Abstract
Study Design:
Systematic review.
Objective:
To evaluate the impact of riluzole on neurobehavioral outcomes in preclinical models of nontraumatic and traumatic spinal cord injury (SCI).
Methods:
An extensive search of the literature was conducted in Medline, EMBASE, and Medline in Process. Studies were included if they evaluated the impact of riluzole on neurobehavioral outcomes in preclinical models of nontraumatic and traumatic SCI. Extensive data were extracted from relevant studies, including sample characteristics, injury model, outcomes assessed, timing of evaluation, and main results. The SYRCLE checklist was used to assess various sources of bias.
Results:
The search yielded a total of 3180 unique citations. A total of 16 studies were deemed relevant and were summarized in this review. Sample sizes ranged from 14 to 90, and injury models included traumatic SCI (n = 9), degenerative cervical myelopathy (n = 2), and spinal cord-ischemia (n = 5). The most commonly assessed outcome measures were BBB (Basso, Beattie, Besnahan) locomotor score and von Frey filament testing. In general, rats treated with riluzole exhibited significantly higher BBB locomotor scores than controls. Furthermore, riluzole significantly increased withdrawal thresholds to innocuous stimuli and tail flick latency following application of radiant heat stimuli. Finally, rats treated with riluzole achieved superior results on many components of gait assessment.
Conclusion:
In preclinical models of traumatic and nontraumatic SCI, riluzole significantly improves locomotor scores, gait function, and neuropathic pain. This review provides the background information necessary to interpret the results of clinical trials on the impact of riluzole in traumatic and nontraumatic SCI.
Keywords
Introduction
Nontraumatic and traumatic injuries to the spinal cord initiate a cascade of pathophysiological changes that may impair normal motor, sensory, and autonomic functions and cause irreversible tissue damage. 1,2 Surgical intervention is recommended as the preferred treatment strategy for patients with moderate to severe degenerative cervical myelopathy (DCM) as it can halt neurologic decline and significantly improve functional impairment, disability, and quality of life. 3,4 Furthermore, early surgical management of traumatic spinal cord injury (SCI) is increasingly prioritized due to emerging evidence that patients decompressed and stabilized within 24 hours of injury exhibit superior outcomes. 5,6
There is also an opportunity to explore adjuvant treatments for the management of traumatic and nontraumatic SCI, including pharmacological strategies. Compression of the spinal cord alters its micro- and macro-vasculature, results in ischemia, and disturbs ionic homeostasis through the activation of voltage-gated sodium channels. 7 An influx of sodium results in cellular swelling, edema, and an increase in the activity of the sodium-calcium exchanger on the neuronal cell membrane. 8 Calcium entry pre-synaptically triggers the release of glutamate, which causes excitotoxicity and neuronal death. A neuroprotective drug such as riluzole may be effective at halting this cascade and preserving the structural integrity of the spinal cord.
Riluzole has neuroprotective, anti-ischemic, and anti-epileptic properties as well as several proposed mechanisms of action. 9 Specifically, it is a sodium channel blocker, a regulator of glutamate release, an antagonist at both NMDA and non-NMDA receptors, and an inhibitor of GABA reuptake. 10 -12 Randomized controlled trials have demonstrated that riluzole increases survival, decreases the necessity for tracheostomy, and attenuates muscle deterioration in patients with amyotrophic lateral sclerosis. 13,14 Riluzole has also demonstrated promising results in other neurological conditions, including Huntington’s disease, cerebellar ataxia, and cervical SCI. 15 -18 Furthermore, in vitro studies have indicated that riluzole significantly improves axonal conduction, prevents cellular necrosis and apoptosis, and enhances nerve fiber regeneration. 19 Given its mechanism of action and the cellular changes that follow compression of the spinal cord, there may be a role for riluzole as an adjuvant therapy in the management of DCM and SCI.
This systematic review aims to evaluate the impact of riluzole on neurobehavioral outcomes in preclinical models of nontraumatic and traumatic SCI. This review serves as an initial step in evaluating the suitability of riluzole for the management of DCM and SCI.
