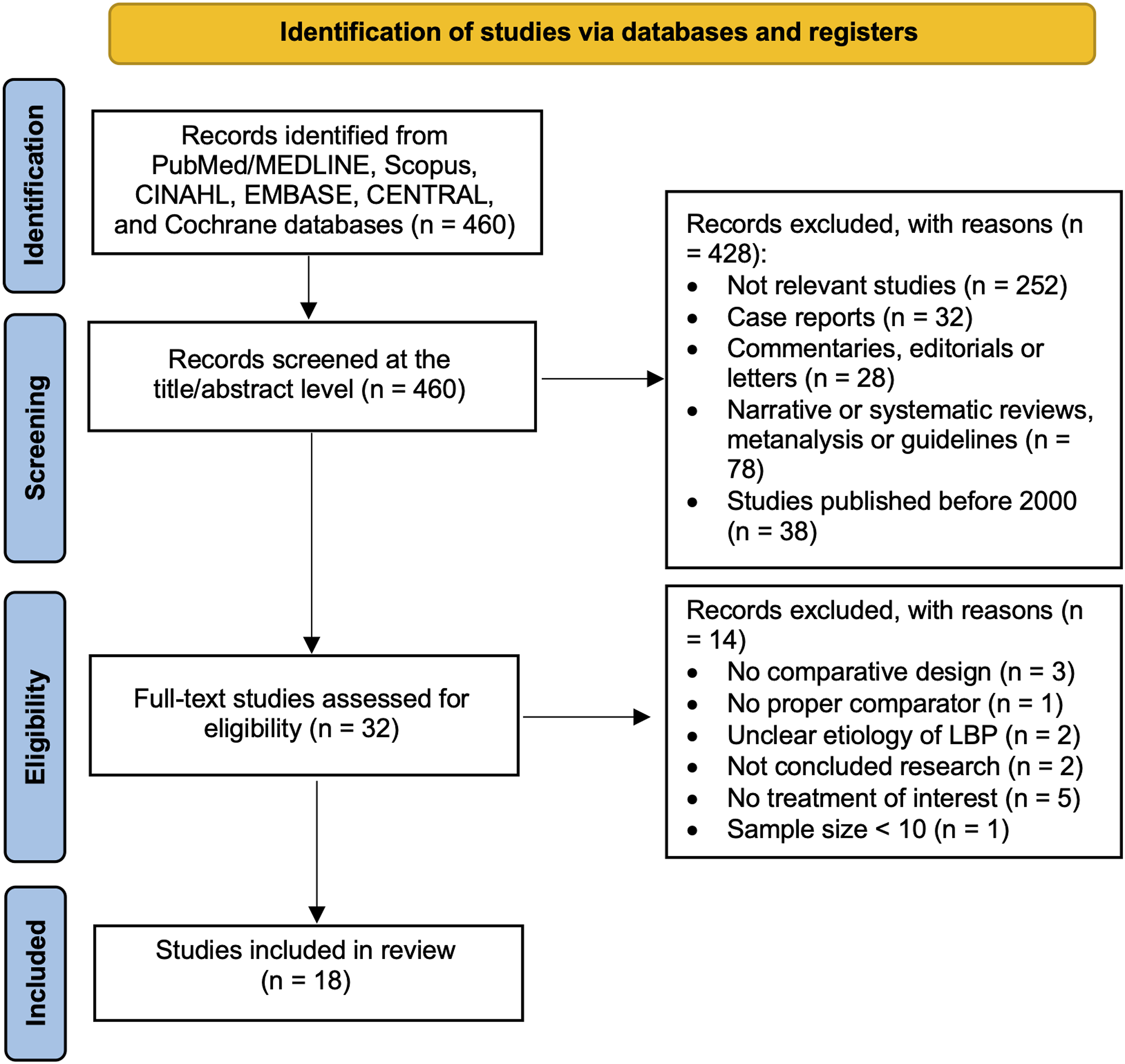
Data Extraction
General study characteristics extracted were: authors, country, sample size, mean age, mean follow-up, study design, level of evidence (LOE), year of publication, intervention and comparison procedures, interventional techniques, and patient-reported outcomes (PROs) including Visual Analogue Scale (VAS), Numeric Rating Scale (NRS), Patient Disability Questionnaire (PDQ), Odom’s criteria, North American Spine Society (NASS) patient satisfaction questionnaire, European Quality of Life 5 Dimensions (EQ-5D) questionnaire, Global Perceived Effect (GPE) scale, Oswestry Disabiliy Index (ODI), Roland-Morris Questionnaire (RMQ), RAND 36-Item Health Survey, World Health Organization (WHO) analgesics intake score, and West Haven-Yale Multidimensional Pain Inventory (MPI). In addition, complications and conclusions of the studies were reported.
Risk of Bias
Given the observational design of included studies, the Risk of Bias 2 tool (RoB-2) 25 for RCTs and the Methodological Index for Non-Randomized Studies (MINORS) 26 tool for non-randomized RCTs (NRCTs) were utilized to assess the risk of bias. In order to avoid imprecisions, selected papers were rated independently by two reviewers (F.R. and L.A.) and verified by a third one (G.P.). The “Robvis” tool was utilized to generate the traffic light plot in accordance with Cochrane recommendations.
