Abstract
Study Design
Systematic Review.
Objectives
Infection following vertebral augmentation using Percutaneous vertebroplasty (PV) and percutaneous balloon kyphoplasty (PBK) is an exceedingly rare, but potentially life-threatening complication. This systematic review evaluates the microbiological characteristics, risk factors, treatment strategies and outcomes of these infections.
Methods
PubMed, Scopus, and Mendeley databases (2000-2025) were systematically reviewed. Inclusion criteria involved case cohorts of more than 5 adult patients with infections following PV or PBK. Data on the initial procedure, demographics, infection biomarkers, neurologic status, comorbidities, causative pathogens, treatment protocols, and outcomes were evaluated.
Results
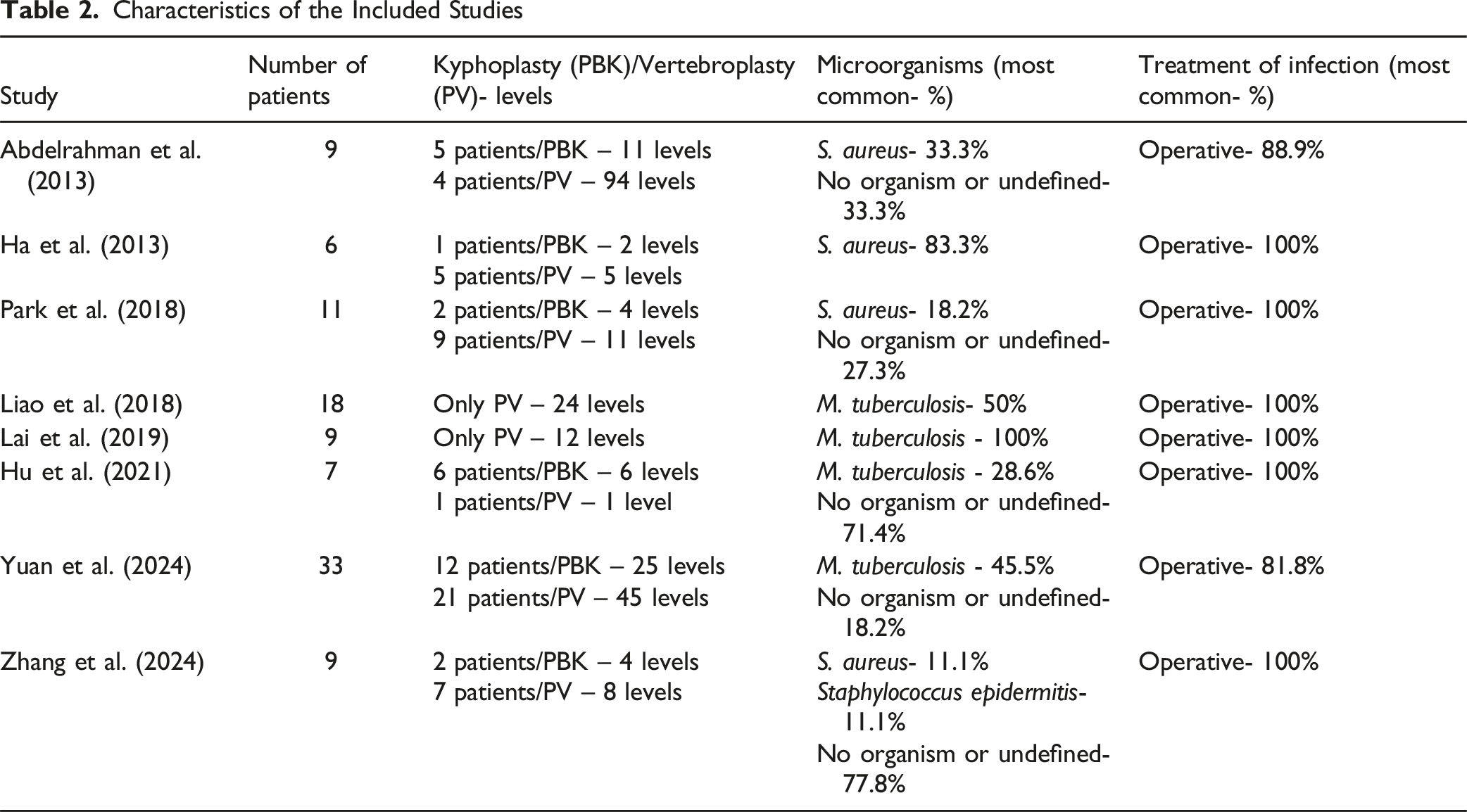
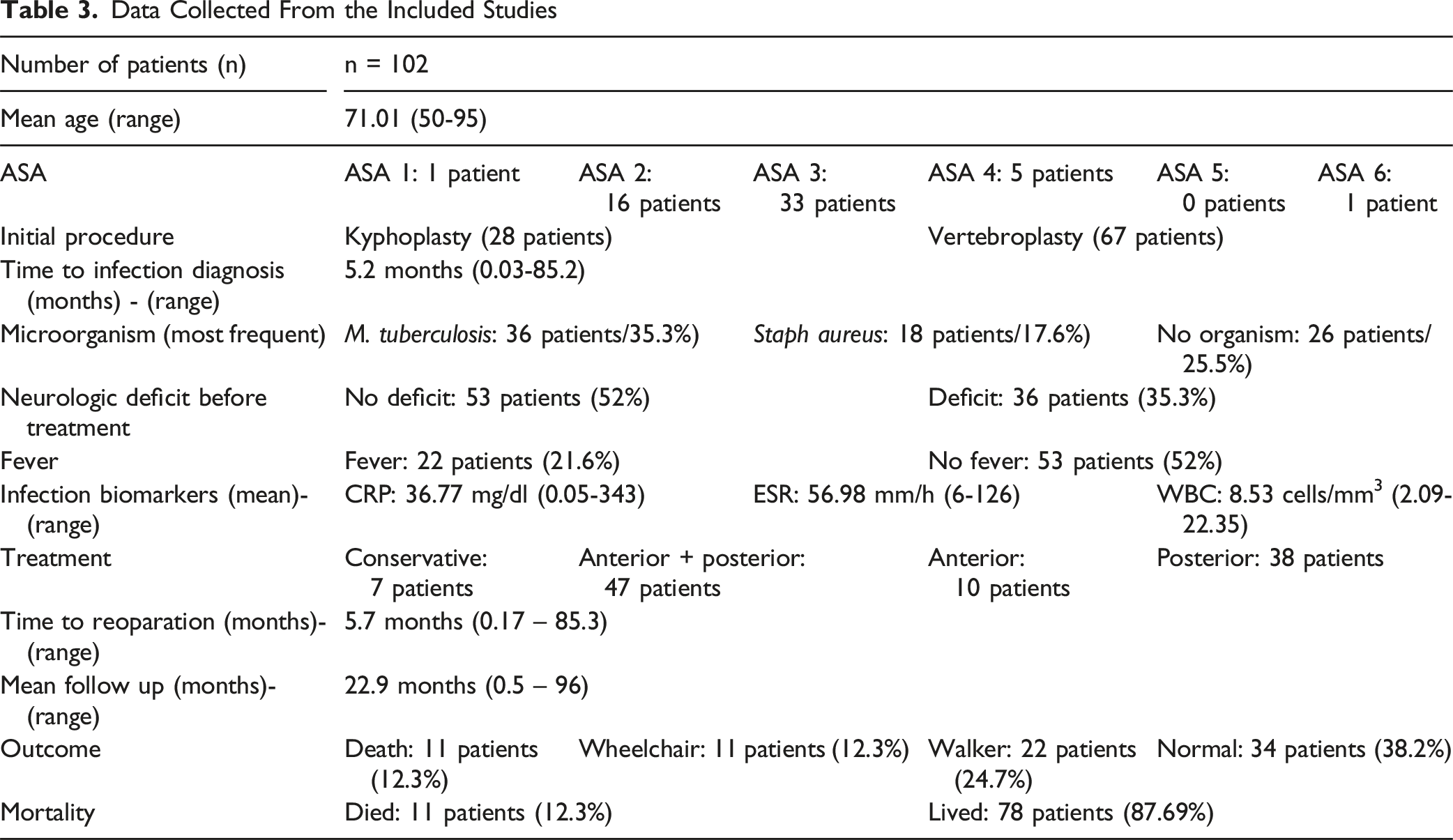
A total of 102 patients from 8 studies were included (mean follow-up: 22.9 months). Most were female (64.7%) with a mean age of 71.0 years (range: 50-95). The mean time interval from the initial procedure to infection diagnosis was 5.2 months (range: 0.03-85.2). The most common pathogen was Mycobacterium tuberculosis (35.3%). The mean infection biomarkers were CRP 36.77 mg/dL (range: 0.05-343), ESR 56.98 mm/h (range: 6-126), and WBC 8.53 cells/mm3 (range: 2.09-22.35). Seven patients (6.1%) were treated conservatively, while 95 patients (93.1%) underwent revision surgery. Outcomes were reported for 89 patients (87%). Final follow-up showed that 34 patients (38%) achieved normal mobility, 22 patients (25%) had assisted mobility (crutches or walker), and 11 patients (12%) required a wheelchair. The mobility status was unclear for 11 patients (12%). The mortality rate was 12.3%. Post-treatment mobility was strongly dependent on preoperative neurologic status (OR = 6.0, P = 0.001).
Conclusions
Post-augmentation spinal infections are rare but serious. Outcomes are strongly influenced by preoperative neurological status, while pathogen type and patient comorbidities have limited prognostic impact.
Keywords
Introduction