Methods
Eligibility Criteria
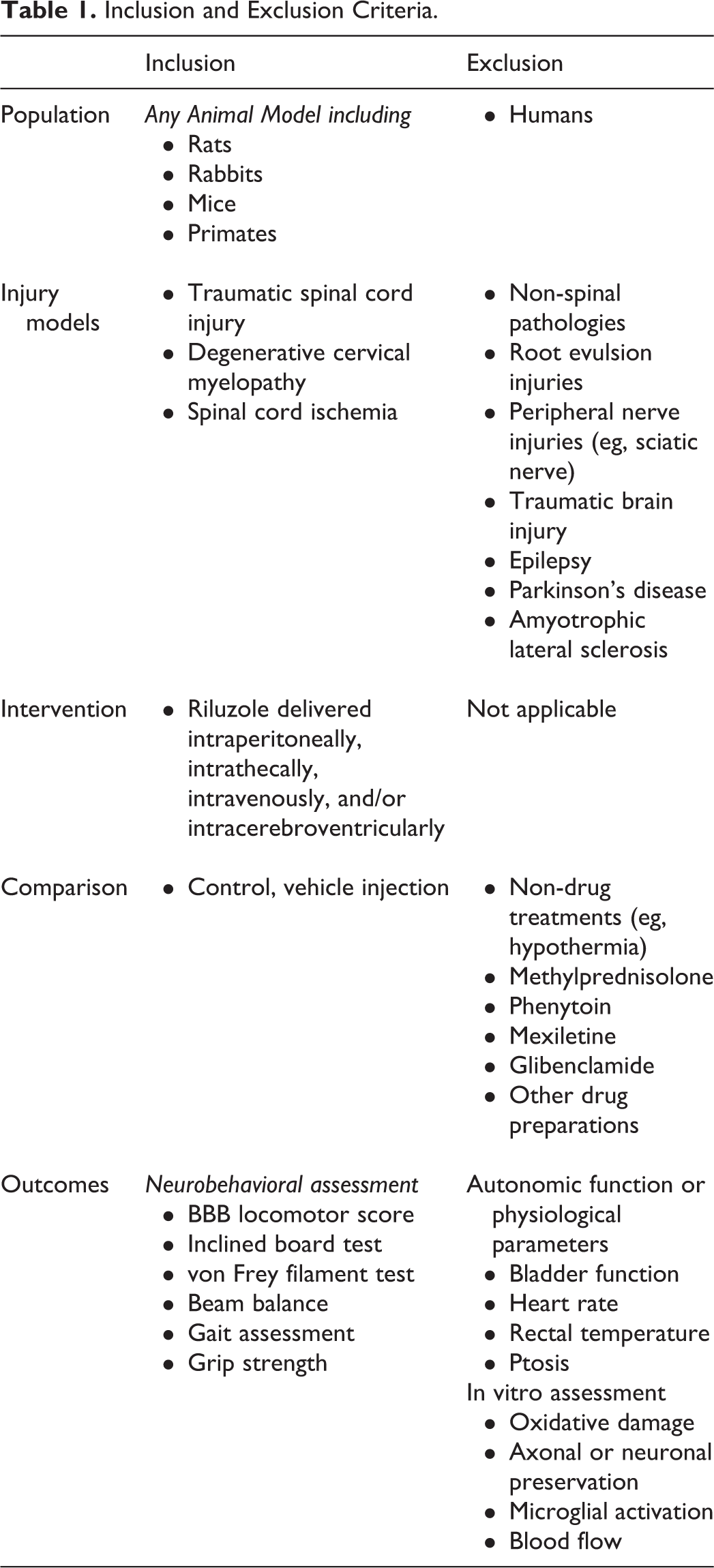
Table 1 provides a detailed summary of the inclusion and exclusion criteria for this review.
Inclusion and Exclusion Criteria.
Population and Injury Model
This review targeted studies on animal models (eg, rats, mice, rabbits, primates) of traumatic and nontraumatic SCI. Studies were excluded if they consisted of humans or if the animal model mimicked root avulsion or peripheral nerve injuries, traumatic brain injury, epilepsy, Parkinson’s disease, or amyotrophic lateral sclerosis.
Intervention and Comparison
The intervention of interest was riluzole, injected intravenously, intraperitoneally, intrathecally, or intracerebroventricularly. There were no limitations on the dosing, timing of administration, or duration of treatment. Studies were only included if they had a control group (eg, vehicle injection) and specifically evaluated outcomes with respect to this group. Studies were excluded if they only compared the efficacy of riluzole to other treatments (eg, hypothermia) or drug regimens (eg, methylprednisolone, phenytoin, glibenclamide, mexiletine).
