Abstract
Fehlings MG, Badhiwala JH, Ahn H, et al. Safety and efficacy of riluzole in patients undergoing decompressive surgery for degenerative cervical myelopathy (CSM-Protect): a multicentre, double-blind, placebo-controlled, randomised, phase 3 trial. Lancet Neurol. 2020.
Study Type 
Study Quality: 
Overall Risk of Bias Rating 
Visual Abstract 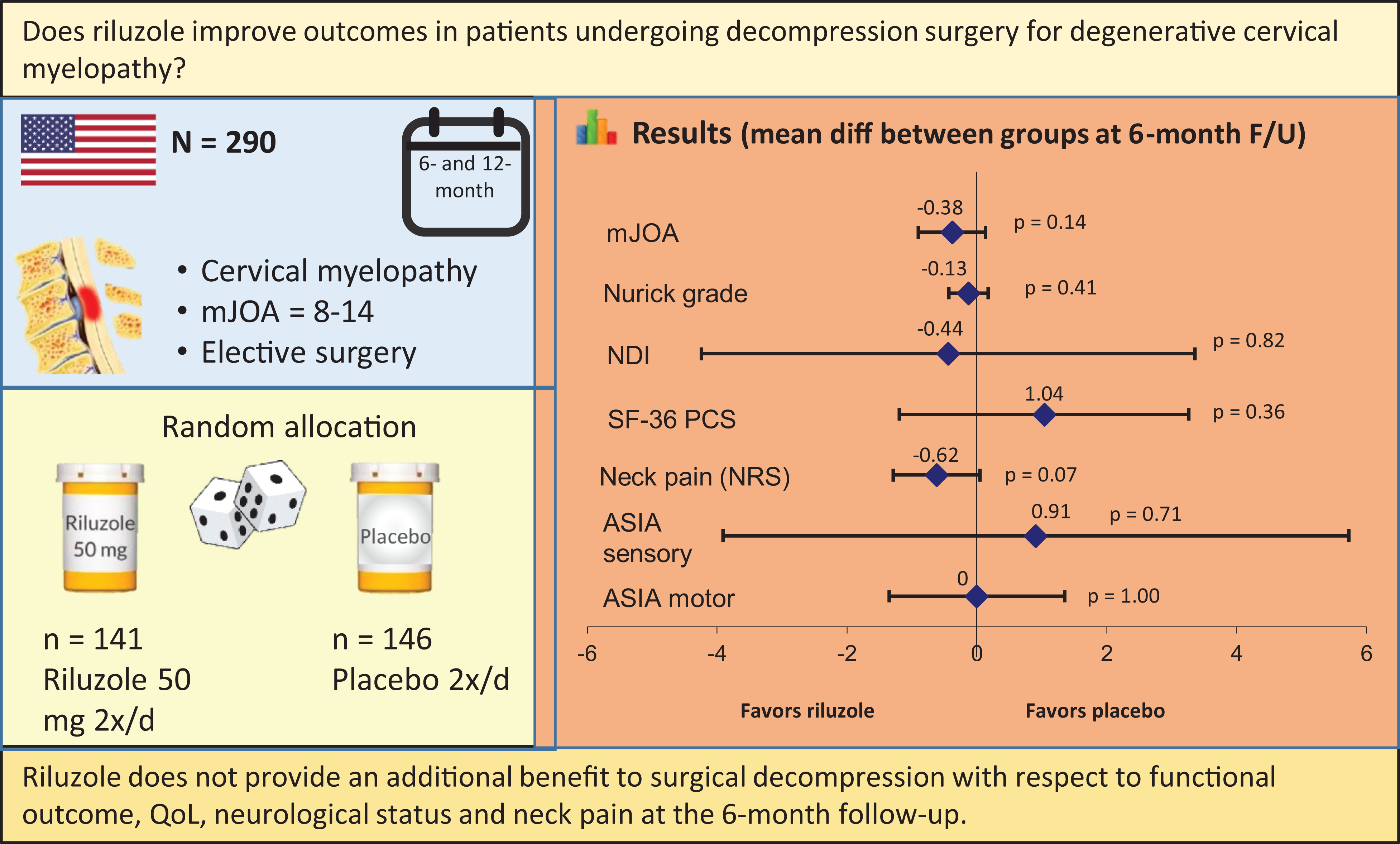
Why Is This Study an Important Topic?