Osteoporotic vertebral compression fractures (OVCFs) are the most common fragility fractures and have a significant impact on quality of life in older individuals. 1 Vertebral augmentation procedures include minimally invasive techniques widely utilized as operative management for OVCF. 2 Percutaneous vertebroplasty (PV), first described by Galibert in 1987, involves percutaneous injection of bone cement, usually polymethylmethacrylate (PMMA), directly into the body of the collapsed vertebra. This aims to alleviate pain and prevent further progression of kyphotic deformity. 3 Percutaneous ballon kyphoplasty (PBK), a later modification of vertebroplasty, involves the use of an inflatable balloon tamp within the vertebral body before cement introduction, theoretically offering the advantage of restoring vertebral height and reducing the possibility of cement leakage.3,4
While both PV and PBK are considered cost-effective, safe procedures in terms of pain reduction and functional improvement, they have been linked to potential complications, such as adjacent vertebral fractures, bleeding, cement leakage, neurologic deficit, allergic reaction to bone cement, cardiovascular collapse, pulmonary embolism and infections.5-9 The risk of infection following PV or PBK is exceptionally low with an incidence ranging from 0.36 to 0.46%.10,11 Current literature mostly consists of scarce data from case reports and small series. Nonetheless, these infections may be life-threatening complications and their diagnosis can be challenging, requiring a combination of clinical assessment, imaging modalities and laboratory tests.12-14 Delayed identification of post-augmentation infections can lead to detrimental consequences, including increased mortality rates. 13 In addition, treatment strategies remain unclear. The mainstay of treatment involves surgical debridement and stabilization, while some authors advocate stand-alone conservative treatment with antibiotics.10,11,15,16
The primary objective of this systematic review was to evaluate the clinical outcomes of spinal infection treatment following percutaneous PBK or PV, with specific attention to mortality, mobility, and pain. The secondary objective was to correlate clinical outcomes with patient characteristics such as past medical history (PMH), neurological and functional status, isolated pathogens, and management-related factors, including the type of treatment.
