Abstract
Study Design
Basic research
Objective
To prepared 3 DNPM/chitosan hybrid hydrogels and chose the best DNPM/chitosan hybrid hydrogel for NP tissue engineering.
Methods
Three DNPM/chitosan hybrid hydrogels were fabricated by changing the ratio of the decellularized NP matrix to chitosan and crosslinking with genipin. NP stem cells (NPSCs) were cultured on the hybrid hydrogels and their proliferation, morphology, and gene expression were evaluated. Finally, an in vivo experiment was performed to evaluate the immune response to the hydrogels.
Results
The adhered NPSCs proliferated well on the hybrid hydrogel. The gene expression of NP-related collagen type II, aggrecan, and Sox-9 from NPSCs cultured on DNPM/chitosan hybrid hydrogel-1 was greater than from cells cultured on DNPM/chitosan hybrid hydrogel-2 and DNPM/chitosan hybrid hydrogel-3. Few inflammatory cells were observed during the in vivo experiment with DNPM/chitosan hybrid hydrogel-1.
Conclusions
DNPM/chitosan hybrid hydrogel-1 is a potential candidate scaffold for NP tissue engineering.
Keywords
Introduction
Degenerative disc disease has a high incidence rate and high disability rate. 1 Intervertebral disc degeneration could lead to intervertebral disc herniation, spinal canal stenosis, or degenerative spondylolisthesis, seriously affecting patient quality of life. Surgical treatments include a discectomy, or interbody fusion and fixation when conservative treatment is ineffective. 2 However, these treatments only temporarily alleviate the symptoms and do not offer a cure, resulting in the potential for relapse. Additionally, the treatments can accelerate the degeneration of adjacent segments, leading to a poor long-term result. Therefore, it is necessary to explore ideal reconstructions and repair measures to avoid or delay the degeneration of intervertebral discs. 3 In recent years, tissue engineering has become a research hotspot in the field of intervertebral disc degeneration, and it is expected to provide an ideal treatment. 4
Seed cells play an important role in nucleus pulposus (NP) tissue engineering, and NP cells and chondrocytes have been used as seed cells.5,6 Risbud et al 7 first reported NP stem cells (NPSCs) in human NP. The NPSCs have stem cell potential and unique advantages, such as better adaptability to the microenvironment of the intervertebral disc and strong chondrogenic differentiation potential, which makes them ideal seed cells for the regeneration and repair of degenerative intervertebral disc NP.
Hydrogels are the most suitable scaffold for NP tissue engineering because their rheological and hydrophilic properties are similar to native NP.8,9 Sun et al 10 successfully constructed tissue-engineered NP in vitro using nano peptide hydrogel scaffolds and rabbit NP cells. However, the extra cellular matrix (ECM)synthesis hould be tailored to enhance ECM synthesis for NP tissue engineering.
In recent years, decellularized matrix scaffolds have been used in tissue engineering. The decellularized NP matrix (DNPM) was expected to be a new scaffold material for NP tissue engineering because it provides a biological environment similar to native NP for seed cells. However, the hydrogel generally has poor mechanical properties, which limits its application. 11 Chitosan could make up for this disadvantage. 12 Additionally, the structure of chitosan is similar to glycosaminoglycan, the main component of the NP.13,14 We previously demonstrated a DNPM/chitosan hybrid hydrogel manufactured with genipin as a promising tissue-engineered NP scaffold. 15 However, the optimal ratio of decellularized DNPM and chitosan has not been identified.
In this study, we prepared 3 DNPM/chitosan hybrid hydrogels by changing the ratio of DNPM and chitosan. We then characterized the 3 hybrid hydrogels, investigated the NP-related gene expression of collagen II, aggrecan, and Sox-9 from NPSCs on the hydrogels, and evaluated the inflammatory reaction of the hydrogels in vivo.
Materials and Methods
Preparation of the DNPM/Chitosan Hybrid Hydrogels
Three kinds of DNPM/Chitosan hybrid hydrogel.

Rheological Properties
The hybrid hydrogels were placed in phosphate buffer saline (PBS) for 3 days to achieve homogenization. To evaluate the rheological properties of the hydrogel, water was placed on the surface of the hydrogel and immediately wiped off. The hydrogel sample was then characterized using a rheometer (MCR301, Anton Paar, Austria). The complex modulus of the hydrogel was measured in the linear viscoelastic region at a constant strain of 1% and different frequencies (1, 10, and 100 rad/s).
Atomic Force Microscope Test
A small amount of PBS was added to the hybrid hydrogel to keep the hydrogel moist. The surface microstructure of the 3 hybrid hydrogels were analyzed using an atomic force microscope (SPM-9700, Shimadzu Co Ltd, Japan).
Scanning Electron Microscopy
The DNPM/chitosan hybrid hydrogels were sputter-coated with gold and imaged using a scanning electron microscope (SEM; S-4800, Hitachi Co Ltd, Japan) at an accelerating voltage of 3 kV.
Contact Angle Test
The contact angles of the DNPM/chitosan hybrid hydrogels were measured with a deionized water bubble method using a SDC-100S contact angle tester (Dingsheng Precision Instrument Company, China). Five samples were chosen from each type of hydrogel, and the average contact angle was calculated.
