Abstract
Study Design:
Prospective cohort study.
Objectives:
The purpose of this prospective study was to evaluate a protocol for radiation-sparing kyphoplasty by assessing dosemetrically recorded radiation exposures to both patient and surgeon.
Methods:
This prospective clinical study examines the radiation exposure to patient and surgeon during single-level kyphoplasty in 32 thoracolumbar osteoporotic vertebral body fractures (12 OF 2, 9 OF 3, 11 OF 4 types) using a radiation aware surgical protocol between May 2017 and November 2019. The radiation exposure was measured at different locations using film, eye lens and ring dosemeters. Dose values are reported under consideration of lower detection limits of each dosemeter type.
Results:
A high proportion of dosemeter readings was below the lower detection limits, especially for the surgeon (>90%). Radiation exposure to the surgeon was highest at the unprotected thyroid gland (0.053 ± 0.047 mSv), however only slightly above the lower detection limit of dosemeters (0.044 mSv). Radiation exposure to the patient was highest at the chest (0.349 ± 0.414 mSv) and the gonad (0.186 ± 0.262 mSv). Fluoroscopy time, dose area product and number of fluoroscopic images were 46.0 ± 17.9 sec, 124 ± 109 cGy×cm2, and 35 ± 13 per kyphoplasty, respectively. Back pain significantly improved from 6.8 ± 1.6 to 2.5 ± 1.7 on the numeric rating scale on the first postoperative day (P < 0.0001).
Conclusions:
The implementation of a strict intraoperative radiation protection protocol allows for safely performed kyphoplasty with ultra-low radiation exposure for the patient and surgeon without exceeding the annual occupational dose limits.
Trial registration:
The study was registered in the German Clinical Trials Register (DRKS00011908, registration date 16/05/2017).
Keywords
Introduction
Vertebral body fractures cause a substantial loss of quality of life due to pain, impaired mobility, reduced physical function and secondary late complications. With the population continuing to age, the challenges of treating osteoporotic vertebral body fractures will also increase. Vertebral fractures occur in 30%-50% of people over 50 years of age, with an increasing incidence in both female and male after age 50. 1 Conservative treatment of osteoporotic vertebral fractures by means of analgesia, rehabilitation, and brace has shortcomings. Minimally invasive percutaneous procedures such as balloon kyphoplasty have been shown to be favorable in treating pain and improving function in selected patients while preventing progression of kyphosis. 2 In percutaneous balloon kyphoplasty, almost all steps of the procedure, such as insertion of the instruments, inflation of the kyphoplasty balloon, and cement injection, are performed under fluoroscopic monitoring, which raises the question of the associated radiation exposure.
Generally, attention in spinal surgery is more and more focused on radiation awareness, especially as the dose limits are increasingly adjusted downward. At the same time, dosemetric data on intraoperative radiation exposure to the surgeon and patient are hardly available.3-5 Often only fluoroscopy times have been reported,6-8 but their extent can be underestimated. For instance, taking into account the current recommendations of the International Commission on Radiological Protection (ICRP) 9 for occupationally exposed persons, a previous report with a fluoroscopy time of 27.7 min and surgeon’s radiation exposure to the eye lens of 0.328 mSv per vertebroplasty 3 implicated that the annual dose limit for the surgeon would already be exceeded after 61 vertebroplasty procedures.
Therefore, the aim of this study is to evaluate a radiation-sparing protocol for percutaneous kyphoplasty by measuring the intraoperative radiation exposure to the patient and surgeon using dosemeters at various locations.
Methods
Design, Patient Characteristics
This prospective, single-institution study investigates the intraoperative dosemetrically assessed radiation exposure to the surgeon and patient during 32 single-level fluoroscopy-guided kyphoplasty procedures of the thoracolumbar spine between May 2017 and November 2019. Inclusion and exclusion criteria are given in Table 1.10,11
Inclusion and Exclusion Criteria.