Outcomes
This review primarily focused on neurobehavioral outcomes such as the Basso, Beattie, Besnahan (BBB) locomotor score, the inclined board test, the von Frey filament test, beam balance, gait assessment, and grip strength. Studies were excluded if they only discussed autonomic function or physiological parameters (eg, bladder function, heart rate, blood pressure) or if they evaluated in vitro changes (eg, oxidative damage, microglial activation, axonal loss) following riluzole administration.
Information Sources
A systematic search was conducted of MEDLINE, MEDLINE In-Process, and EMBASE to identify relevant studies. The search was completed on November 13, 2017.
Search Strategy
A search strategy was constructed with the assistance of a librarian at the Toronto Western Hospital. The strategy was originally prepared in MEDLINE and then appropriately modified for EMBASE. The terms used to search both databases are provided in Appendix A, available online. Only studies involving animal models of spinal pathologies and in English were considered for inclusion, with no other limits applied.
Study Selection
Duplicates, conference proceedings, editorials, and reviews were first excluded in Endnote. The remaining abstracts were reviewed independently by 2 of the authors and sorted based on predefined inclusion criteria (MZ and LT). In some cases, full text investigation was required to clarify whether the study was relevant. Discussion was used to resolve disagreement between reviewers.
Data Extraction and Synthesis
The following data were extracted from each article: author, year and location of investigation; sample characteristics, including sample size, type and weight of animals, and level of injury; injury model; intervention, including dose and route of drug administration; outcomes evaluated and timing of assessment; and statistical methods. Main study conclusions were also extracted if they highlighted the impact of riluzole on neurobehavioral outcomes compared to controls.
Assessment of Risk of Bias and Study Quality