Cell Proliferation Test
Rat NPSCs were harvested as previously described 16 and they were seeded on the DNPM/chitosan hybrid hydrogels at a density of 2000 cells per well in a 24-well plate. One, 3, 5, and 7 days after inoculation, the cell proliferation was detected using a CCK-8 assay. The NPSCs cultured on the DNPM/chitosan hybrid hydrogels and tissue culture plates were washed twice with a PBS solution, and then incubated with 200 μL PBS and 20 μL of a MTS solution for 4 hours at 37°C. The well absorbance was measured at 450 nm using a microplate reader, and 5 replicate wells were evaluated for each group at each time point.
Morphological Observation
The NPSCs were seeded on the 3 DNPM/chitosan hybrid hydrogels at a density of 5000 cells per well on a 24-well plate. FITC-phalloidin and 4′,6―diamidino-2―phenylindole (DAPI) staining were used to observe the cytoskeletal and nuclear morphology of the NPSCs after 3 days of inoculation. 15 The cellular morphology was observed using an inverted immunofluorescence microscope (Zeiss Axiovert 200, Carl Zeiss Inc, Thornwood, NY, USA).
Gene Expression Analysis
The Sequence of the genes.

Evaluation of Degradation in a Subcutaneous in Vivo Model
To evaluate the degradation in vivo, the DNPM/chitosan hybrid hydrogels were cut into .5 cm diameter circles and implanted in the subcutaneous tissue of the backs of 4 rats. After 4 weeks, implants were harvested and stored in a 2.5% glutaraldehyde solution. The samples were then prepared for hematoxylin