Results
Study Selection
A total of 460 studies were found. Of these studies, 428 articles were excluded through title and abstract screening. Then, 32 full-text articles were screened. Out of these studies, 14 were excluded (no proper comparator, n = 1; unclear etiology of LBP, n = 2; intervention treatments inappropriate for the review question, n = 5; unconcluded research, n = 2; sample size < 10, n = 1, no comparative design, n = 3). After this process, 18 articles were included in our study. The review protocol has been approved by the International prospective register of systematic reviews (PROSPERO) under the ID CRD42022343162. The high heterogeneity between studies (in terms of type of intervention and comparison technique as well as outcomes) precluded an effective meta-analysis from being conducted.
Study Characteristics
Study Design and Patient Demographics of Included Studies.
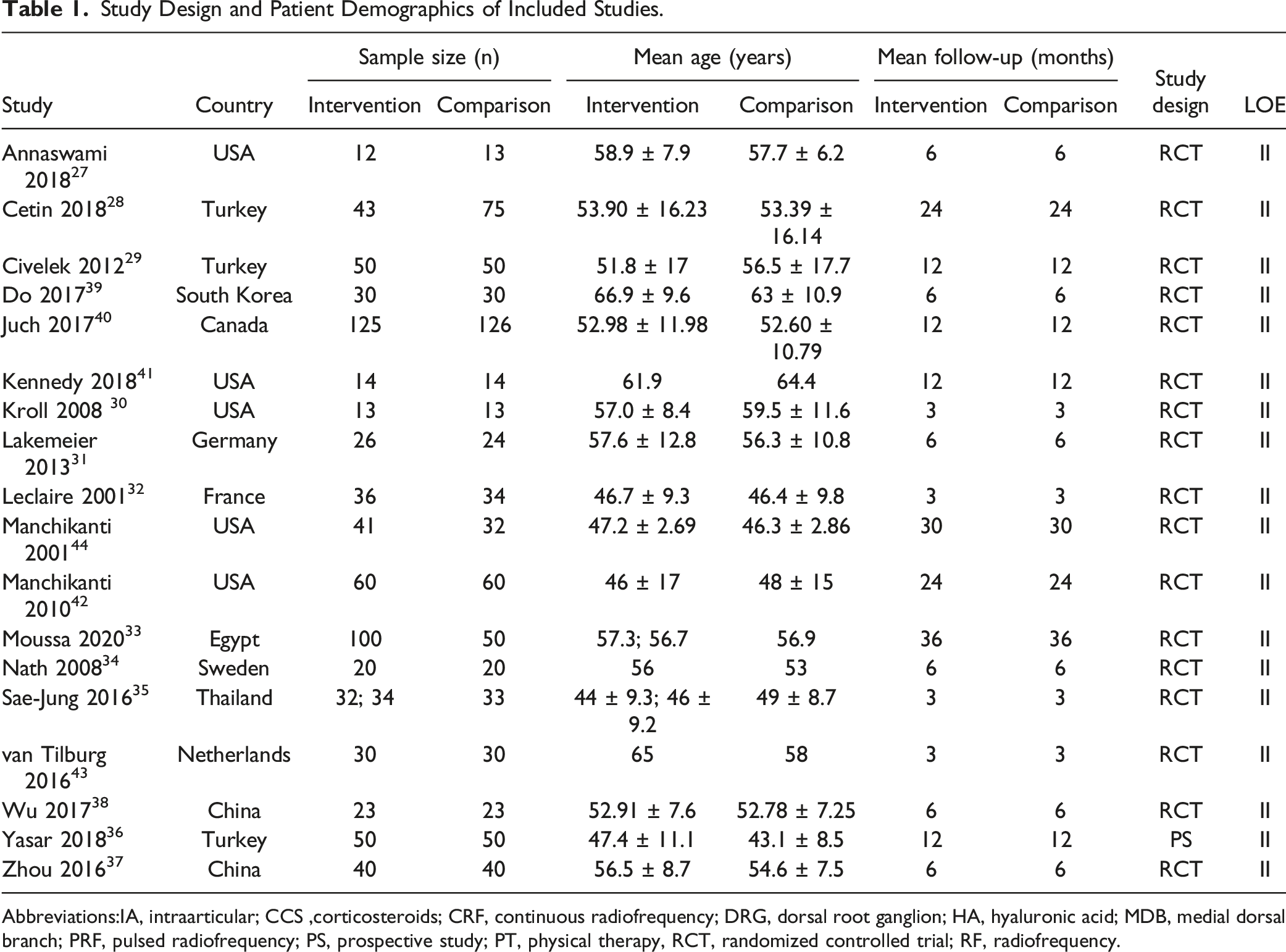
Abbreviations:IA, intraarticular; CCS ,corticosteroids; CRF, continuous radiofrequency; DRG, dorsal root ganglion; HA, hyaluronic acid; MDB, medial dorsal branch; PRF, pulsed radiofrequency; PS, prospective study; PT, physical therapy, RCT, randomized controlled trial; RF, radiofrequency.