Materials and Methods
Search Strategy
PubMed, Scopus and Mendeley databases were systematically searched from January 2000 to January 2025. The used key terms were “vertebral augmentation procedures”, “cement augmentation”, “vertebroplasty”, “kyphoplasty”, “cementoplasty” and “infection.” This systematic review was registered in PROSPERO and was prepared in accordance with Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) 2020 guidelines. 17 Moreover, the PICOS (population, intervention, comparison, outcome, study type) question format was used to plan the analysis. 18 Thus, prospective or retrospective studies reporting adult patients who developed spinal infection after PV or PBK were included in the analysis. Duplicate records were removed using the Mendeley Reference Manager program. The quality of the studies was assessed by 2 authors using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Case Series. 19 In case of disparity between the 2 authors during the screening and data extraction process, a third reviewer was consulted in order to achieve consensus.
Inclusion Criteria
1. Adult population. 2. English language. 3. Full text available. 4. Prospective or retrospective cohort studies reporting patients who developed infection after PV or PBK at any time after the initial procedure. 5. Low risk of bias according to JBI Critical Appraisal Checklist for case series (score of at least 7/10 YES responses) 6. Case series of more than 5 patients.
Exclusion Criteria
l. Clinical studies including high energy trauma treated with PV or PBK. 2. Review articles, case reports, technical notes, author opinions and short communication. 3. Experimental in vitro or in vivo studies.
The patients’ demographics, the (American Society of Anesthesiologists) ASA score, the initial operative procedure (PV or PBK), the microorganism identified, regardless of the isolation method, infection biomarkers (White blood cells- WBC, Erythrocyte Sedimentation Rate- ESR, C-reactive protein- CRP), and the neurological status before and after infection management were recorded. Neurological status was described as normal or abnormal. No grading of the neurologic deficit was performed. The risk factors related to patient PMH (diabetes mellitus, heart disease, cancer, bacteremia, obesity, chronic kidney disease (CKD), chronic respiratory disease, smoking, pulmonary tuberculosis (TB), urinary tract infection (UTI), bacteremia and autoimmune disease), which can potentially affect the clinical outcome, were also recorded.
