Abstract
Study design:
Preclinical ovine model.
Objective:
To assess the in vivo efficacy and safety of the P-15 L bone graft substitute and compare its performance to autologous iliac crest bone graft (ICBG) for lumbar interbody fusion indications.
Methods:
Thirty skeletally mature sheep underwent lumbar interbody fusion surgery. Half of the sheep received autologous ICBG and the other half the peptide enhanced bone graft substitute (P-15 L). Following termination at 1, 3, and 6 months after surgery, the operated segments were analyzed using micro computed tomography (µCT), histology, and destructive mechanical testing. Additional systemic health monitoring was performed for the P-15 L group.
Results:
One month after surgery, there was only minor evidence of bone remodeling and residual graft material could be clearly observed within the cage. There was active bone remodeling between 1 and 3 months after surgery. At 3 months after surgery significantly denser and stiffer bone was found in the P-15 L group, whereas at 6 months, P-15 L and ICBG gave similar fusion results. The P-15 L bone graft substitute did not have any adverse effects on systemic health.
Conclusions:
The drug device combination P-15 L was demonstrated to be effective and save for lumbar interbody fusion as evidenced by this ovine model. Compared to autologous ICBG, P-15 L seems to expedite bone formation and remodeling but in the longer-term fusion results were similar.
Introduction
With a lifetime prevalence of 40-80%, low back pain is a major healthcare problem.1,2 Lumbar interbody fusion is a routine treatment to realize satisfactory pain relief when chronic back pain originates from segment instability and is refractory to conservative treatments.3-5 The frequency of spinal fusion surgery has increased rapidly in the last 2 decades, partly accelerated by the development of new fixation devices and the introduction of alternative bone graft materials. 6 Despite these new innovations, autologous iliac crest bone grafting still remains the gold standard in spinal interbody fusion surgery because of its osteogenic, osteoconductive, and osteoinductive properties. 7 However, harvesting iliac crest bone graft (ICBG) is associated with donor-site morbidity, 8 the amount of ICBG may be insufficient, and the ICBG quality and regenerative capacity are not uniform as they are donor-dependent.
Alternatives to ICBG have been proposed and (pre)clinically tested.9,10 i-FACTOR peptide enhanced bone graft (Cerapedics, Inc., Westminster, CO) is one of those alternatives to autologous ICBG. i-FACTOR consists of synthetically produced 15-amino acid long polypeptides (P-15) chemically bound to anorganic bone mineral (ABM) particles, embedded in a hydrogel carrier.11,12 The P-15 peptides are replicates of the cell-binding domain as naturally found on type-I collagen, 13 whereas the ABM particles serve as osteoconductive 3-dimensional (3D) scaffolds. The proposed mechanism of action following attachment of cells to an ABM/P-15 complex has been described in detail previously.14-17 In short, the cell attachment is proposed to initiate a cascade of cell-signaling that modulates synthesis of extracellular matrix and growth factors to orchestrate cell proliferation, differentiation, and osteogenesis.