Risk of Bias
The RoB-2 tool for RCTs and the MINORS score for NRCTs, were used to assess the risk of bias of each study. For RCTs, we found 5 studies with an overall risk of bias identified as “low”, 7 as “some concerns” and 5 as “high risk” (Figure 2). The MINORS tool was adopted to assess the quality of evidence of the only included NRCT, with a total score of 18/24 (Supplementary Table). Risk of bias as assessed by the Risk of Bias of Randomized Controlled Trials (RoB 2) tool based on the Cochrane Handbook for Systematic Reviews of Intervention.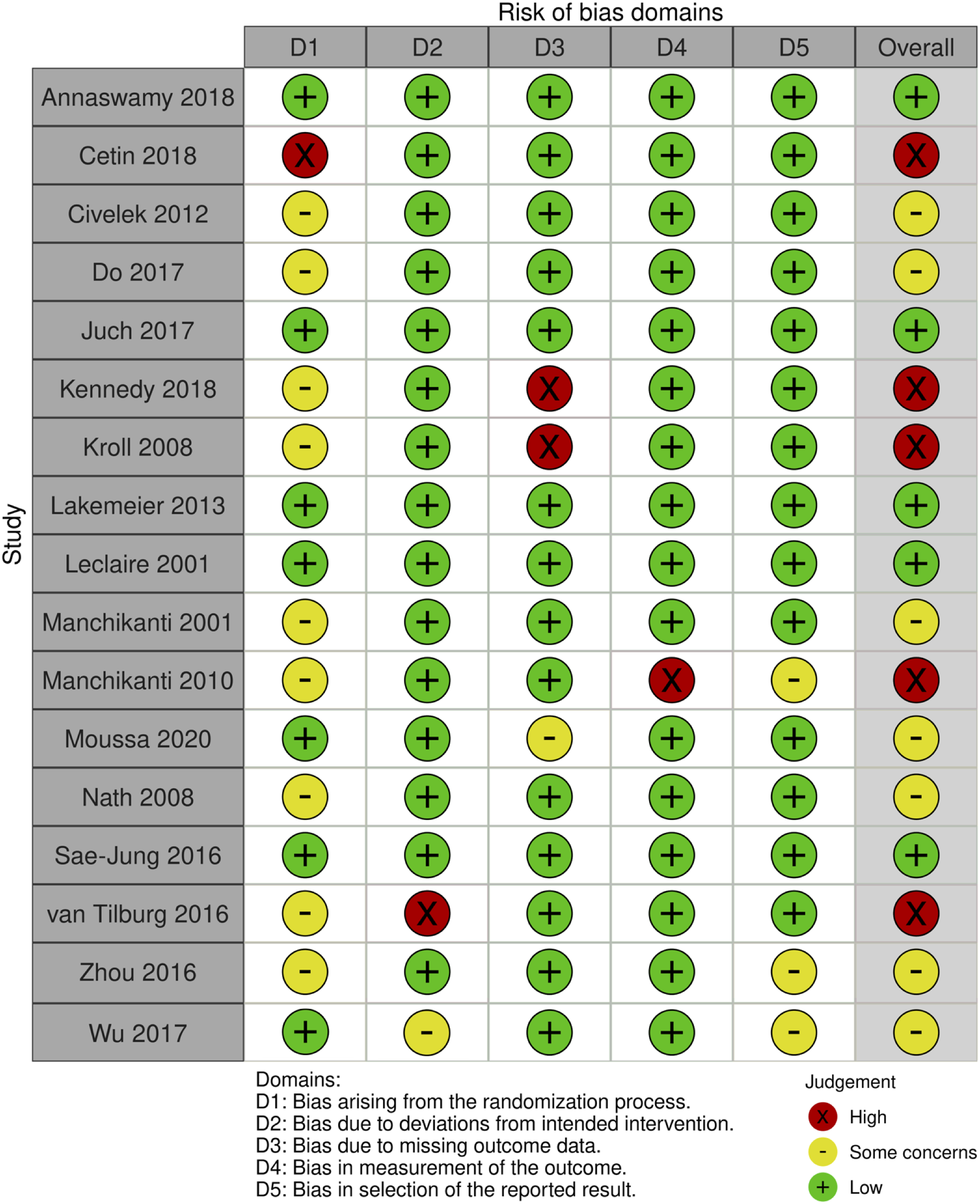
Results of Individual Studies
Intervention Characteristics of Included Studies.