Included patients (age: 78.5 ± 7.4 years; body mass index: 26.1 ± 5.9 kg/m 2 ) underwent kyphoplasty for 12 OF 2, 9 OF 3 and 11 OF 4 vertebral body fractures according to the osteoporotic fracture (OF) classification system of the DGOU (German Society for Orthopaedics and Trauma). 10 Treated were fractures from T6 to L4, with the vertebral body of T12 (n = 8) being most frequently affected, followed by L4 (n = 6) and L1 (n = 5).
Ethics Statement
The local ethics committee approved the study (reference number 64/17). The study was registered in the German Clinical Trials Register (DRKS00011908, registration date 16/05/2017). All patients gave their informed consent to the procedure and participation in the study.
Outcome Measures
The primary outcome was the dosemetrically assessed radiation exposure to the surgeon and the patient using film dosemeters (AWST GD 60, Hp(10)), eye lens dosemeters (Eye-D with LiF: Mg, Cu, P detectors, Hp(3)) and ring dosemeters (AWST TD 60, with LiF: Mg, Ti detectors, Hp(0,07)).12,13 The surgeon wore X-ray protection equipment including lead apron (lead equivalent front part Pb 0.7 mm; Mavig, Munich, Germany), thyroid protection (lead equivalent: 0.5 mm; Mavig, Munich, Germany) and lead glass goggles (lead equivalent: 0.75 mm: X-Ray Protective Glasses BR118, Mavig, Munich/Germany). Film, eye lens and ring dosemeters were attached unprotected to the trunk, forehead (using a headband) or fingers in order to represent a worst-case scenario when recording the radiation exposure during single-level fluoroscopy-guided kyphoplasty. A further, protected film dosemeter was placed below the lead apron at chest level. On the patient, unprotected film dosemeters were attached to different locations of the trunk (thyroid, chest, pubic symphysis as approximation for gonads of either sex). Two additional dosemeters of each dosemeter type were positioned outside the operating room for background subtraction. Each dosemeter was read in the Individual Monitoring Service of the Helmholtz Zentrum München (German Research Center for Environmental Health, Munich, Germany). For each kyphoplasty an unused dosemeter set was utilized.
Dosemetric evaluation is always associated with Physics-related difficulties such as appropriate background subtraction and measurement accuracy. Therefore, we included uncertainty models concerning the contribution of background subtraction as well as energy and angular dependence, among others, to derive detection limits of dosemeters for the obtained net doses. 14 On this basis we assumed a lower detection limit of 0.044 mSv for film dosemeters, and 0.030 mSv for eye lens and ring dosemeters, as in previous studies. 15
Secondary outcome measures were fluoroscopy time, dose area product, number of fluoroscopic images per kyphoplasty, improvement of local pain (numeric pain rating scale) and complications such as cement extravasation. Furthermore, we calculated the maximum allowable annual number of operations according to the surgeon’s average radiation exposure and the occupational dose limits of the ICRP. 9
Surgical Technique
The patient was placed in prone position under general anesthesia and equipped with dosemeters. The principal surgical steps are shown in Table 2 and Figure 1.
Principal Surgical Steps of Radiation-Sparing Kyphoplasty in Brief (see also Figure 1).