Sherman et al were the first to analyze the efficacy of i-FACTOR compared to iliac crest autologous bone for lumbar interbody fusion indications by conducting a small prospective study in an ovine model. 18 In a human clinical study evaluating fusion in single and multilevel anterior lumbar interbody fusion (ALIF), Mobbs et al found i-FACTOR to be safe and effective in a cohort of 110 consecutive patients. 19 Lauweryns and Raskin conducted a prospective comparative analysis in which patients undergoing posterior lumbar interbody fusion (PLIF) received local autologous bone in one cage and i-FACTOR in the other enabling intra-patient control. Bony bridging was realized earlier in the cage enriched with i-FACTOR. 20 Jacobsen et al recently concluded that i-FACTOR graft significantly increased fusion rates compared to allograft when being used as graft extender in non-instrumented lumbar posterolateral gutter fusion. 21 Additionally, the product was already proven to be non-inferior to autologous bone for single-level anterior cervical discectomy and fusion (ACDF) in a multicenter food and drug administration (FDA) investigational device exemption (IDE) study. 22 Moreover, the IDE clinical study demonstrated that i-FACTOR was statistically superior to autologous bone at both 1-year and 2-years following surgery in terms of a composite endpoint defining overall clinical success.22,23
A new iteration of i-FACTOR, called P-15 L (i-FACTOR+ Matrix), has been developed. This product incorporates P-15 coated ABM particles within a bovine collagen carrier to offer alternate handling properties compared to the existing i-FACTOR Putty. The purpose of this study was to assess the in vivo efficacy and safety of P-15 L and compare its performance to iliac crest autologous bone graft in lumbar interbody fusion. Bone remodeling was monitored in an ovine interbody fusion model using post-mortem micro computed tomography (µCT), histomorphometry, histopathology, and destructive testing following termination at 3 different time points after surgery. In parallel, this study evaluated any impact of P-15 L on systemic tissues and organs. Additionally, hematology and serum biochemistry was performed.
Methods
Animal Model and Study Design
All procedures were reviewed and approved by the testing facility’s institutional animal care and use committee (AccelLab, Boisbriand, QC, Canada). Thirty skeletally mature, female Dorset X Rideau Arcott hybrid sheep were included (range 35-76 kg). The first cohort (n = 6) underwent spinal fusion at a single intervertebral level (L2-L3) while the second and third cohort (n = 12 for both) underwent spinal fusion at 2 non-contiguous intervertebral levels (L2-L3 and L4-L5). All operated levels in this study were treated with a polyetheretherketone (PEEK) interbody cage (22x10x7 or 22x10x8 mm). Within each cohort, half of the sheep received the cages grafted with ICBG and half received the cages grafted with P-15 L. The 3 cohorts were terminated after a period of 1, 3, and 6 months, respectively.
Surgical Procedure
A retroperitoneal approach allowed access to the lumbar spine. Discectomy and distraction were completed and trial implants were used to determine the appropriate implant size. For the ICBG group, an incision was made over the iliac crest in order to harvest cortico-cancellous bone and the site was flushed with sterile saline and closed in layers using appropriate sutures. For the P-15 L group, the putty was hydrated with saline and kneaded for approximately 30 seconds to prepare for implantation. An appropriate amount of graft material was placed to completely fill the PEEK cage in both groups. The cage was implanted into the prepared disc space and a plastic intervertebral plate was secured into place using 4 metal screws.
Follow-Up and Termination
Animals were group housed where hay and tap water were provided ad libitum. General welfare monitoring and observation was performed at least twice a day. At the designated time point, the animals were euthanized by a lethal injection of saturated potassium chloride. All animals underwent necropsy. For the P-15 L group only, organ sections (heart, kidney, lung, liver, and spleen) were harvested for semi-quantitative histopathological evaluation and blood samples were withdrawn for hematology and serum biochemistry. The entire lumbar spinal column was removed en-bloc and connecting soft tissue and transverse processes were removed from operative segments. Segments L4-L5 were used for mechanical testing while segments L2-L3 were used for µCT scanning and histological analysis. Scientists were blinded to the bone graft that was used, and only received the segments and their corresponding unique identifier.
µCT Morphology and Stiffness Analysis
All L2-L3 segments were scanned at a 25 µm isotropic resolution in a µCT system (Nikon XT H 225, Nikon, Tokyo, Japan). Images were converted to Scanco proprietary format and evaluated in associated software (SCANCO Medical AG, Brüttisellen, Switzerland) and downscaled to a resolution of 50 µm. After noise suppression by a 3D Gaussian filter (σ = 0.8, support = 1 voxel), bone tissue was segmented based on voxel reference values for air and dense cortical bone which were calculated within each image dataset separately. Since metal screws were used to constrain the plastic plate and tantalum markers were embedded in the PEEK cages, the region around the screws and a few millimeters in the central fusion region were excluded from all analyses due to metal streak artifacts (Figure 1).