Statistical Analysis
The overall visual analogue scale for pain (VAS) score and the overall mortality risk were calculated using the random effects model. The heterogeneity between studies was measured by means of the Higgins I2 index. 20 The correlation of mortality to microorganism and to the type of operative treatment was estimated using the Pearson’s chi-square test. The binary logistic regression model was conducted to investigate the association of mortality to the infection biomarkers (WBC, ESR, CRP), age, ASA score, pre and post-operative neurological status, and PMH risk factors (diabetes mellitus, heart disease, cancer, bacteremia, obesity, chronic kidney disease (CKD), chronic respiratory disease, smoking, pulmonary tuberculosis (TB), urinary tract infection (UTI), bacteremia and autoimmune disease).
The Pearson’s chi-square test was used to investigate the correlation of post-treatment mobility to the preoperative neurologic deficit and to the type of operative treatment. Abnormal mobility was defined as the use of a walker or wheelchair, with the latter category also encompassing patients who were completely non-ambulatory (eg, bedridden). The binary regression model was conducted to evaluate the correlation of post-treatment mobility to the infection markers, patients’ demographics, ASA score and PMH risk factors. The Nagelkerke R square was used to measure the goodness of fit in the logistic regression process. The statistical analysis was performed using the SPSS 28.0 program.
Results
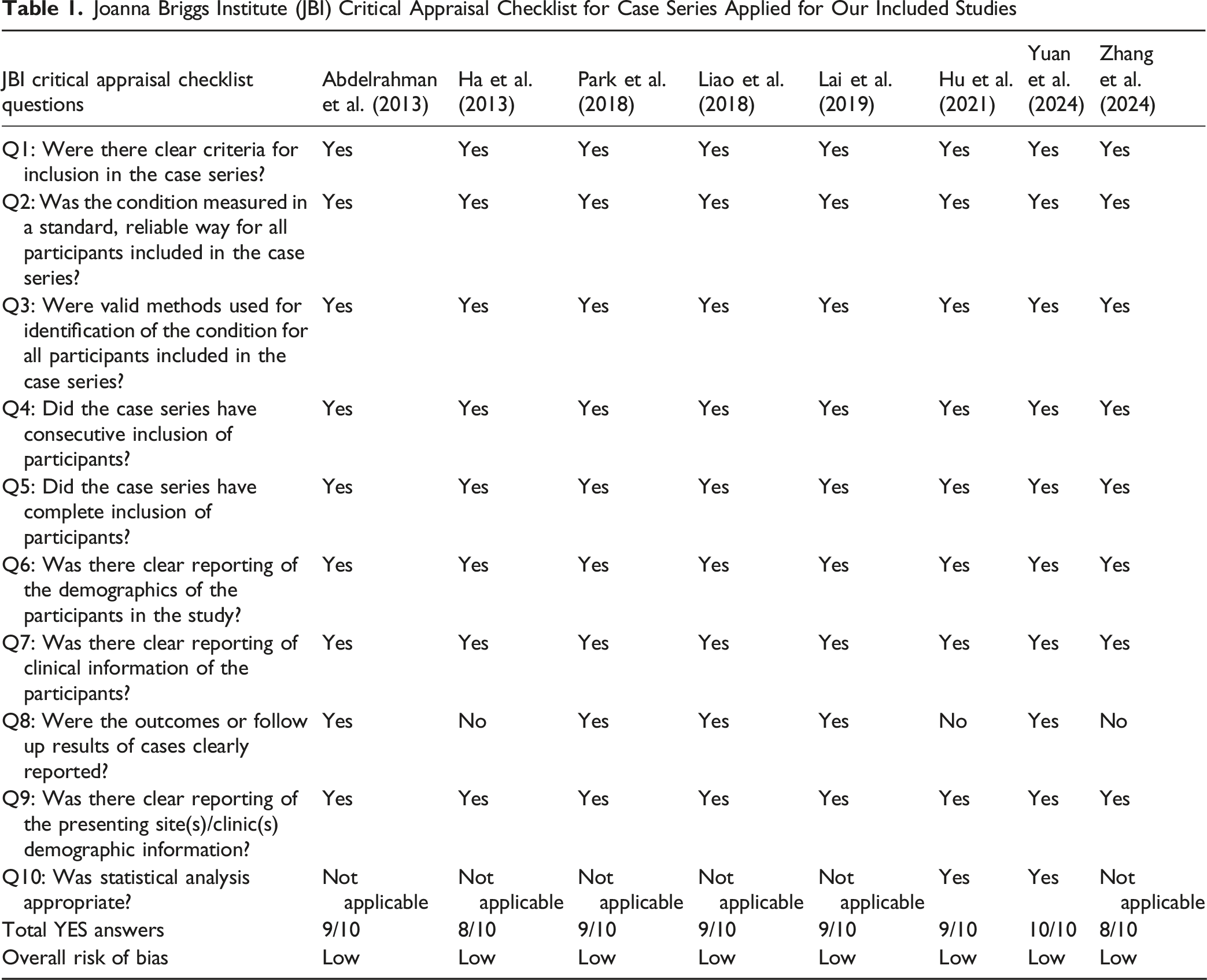
The search initially attributed 1.338 studies. All duplicate records (n = 295) were identified and removed using the Mendeley Reference Manager Program. The “English” filter was applied to the Scopus search engine, and as a result, 95 papers were excluded automatically. Nine hundred fifty articles were excluded by title and abstract leaving 56 articles. Of those, 8 studies were compatible with the eligibility criteria including 102 patients in total and they were used for the quantitative synthesis (Figure 1).10,11,13-16,21,22 Table 1 presents the quality assessment for each study using the JBI Critical Appraisal Tool for Case Series. All the included studies demonstrated high methodological quality and were assessed as low risk of bias. Prisma 2020 Flow Diagram for the Search Strategy Used to Review Infections Following Vertebral Augmentation Procedures Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Case Series Applied for Our Included Studies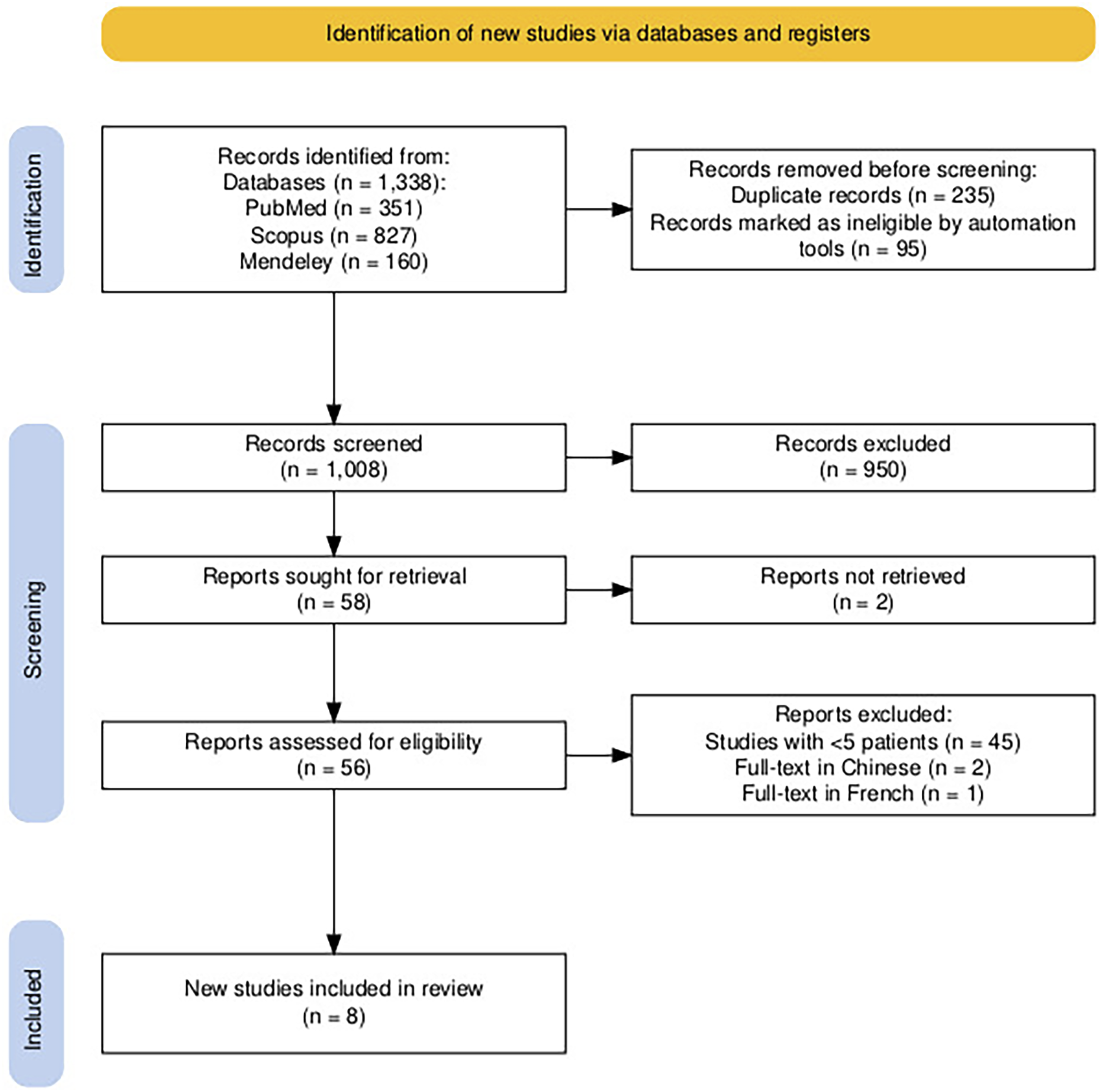