The risk of bias of each study was evaluated using the SYRCLE tool (Systematic Review Center for Laboratory Animal Experimentation). 20 This checklist, presented in Table 2, was adopted from the Cochrane Collaboration risk of bias tool and modified to encompass certain biases that are relevant to animal experiments. It consists of 10 domains related to 6 types of bias: selection, performance, detection, attrition, reporting, and other biases. 20 Signaling questions provided by Hooijmans et al were used to assist in judging whether the experiment had a low, moderate, or high risk of bias for each entry. 20 The authors of this study also recommended not to compute a summary score as that would involve assigning weights to each domain.
Systematic Review Center for Laboratory Animal Experimentation (SYRCLE) Toola.

a Derived from Hooijmans et al. 20
Reporting
This review was formatted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist. 21
Results
Study Selection
The search yielded a total of 3180 unique citations. Using Endnote, 1799 articles were excluded because they were either not in English (n = 196) or were conference proceedings, editorials, literature reviews, or commentaries (n = 1603). After review of the remaining titles and abstracts, 1340 studies did not meet the inclusion criteria. Following full text investigation, an additional 25 studies were excluded; reasons for exclusion are provided in Appendix B. A total of 16 studies were considered relevant following this review process (Figure 1).
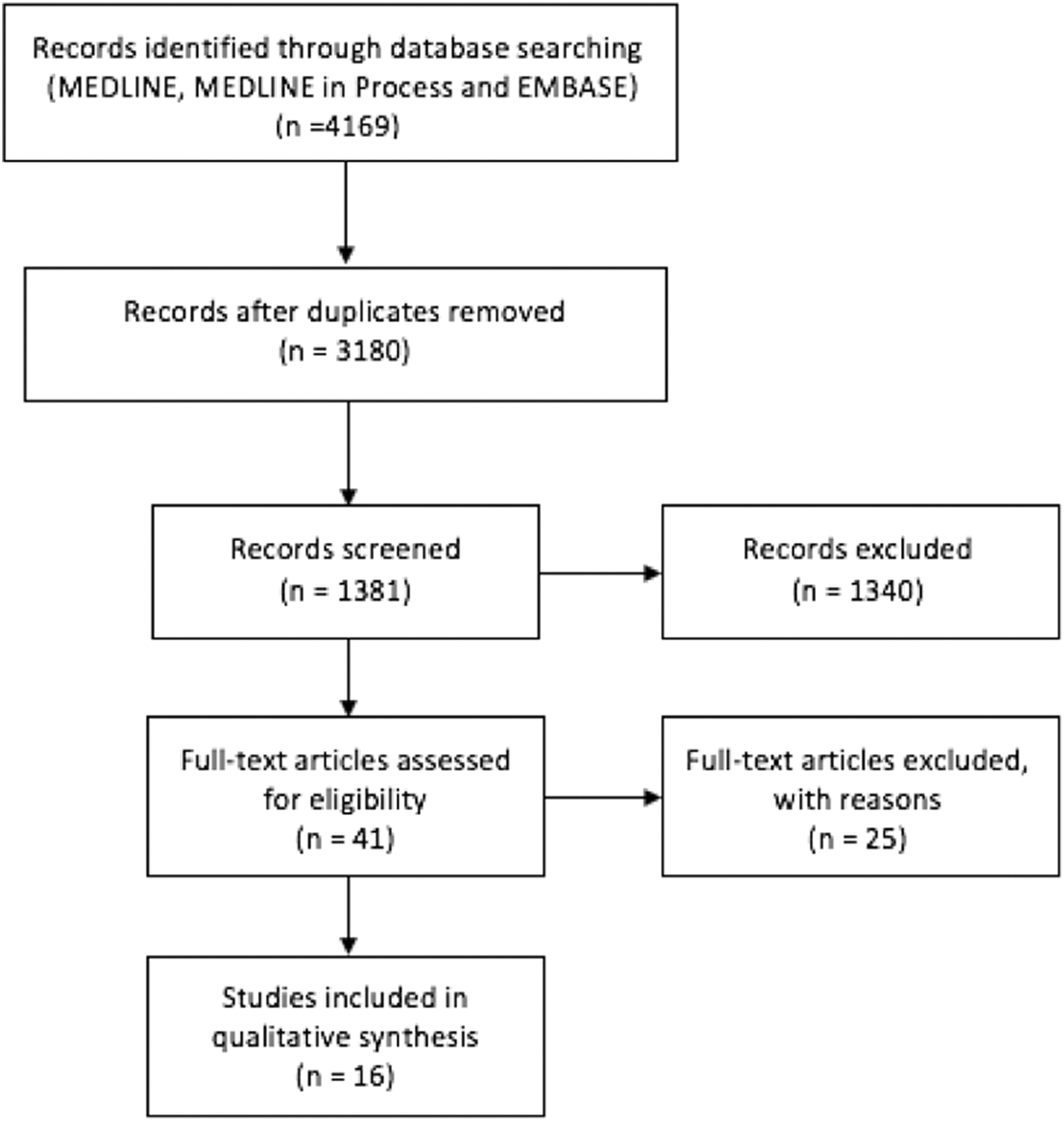
Overview of Study Selection.
Study Characteristics
Sixteen studies evaluated the impact of riluzole on neurobehavioral outcomes in either rat (n = 14) 9,10,12,19,22 -31 or rabbit (n = 2) 11,32 models of spinal cord pathology. Injury models included acute SCI via weight dropping, micro-scissors, or vascular clips (n = 9) 9,12,19,22,24,27 -30 ; DCM through progressive compression of the cord (n = 2) 26,33 ; and spinal cord ischemia via occlusion of the aorta (n = 5). 10,11,25,31,32 Of the models of acute SCI, 4 were at the thoracic level, 3 were cervical, 2 were cervicothoracic (C7-T1), and 1 was sacral. The most commonly assessed outcome measures were BBB locomotor score (n = 7) and gait analysis (n = 4). Table 3 summarizes the tools used to evaluate outcomes. Table 4 provides an overview of the included studies.
An Overview of the Tools Used to Evaluate Outcomes.

Summary of Included Studies.