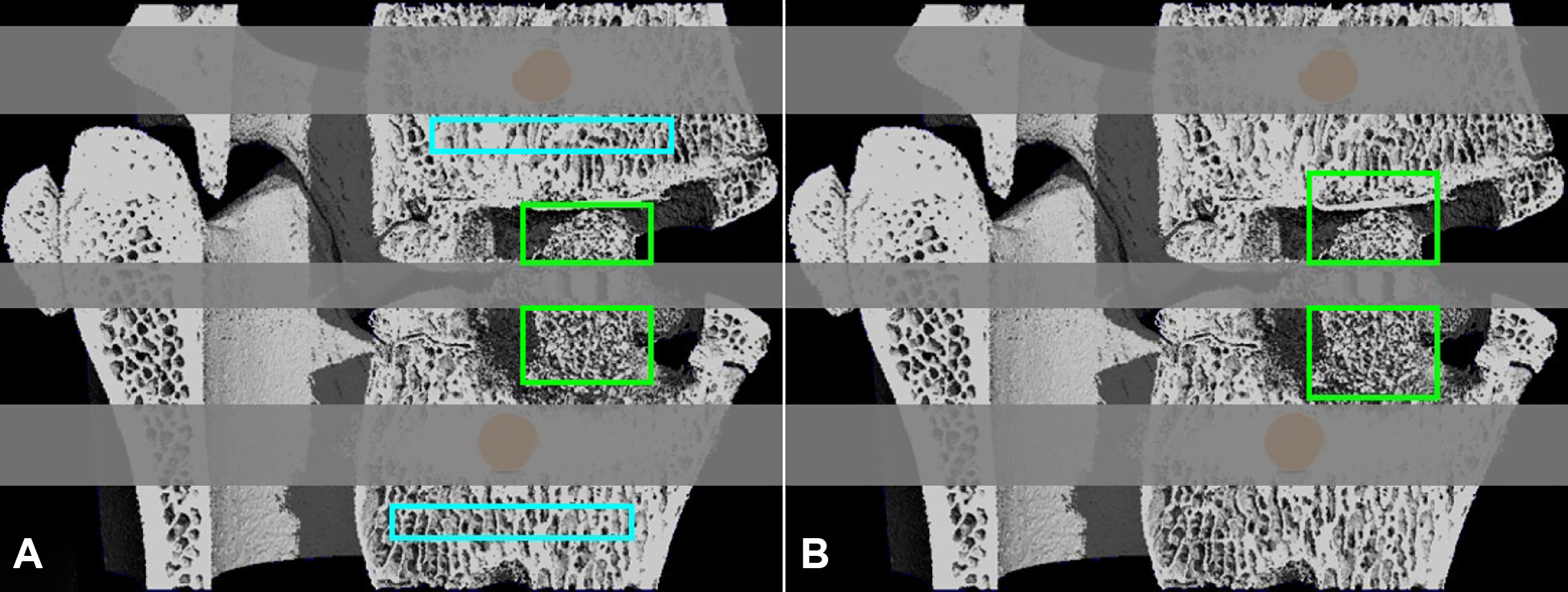
Sagittal cut of the 3D rendered segmentation of a representative sample of the 1 month cohort. The regions excluded due to metal artifacts are greyed out. (a) The artifact-free region of the graft window in green and the unaffected cancellous bone reference regions in cyan. (b) Both regions of the graft window were exteriorly propagated to ensure a uniform height of 4 millimeters for the μFE analysis.
Axial slices were manually contoured, segmenting the complete graft window of the cage. Slices without a complete footprint of the cage were excluded from the segmentation as these likely contain original bone structures. For the artifact-free region of the graft window, the bone volume over total volume (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb.Th), trabecular separation (Tb.Sp), and connectivity density (Conn.D) were determined. To obtain reference values for these morphological parameters, the same parameters were calculated for unaffected cancellous bone regions in the vertebrae adjacent to the cage (Figure 1A).