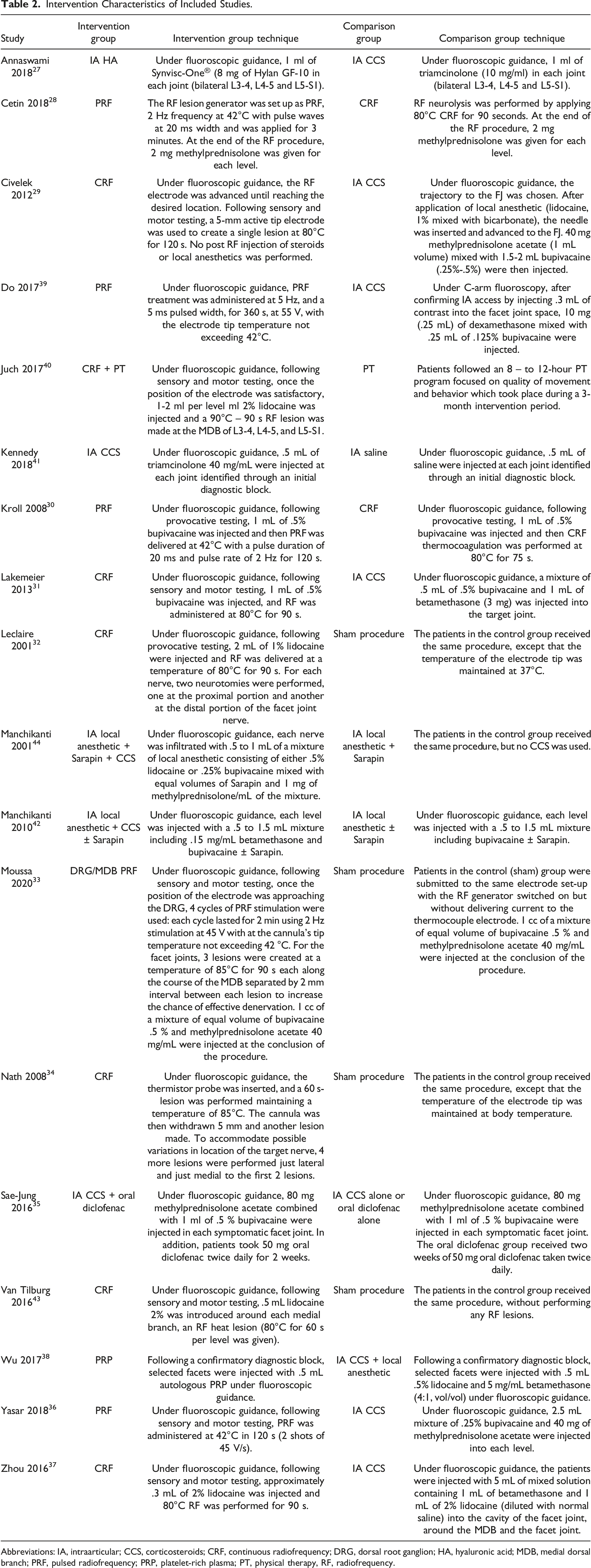
Abbreviations: IA, intraarticular; CCS, corticosteroids; CRF, continuous radiofrequency; DRG, dorsal root ganglion; HA, hyaluronic acid; MDB, medial dorsal branch; PRF, pulsed radiofrequency; PRP, platelet-rich plasma; PT, physical therapy, RF, radiofrequency.
Outcomes, Complications, and Conclusions of Included Studies.

Abbreviations: CCS, corticosteroids; CRF, continuous radiofrequency; CT, combined treatment; DRG, dorsal root ganglion; EQ-5D, European Quality of Life 5 Dimensions; FJ, facet joint; FJS, facet joint syndrome; GPE, Global Perceived Effect; HA, hyaluronic acid; IA, intraarticular; LBP, low back pain; MDB, medial dorsal branch; NRS, numeric rating scale; OD, oral diclofenac; ODI, Oswestry Disability Index; PDQ, Patient Disability Questionnaire; PRF, pulsed radiofrequency; PRP, platelet-rich plasma; PT, physical therapy, RF, radiofrequency; RMQ, Roland-Morris Questionnaire; VAS, visual analogue scale, WHO, World Health Organization.
IA HA
The study by Annaswamy et al. 27 evaluated the effect of IA hyaluronic acid (8 mg Hylan GF-10) compared to IA CCS (1 ml of 10 mg/ml triamcinolone). No significant intergroup differences in terms of VAS, PDQ and overall satisfaction at 6 months, both before and after the treatment, were reported. Regarding within-group analyses, the only statistically significant change noted was in the HA group, which showed an improvement of VAS only at the 1 month-time point compared to baseline (70 ± 20 vs 45 ± 25, P = .008). Regarding PDQ scores, patients in the CCS group showed a statistically significant improvement only at 1 month (100 ± 23 vs 77 ± 30, P = .009) whereas patients in the HA group showed significant improvements from baseline (102 ± 28) at all time points: 1 month (74 ± 34, P = .002), 3 months (74 ± 36, P = .037) and 6 months (52-99.5, P < .001). Failed injection was reported in 3/13 patients in the CCS group and 1/12 patients in the HA group, while 3/13 and 2/12 reported a transient LBP increase in the former and latter groups, respectively.
RF FJ Denervation
The efficacy of pulsed radiofrequency (PRF) compared to continuous radiofrequency (CRF) facet joint denervation was assessed in 2 studies.28,30
Cetin et al. 28 demonstrated that both PRF and CRF decreased VAS score compared to baseline (8.44 ± .93 and 8.10 ± .95, respectively). However, CRF was associated with lower pain scores compared to PRF at 1 month (CRF: 3.27 ± 1.24; PRF: 3.79 ± 1.05, P = .027), 3 months (CRF: 3.39 ± 1.04; PRF: 4.09 ± 1.11, P = .001) and 6 months (CRF 3.79 ± .67; PRF: 5.66 ± 1.14:, P = .001), while VAS scores were higher in the CRF group at 1 year (CRF: 4.04 ± .81; PRF: 3.69 ± .86, P = .001) and 2 years (CRF: 4.09 ± .78; PRF: 5.20 ± 1.06, P = .001). In terms of satisfaction, the Odom’s criteria reported excellent/good outcomes in 38/43 patients in the CRF group and 8/75 patients in the PRF group, while fair/poor results were reported in 4/43 and 67/75 patients treated with CRF and PRF, respectively. Furthermore, neuropathic pain developed in 1 patient within the CRF group and 2 patients within the PRF group.