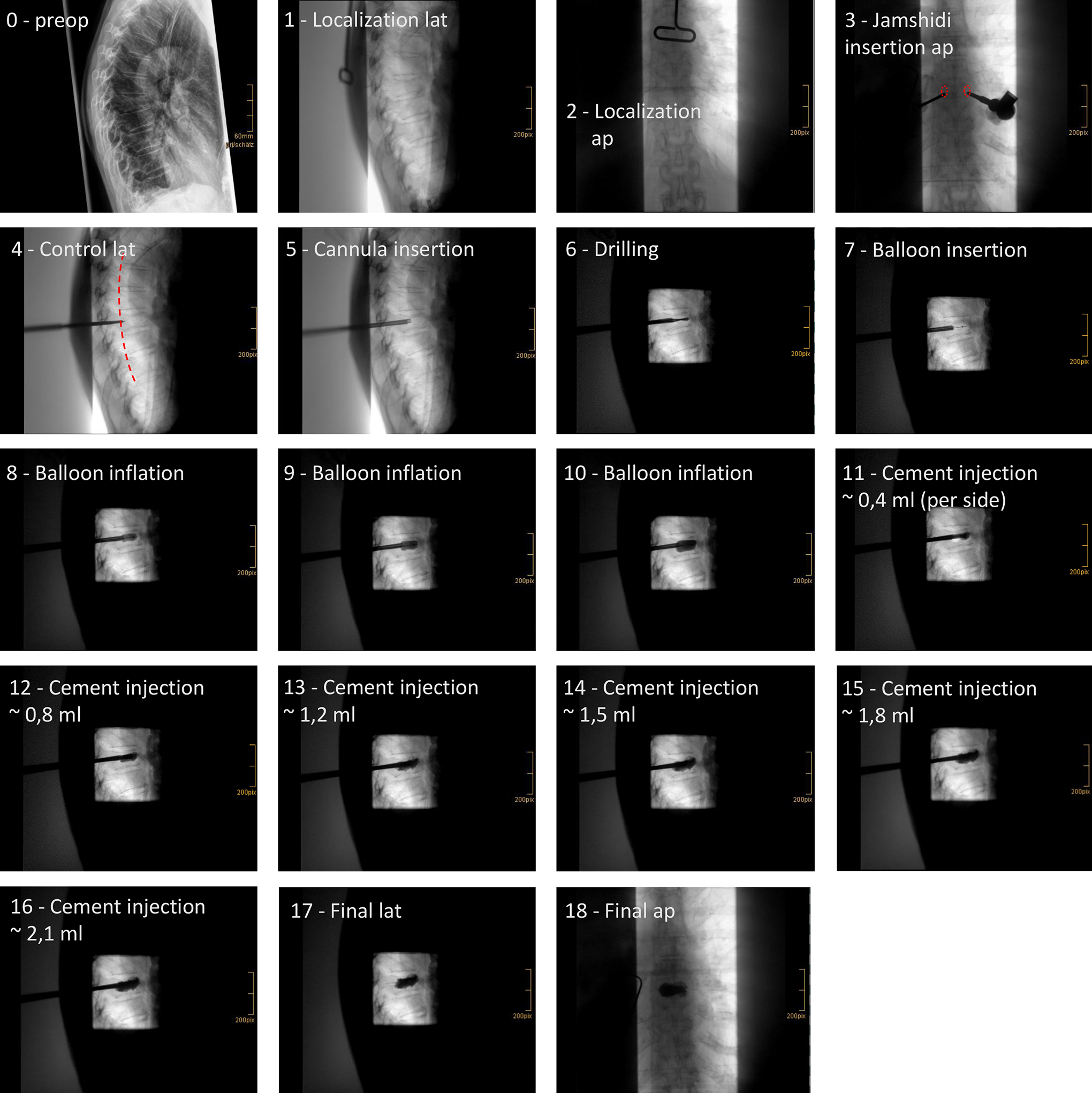
The figure demonstrates the application of the protocol for radiation-sparing kyphoplasty in an osteoporotic fracture (OF 3) of T8 (patient no. 21). Shown are 18 of 28 intraoperative single-shot fluoroscopic images required in this case (fluoroscopy time: 37 sec, dose area product: 27.5 cGy/cm 2 ). After localization (1, 2), bone needles were inserted on both sides in anterior-posterior (ap) projection up to the medial margin of the pedicles (2, pedicles superimposed in red). The correct intrapedicular position was verified in lateral (lat) projection (4), posterior wall of the vertebral bodies is superimposed in red). This was followed by positioning of the working cannulas in the posterior third of the corpus vertebrae (5), drilling of a working channel (6), insertion of the kyphoplasty balloon (7) with gradual inflation of the balloon (8-10). The cement distribution was monitored with single-shot fluoroscopic images after every 0.3 to 0.4 ml of cement injection per side in this case (11-16), without evidence of intraspinal or prevertebral cement extravasation. Continuous fluoroscopy was not used at any time. Note the collimation from both sides at the beginning (1-5), and then the maximum collimation applied in the further course (6-17). The operating room staff kept distance to the radiation source for each X-ray exposure and stepped back behind mobile lead glass walls.