and eosin (H&E) staining and imaged with a Axiovert 200 inverted phase-contrast microscope (Carl Zeiss, Germany).
Results
DNPM/Chitosan Hybrid Hydrogel Characterization
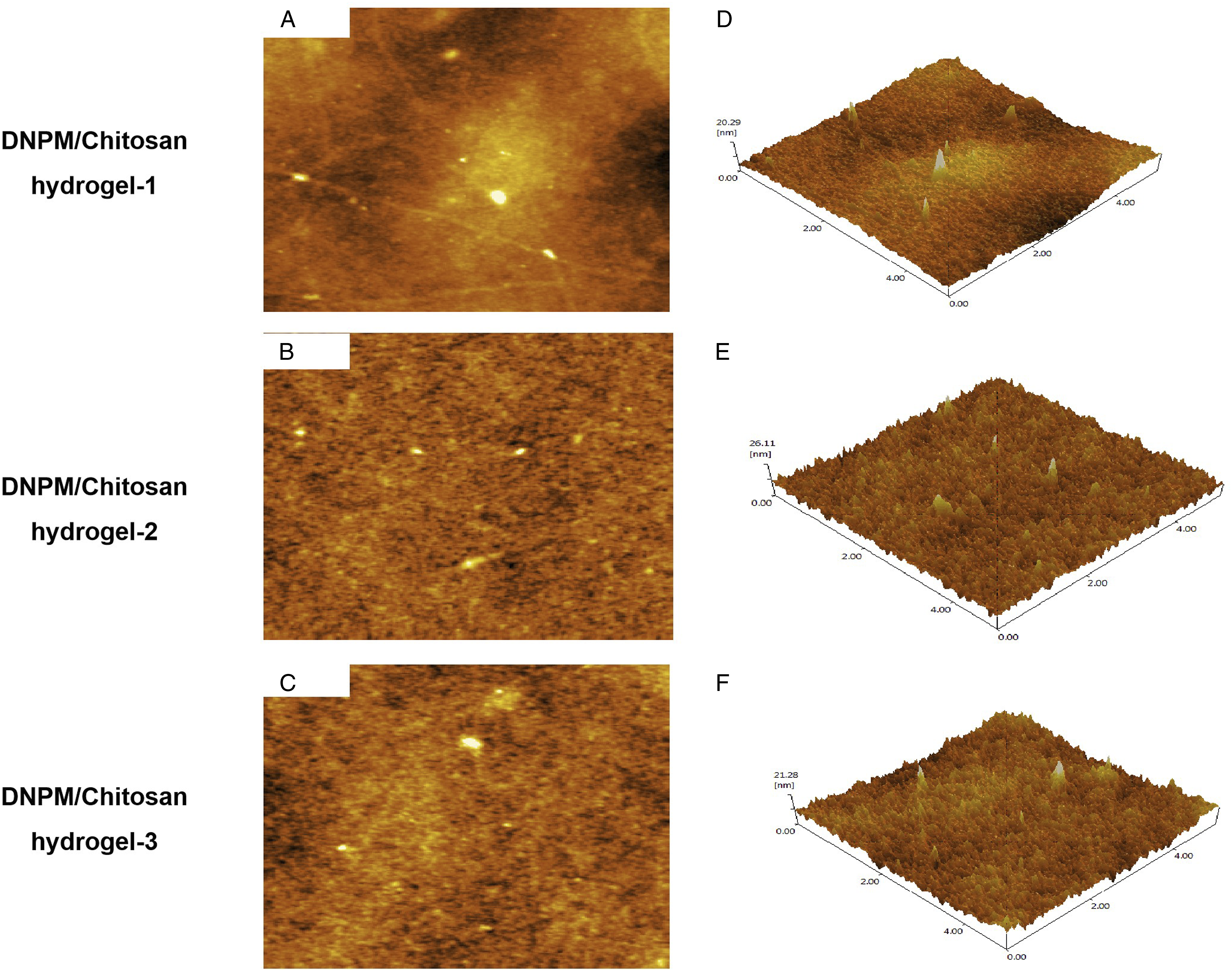
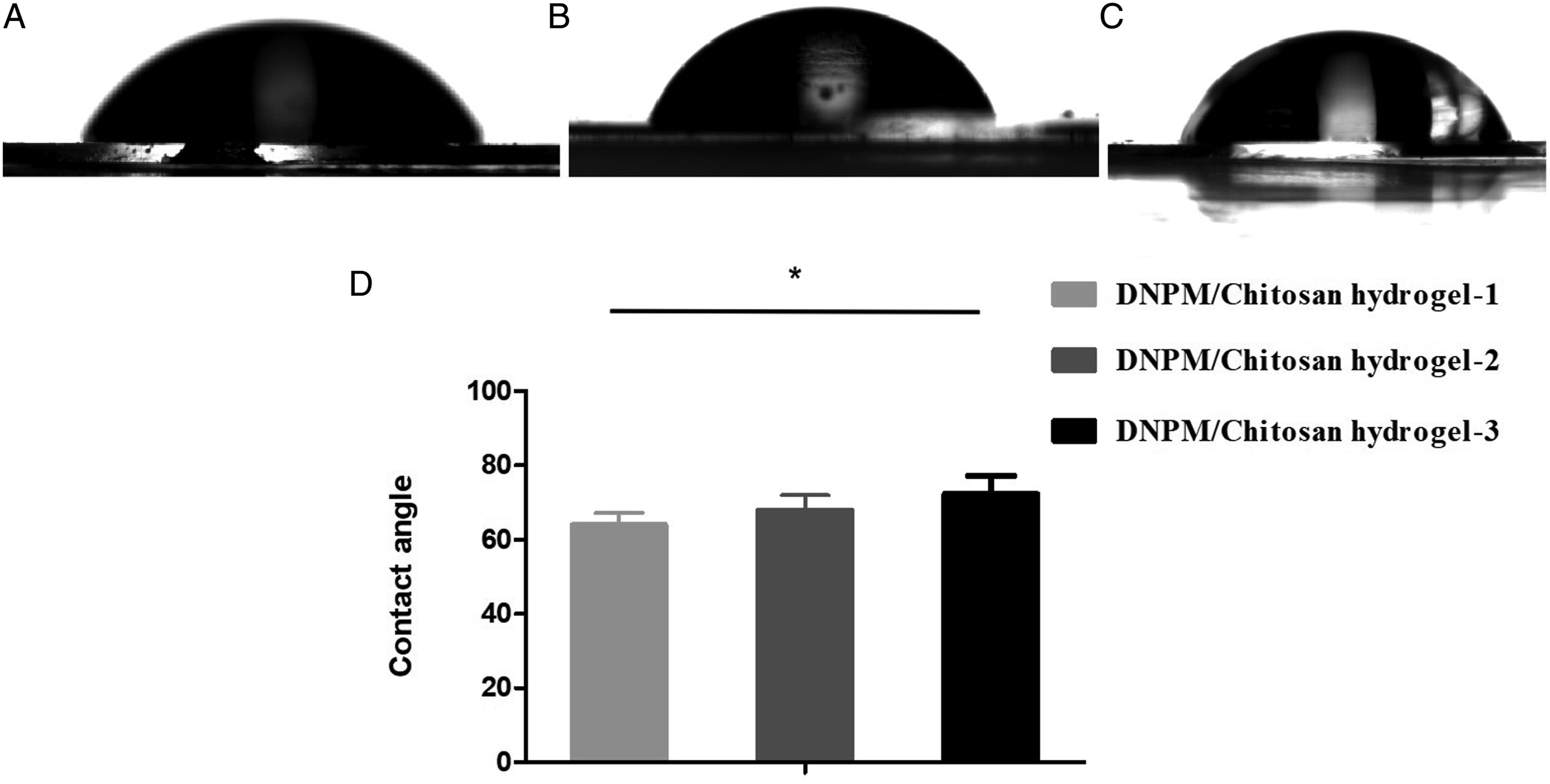
The rheological modulus values of the DNPM/chitosan hybrid hydrogels were 76.3 Shear modulus of 3 DNPM/chitosan hybrid hydrogels. AFM images of the (A-C) surface and (D-F) 3D DNPM/chitosan hybrid hydrogels. SEM images of the 3 DNPM/chitosan hybrid hydrogels. Scale bar: 50 μm. The contact angle of the 3 DNPM/chitosan hybrid hydrogels. *indicates statistically significant (P < .05), n=5.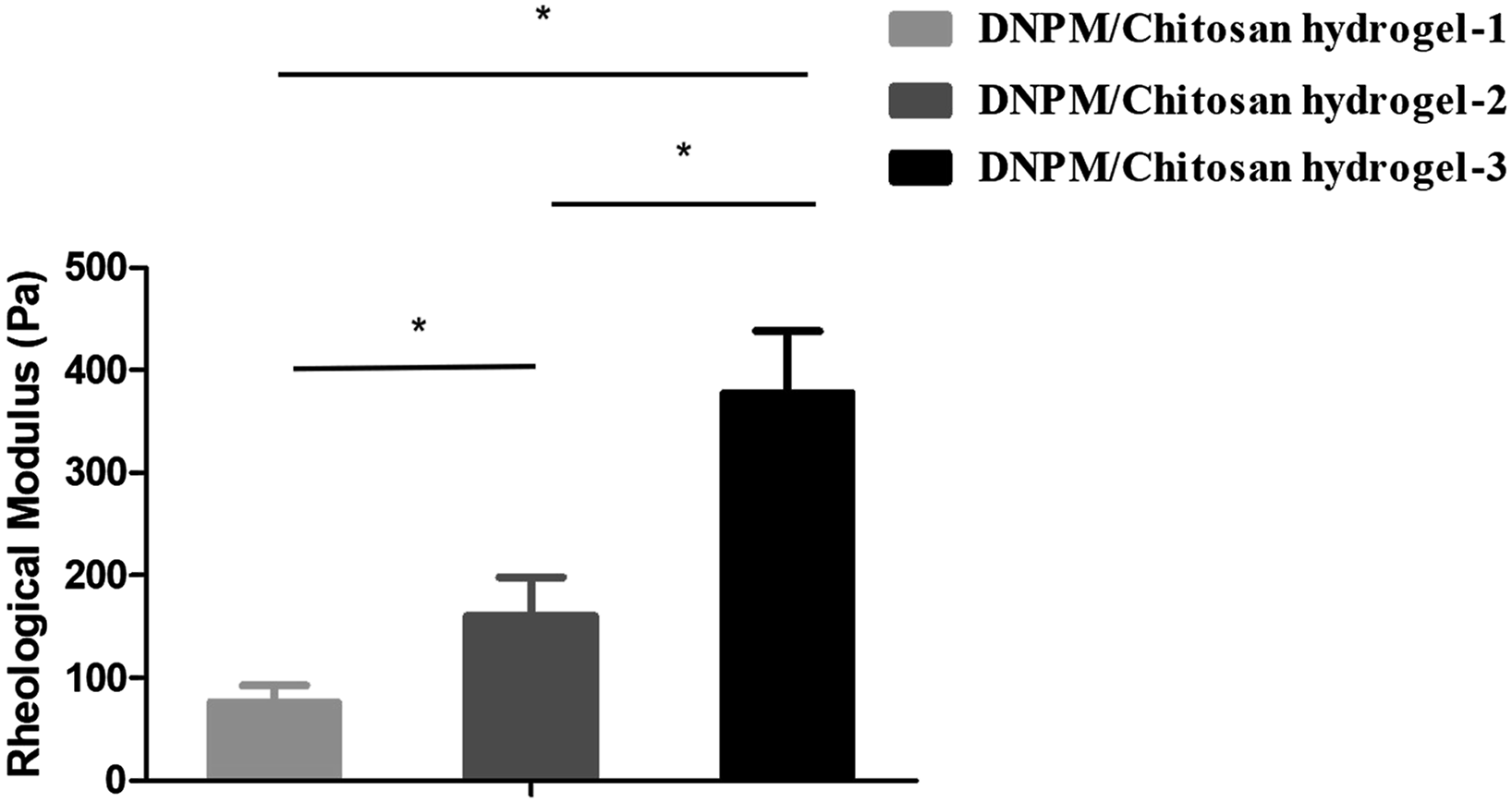

Cellular Morphology and Metabolic Activity
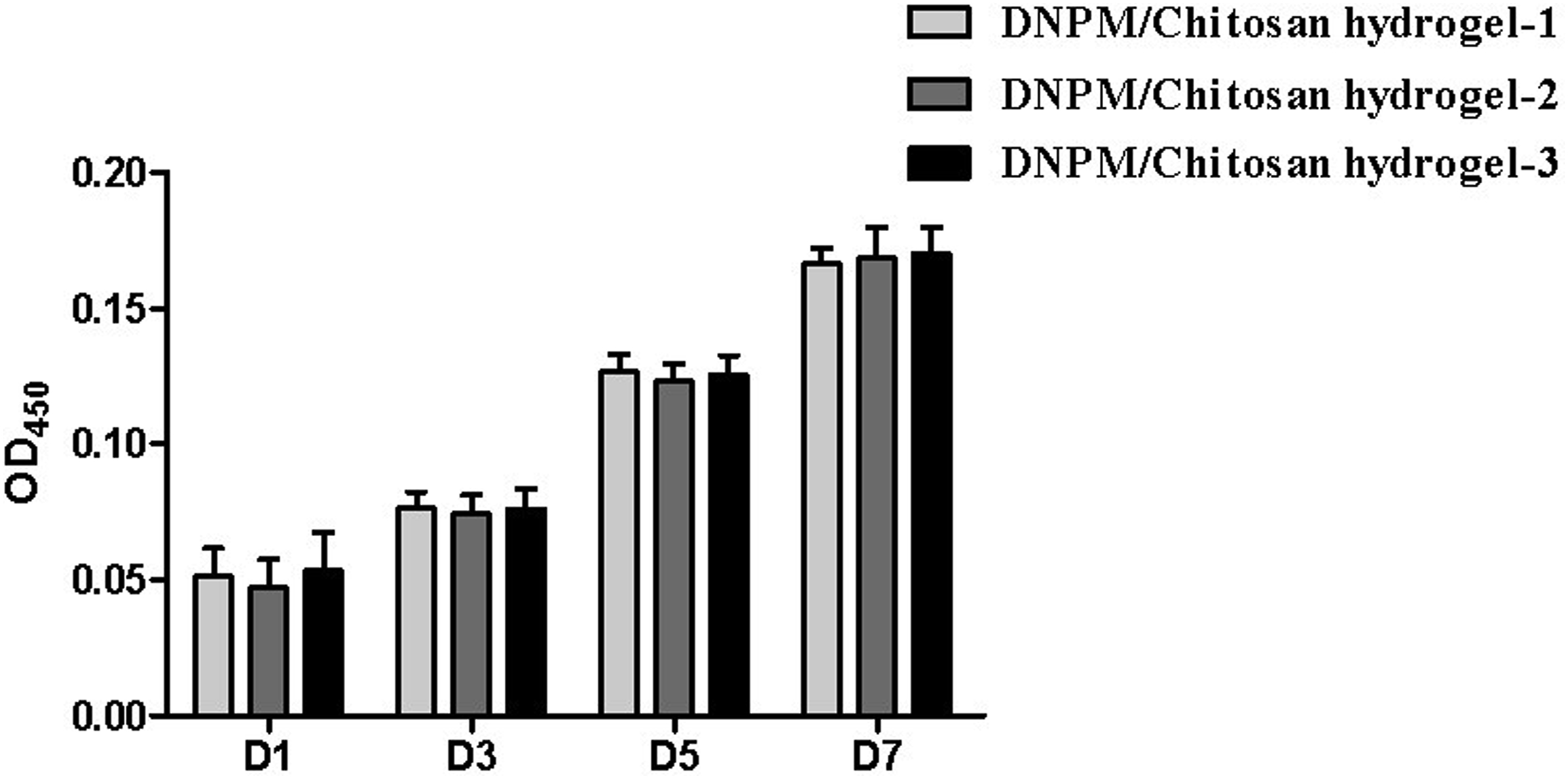