Conversely, Kroll et al. 30 reported no significant intergroup differences in terms of pain and disability before and after the treatments. Within the CRF group, a significant improvement of VAS and ODI scores were found at 3 months compared to baseline (51.9 ± 27.4 vs 76.2 ± 16 and 41.7 ± 16.9 vs 52 ± 17.3, respectively), while no difference was retrieved in the PRF group.The efficacy of RF compared to locally injected CCS was assessed in 5 studies.29,31,36,37,39
In the study from Civelek et al. 29 , both groups showed a significant improvement of mean VAS scores vs the baseline (CCS: 8.5; CRF: 8.2) at all timepoints. While VAS was lower in the CCS group soon after the procedure (CCS: 1.2; CRF: 2.4, P = .00), pain scores were significantly decreased in the CRF group at 1 month (CCS: 3.4; CRF: 2.2, P = .00), 6 months (CCS: 4.4; CRF: 2.5, P = .00) and 12 months (CCS: 4.9; CRF: 2.6, P = .00). Conversely, no significant intergroup difference was found in terms of EQ-5D and NASS scores, except for the latter at 12 months (CCS: 2; CRF: 1.5, P = .04). Burning-like dysesthesias were reported in 2 patients in the CRF group.
Lakemeier et al. 31 reported that, while both groups showed an improvement of VAS, RMQ and ODI following the procedure, no significant intergroup difference in any score was reported at the 6-month follow-up.
Yasar et al. 36 found that VAS scores decreased compared to baseline in both groups (CCS: 6.4 ± .9; PRF: 5.8 ± 1) and were significantly lower in the PRF group at 3 months (CCS: 3.3 ± 1; PRF: 2.5 ± 1, P < .05) and 6 months (CCS: 3.3 ± .9; PRF: 2.3 ± 1.4, P < .05). However, no statistically significant difference was found at 9 months (CCS: 2.5 ± 1; PRF: 2.7 ± .9) and 12 months (CCS: 3 ± 1.5; PRF: 3 ± 1.1). Similarly, ODI improved from preoperative (CCS: 57.2 ± 13.9; PRF: 58.5 ± 13.1) and was significantly lower in the PRF group at 3 (CCS: 24.1 ± 8.7; PRF: 18.9 ± 5.7, P < .05) and 6 months (CCS: 24.8 ± 9.5; PRF: 14.9 ± 7, P < .05), while scores were comparable at subsequent follow-ups (9 months: CCS: 12.2 ± 3.8; PRF: 10.4 ± 2.8;12 months: CCS: 12.1 ± 4.4; PRF: 17.2 ± 6.4). Post-injection pain was experienced in 4% of study participants.
Zhou et al. 37 reported that mean VAS score significantly decreased from baseline (CCS: 6.8; CRF: 6.7) in both groups, but no intergroup difference was found at 30 minutes (CCS: 3.6; PRF: 3.5), 1 day (CCS: 2.7; PRF: 2.7) and 1 week (CCS: 1.9; PRF: 1.4) following the procedure. Conversely, VAS was significantly lower in the PRF group after 1 month (CCS: 3.6; PRF: 1.4, P < .05) and 12 months (CCS: 5.8; PRF: 1.7, P < .05). With regards to the mean Schober index, no significant difference was reported compared to the baseline (CCS: 6.85 CRF: 6.6) at any timepoint and between groups at 1 week (CCS: 8.9; PRF: 9), while it was significantly higher in patients treated with PRF after 1 month (CCS: 7.3; PRF: 8.6, P < .01) and 12 months (CCS: 6.2; PRF: 8.8, P < .05). Considering the patient-reported half-year efficacy assessed with the Surgical Efficacy Criteria of the Spine Surgery Group (Orthopedic Branch of Chinese Medical Association), an excellent outcome was reached by 12.5% patients in the CCS group and 62.5% in the PRF group; a good outcome in 30% of the patients treated with CCS and 27.5% with PRF; an eligible outcome in 5% and 7.5%, respectively; a poor outcome in 52.5% in the former and 2.5% in the latter.
In the study by Do et al., 39 NRS scores significantly improved compared to the baseline (CCS: 5.0 ± .8; PRF: 4.9 ± .8) at all timepoints after the procedure. However, NRS was significantly lower in the CCS group at 2 weeks (CCS: 1.4 ± .8; PRF: 2.3 ± 1.4, P < .05) and 1 month (CCS: 1.8 ± 1.2; PRF: 2.5 ± 1.4, P < .05), but not at later follow-ups (2 months: CCS: 2.9 ± 1.4, PRF: 2.5 ± 1.3; 6 months: CCS: 3.2, PRF: 2.7 ± 1.5). In the CCS group, 1 patient developed hyperglycemia (> 300 mg/dL).
Juch et al. 40 compared the efficacy of CRF combined with physical therapy (PT) vs PT alone. Authors reported that the satisfaction rate calculated with GPE in the CRF group was significantly higher at 3 (PT: 4.95%, CRF: 29.63%, P < .001) and 6 weeks (PT: 9.32%; CRF: 29.41%, P = .005), but not at other timepoints (3 months: PT: 23.68%, CRF: 26.13%; 6 months: PT: 36.11%, CRF: 40.70%; 9 months: PT: 40%, CRF: 38.67%; 12 months: PT: 39.22%, PRF: 42.71%. In addition, no significant intergroup differences in terms of NRS, RAND-36, EQ-5D, MPI and ODI were retrieved.