In all cases, percutaneous bilateral kyphoplasty was performed using a commercially available balloon kyphoplasty system (KMC Kyphoplasty System, Shanghai Kinetic Medical Co., Ltd., Shanghai, China) and a state-of-the-art mobile C-arm with digital flat-panel detector (Ziehm Vision RFD 3D, Ziehm Imaging GmbH, Nuremberg, Germany).16,17
To minimize radiation exposure, we strictly adhered to the ALARA principle (as low as reasonably achievable) and established a protocol for radiation-sparing kyphoplasty (Table 3, Figure 1), whose radiation-sparing surgical principles have been strictly followed.
Protocol for Radiation-Sparing Kyphoplasty.

Statistical Analysis
The radiation dose values were calculated as follows: First, subtraction of the average radiation dose of the 2 respective background dosemeters from the gross radiation dose value of each individual dosemeter reading. Second, if this subtracted value was below the lower detection limit for the net dose, then setting the final radiation dose value conservatively to the respective lower detection limit (0.030 mSv or 0.044 mSv). By using the (metrologically based) maximum expected dose value, this approach prevents reporting false low dose values.
Statistical analysis was performed using Prism 6 for Mac (GraphPad Software Inc., La Jolla, USA). Results were expressed as mean ± standard deviation. Variables for numeric pain rating scale rejected normality (Shapiro-Wilk normality test) and were analyzed using Wilcoxon matched-pairs signed rank test. A P value of <0.05 was considered statistically significant.
Results
Radiation Exposure Data