Characteristics of the Included Studies
Data Collected From the Included Studies
Reporting on antibiotic treatment was inconsistent. Zhang et al. reported culture-guided intravenous antibiotics for 2 weeks followed by oral antibiotics for 8 weeks, with empirical therapy if cultures were negative. 14 Hu et al. stated that all of their patients received either antibiotics or anti-tuberculous therapy for a long period but did not specify the regimen or duration. 16 Lai et al. mentioned 6 months of anti-tuberculous therapy in all but 1 patient who died, without providing details on the exact protocol. 22 Park et al. mentioned antibiotic use in 2 surgically treated patients who were presented in detail. They also noted that conservative treatment included antibiotics, but without offering information regarding the agents or duration. No data were provided for the remaining surgically treated cases. 11 The remaining 4 studies did not provide any details on whether antibiotics were administered in their included cases.10,13,15,21
The pooled mean VAS score was 2.43, 95% Confidence Interval (C.I.: 1.85 – 3.00), I2: 0.75 (75% heterogeneity) (Figure 2). The pooled risk of mortality was 0.123 (12.3%), 95% C.I.: −0.01-0.21. Forest Plot of Pooled Mean Postoperative VAS Scores. Squares Represent Mean Scores From Individual Studies With 95% Confidence Intervals, and the Diamond Shows the Overall Pooled Estimate Calculated Using a Random-Effects Model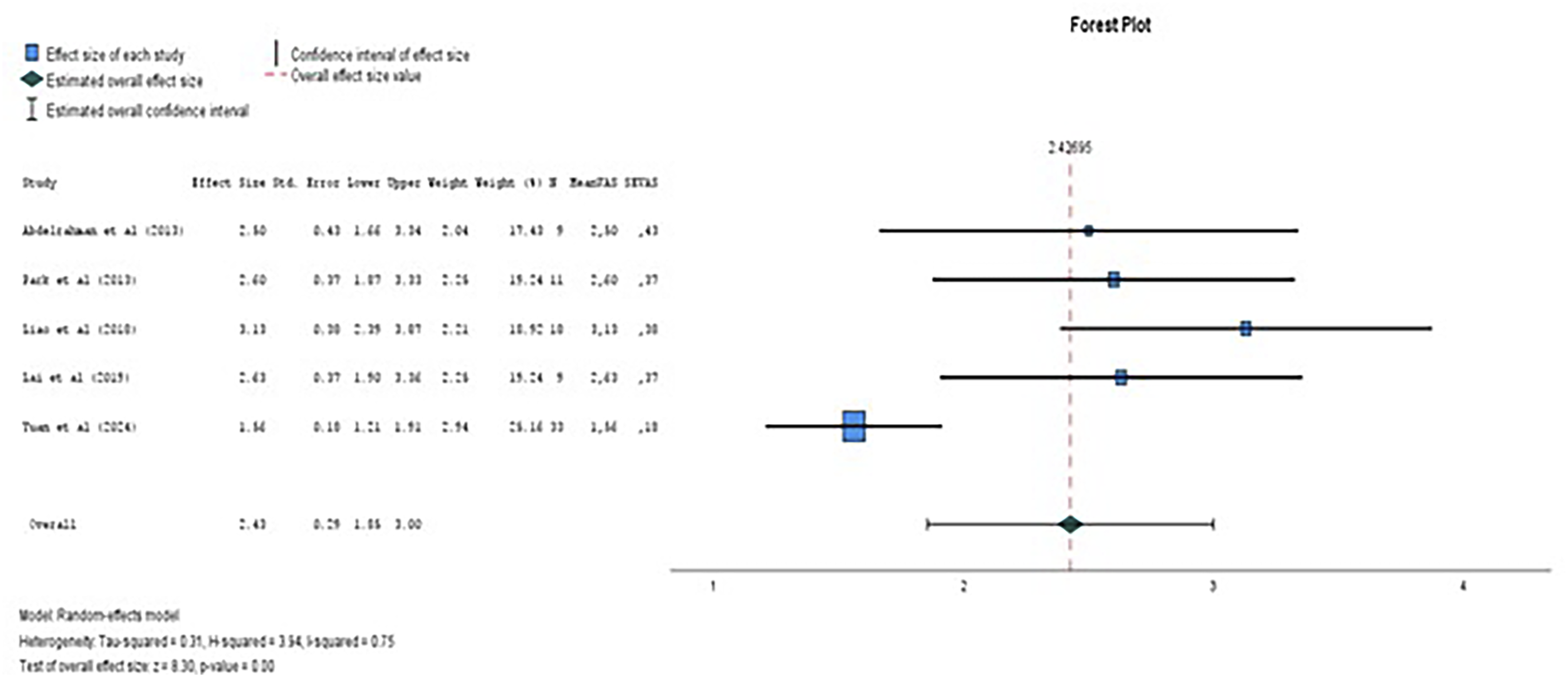
No statistically significant impact of the identified bacteria to mortality was found (P = 0.36). Four out of ten patients (40%) undergoing anterior surgical approach passed away, while none of the 38 patients who were treated through a posterior approach had such an outcome. Three out of 47 patients (6%) who underwent a combined anterior and posterior approach and 2 of 7 (29%) patients who were treated conservatively died. The difference regarding mortality among the former surgical approaches was statistically significant (P – value: 0.001). The logistic regression model found no significant correlation between ASA score, PMH risk factors, pre or post-operative neurological deficit, nor infection biomarkers, and mortality. The addition of the aforementioned independent variables to the model explained the 79.7% (R square: 0.797) of the variability of the mortality being the dependent variable.