The effect of RF FJ denervation was compared to a sham control in 4 studies.32-34,43
Leclaire et al. 32 showed that patients in the CRF group reported a significant improvement of ODI (sham: 34.4; CRF: 35.6, P < .05) and RMQ (sham: 49.5; CRF: 44.5, P < .05) at 1 month compared to baseline (ODI: sham: 36.4 ± 14.6, CRF: 38.3 ± 14.7; RMQ: sham: 51.6 ± 22.8; CRF: 52.9 ± 18.2) but not at 3 months (ODI: sham: 33.7, CRF: 33.6; RMQ: sham: 44.4, CRF: 43.1). Similarly, no significant intergroup differences in terms of VAS, low back mobility (flexion, extension, side-bending, and rotations, maximal strength against resistance, and angular speed against 25% of the maximum strength resistance) and return to work were encountered.
Likewise, van Tilburg et al. 43 reported a decrease of pain scores following the treatment but no significant differences between the two groups regarding NRS and GPE.
In their study, Moussa et al. 33 compared PRF of the dorsal root ganglion (DRG) or the medial dorsal branch (MDB) with a sham procedure. Patients undergoing DRG RF showed a significantly higher improvement of VAS scores at all timepoints compared to other groups (3 months: PRF: 8.3 ± 1 (DRG), 5.2 ± 1 (MDB), sham: 5.1 ±1.3, P = .047; 6 months: PRF: 8.1 ± 1.2 (DRG), 5 ± 1.1 (MDB), sham: 2.1 ± .4, P = .031; 1 year: PRF: 7.9 ± 1 (DRG), 4.8 ± 1 (MDB), sham: .6 ± .4, P = .008; 2 years: PRF: 7.7 ± .7 (DRG), 2.1 ± .5 (MDB), sham: .5 ± .2, P = .006; 3 years: PRF: 7.5 ± 1 (DRG), 2 ± .6 (MDB), sham: .3 ± .2, P = .003). Similarly, mean changes of ODI scores were significantly higher in the DRG group compared to the others (3 months: PRF: 50.5 (DRG), 34.9 (MDB), sham: 33.6, P = .048; 6 months: PRF: 48.1 (DRG), 30.3 (MDB), sham: 10.8, P = .032; 1 year: PRF: 43.9 (DRG), 26.4 (MDB), sham: 5.5, P = .011; 2 years: PRF: 39.3 (DRG), 15.3 (MDB), sham: 3.7, P = .008; 3 years: PRF: 39.2 (DRG), 6.3 (MDB), sham: 2, P = .004). Patients receiving DRG RF also reported a higher rate of GPE improvement (> 50%) when compared to MDB RF and sham groups (3 months: PRF: 80 (DRG), 60 (MDB), sham: 50, P = .048; 6 months: PRF: 74 (DRG), 42 (MDB), sham: 12, P = .035; 1 year: PRF: 70 (DRG), 30 (MDB), sham: 4, P = .017; 2 years: PRF: 64 (DRG), 10 (MDB); sham: 2, P = .005; 3 years: PRF: 62 (DRG), 6 (MDB), sham: 0, P = .001). In addition, a more consistent mean reduction of the WHO analgesics intake score was recorded in patients following RF, especially in the DRG group (3 months: PRF: 2.4 (DRG), 2 (MDB); sham: 2, P = .042; 6 months: PRF: 2.3 (DRG), 1.9 (MDB); sham: .8, P = .035; 1 year: PRF: 2.1 (DRG), 1.8 (MDB), sham: .2, P = .03; 2 years: PRF: 2 (DRG), .6 (MDB); sham: .1, P = .007; 3 years: PRF: 1.9 (DRG), .5 (MDB), sham: .1, P = .003).
Nath et al. 34 reported a more consistent reduction of VAS scores in the CRF group at 6 months compared to baseline (CRF: 4.10 vs 6.03; sham: 3.98 vs 4.35), and this difference was statistically significant (P = .02). Similarly, patients in the CRF group displayed a significant improvement of 6-point scales regarding analgesic intake (CRF: 2.55 vs 3.95; sham: 3.2 vs 3.80, P = .04) and subjective global assessment (CRF: 2.77 vs 3.85; sham: 3.05 vs 3.35, P = .004) at the same timepoint.
IA CCS
Kennedy et al. 41 compared the injection of IA CCS (20 mg triamcinolone) with IA saline. As > 75% of enrolled patients in both groups underwent RF neurotomy before the first scheduled follow-up at 6 weeks, no data pertaining NRS and ODI were presented. However, no statistically significant difference in either the need to progress to or time to RF was reported in any group.
Manchikanti and colleagues 44 evaluated the effect of an IA injection of a mixture of methylprednisolone, lidocaine or bupivacaine and Sarapin compared to the same preparation without CCS. No significant intergroup differences in terms of pain, overall health, narcotic intake, and employment status were encountered. In another study, the same authors utilized a blend of bupivacaine and betamethasone with or without Sarapin compared with bupivacaine alone or mixed with Sarapin. 42 Similar to their previous study, no statistically significant difference regarding NRS, ODI, narcotic assumption and employment was reported between groups.