The dosemetric results of the intraoperative radiation exposure to the surgeon and patient after background subtraction of the reference dosemeters are shown in Figure 2. The dosemeter readings for the surgeon were predominantly below the lower detection limits, at least 83%. Mean radiation exposure to the surgeon was highest at the unprotected thyroid gland (0.053 ± 0.047 mSv), however only slightly above the lower detection limit of the corresponding dosemeter type (0.044 mSv). During one single kyphoplasty of T8, a maximum dose value at the surgeon’s unprotected thyroid gland of 0.307 mSv was measured. Mean radiation exposure to the patient was highest at the chest (0.349 ± 0.414 mSv) and the gonad (0.186 ± 0.262 mSv). During one single kyphoplasty of L1, a maximum dose value of 1.946 mSv was measured at the patient’s chest.
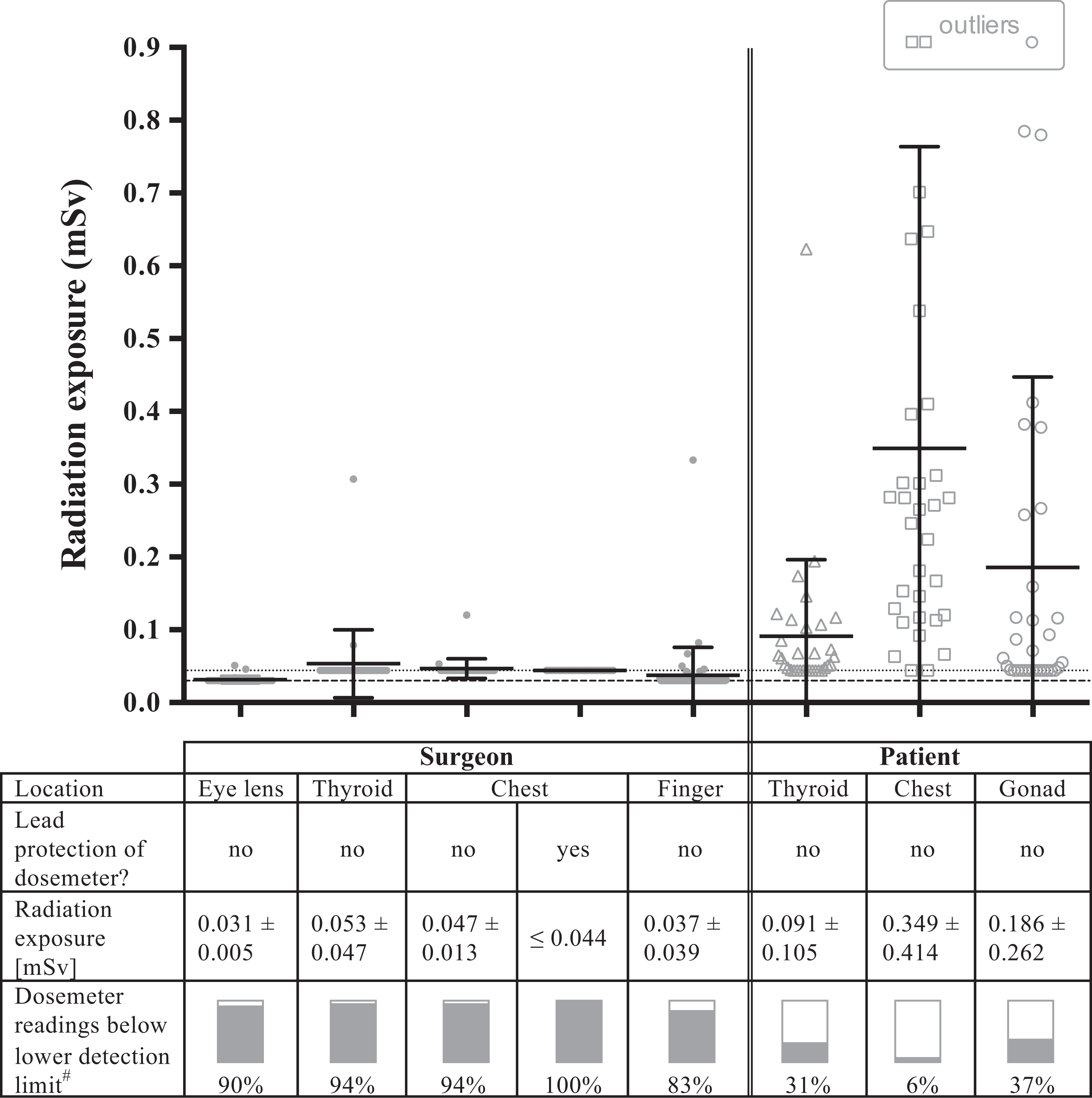
Dosemetrically assessed radiation exposure to the surgeon and patient during 32 single-level fluoroscopy-guided kyphoplasty procedures at various locations in consideration of lower detection limits of dosemeters. Besides the individual dose values, the mean radiation exposures ± standard deviations are shown as bars. # Lower detection limits: 0.030 mSv for eye lens and finger dosemeters (dashed line), and 0.044 mSv for film dosemeters (dotted line), respectively.
Secondary Outcome Data
The operation time per kyphoplasty was 34.6 ± 9.8 min. On average, 2.5 ± 0.8 ml of bone cement was injected on each side per vertebral body. The mean fluoroscopy time per kyphoplasty was 46.0 ± 17.9 sec with 35 ± 13 single fluoroscopic images and a dose area product of 124 ± 109 cGy×cm 2 . All patients reported reduced back pain on the first postoperative day (numeric pain rating scale 2.5 ± 1.7 versus 6.8 ± 1.6 preoperatively; P < 0.0001).
Complications
Intraoperatively, the anesthesiologists were questioned and reported no hemodynamic irregularity during cement injection as a potential sign of pulmonary embolism. Low-grade cement leakage without clinical sequelae was observed in 4 patients, of which 3 were prevertebral and one intradiscal. No epidural cement extravasation was observed.