Degenerative cervical myelopathy represents a non-traumatic form of spinal cord injury for which surgical decompression is often indicated. Many patients end up with substantial residual postoperative disability. Therefore, examining pharmacological therapies that could be combined with surgery to improve long-term patient outcomes is warranted.
What Was the Primary Clinical Question?
In patients who undergo decompression surgery for degenerative cervical myelopathy, does riluzole improve postoperative outcomes?
Study Characteristics

Results
Result 1. Change in mJOA from baseline. 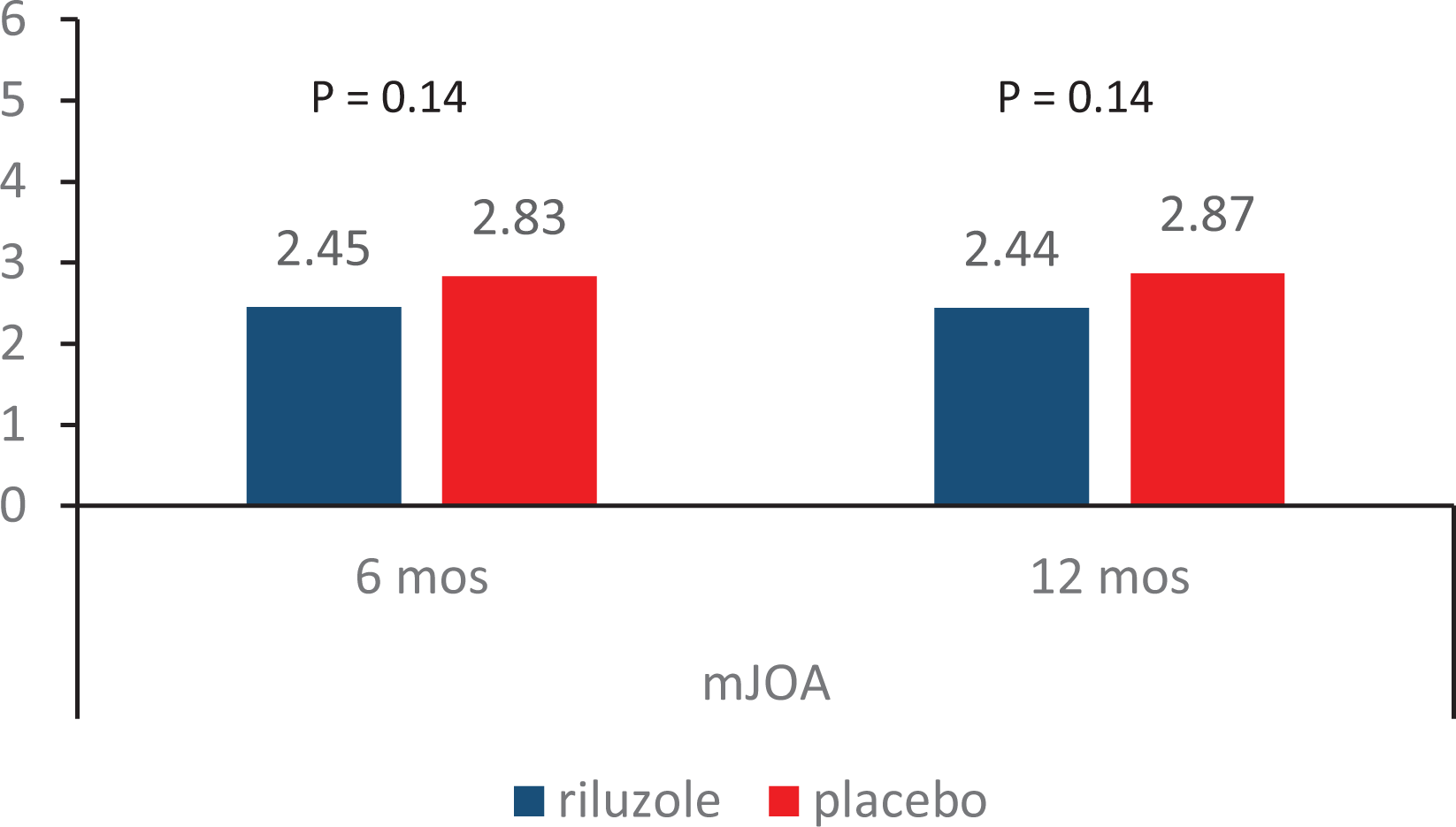