The cytoskeleton staining results showed that the NPSCs adhered and spread well on the DNPM/chitosan hybrid hydrogels (Figure 5). In addition, the CCK-8 assay results indicated that the NPSCs proliferated well on the hydrogel and there were no significant differences between the 3 hybrid hydrogels (Figure 6). Cytoskeleton staining image of the NPSCs on the 3 DNPM/chitosan hybrid hydrogels. Scale bar: 400 μm. The proliferation of NPSCs on the 3 DNPM/chitosan hybrid hydrogels after 1, 3, 5, and 7 days of culture.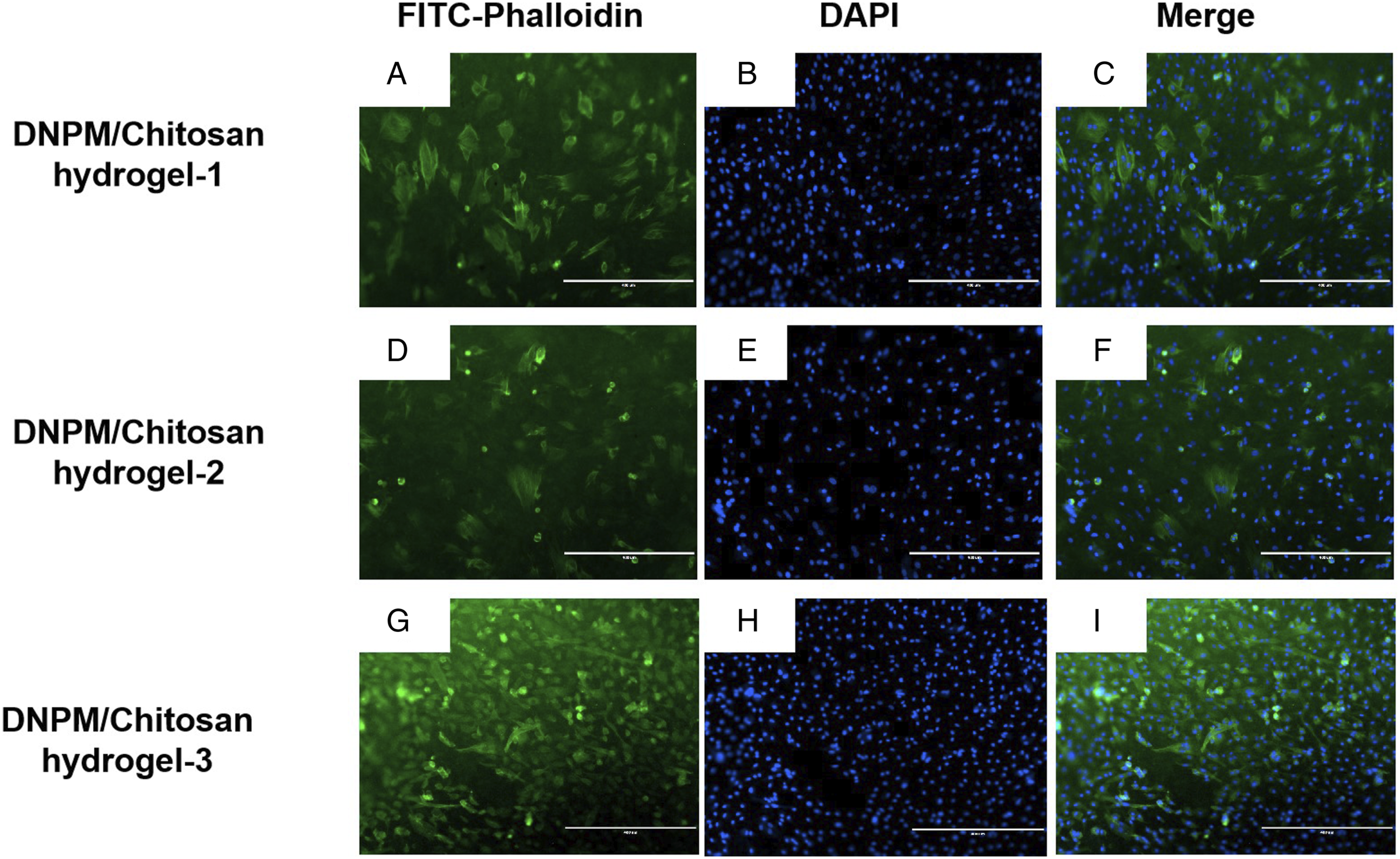
NP-Related Gene Expression Analysis
We quantified the expression of the NP-related proteins, Col-II, aggrecan, and Sox-9, in the hydrogels using RT-qPCR. As shown in Figure 7 the NPSCs