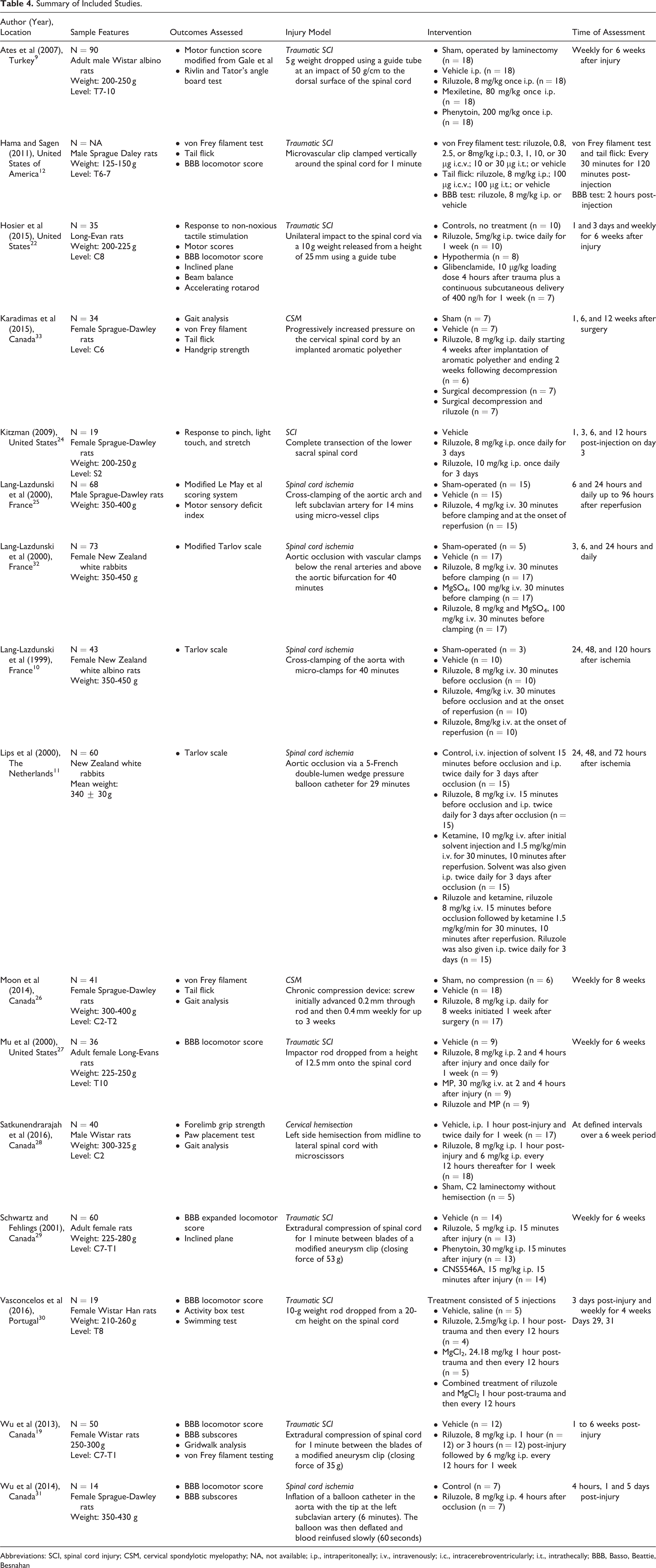
Abbreviations: SCI, spinal cord injury; CSM, cervical spondylotic myelopathy; NA, not available; i.p., intraperitoneally; i.v., intravenously; i.c., intracerebroventricularly; i.t., intrathecally; BBB, Basso, Beattie, Besnahan
Risk of Bias
The SYRCLE tool evaluated risk of bias across studies. In the majority of studies, allocation sequence was adequately generated, applied (n = 16), and concealed (n = 15). Fifteen studies randomly selected animals for outcome assessment. Investigators were blinded from the intervention in 6 studies and outcome assessors were blinded in 11 studies. Animals were randomly housed during the experiment in only 6 studies and incomplete outcome data was only addressed in 3 studies. Finally, it was unclear whether outcomes were selectively reported in any of the studies (Appendix C).
What Is the Impact of Riluzole on Neurobehavioral Outcomes?
The main results are summarized in Table 5.