For a mechanical evaluation, the regions cranial and caudal to the marker artifacts used for morphology analysis were both propagated exteriorly to realize a consistent region height of 4 millimeters for all datasets (Figure 1B). Inclusion of normal bone as result of these extensions was inevitable. However, inclusion of these structures will not adversely affect the eventual analysis as the stiffness of an inhomogeneous structure is primarily determined by the weakest substructure, i.e. the newly formed bone within the cage. The bone voxels within the regions were converted to 3D brick elements to enable micro finite element (µFE) modeling. Bone was assumed to be isotropic linear elastic with a Young’s modulus of 10 GPa and a Poisson’s ratio of 0.3. Boundary conditions applied represented a 1% compressive strain in the longitudinal direction with “glued” interface conditions. The stiffness of both the region cranial and caudal to the artifacts were calculated as the reaction force over the applied deformation.
Histological Analysis
Following µCT scanning, the undecalcified blocks were dehydrated in graded ethanol series, infiltrated with methylmethacrylate, and polymerized. A midsagittal cut extending through the graft region was obtained, microgrounded, and polished down to approximately 60 µm (Exakt technologies, Inc., Oklahoma City, OK, USA). Next, the section was stained with Goldner’s Trichrome for osteoid and mineralized bone differentiation. Histomorphometric analysis using Image-Pro Premier software (Media Cybernetics, Inc., Rockville, MD, USA) quantified the bone with marrow area within and normalized to the intervertebral region of interest (ROI). The ROI was defined from one endplate to the other, restricted to the width of the internal graft window of the cage.
Stained sections of the intervertebral segments were all evaluated for efficacy and safety by a certified pathologist. Sections were graded from 0 to 4 for inflammatory cell type (lymphocytes, macrophages, polymorphonuclear, and giant cells) and tissue response (neovascularization, fibrosis, fatty infiltrate, peri-implant hemorrhage, cartilage formation, and residual graft) based on ISO 10 993-6. 24 Additionally, inflammation response, bone formation and quality of bone bridging were assessed with a qualitative scale from 0 to 3. 25
Destructive Tensile Testing
Spinous processes, laminas, pedicles, and plastic plates were removed from the L4-L5 segments providing the isolated interbody fusion region. The segments were anchored using stainless steel screws and secured into custom metal fixtures with potting material, ensuring horizontal and concentric alignment. The potted samples were mounted into a testing machine (ElectroPuls E3000, Instron, Norwood, MA, USA) and a uniaxial tensile load was applied to the upper vertebra at a constant cross-head speed of 0.5 mm/min until failure of the implant-bone interface. Peak load and corresponding ultimate displacement were derived from the force-displacement curve which was digitally recorded at a rate of 10 Hertz.
Statistical Analysis
All values are expressed as mean ± standard deviation (SD). The 2 different graft groups were compared within the 1, 3, and 6 month follow-up cohort. For the µCT analysis, results from the regions cranial and caudal to the marker artifact were first averaged within the sample as they were found to be similar. Normality and equal variance tests were performed for continuous data. When both were not violated, unpaired, 2-tailed t tests were performed. In case of non-normality or no equal variances, a Mann-Whitney U test was performed. A Mann-Whitney rank sum test was performed in case of ordinal data. Statistical significance was set at p < 0.05.
Results
Surgical procedures were without any adverse events and all animals completed the experiment according to protocol. Necropsy and additional histopathological evaluation of systemic organs were normal and consistent with surgical intervention.