encapsulated in the DNPM/chitosan hybrid hydrogel-1 expressed the highest level of Col-II and Sox-9. The differences were statistically significant. For the aggrecan gene expression, NPSCs grown on DNPM/chitosan hybrid hydrogel-1 and DNPM/chitosan hybrid hydrogel-3 were significantly greater than those on DNPM/chitosan hybrid hydrogel-2. However, there was no significant difference between DNPM/chitosan hybrid hydrogel-1 and DNPM/chitosan hybrid hydrogel-3. (A) Col-II, (B) aggrecan, and C) Sox-9: Gene expression of NPSCs on the 3 DNPM/chitosan hybrid hydrogels. *indicates statistically significant (P < .05).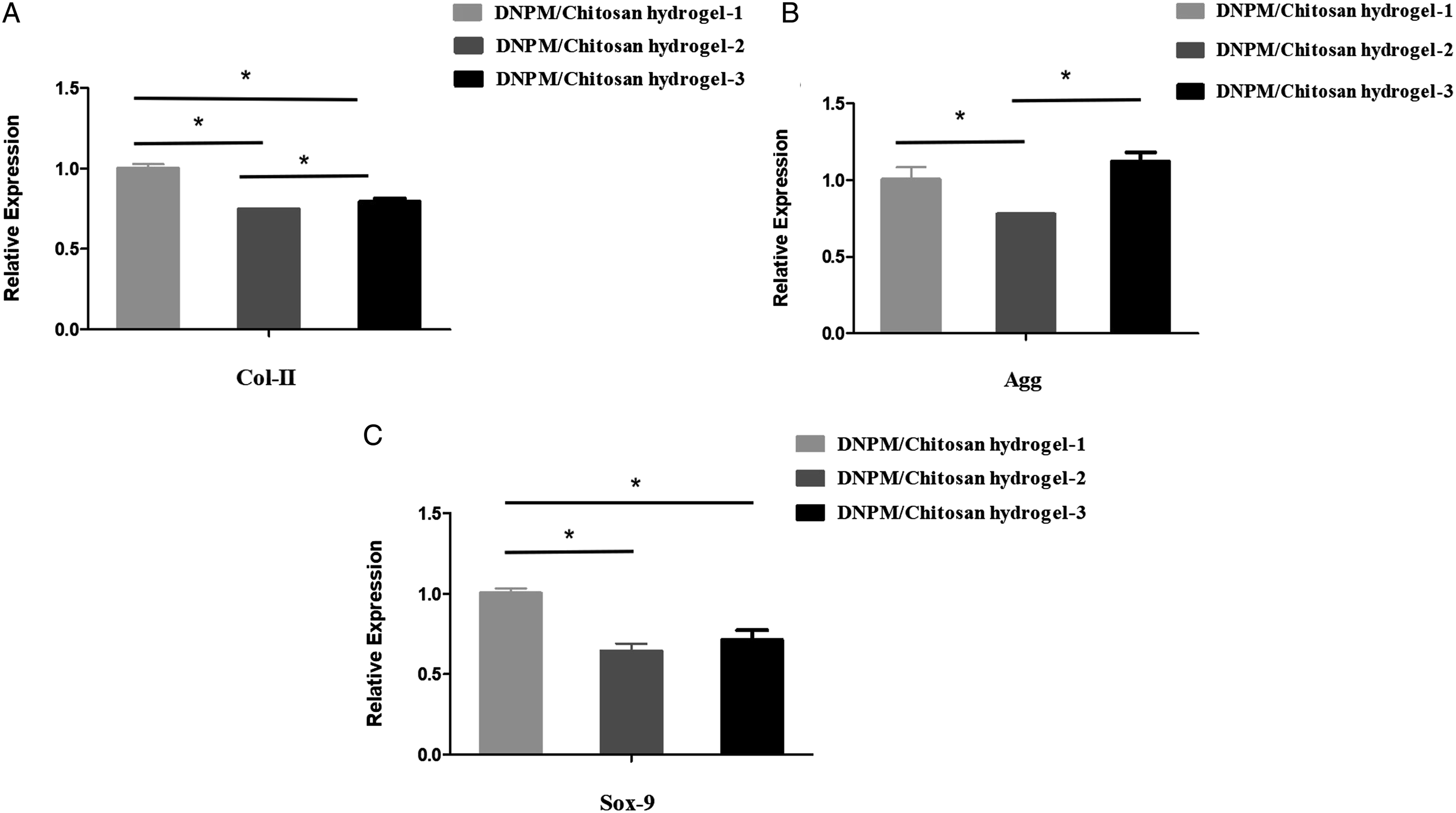
Evaluation of the Scaffold Degradation in Vivo