Statistical Analysis and Main Conclusions of Included Studies
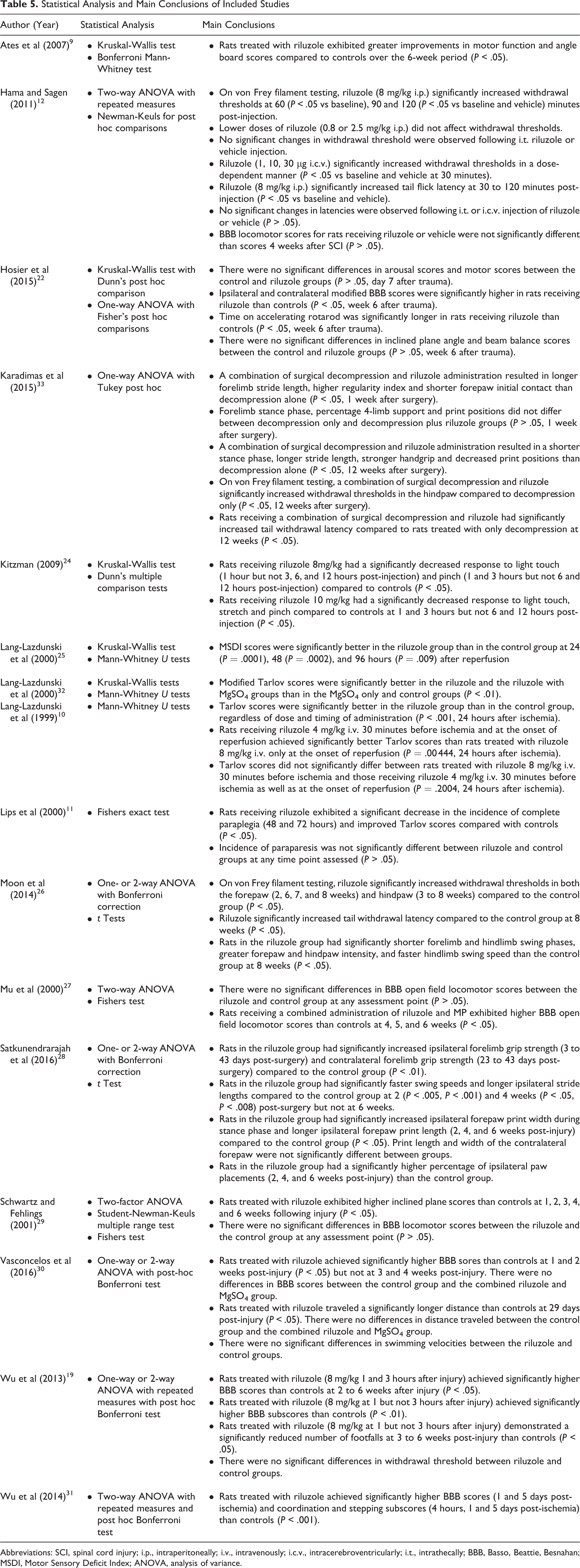
Abbreviations: SCI, spinal cord injury; i.p., intraperitoneally; i.v., intravenously; i.c.v., intracerebroventricularly; i.t., intrathecally; BBB, Basso, Beattie, Besnahan; MSDI, Motor Sensory Deficit Index; ANOVA, analysis of variance.
Basso, Beattie, Besnahan Locomotor Score
Six studies evaluated the impact of riluzole on BBB locomotor scores in rats with SCI. 12,19,22,27,29,30 In a study by Hosier et al, rats treated with riluzole exhibited significantly higher BBB scores than controls in both the ipsilateral and contralateral limbs at 6 weeks following injury. 22 Vasconcelos et al also demonstrated improved BBB scores in a riluzole group at 1 and 2 weeks postinjury, but not at 3 or 4 weeks. 30 Furthermore, only rats in the riluzole group were able to achieve plantar weight support at 3 weeks. In a third study, rats treated with 8 mg/kg riluzole intraperitoneally at 1 and 3 hours after injury exhibited significant improvements in BBB score compared to controls at 2 to 6 weeks following injury. 19 However, only rats administered with riluzole 1 hour after injury demonstrated significant improvement on the BBB subscores. In contrast, 3 studies indicated no association between BBB score and riluzole administration in preclinical models of SCI. 12,27,29
A single study by Wu et al examined BBB score and subscores in rats treated with riluzole 4 hours after occlusion of the aorta. 31 Based on their results, riluzole preserved function at 1 and 5 days following ischemia. Furthermore, rats treated with riluzole had significantly higher stepping and coordination subscores than controls.