Sagittal µCT cuts for both groups at the different evaluation points can be found in Figure 2. Note that residual graft material can be recognized in the P-15 L group as hyper-opaque particles. Morphometric analysis revealed higher bone volume, higher trabecular thickness, higher trabecular number and lower trabecular separation in the P-15 L group compared to the ICBG group 1 month after surgery. Three months after surgery, these differences are more pronounced for the bone volume and trabecular thickness, and less pronounced for the trabecular number and separation. In addition, lower values for the connectivity density and higher values for the stiffness of the bone were found for the P-15 L compared to the ICBG group. The stiffness and all morphological parameters but trabecular thickness were shown to eventually reach similar values for both groups after 6 months. A continuous increase of the stiffness of the new bone was predicted for both groups. Independent of graft group, the connectivity density increases first, after which it slightly decreases again. For the P-15 L group, changes in other morphological parameters are especially observable between the 1 and 3 month time point suggestive of rapid bone formation and remodeling. Although similar trends are found in the ICBG group regarding trabecular number and separation, the bone volume and trabecular thickness seem to change more gradually over time. In contrast, the morphology parameters of the unaffected cancellous bone in the reference region do not change over time. At every time point, both graft groups differ from the reference region for at least one of the morphological parameters (Figure 3).
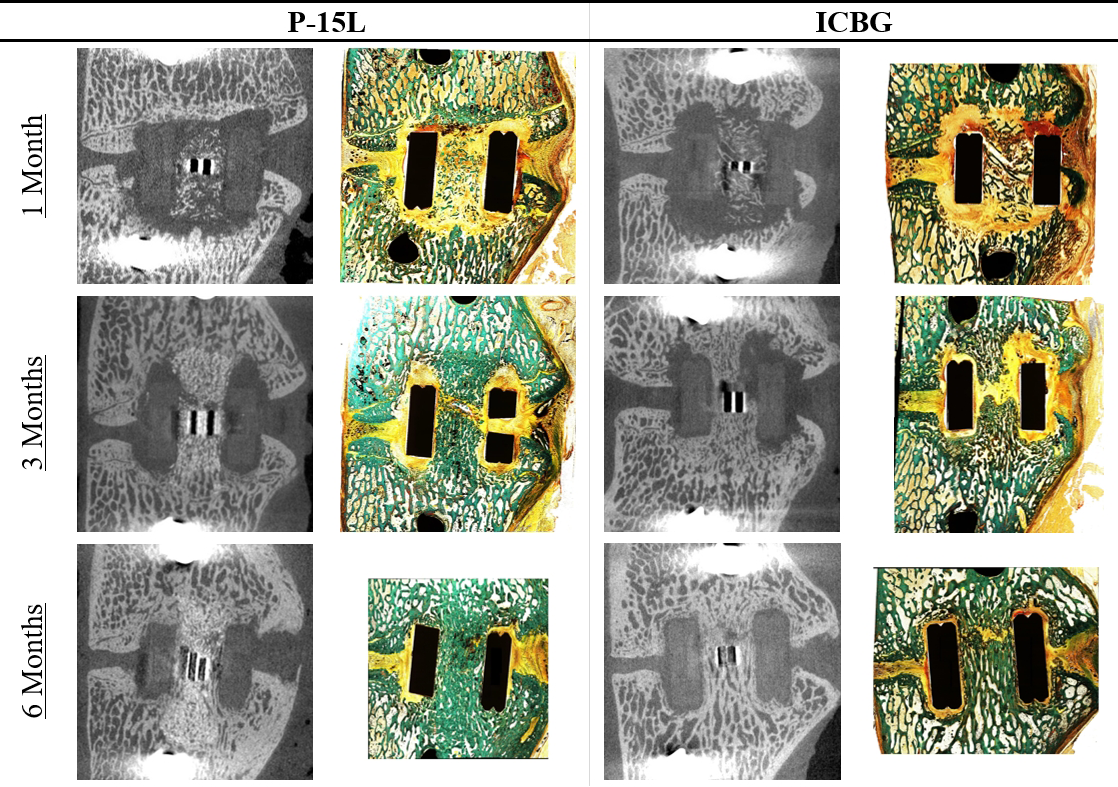
Sagittal μCT and histology cuts of both groups 1, 3, and 6 months after surgery. Residual P-15L graft can be recognized as hyper-opaque particles in μCT scans. For both groups, marker artifacts obscure the central region of the cage in the scans. Corresponding histological sections reveal there are no unexpected deviations in these artifact regions.