Four weeks after implantation, H&E staining revealed few inflammatory cells around the DNPM/chitosan hybrid hydrogel-1, whereas more inflammatory cells surrounded DNPM/chitosan hybrid hydrogel-2 and DNPM/chitosan hybrid hydrogel-3. There was no obvious degradation of the 3 hybrid hydrogels (Figure 8). H&E staining of the 3 DNPM/chitosan hybrid hydrogels after implantation in rats for 4 weeks. The inflammatory cells are indicated by a red arrow. Scale bar: A-C 75 μm, D-F 50 μm.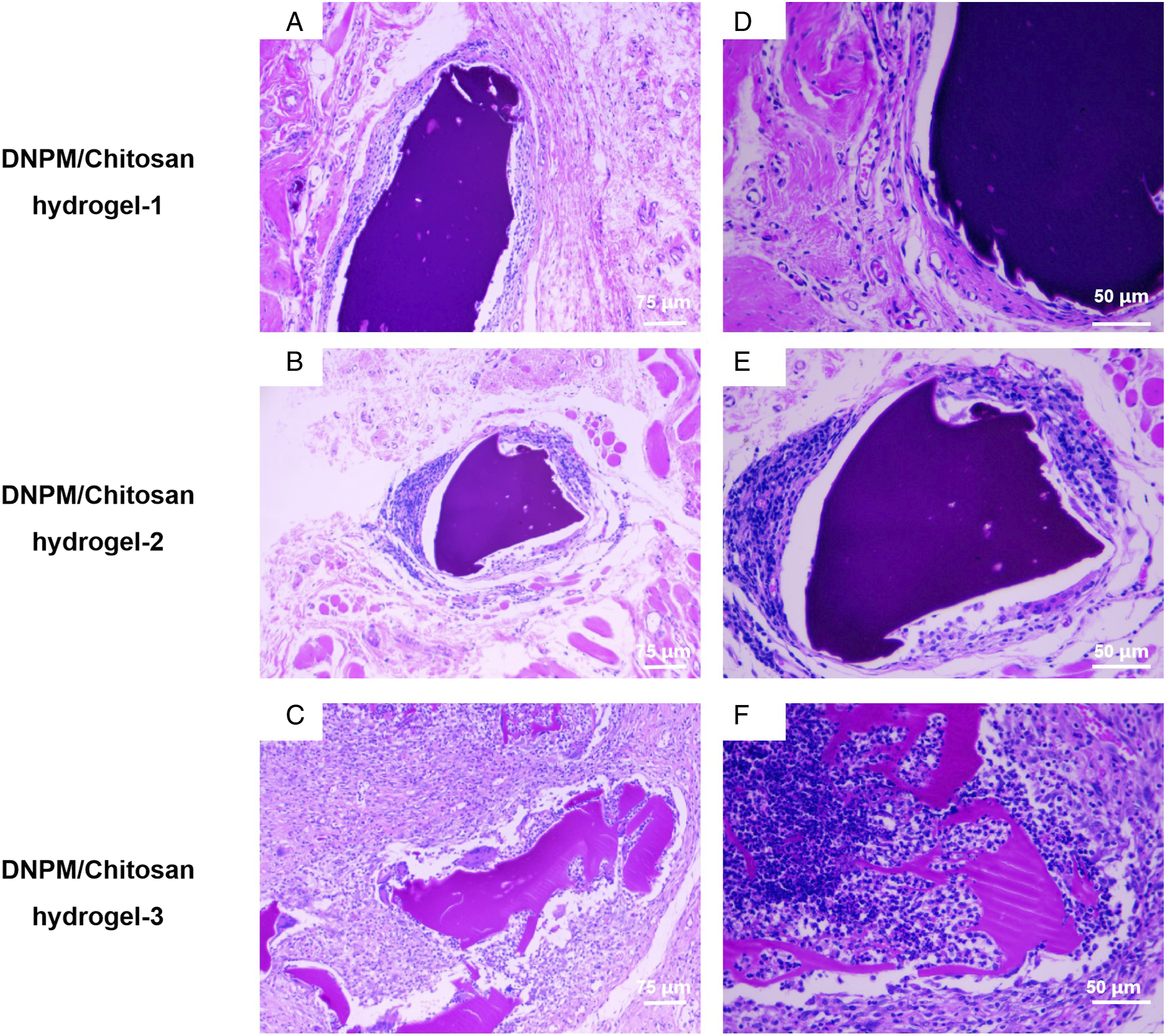
Discussion
In this study, we manufactured 3 DNPM/chitosan hybrid hydrogels by changing the ratio of DNPM to chitosan and crosslinking with genipin. NPSCs expressed more NP-related genes, such as Col-II, aggrecan, and Sox-9, on DNPM/chitosan hybrid hydrogel-1 than the other hydrogels.