Sae-Jung et al. 35 compared the efficacy of IA CCS (80 mg methylprednisolone) and oral diclofenac (50 mg twice daily for 2 weeks) vs oral diclofenac (OD) or IA CCS alone. Patient receiving the combined treatment (CT) showed a higher improvement of ODI compared to other groups at 1 month (OD: 30.1 ± 8.1; CCS: 20.2 ± 8, CT: 15.1 ± 5.5; P < .001 CT vs OD; P = .02 CT vs CCS) and to the OD group at 3 months (OD: 42.4 ± 9; CCS: 32.2 ± 15.6, CT: 26.2 ± 11.7, CT vs OD, P < .001 CT vs OD; P = .15 CT vs CCS). Likewise, VAS scores were significantly lower in the CT group compared to the OD group (but similar to CCS alone) at 1 month (OD: 5.3 ± 1.4; CCS: 3.6 ± .7, CT: 3.3 ± 1.1; P < .001 CT vs OD; P = .76 CT vs CCS) and 3 months (OD: 42.4 ± 9, CCS: 32.2 ± 15.6, CT: 26.2 ± 11.7, P = .002 CT vs OD; P = .08 CT vs CCS). 12% of patients receiving OD presented dyspepsia, while post-injection discomfort was reported in 44% of patients following IA CCS.
IA PRP
Wu et al. 38 conducted a prospective RCT comparing IA FJ injection of .5 mL autologous PRP with .5 ml CCS and local anesthetic (.4 mL .5% lidocaine and .1 mL 5 mg/mL betamethasone). Significantly lower VAS scores were encountered in both groups at all timepoints (1 week, 1 month, 2 months, 3 months and 6 months, P < .01) except immediately following injection. VAS decreased more consistently in the group treated with CCS and lidocaine at 1 week and 1 month (P < .01), although the group treated with PRP showed a better pain relief at 3 and 6 months (P < .01). The ODI and RMQ showed a similar trend, with higher scores at earlier timepoints in the former group (P < .01) and at later follow-up intervals in the latter (P < .01). Regarding satisfaction, modified MacNab criteria showed no significant intergroup difference except at 6 months, when the number of patients in the PRP group declaring “excellent” (52.38%) and “good” (28.57%) outcomes were significantly higher compared to individuals in the other group (20% and 30%, respectively; P < .05). The 50% pain improvement threshold was reached by 85% of patients treated with CCS and lidocaine at 1 week and 1 month, while it was reported by 80.95% in the PRP group at 3 and 6 months. Furthermore, 40% of patients in the former group used acetaminophen for pain relief, whereas only 23.81% utilized in the PRP group.
Discussion
LBP represents a major global problem affecting more than 500 million people, being the leading global cause of disability. FJS is often a misdiagnosed and inadequately treated condition but constitutes a common source of LBP that must be properly addressed. Conservative treatment can provide a valid alternative for the treatment of FJS. In this regard, several minimally invasive nonsurgical procedures have been developed to deliver specific therapeutics in proximity of FJs.
For selected studies with FJS associated with chronic LBP, the use of these approaches overall produces a significant long-term improvement in pain and functional scores, although with considerable differences among the different techniques. CCS play a dual role in the management of pain in osteoarthritis, as they interrupt nociceptive inputs at central and peripheral level and mitigate the pro-inflammatory environment in affected joints. 45 In our analysis, we included 4 studies evaluating FJ injections with CCS compared to a control group without CCS.35,41,42,44 Among these, 1 study reported a lack of efficacy in reducing the need for subsequent RF, 41 while other 2 studies showed no significant intergroup differences in terms of pain, overall health, analgesics intake and employment status.42,44 Only 1 study reported a significant improvement of VAS and ODI at 1 and 3 months with the use of IA CCS, although in combination with oral diclofenac. 35
Overall, clinical outcomes after FJ block appear very heterogeneous, and may be affected by the use of different steroids and local anesthetics in the treatment and control groups. Furthermore, all reported studies presented a short follow-up with a maximum of 6 months. Given these limitations, it is difficult to assess the role of FJ injections in the management of FJ-induced LBP and the level of evidence remains low (III).
Compared to CCS, HA has a different therapeutic activity, as it aims to rebalance the elastic and viscous properties of the synovial fluid. 46 One study showed the efficacy of IA with HA in providing an overall VAS improvement of 42% at 6 months, without any significant difference compared with CCS, although HA resulted in a significant functional benefit at 6 months. 27 The clinical efficacy of HA has been tested in other clinical settings, such as knee osteoarthritis. 47 However, the efficacy of HA may be limited by the anatomy of FJs, since this compound is injected a restricted intracapsular anatomical space and it is difficult to establish how much actually reaches the joint surface between the two articular processes in order to produce clinical benefits.
The use of PRP for FJS was reported only in 1 study. 38 PRP is a blood derivative mainly containing a large number of growth factors, including including platelet-derived growth factor (PDGF), transforming growth factor-β (TGF-β), fibroblast growth factor (FGF), insulin-like growth factor 1 (IGF-1), connective tissue growth factor (CTGF), and epidermal growth factor (EGF), as well as additional bioactive factors that have shown anabolic and anti-inflammatory properties in several tissues, including cartilage, tendon and the intervertebral disc. 48 Therefore, it is widely used to treat an increasing range of degenerative musculoskeletal disorders, especially osteoarthritis and tendinopathies. 49 In the RCT conducted by Wu et al., 38 PRP was compared to CCS and a local anesthetic. Although LBP transiently increased in both groups following injection, pain and disability improved more significantly in the latter group at earlier timepoints (until 2 months), while patients receiving PRP showed better oucomes at later follow-ups (3 and 6 months). Similarly, the satisfaction rate at 6 months was significantly higher in patients in the PRP group. These differences may indicate that CCS are effective in the short term, whereas PRP may present a longer duration efficacy.