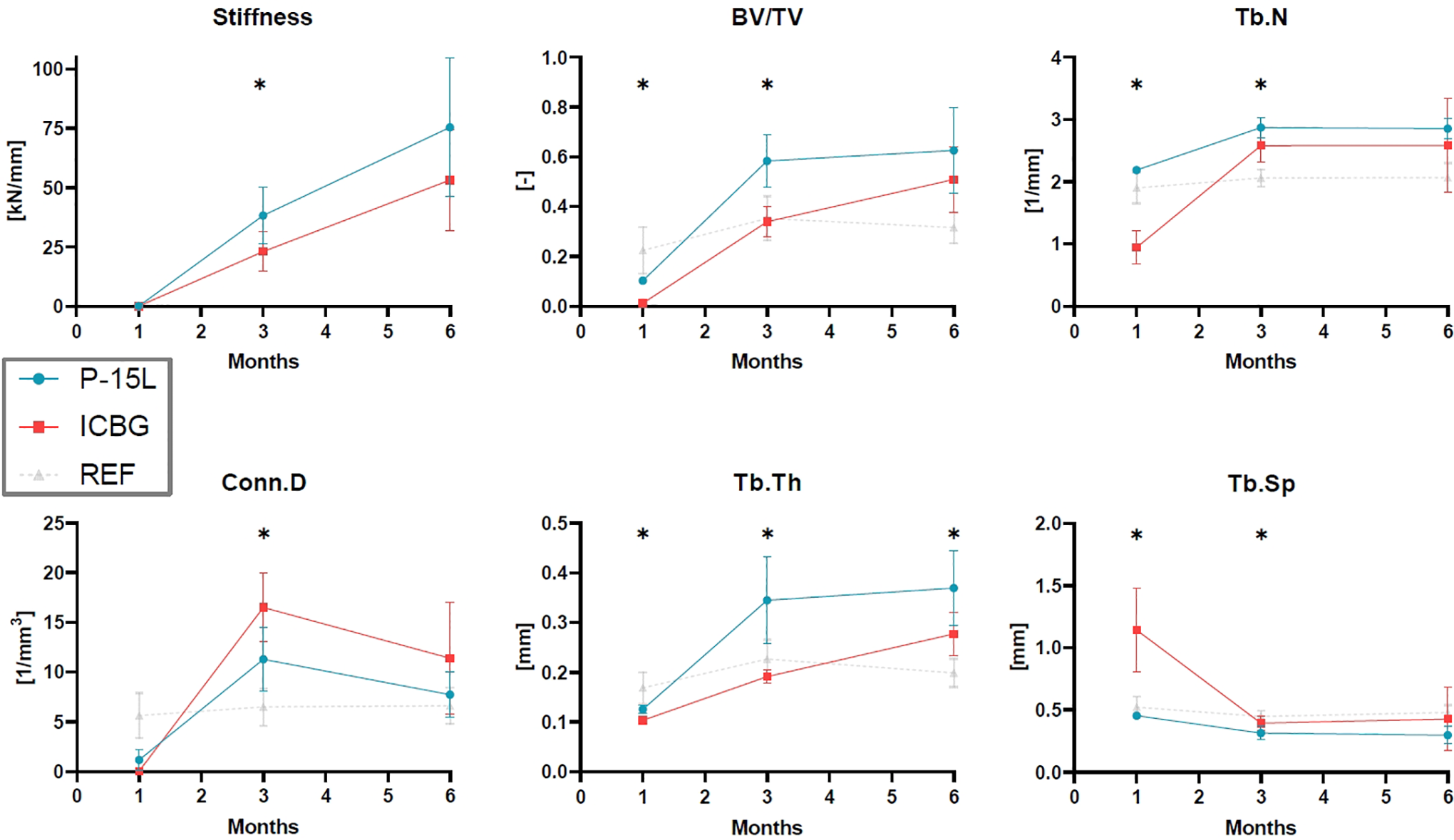
Changes over time in morphological parameters of the bone within the graft window and associated stiffness as determined by μFE. Time points marked with a star (*) indicate a significant difference between the P-15L and ICBG group. Morphological parameters of unaffected cancellous bone are shown as reference (REF). Abbreviations: BV/TV, bone volume over total volume; Tb.N, trabecular number; Tb.Th, trabecular thickness; Tb.Sp, trabecular separation; Conn.D, connectivity density.