Discussion
Attention and awareness of medically related radiation exposure has strongly increased in recent years. Any application of ionizing radiation must be critically evaluated in advance to avoid unnecessary radiation exposure. Finally, there is no threshold dose below which ionizing radiation certainly poses no risk according to the stochastic model for radiation damage.18,19
In general, a distinction must be made between the radiation exposure of the patient and that of the surgeon. The patient is usually exposed to a one-time surgery-related radiation exposure. The surgeon, on the other hand, is exposed on a daily basis and therefore requires particular protection. Especially the eye lens of occupationally exposed personnel has become the focus of attention over the past few years as the dose limit for radiation-related late manifestations of the eye lens is estimated to be lower than previously assumed. Consequently, the ICRP recently revised the recommended dose limit for the eye lens from 150 mSv for occupationally exposed personnel to 20 mSv per year. 9 This may explain why dosemetry of the eye lens was rarely carried out in the early 2000s. Radiation aware surgeons as in the present study would exceed this annual dose limit at the earliest after 646 kyphoplasty procedures. However, we intentionally examined the worst-case scenario of completely unprotected eye lenses so that a further reduction of radiation exposure is achievable by wearing lead glass goggles. 20 Consequently, a considerably higher annual number of permissible kyphoplasty procedures can be assumed when wearing lead glass goggles. The annual occupational dose limit of 500 mSv for the unprotected skin 9 would first be exceeded after 9,434 kyphoplasty procedures (location of the thyroid gland). Nevertheless, it is recommendable to wear thyroid protection in order to further reduce the radiation dose.
Several studies report only the fluoroscopy time during cement augmentation procedures and do not provide details on the surgical procedure or the way the C-arm device and thus X-rays are used. Very high fluoroscopy times of up to 36 times (27.7 versus 0.77 min in the present study) 3 or even more 21 have been reported in the literature compared with the presented study. It can therefore be assumed that the recommendations for radiation-sparing practices listed in Table 3 were not sufficiently applied. For example, it is not necessary to visualize every step of the procedure fluoroscopically. Moreover, we do not support the opinion that some instruments must be held by the surgeon during the application of ionizing radiation, as is still advocated even in recent reports. 22 Furthermore, the permanent radiographic control of cement injection by means of continuous fluoroscopy, as recommended in the early 2000s, must be considered obsolete.