Result 2. Change in function from baseline. 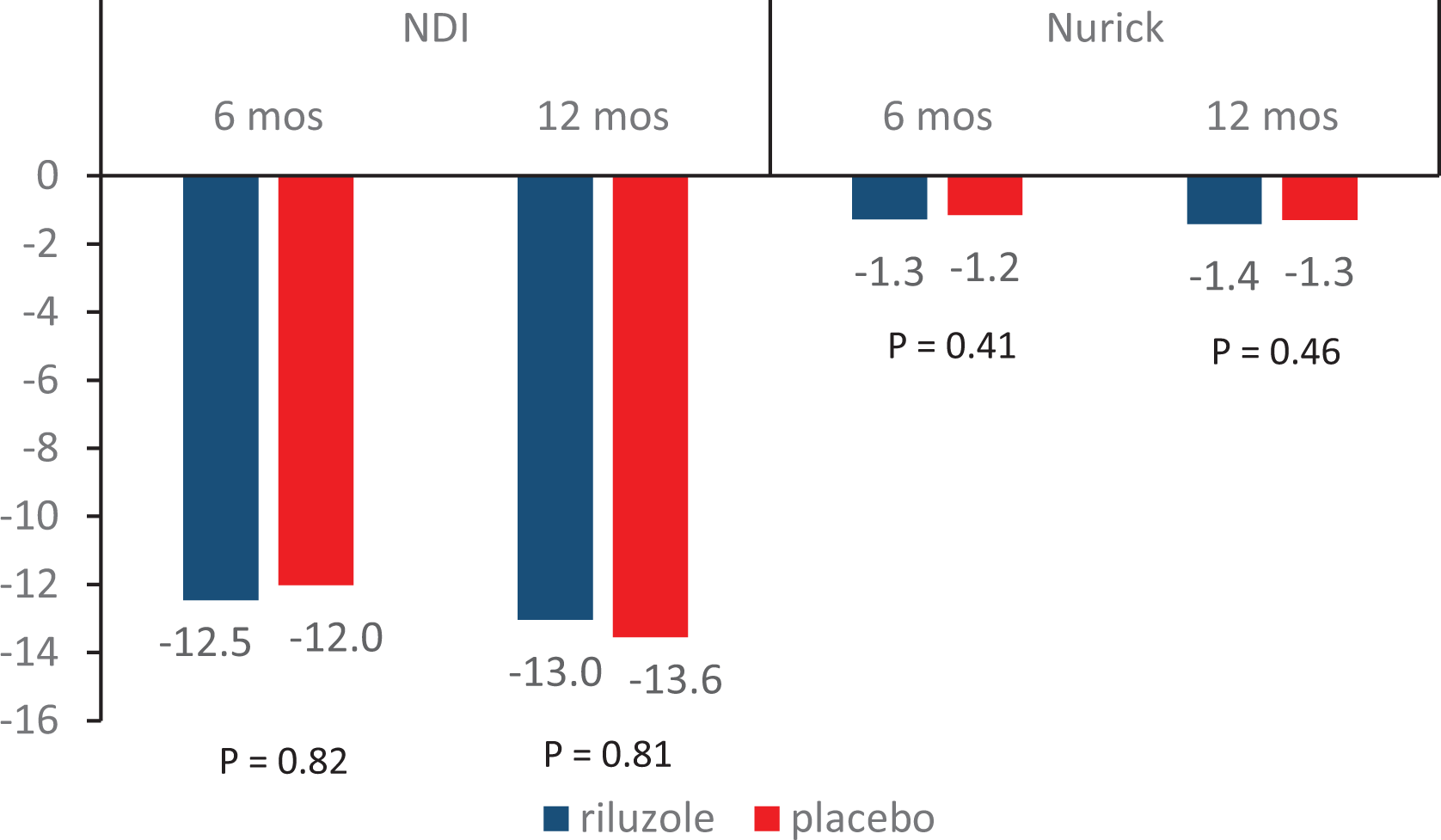
Result 3. Change in QoL from baseline. 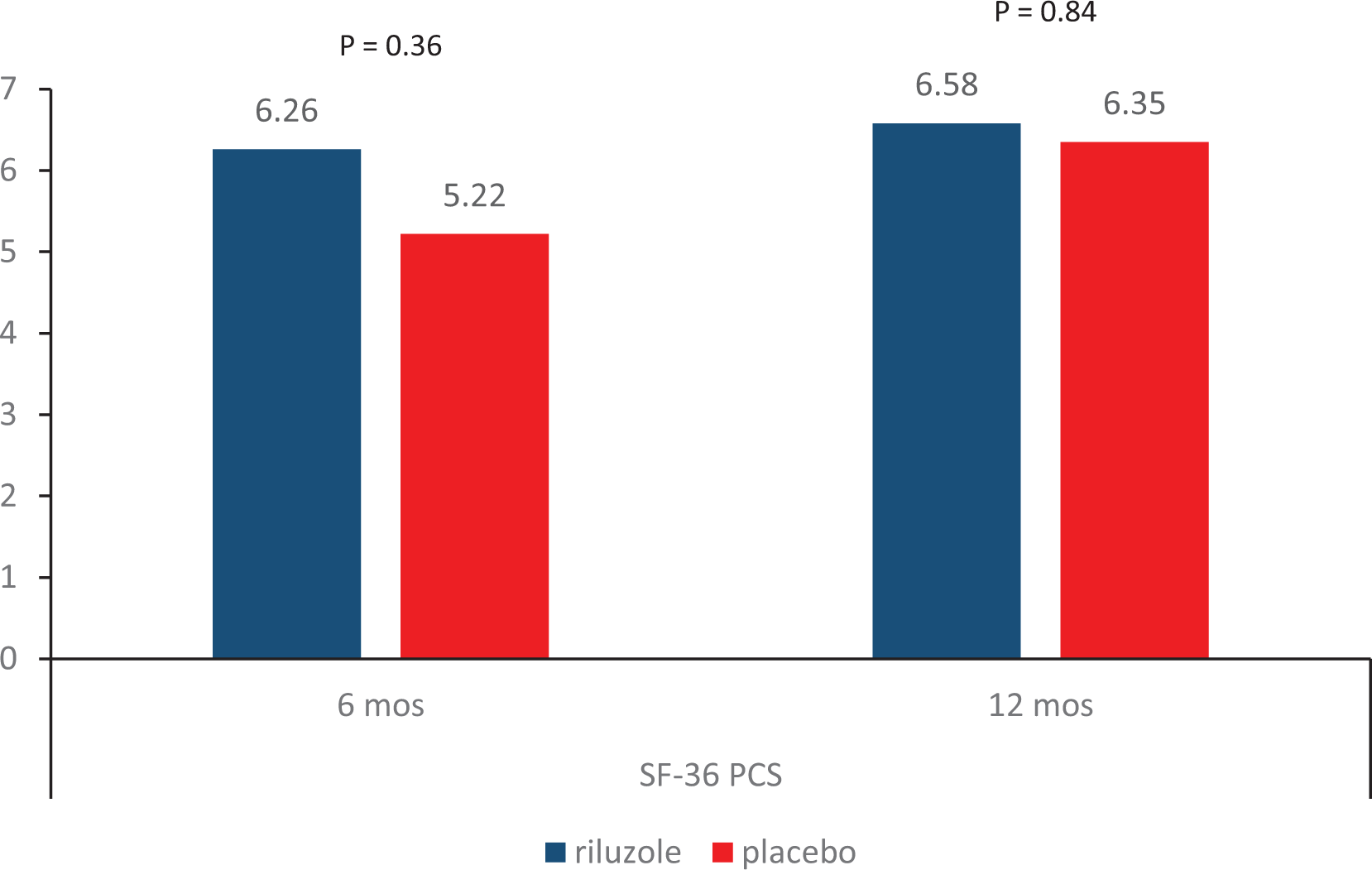
Result 4. Change in neck pain from baseline. 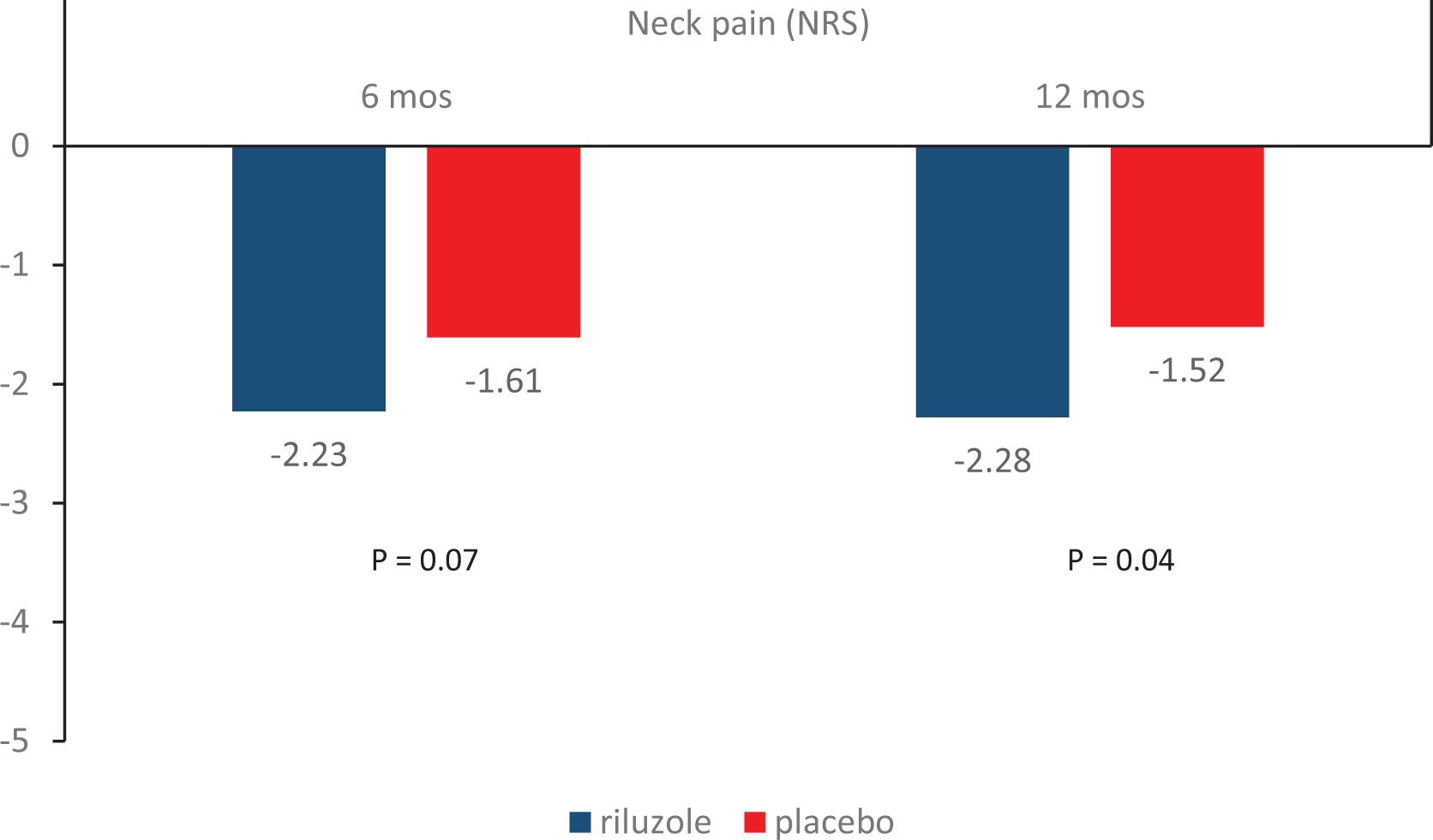