Sagittal sections stained with Goldner’s Trichrome can be found in Figure 2 for both groups at the different evaluation points. The bone with marrow area within and expressed as percentage of the total intervertebral ROI (BMfrac) can be found in Table 1. The value strongly increases between 1 and 3 months after which it stabilizes. BMfrac displays similar values for both groups at every time point. The peak load and ultimate displacement as found by destructive tensile testing are not significantly different between graft groups within both the 3 and 6 month cohort. Peak load values appear to be higher and more variable at the 6 months cohort.
Overview of the Bone Including Marrow Area Expressed as Percentage of the Intervertebral Region of Interest (BMfrac).
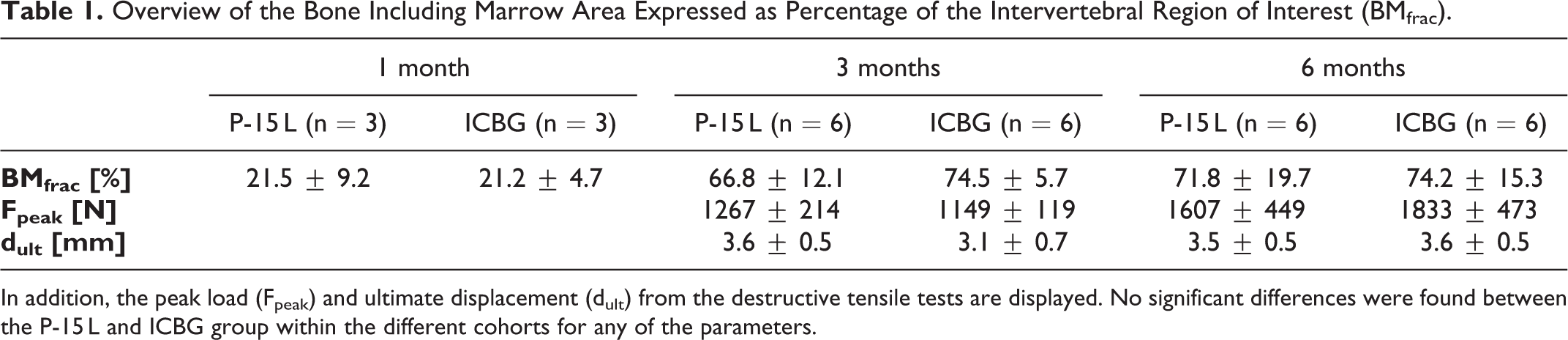
In addition, the peak load (Fpeak) and ultimate displacement (dult) from the destructive tensile tests are displayed. No significant differences were found between the P-15 L and ICBG group within the different cohorts for any of the parameters.
The inflammatory cell infiltration, inflammation, neovascularization, fibrosis, cartilage formation and new bone formation were similar for both groups at all evaluation points. For both groups, data demonstrated no significant inflammatory responses. New bone formation increased from 1 to 3 months and remained similar at 3 and 6 months for both groups. Cartilage formation increased from 1 to 3 months and then tended to slightly decrease from 3 to 6 months. The residual graft score remained relatively similar over time in the P-15 L group while the graft lost trace over time in the ICBG group. At 3 and 6 months after surgery, the resulting graft score was significantly higher for the P-15 L group compared to the ICBG group.