Table 4 gives an overview of selected studies on cement augmentation procedures from the past 2 decades. The table shows large differences in fluoroscopy times and radiation exposures, which can be explained by a heterogeneous use of ionizing radiation and radiation protection measures. With the exception of the publication of Zhang et al, 23 all authors report fluoroscopy times of at least one minute,2,8,24-27 mostly more than 5 minutes3,22,28-34 or even up to 60 minutes (Mehdizade et al). 21 Correspondingly, high dosemetrically recorded radiation exposures have been described at the surgeon’s eye lens. For example, in a setting reported by Fitousi et al 3 the annual dose limit would be exceeded after 61 vertebroplasties.
Overview of Studies on Radiation Data During Cement Augmentations per Vertebral Body.
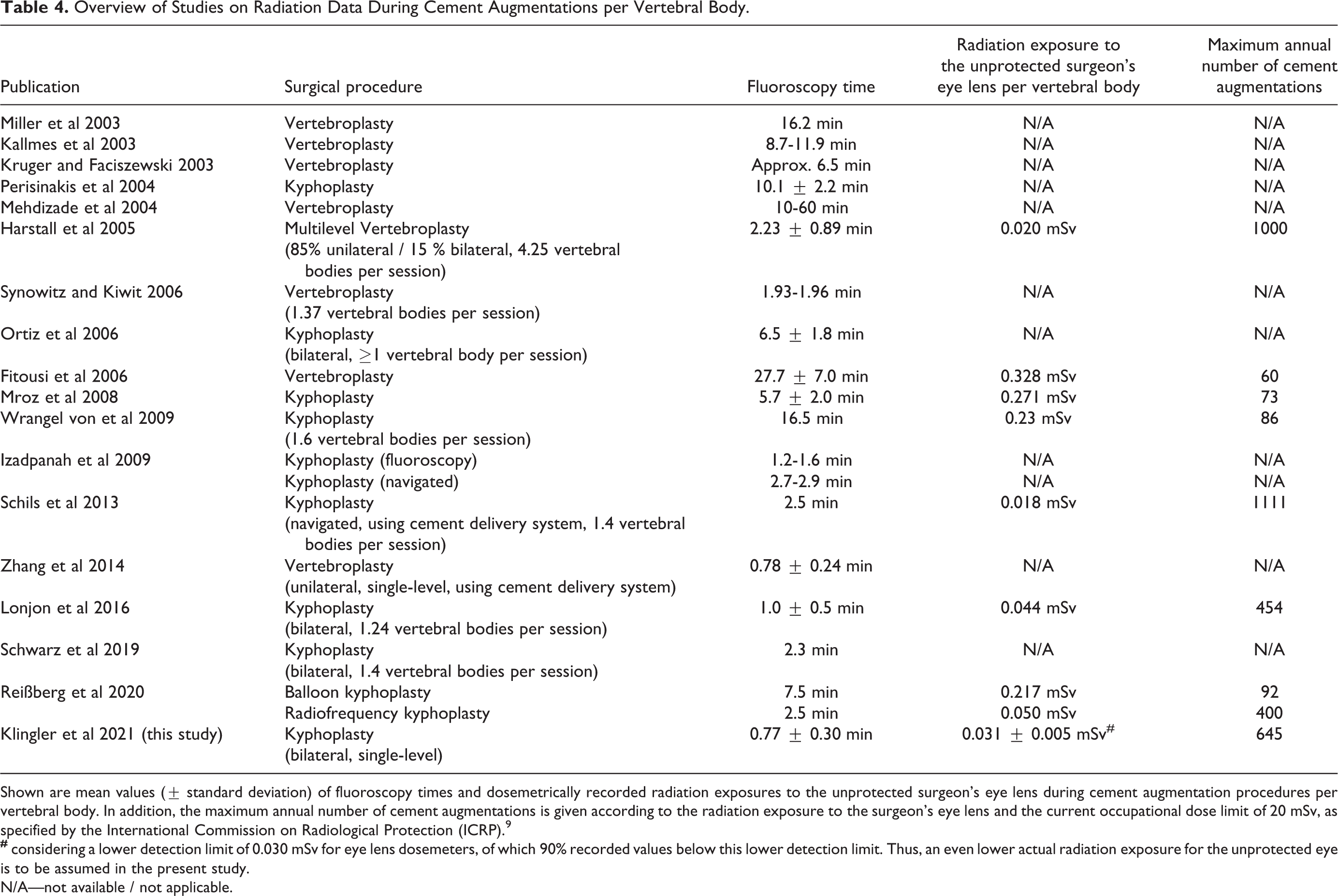
Shown are mean values (± standard deviation) of fluoroscopy times and dosemetrically recorded radiation exposures to the unprotected surgeon’s eye lens during cement augmentation procedures per vertebral body. In addition, the maximum annual number of cement augmentations is given according to the radiation exposure to the surgeon’s eye lens and the current occupational dose limit of 20 mSv, as specified by the International Commission on Radiological Protection (ICRP). 9
N/A—not available / not applicable.
However, the listed studies are not directly comparable; some are quite inhomogeneous and involve varying numbers of augmented vertebras, which can lead to a comparatively lower radiation exposure calculated per treated vertebral body. Likewise, unilateral approaches or dedicated cement delivery systems can lead to comparatively lower fluoroscopy times or radiation exposures. It is also important to note that kyphoplasty, compared to vertebroplasty, involves additional steps that are typically performed under fluoroscopic control.
Few reports of acceptable radiation exposure to surgeons are available. The studies of Harstall et al 24 and Schils et al 2 have demonstrated low radiation exposures at the surgeon’s eye lens (0.020 and 0.018 mSv), but still reported fluoroscopy times of more than 2 minutes. This suggests radiation-conscious behavior of the surgeon, e.g. by stepping back from the patient during the application of fluoroscopy. At the same time, however, it would have been desirable if the authors had also been able to further reduce the fluoroscopy time in order to lower the radiation exposure for the patient as well. In the presented study, however, we were able to achieve both of these objectives by evaluating a radiation-sparing protocol with both a reduction in fluoroscopy time to 0.77 min and a very low exposure of the surgeon’s unprotected eye lens of 0.031 mSv.