The logistic regression model found no significant correlation between PMH risk factors and infection biomarkers to post treatment mobility. The addition of the aforementioned independent variables to the model explained the 76.2% (R square: 0.762) of the mobility variability as the dependent variable. The post treatment mobility was strongly correlated to neurologic status prior to treatment (P – value: 0.001). The odds ratio (OR) was 6.00 (95% C.I.: 2.0-18.2), meaning the existence of preoperative neurologic deficit markedly increases the risk for post-treatment abnormal mobility by 6 times as opposed to the risk for abnormal mobility when there is no deficit prior to treatment (Figure 3). No difference regarding mobility was found among different types of surgical approaches employed (P – value: 0.477). Bar Chart Showing the Association of Post Treatment Mobility to the Neurological Status Before Treatment. Blue Bars Represent Patients Without Neurological Deficit, and Red Bars Represent Patients With a Neurological Deficit; Mobility Outcomes are Categorized as Normal or Abnormal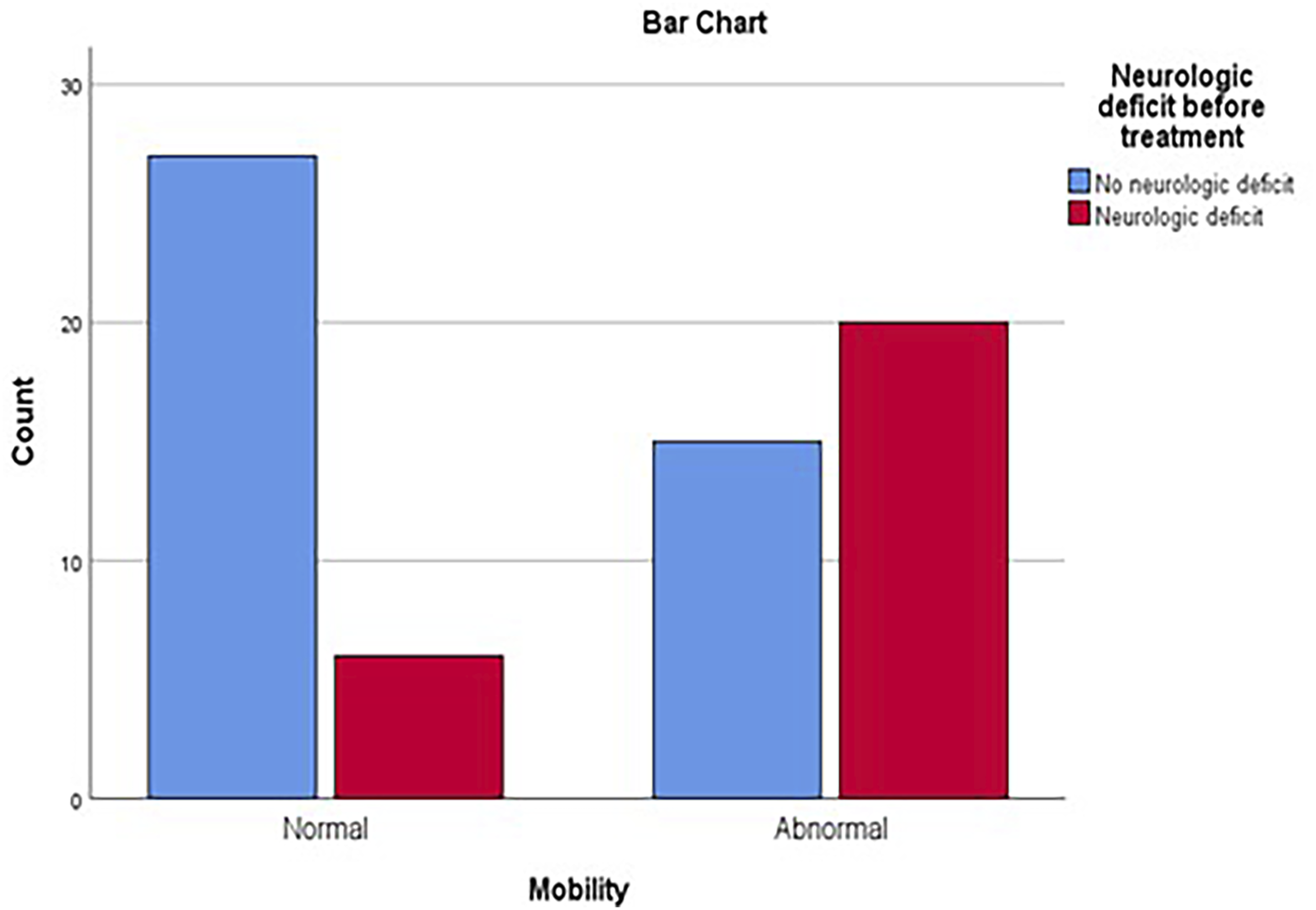
Discussion
This systematic review represents the most comprehensive synthesis to date of infections following PV and PBK, encompassing 102 patients from 8 studies. The primary findings indicate that although such infections are exceedingly rare, they carry a notable risk of morbidity and mortality.10,11 The most commonly isolated pathogen was Mycobacterium tuberculosis (35.3%). Surgical intervention, particularly through anterior or combined approaches, was associated with higher mortality (P = 0.001), whereas conservative management was reserved for a small minority. Importantly, preoperative neurological status emerged as the most significant predictor of post-treatment mobility (OR = 6.0, P = 0.001), while infection biomarkers and patient comorbidities did not significantly influence clinical outcomes. These findings underscore the diagnostic challenges, treatment variability, and need for early recognition and individualized surgical decision-making in managing these infections. The time interval between the initial procedure and diagnosis of infection was inconsistently reported, as this information was provided by only 2 studies. The mean time for diagnosis was 5.2 months, with a wide range spanning from 1 day to 7 years, reflecting the variety of clinical scenarios and advocating the required high index of suspicion required for detection of possible infection. Revision surgery was relatively delayed, with a mean of 5.7 months derived from a wide range of timelines matching the mean time to diagnosis of infection. The small subset of patients that were treated conservatively can be potentially attributed to patients being unfit for any surgical intervention.