Von Frey Filament
Four studies assessed sensitivity to innocuous mechanical stimulation using von Frey filament testing. 12,19,26,33 In a study by Haman and Sagen, riluzole administered intraperitoneally significantly increased withdrawal thresholds at 60, 90, and 120 minutes posttreatment. 12 Furthermore, intracerebroventricular injection of riluzole increased withdrawal thresholds in a dose-dependent manner. 12 In contrast, lower doses of intraperitoneal riluzole (0.8 or 2.5 mg/kg) or riluzole administered intrathecally did not affect the response to mechanical stimuli. A second study by Moon et al also demonstrated increased withdrawal thresholds in both paws in rats treated intraperitoneally with riluzole. 26 Furthermore, a combination of decompression surgery and riluzole was superior at attenuating mechanical allodynia as compared to decompression alone. 33 Finally a study by Wu et al failed to identify significant differences in response to mechanical stimuli between rats injected with riluzole and controls. 19
Tail Flick
Three studies assessed thermal hyperalgesia using the tail flick test. 12,26,33 Intraperitoneal injection of riluzole significantly increased tail flick latency compared with controls. 12,26 Furthermore, rats receiving a combination of surgical decompression and riluzole had significantly reduced thermal allodynia compared with decompression alone. 33 There were, however, no significant changes in latencies between controls and rats receiving intrathecal or intracerebroventricular riluzole. 12
Tarlov Score
Three studies evaluated the impact of riluzole on Tarlov scores. 10,11,32 Based on their results, rats receiving riluzole exhibited significantly improved Tarlov scores compared with controls. Furthermore, the incidence of complete paraplegia, defined as a Tarlov score of 0, was significantly lower in rats treated with riluzole. 11 In contrast, the incidence of paraparesis, defined as a Tarlov score of 1, 2, or 3, was not significantly different between riluzole and control groups. 11 Furthermore, results from the study by Lang-Lazdunski et al indicated that riluzole given before ischemia is more effective than riluzole injected at the onset of reperfusion. 10
Gait Analysis and Grip Strength
Gait analysis was performed in 4 studies. 19,26,28,33 Compared with controls, riluzole resulted in significantly shorter limb swing phases; greater paw pressure; longer ipsilateral stride length, print width during stance phase and print length; reduced number of footfalls; and a higher percentage of ipsilateral paw placements. 19,26,28 Print length and width of the contralateral forepaw, however, did not significantly differ between riluzole and control groups. 28 In a study by Karadimas et al, a combination of surgical decompression and riluzole resulted in longer forelimb stride length, higher regularity index, shorter forepaw initial contact, shorter stance phase, stronger hand grip, and decreased print positions than decompression alone. 33 Finally, ipsilateral and contralateral grip strength were significantly higher in rats treated with riluzole than controls. 28
Inclined Plane Scores
Three studies assessed the association between riluzole and inclined plane scores. 9,22,29 In a study by Schwartz and Fehlings, rats treated with riluzole exhibited higher inclined plane scores than controls at 1 to 4 and 6 weeks following injury. 29 This positive finding was confirmed by Ates et al. 9 In contrast, the study by Hosier et al did not detect significant differences in inclined plane scores between the control and riluzole groups in a model of unilateral cervical SCI. 22
Other Measures
Based on single studies, rats receiving riluzole exhibited significantly longer time on accelerating rotarod, improved motor sensory deficit index, and a longer distance traveled on an activity test than controls. 22,25,30 Furthermore, a study by Kitzman demonstrated that rats receiving 10 mg/kg of riluzole had a significantly decreased response to light touch, stretch, and pinch compared with controls at 1 and 3 hours, but not at 6 and 12 hours, post injection. 24 In contrast, single studies identified no association between treatment with riluzole and beam balance scores or swimming velocities. 22,30
Discussion
This systematic review aims to evaluate the association between riluzole and neurobehavioral outcomes in preclinical models of traumatic and nontraumatic SCI. Based on the results, riluzole has a significant impact on locomotor scores, gait parameters, and measures of hyperalgesia and mechanical allodynia.