Besides, Harstall et al 24 treated an inhomogeneous group of 30 patients in 32 sessions, in which a total of 136 vertebral bodies (4.25 vertebral bodies per session) were augmented, mostly via a unilateral access (85%). As a result of these 2 facts, lower radiation data are in principle achievable compared to the presented investigation, in which solely bilateral single-level kyphoplasty was performed. Likewise, the studies of Schils et al 2 and Lonjon et al 26 included bilevel cement augmentation. Zhang et al 23 achieved a fluoroscopy time of less than 1 minute for single-level vertebroplasty, though using a dedicated cement delivery system and a unilateral access. Unfortunately, these authors did not perform eye lens dosemetry.
In the presented study, we demonstrate the feasibility and efficacy of the proposed radiation-sparing protocol leading to ultra-low fluoroscopy times and radiation exposures to both patient and surgeon. Overall, the highest priority must be given to the safety of the patient. It is therefore not a question of achieving a minimization of radiation exposure at all costs, but rather, depending on the expertise of the surgeon, a reduction in radiation exposure while maintaining a favorable surgical result without complications. The present study shows that the dosage of radiation to the patient and the surgeon does not have to be conflicting. Rather, it proved that a considerable reduction in radiation exposure is achievable for both the patient and the surgeon at the same time and should therefore ideally be aimed for. None of the observed low-grade cement leakages (4 of 32 thoracolumbar fractures) was epidural or led to clinical sequelae. Moreover, the anesthesiologists did not report any hemodynamic irregularity during cement injection as a potential sign of pulmonary embolism. The mean pain reduction from 6.8 to 2.5 on the first postoperative day confirmed the efficacy of the proposed kyphoplasty technique and is comparable to the results reported in the literature.
Limitations
First, there is no direct comparative group in our study. Second, if the dosemeters are read after each kyphoplasty, as in the presented study, the dose values may be close to or below the lower detection limit—in contrast to the reading of accumulated dosemeter values after multiple procedures. Although assuming the maximum expected radiation doses might have masked small differences, false low radiation doses are avoided, which is of utmost importance. Third, several surgeons performed the kyphoplasty procedures. On the one hand, this leads to certain heterogeneity of the surgical techniques, but on the other hand this situation reflects the actual training situation in a tertiary university center and furthermore, that also residents can implement a radiation aware surgical technique.
Conclusions
The presented radiation-sparing protocol for percutaneous kyphoplasty implied radiation-conscious behavior and only essential use of the imaging device. With this given protocol, it is estimated that at least 645 cases may be performed without exceeding the annual occupational dose limits.
Footnotes
Authors’ Note
Conception and design: Klingler, Hubbe; Acquisition of data: all authors; Provision and evaluation of dosemeters: Brönner, Hoedlmoser; Analysis and interpretation of data: Klingler, Brönner, Hoedlmoser, Naseri; Drafting the manuscript: Klingler; Critically revising the manuscript: all authors; Approving the final manuscript: all authors. The study was approved by the local ethics committee (64/17) and registered in the German Clinical Trials Register (DRKS00011908). Parts of this work were presented at the Annual Meeting of the Spine Section of the German Society of Neurosurgery (DGNC) September 6-7, 2019, Gießen, Germany, and at the Annual Meeting of the German Spine Society (DWG) November 28-30, 2019, Munich, Germany. In addition, parts of this work were accepted for presentation at the Annual Meeting of the German Society of Neurosurgery (DGNC) June 21-24, 2020, Lübeck, Germany.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The clinic had a cooperation agreement for system development with Stryker and Ziehm. Ulrich Hubbe stated max. 1.000 USD from B.Braun Aesculap for consulting.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing charge was funded by the Baden-Württemberg Ministry of Science, Research and Art and the University of Freiburg in the funding programme Open Access Publishing.