The etiology of surgical site infection (SSI) after cement augmentation can be classified as: (1) preexistent spondylitis mistakenly diagnosed as a fracture, (2) infection acquired by direct inoculation during surgical procedure, and (3) infection caused by hematogenous spread. 11 Amongst all patients, only 3 (2.9%), reported by Hu et al., had infections prior to cement augmentation that was initially misdiagnosed as OVCFs. 16 A literature review conducted by Park et al. reported a misdiagnosis rate of 14.3%, which is substantially higher than the findings of this review. 11 However, an early onset infection might be the consequence of cementation in the setting of preexisting infection, especially in cases with tuberculous (TB) spondylitis.11,15,16 This raises the question of whether routine biopsies should be obtained during the initial procedure, which may identify an occult infection or malignancy. The existing evidence regarding this question remains inconsistent. While Pneumaticos et al. do not support the routine use of transpedicular biopsies, a more recent systematic review by Osterhoff et al. suggests that routine biopsies can be considered, especially when preoperative imaging is absent or inconclusive.23,24 In the present systematic review, 12 patients (11.7%) were diagnosed with infection within 30 days after the initial operation, suggesting a possible higher prevalence of misinterpreted preexisting infections as OVCFs.
According to the presented data, the most common pathogen was M. tuberculosis with a rate of 35.3% followed by S. aureus, accounting for 17.6% of these infections. No organism was isolated in a major subset of 25%, suggesting diagnostic difficulties and absence of causative antimicrobial treatment in these cases. Regarding the association between mortality and identified pathogens, the current study showed that the isolated bacteria did not have a statistically significant effect on mortality (P-value = 0.36). TB used to be the most common cause of spinal infections a few decades ago. 25 Recently, however, the microbiological profile of primary spinal infections has shifted, making pyogenic infections the leading cause. S. aureus is the most common causative microorganism for pyogenic spondylitis and spinal epidural abscess, responsible for up to 80% of reported cases.25-28 Staphylococcus species are also the most abundantly invading organisms in SSI following both instrumented and non-instrumented spinal procedures.29,30 However, no consensus exists regarding the leading causative pathogen in SSI following vertebral augmentation procedures. Abdelrahman et al. advocated that S. aureus is the most isolated bacterium. 10 Park et al. reported equal contribution of S. aureus and M. tuberculosis with an incidence of 19% each. 11 On the contrary, Yuan et al in the largest case series study to date suggested that M. tuberculosis was the leading cause of post-augmentation infection with a 45.5% rate. 13 The increased prevalence of M. tuberculosis in the presented results may reflect a plethora of factors. Lai et al. solely included patients with TB spondylitis following PV or PBK; thus, failing to eliminate selection bias in subject inclusion. 22 Moreover, the studies that met the eligibility criteria mainly examined country populations with high TB prevalence, such as Taiwan, China and South Korea.22,31,32 The total sample size including all 102 patients is limited, hindering provision of robust representation of the general trend. Consequently, concluding that M. tuberculosis is the most common cause of infection after PV or PBK is perhaps precarious. Screening for TB prior to cement augmentation procedures may be beneficial in endemic countries. Additionally, ruling out a pre-operative infection that could potentially be misdiagnosed as a fracture is strongly recommended.
Laboratory infection biomarkers, including CRP, ESR and WBC were reported for the majority of patients. ESR and CRP were elevated in 84% and 92% of patients respectively, and serve as supportive tools in the diagnosis of infection. On the other hand, WBC was abnormal in just 22% of the patients, so it is not a reliable biomarker to be used on its own. Despite their use in diagnosis or monitoring, this study demonstrates that infection biomarkers have poor correlation to mortality or mobility.
Individuals with risk factors related to PMH, such as immunosuppression, diabetes mellitus, heart disease, CKD, malignancy or pulmonary TB, have increased susceptibility to spinal infections.10,13,16,21,26,33 Nonetheless, the analysis demonstrates that since infection is established, PMH does not significantly influence mortality or mobility.