Various materials, including gelatin, collagen type II, alginate, and hyaluronic acid, have been used in NP tissue engineering.17-20 However, the low mechanical strength of those scaffolds leads to failure of the transplant because of the inability to bear the pressure of normal NP in the early stage. Additionally, seed cells cannot secrete as much ECM on the scaffold as in native NP tissue, which may also cause failure. DNPM reduces the immune response and also regulates the adhesion, proliferation, differentiation, and phenotypic maintenance of seed cells. 21 In our previous study, we manufactured a DNPM/chitosan hybrid hydrogel with TGF-β3 and demonstrated that the DNPM/chitosan hybrid hydrogel may be a promising candidate scaffold for NP tissue engineering. 22 In this study, NPSCs on DNPM/chitosan hybrid hydrogel-1 expressed more col-II, aggrecan, and Sox-9 than cells on the other hydrogels. The gene expression may be attributed to more DNPM in the DNPM/chitosan hybrid hydrogel-1 than in the other hydrogels, as shown in the AFM test. Specifically, collagen fiber was only observed in DNPM/chitosan hybrid hydrogel-1. A previous study proved that the decellularized matrix from different tissues or organs may impact the differentiation of certain mesenchymal stem cells. 23 For example, Liu et al fabricated Decellularized Annulus Fibrosus Matrix/PECUU blended scaffolds and found DAFM promoted the synthesis of annulus fibrosus-related ECM. However, the expression level of NP-related genes was not directly proportional to the DNPM content or the shear modulus of the hybrid hydrogel. A similar result was observed in a study performed by Xu et al, 24 who prepared 3 hydrogels made of collagen hydrolysate gelatin and methacrylate (GelMA): 5% GelMA hydrogel, 10% GelMA hydrogel, and 15% GelMA hydrogel. The compression modulus was 46.78 kPa for the 5% GelMA hydrogel and 82.05 kPa for the 15% GelMA hydrogel, and the Col-II and aggrecan gene expressions of NPs from the 5% GelMA hydrogel were significantly greater than those from the other hydrogels.
NPSCs are a potential seed cell for NP tissue engineering. For example, Chen et al transplanted NPSCs to repair rabbit degenerative NP. The signal of the NP was enhanced with MRI detection, and the expressions of proteoglycan and type II collagen were significantly increased. 25 In this study, cytoskeleton staining showed that the NPSCs adhered and spread well on the surface of the hybrid hydrogel, and cell proliferation was verified. The biocompatibility of the hybrid hydrogel could contribute to the low contact angle, as shown in Figure 4. The low contact angle confirms the hybrid hydrogel has high hydrophilicity, which is conducive to the adhesion and proliferation of seed cells. At the same time, Figure 4 also indicated that more DNPM in the hybrid hydrogel reduced the contact angle, which is expected because of the hydrophobic nature of collagen. de Deus manufactured PLA, PLA–CUR, PLA–N(CH3)2, and PLA–N(C2H5)2 electrospun scaffolds and found the contact angles correlated with the hydrophobic curcuminoids. 26
NP tissue engineering scaffolds ultimately need to be implanted in vivo. The in vivo degradation and inflammatory response are also concerns of tissue engineering scaffolds. There was no obvious degradation 4 weeks after the hybrid hydrogel was placed in the subcutaneous tissue of rats, which may be attributed to the short time in vivo. Additionally, there were fewer inflammatory cells near the DNPM/chitosan hybrid hydrogel-1 than with other hydrogels. Therefore, more DNPM in the hybrid hydrogel caused less inflammatory reaction. A similar phenomenon was observed in our previous study. The gene expression of inflammatory factors, such as TNF-αin and IL-6, from rabbit AFSCs grown on DAFM/PECUU fibrous scaffolds was less than cells on PECUU scaffolds in vitro. 27 Additionally, we implanted 2 DAFM/PECUU fibrous scaffolds in rabbit and observed a light inflammatory reaction around the implant. 28
There are some limitations of this study. For example, we did not implant the DNPM/chitosan hybrid hydrogel into the degenerative intervertebral disc to analyze the repair. In a future study, we will use DNPM/chitosan hybrid hydrogel as a NP tissue engineering scaffold and DAFM/PECUU aligned and blended electrospun scaffolds with different modulus as fibrous annulus scaffolds to construct a NP–AF integrated tissue engineering intervertebral disc.
Conclusion
In summary, we synthesized 3 DNPM/chitosan hybrid hydrogels by changing the ratio of DNPM and chitosan. NPSCs adhered and proliferated well on all of the DNPM/chitosan hybrid hydrogels because of the low contact angle. DNPM/chitosan hybrid hydrogel-1 had the best performance for Col-II, aggrecan, and Sox-9 expression and immune reaction in vivo. The results indicate that the DNPM/chitosan hybrid hydrogel-1 may be a promising scaffold for NP regeneration in the treatment of DDD.
Footnotes
Acknowledgments
Authors' contributions
YZ wrote the main manuscript text. YZ and YF prepared figures 1-8. CL and XF participated in the project conceptualization and the guarantor of the article. All authors reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from the National Natural Science Foundation of China (82172427, 81702158); Foreign Science and Technology Cooperation of Anhui Province (202004b11020027); “Peak” Training Program for Scientific Research of Yijishan Hospital, Wannan Medical College (GF2019G07, GF2019G12); and Young and middle-aged scientific research fund of Wannan Medical College (WK2020F22, WK2021ZF14).
Availability of data and material
According to the requirements, data can be obtained from the corresponding author to support the results of this study.