Result 5. Change in ASIA from baseline. 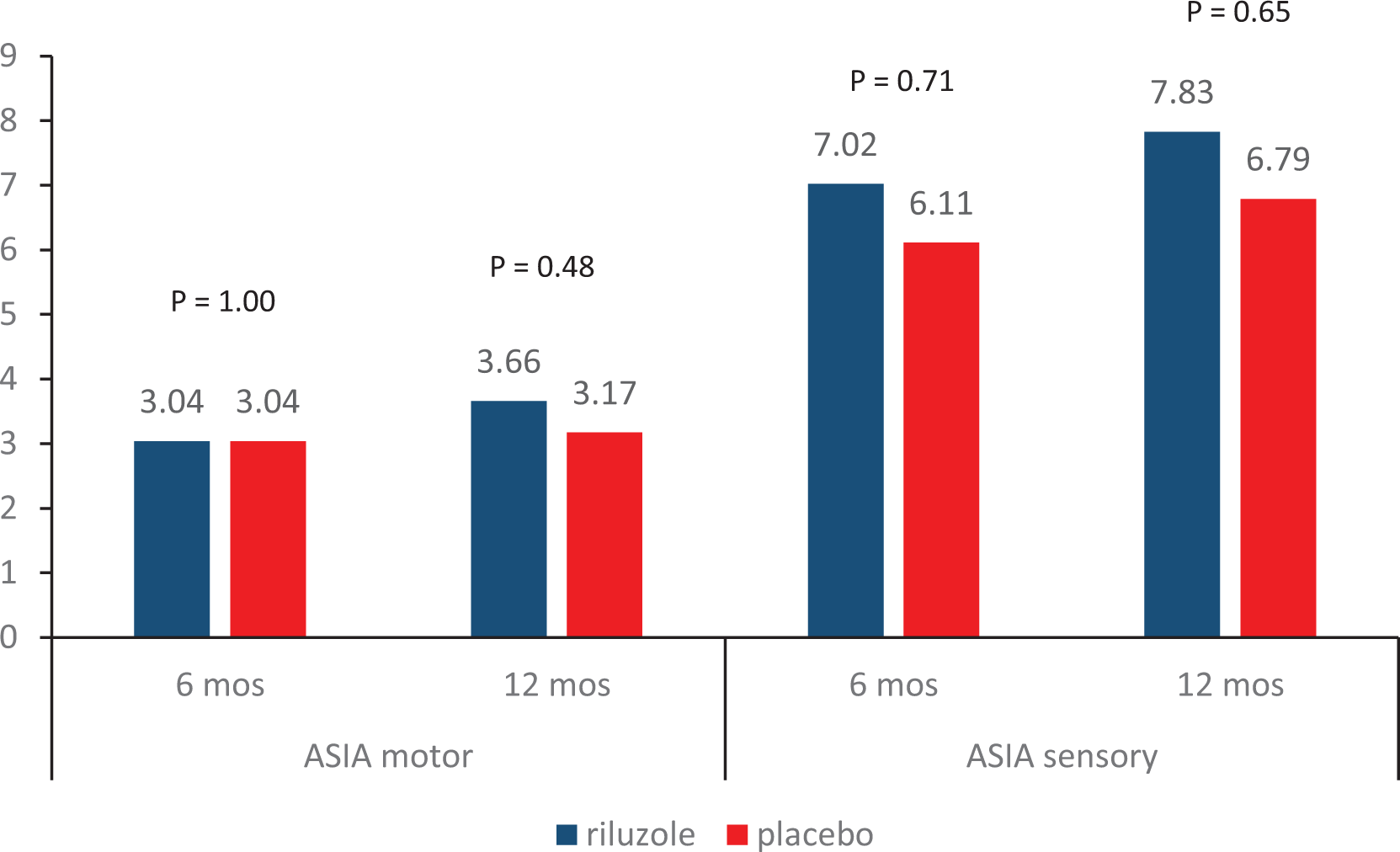
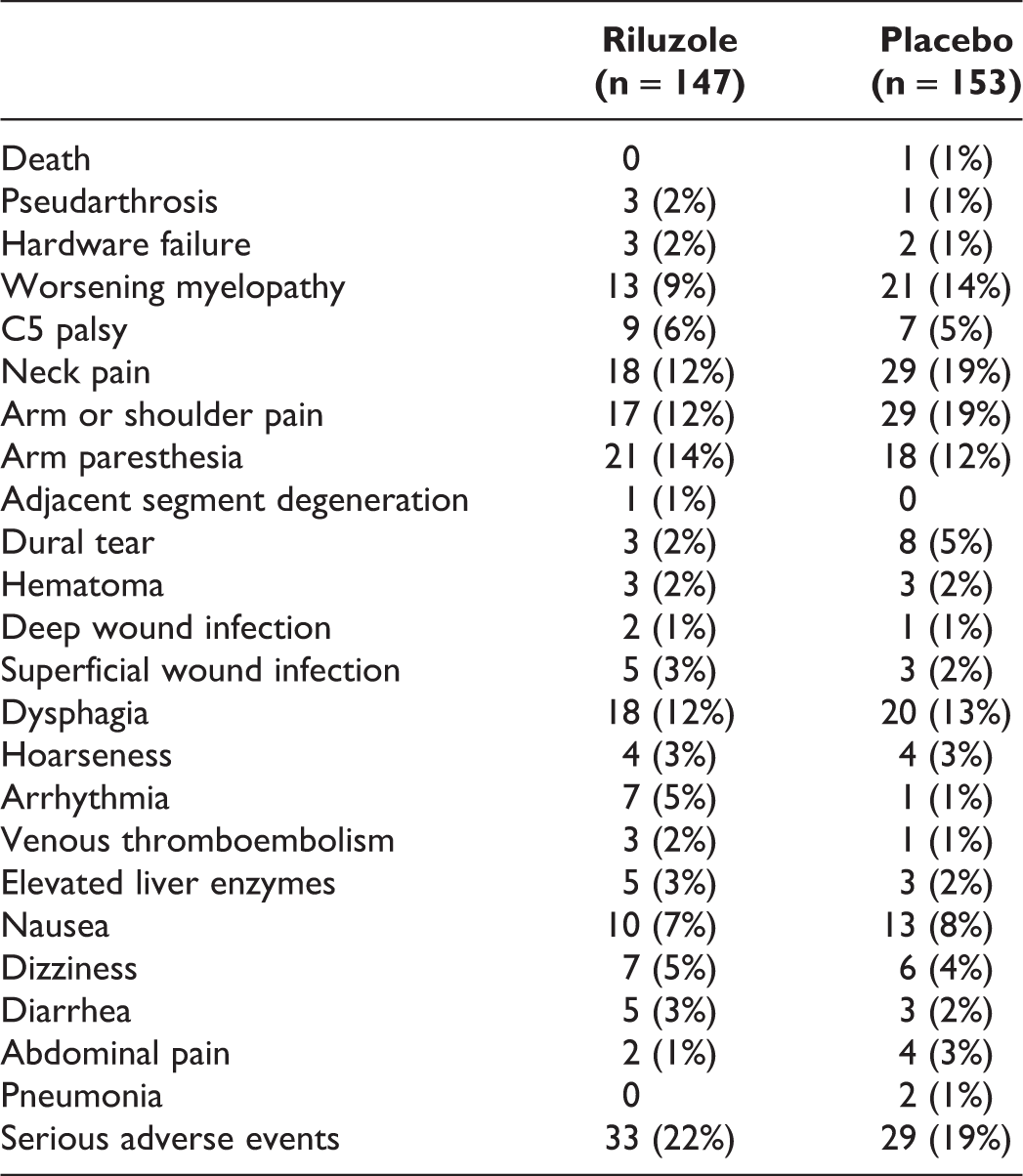
Result 6. Adverse Events. Adverse Events
How Will This Affect the Care of My Patients?
Riluzole does not provide an additional benefit to surgical decompression with respect to functional outcomes, QoL, and neurological status. The potential effect of riluzole on pain outcomes warrants further investigation.