In contrast to SSI following other spinal procedures, the optimum treatment protocol for infections following PV or PBK has not been standardized.29,30 Surgical intervention was the preferred management in 93% of the included patients. The chosen surgical approach is still debatable and management of each individual case should be carefully tailored to any given patient. In the cohorts included, anterior column reconstruction with either a 360° approach (47 patients) or a stand-alone anterior approach (10 patients) was most commonly performed. Posterior approach was applied in 38 patients. None of the patients who were operated through posterior approach passed away. Surgical procedures involving anterior column restoration were proven to be related with statistically significant (P – value: 0.001) higher mortality rate. Perhaps patients requiring extensive surgical approaches suffered more destructive infections with severe ramifications. Moreover, the anterior approach is a much more invasive surgical technique with increased operation time and blood loss, so it carries an inherently higher risk of morbidity and mortality. 34 The surgical approach did not, according to the present analysis, affect postoperative mobility status. Conservative treatment was chosen for only 7 patients, most likely reflecting selection of individuals with advanced age, frailty, or comorbidities. The relatively high mortality observed in this group (29%) may therefore be related to these baseline characteristics and infection severity, rather than the treatment modality itself. However, the absence of detailed treatment protocols, including antibiotic regimens, and the very small sample size limit the interpretation of this finding.
There was a considerable degree of consistency among studies in the outcome measures, with the majority using mortality, mobility and pain. The mean follow-up was almost 2 years, which is generally adequate, although the broad range varied from 15 days to 8 years. The findings of this systematic review suggest that infections after PV or PBK have a mortality rate of 10.1% which is higher than the overall mortality rate of SSI following spinal surgery which ranges between 1.1% and 7.7%. 35 Mortality rates for SSI following minimally invasive spine surgery (MISS) are not well-documented, potentially due to the rarity of these infections. This finding underlines the severity of infections after PV or PBK and could be attributed to the increased patient age or the need for extensive revision surgical procedures.
Pooled mean VAS score was effectively reduced to 2.43 following infection treatment, reflecting a notable improvement in patient-reported pain levels, and suggesting that regardless of the therapeutic approach, successful infection control can lead to notable pain relief. Most of the patients achieved a normal mobility status, followed by patients who used a walker and a wheelchair. Prior neurologic deficit is a strong predictor for post-treatment mobility restriction, as patients with preoperative neurological impairment have a 6-time higher risk of abnormal mobility status, emphasizing the value of early diagnosis and prompt management of these infections.
This study is subject to some limitations. Most case series included were retrospective and had a small sample size. In addition, despite the low risk of bias according to JBI tool, selection and information bias are still considerable in case series studies. Another limitation is the inconsistent reporting of microorganism isolation methods, which restricts detailed analysis of diagnostic pathways and may introduce further reporting bias. Preoperative neurologic status was reported inconsistently and heterogeneously across studies. Some of them noted only presence or absence, others provided descriptive terms (eg, paraparesis, radiculopathy) and only 2 studies documented formal graded scales (ASIA and Frankel). For this reason, we did not attempt to apply a uniform grading system in our analysis. While our results confirm that presence of preoperative neurological deficit is the strongest predictor of postoperative mobility, the variability in reporting and the absence of grading in our statistical analysis should be acknowledged as a limitation. Mobility status before vertebral augmentation and at infection diagnosis was not reported and could only be indirectly inferred from the inconsistently described neurological deficits. Therefore, direct comparison across these time points was not possible. In addition, the included studies provided limited and inconsistent data regarding the type or duration of antibiotic regimens, with several failing to specify whether antibiotics were administered at all. Finally, the interpretation of outcomes should ideally be based on quantitative data such as quality of life and neurological status scores, and in the selected cases, data is incomplete. Nevertheless, this review provides the first structured synthesis of clinical characteristics, treatment strategies, and outcomes for post-augmentation spinal infections, offering valuable guidance for clinical decision-making in this rare but serious complication.
Conclusion
Infections following vertebroplasty or kyphoplasty are rare but serious complications, most commonly caused by Mycobacterium tuberculosis and Staphylococcus aureus, with a significant proportion yielding no identifiable pathogen. Surgical management remains the mainstay of treatment, with anterior or combined approaches linked to higher mortality. Presence of preoperative neurological deficit is the strongest predictor of functional outcome, including mobility status, while patient comorbidities and infection biomarkers show limited prognostic value. Effective infection control was associated with meaningful pain relief, independent of the treatment approach. These findings emphasize the need for early diagnosis, careful patient selection, and standardized outcome reporting in future studies.
Supplemental Material
Supplemental Material - Infection Following Vertebroplasty or Kyphoplasty: A Systematic Review of Risk Factors, Treatment Options and Outcomes
Supplemental Material for Infection Following Vertebroplasty or Kyphoplasty: A Systematic Review of Risk Factors, Treatment Options and Outcomes by Konstantinos Zampetakis, Ioannis M. Stavrakakis, Kosmas Samartzidis, Constantinos Chaniotakis, Christothea-Alexandra Tsiridis, Christos Koutserimpas, Kalliopi Alpantaki, MD, PhD in Global Spine Journal
Footnotes
Authors’ Contribution
Conceptualization, K.A.; methodology, K.Z, I.M.S. C.C., C.K., and K.A.; statistical analysis, I.M.S; investigation, K.Z., C.C., K.S., C.A.T., and C.K.; data curation, K.Z., C.C., K.S., and C.A.T.; writing—original draft preparation, K.Z., I.M.S., and K.A.; writing—review and editing, C.K. C.A.T.; visualization, K.A. C.K. ;supervision, K.A.; All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