Regarding the use of RF ablation, out of the 12 studies included in this analysis, 7 studies demonstrated both short-term and long-term efficacy,28-30,33,34,36,37 2 trials revealed only short-term efficacy,31,39 whereas 3 studies showed a lack of effectiveness.32,40,43 Four studies compared RF ablation with a sham group: in the study by Nath et al., 34 RF was associated with a significant decrease of VAS and analgesic intake at 6 months, while Leclaire 32 and van Tilburg 43 did not find any significant difference in pain scores, ODI and patient satisfaction. However, RF ablation may have different outcomes depending on the nerve structures undergoing neurolysis. Not by chance, Moussa and colleagues 33 investigated two different RF targets, showing that RF performed on the dorsal root ganglion had the best outcomes in term of VAS and ODI at all timepoints compared to RF targeting the medial dorsal branch. Another factor which may influence the efficacy of nerve ablation is the modality of RF, which include continuous (traditional) or pulsed RF, and this has been investigated by 2 of the included studies,28,30 although with different results. Cetin et al. 28 found a better short-term VAS using continuous RF, while better long-term pain scores were showed using pulsed RF. On the contrary, Kroll and colleagues 30 did not find any significant difference between groups.
Five studies compared RF for FJ denervation with CCS IA injections 29,31,36,37,39: most of these found comparable short-term pain and functional outcomes, although RF was demonstrated to be superior at 6 and 12 months in 3 studies,29,36,37 while 2 studies did not found long-term significant differences between groups.31,39 However, despite the better long-term outcome related to the use of RF, 2 cases of postprocedural neuropathic pain have been described by Civelek et al., 29 while other 3 cases by Cetin and colleagues. 28 On the other hand, adverse events of IA CCS may include transient hyperglycemia.39,50
This systematic review revealed a level II evidence for short-term effectiveness of 6 months or less and for long-term effectiveness of 6 months or longer about the use of RF for chronic LBP due to FJS. These results confirm the recommendations from the American Society of Interventional Pain Physicians (ASIPP) guidelines for FJ interventions. 51
This review has several limitations. The inclusion of patients with different comorbidities may have introduced a bias in the evaluation of the results, as pain originating from structures other than FJs may respond differently to the injection therapy. Moreover, previous spinal surgery did not represent an exclusion criterion for most of the included studies. This may represent a source of bias, since surgical complications, such as adhesions, hematomas, bony and soft tissues compressions may lead to LBP, which might occasionally confuse the diagnosis. Furthermore, while statistically significant differences were effectively reported in most studies, mean changes in outcome measures did not always reach clinical significance. For example, the minimal clinically important differences (MCID) of VAS and ODI scores, reportedly 2 52 and 12.88 53 respectively, have not been met by most studies reporting statistically significant improvements of such outcome measures.28,29,32,34-36 Therefore, results of these studies should be interpreted with further caution. Finally, given the lack of quantitative data regarding the IA drugs used for FJ injections, studies reporting outcomes from combined treatments were considered in this review. For the same reason, the heterogeneity of the data analyzed prevented a meta-analysis to be performed.
Conclusion
FJS is a common cause of LBP that can be managed with several different strategies. In the last decade, a growing number of minimally invasive approaches has been developed to either reduce FJ inflammation, enhance joint lubrication, directly target FJ sensory innervation, or a combination of these. According to our analysis, the use of these techniques has provided mixed results, with overall little short-term or no benefits on pain, disability, and other investigated outcomes. This can be partly explained by the substantial heterogeneity affecting utilized techniques and included populations as well as by the complexity of LBP, which may present several pain generators other than FJs alone.
Supplemental Material
Supplemental material - Interventional Minimally Invasive Treatments for Chronic Low Back Pain Caused by Lumbar Facet Joint Syndrome: A Systematic Review
Supplemental material for Interventional Minimally Invasive Treatments for Chronic Low Back Pain Caused by Lumbar Facet Joint Syndrome: A Systematic Review by Luca Ambrosio, Gianluca Vadalà, Fabrizio Russo, Giuseppe Pascarella, Sergio De Salvatore, Giuseppe F. Papalia, Alessandro Ruggiero, Marta Di Folco, Massimiliano Carassiti, Rocco Papalia, and Vincenzo Denaro in Global Spine Journal
Supplemental Material
Supplemental material - Interventional Minimally Invasive Treatments for Chronic Low Back Pain Caused by Lumbar Facet Joint Syndrome: A Systematic Review
Supplemental material for Interventional Minimally Invasive Treatments for Chronic Low Back Pain Caused by Lumbar Facet Joint Syndrome: A Systematic Review by Luca Ambrosio, Gianluca Vadalà, Fabrizio Russo, Giuseppe Pascarella, Sergio De Salvatore, Giuseppe F. Papalia, Alessandro Ruggiero, Marta Di Folco, Massimiliano Carassiti, Rocco Papalia, and Vincenzo Denaro in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