The most common outcome assessment tool used across studies was the BBB locomotor score, which was originally designed to evaluate midline thoracic injuries. 22 Other tests were also conducted to assess upper extremity function in isolation as well as coordination of the forelimbs and hindlimbs; these included grip strength, accelerating rotarod, and beam balance. 22,28 As summarized by this review, riluzole significantly improved motor recovery, locomotion, and functional outcomes in a variety of animal models of traumatic and nontraumatic SCI. Potential explanations for these results include (1) sparing of serotonergic and glutamatergic fibers involved in maintaining posture, initiating locomotion, and/or modulating neuronal circuits and (2) increased neuron counts in the red, reticular, and vestibular nuclei. 29,30 In contrast, a study by Vasconcelos et al reported that riluzole did not affect swimming velocities; this is likely because swimming does not require body weight support due to buoyancy. 30 Furthermore, some studies indicated that riluzole does not have an impact on BBB locomotor score, upper extremity function, or coordination. 12,22,27,29 Timing and duration of riluzole administration may also be a relevant consideration; specifically, rapid and prolonged treatment enables immediate and continued blockage of glutamatergic excitotoxicity and improved neurobehavioral outcomes. 11,19
This review also indicated that riluzole may attenuate neuropathic pain and suppress spasticity. Injury to the spinal cord results in hyperalgesia and mechanical allodynia below the level of injury while increasing spontaneous activity at the dorsal horn. 12,26 These findings were confirmed by increased withdrawal thresholds to innocuous stimuli and an increase in tail flick latency following application of a radiant heat stimuli. 12,26 Moreover, there was a significant decrease in response to noxious and non-noxious stimuli, including stretch, pinch, and light touch. 24 Riluzole affects these outcomes by modulating glutamatergic excitotoxicity in the dorsal horn. 26 Furthermore, an increase in spontaneous activity may be due to a decrease in inhibitory GABAergic interneurons at the dorsal horn as well as a reduction of descending inhibition from the bulbospinal serotonergic and adrenergic neurons. 12 Longer term changes may also occur, including increased gene transcription of voltage gated sodium channels, causing abnormal physiological responses to peripheral stimulation. Finally, the administration dose may be an important consideration as lower doses of riluzole (eg, 0.8 or 2.5 mg/kg) do not improve sensitivity to innocuous mechanical stimuli. 12 The dose response gradient observed on intracerebroventricular injection of riluzole signifies that the brain (in addition to the peripheral nerves) may also be a key site of riluzole’s actions; specifically, it is hypothesized that the ventral posterolateral nucleus of the thalamus may be an important target. 12
Riluzole may also significantly improve gait parameters. In a study by Karadimas et al, surgical decompression was associated with increased blood flow and reperfusion of the spinal cord parenchyma. 33 Reperfusion of the gray matter resulted in chronic and persistent neuronal oxidative damage as well as increased expression of DNA damage repair processes. 33 This study also demonstrated that riluzole can attenuate ischemia reperfusion injury associated with decompression surgery, decrease oxidative damage, and protect against destruction of the mitochondrial membrane. Furthermore, a combination of surgical decompression and riluzole can significantly improve forelimb function and nearly restore a smooth and rhythmic gait pattern. 33 In addition to restoration of motor function, improvement in gait parameters may also reflect a reduction in sensitivity to mechanical stimulation. In a study by Moon et al, rats treated with riluzole had significantly longer contact between the paw and the glass plate. 26
This review provides the background information necessary to interpret the results of clinical trials on the impact of riluzole in traumatic and nontraumatic SCI. Improvements in tissue damage and neurobehavioral outcomes may significantly affect quality of life in these patients.
Strengths and Limitations
This systematic review reflects the first to evaluate the impact of riluzole on neurobehavioral outcomes in preclinical models of traumatic and nontraumatic SCI. Strengths of this review include the following: (1) the search strategy was extensive, (2) 2 reviewers independently evaluated the articles for eligibility, (3) the evidence was assessed using the SYRCLE tool, and (4) the review was formatted using the PRISMA guidelines. Limitations of this review include the following: (1) studies were excluded if they were not in English and (2) it was challenging to assess certain domains of the SYRCLE tool.
Conclusion
In preclinical models of traumatic and nontraumatic SCI, riluzole significantly improves locomotor scores, gait function, and measures of neuropathic pain. This review provides the background information necessary to interpret the results of clinical trials on the impact of riluzole in traumatic and nontraumatic SCI.
Supplemental Material
Supplemental Material, Riluzole_Appendix - The Impact of Riluzole on Neurobehavioral Outcomes in Preclinical Models of Traumatic and Nontraumatic Spinal Cord Injury: Results From a Systematic Review of the Literature
Supplemental Material, Riluzole_Appendix for The Impact of Riluzole on Neurobehavioral Outcomes in Preclinical Models of Traumatic and Nontraumatic Spinal Cord Injury: Results From a Systematic Review of the Literature by Lindsay A. Tetreault, Mary P. Zhu, Jefferson R. Wilson, Spyridon K. Karadimas and Michael G. Fehlings in Global Spine Journal
Footnotes
Acknowledgments
MGF would like to acknowledge support from the Gerry and Tootsie Halbert Chair in Neural Repair and Regeneration as well as the DeZwirek Family Foundation. The authors acknowledge support from the AOSpine Knowledge Forum Spinal Cord Injury for Translational research with riluzole.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