Discussion
This ovine interbody fusion model revealed the in vivo efficacy and safety of a peptide enhanced bone graft substitute (P-15 L) and compared its performance as a stand-alone graft material with respect to iliac crest autologous bone. Although comparable bone morphology and stiffness were found for the P-15 L bone graft substitute and the iliac crest autologous bone 6 months after surgery, the P-15 L substitute expedites bone formation and remodeling as evidenced by superior bone morphology and stiffness 3 months after surgery as compared to the iliac crest autologous bone. No graft migration was observed and P-15 L did not elicit any adverse local or systemic effects, demonstrating the graft substitute is safe to use for spinal interbody fusion indications.
The conclusions drawn in this study mainly rely on the µCT analyses as these accommodate a comprehensive characterization of the full 3D structure of the new bone and can predict resulting functionality in terms of mechanical stability.26,27 A main limitation of this study was the exclusion of the central region of the cage in µCT analyses due to marker artifacts; however, histomorphometric analyses were conducted to verify that these artifact regions did not reveal any unexpected deviations demonstrating the µCT data represents the total segment appropriately. Generally, higher bone volume ratios emerged from histomorphometry as in these analyses the bone with marrow area was quantified as opposed to the µCT analyses which did not include the marrow area. Furthermore, µCT morphometry characterized the complete 3D microstructure of the bone unraveling differences in trabecular bone development which were unnoticed by histomorphometry.
As a consequence, no differences were found between groups for bone including marrow area using histomorphometry while µCT analysis revealed denser bone in the P-15 L group 3 months after surgery. Denser bone was revealed as both more and thicker trabeculae were found in a structure having less redundant connections. Consequently, the P-15 L group showed lower separation between the trabeculae and a higher bone volume over total volume despite the groups having a similar bone including marrow area as determined by histomorphometry. Moreover, the bony construct in the P-15 L group was found to be stiffer 3 months after surgery suggesting more mature remodeling. Six months after surgery, both groups showed similar bone constructs with a higher density compared to unaffected cancellous bone controls. Increasing bone density is a phenomenon commonly observed within the graft area of spinal cages when fusion progresses.18,28
Care should be taken to draw conclusions from the 1 month µCT and histomorphometry analyses as the bone structures at this evaluation point will consist of residual graft material and immature bone. These structures are presumably strongly inhomogeneous challenging appropriate interpretation of parameters describing the trabecular structure. In addition, the volume ratios evaluated at this time point mainly provide insight in the maximal “background noise” generated by the graft. As the relative bone volumes at 1 month were low, it is plausible to attribute the differences found 3 months after surgery solely to ongoing bone growth and remodeling despite the presence of residual graft.
Apart from the differences in trabecular bone morphology and predicted stiffness 3 months after surgery, other histomorphometric and mechanical analysis yielded the same results for both groups. Destructive tensile testing provided strength characteristics of the whole segment while the µFE predicts the compression stiffness of the newly formed, artifact-free bone construct only. As a consequence, it is not possible to directly correlate these mechanical parameters. The increase in peak force and its variance between 3 and 6 months are suggested to result from the varying amount of bone bridges external to the cage. External bone bridges are commonly found in preclinical animal models as well as in clinical situations.29,30
A limitation of this study arose from the difference in traceability of residual P-15 L and ICBG material. Corresponding to previous literature, residual P-15 L was identifiable in this study up to 6 months after surgery in both µCT and histological slices. 18 In contrast, it was impossible to visually distinguish residual ICBG from newly formed bone in µCT slices. Histopathologically, ICBG could be recognized 1 month after surgery as an isolated bone structure but on the longer term, as remodeling proceeds, original ICBG lost trace, i.e. ambiguity arises whether ICGB has been resorbed and replaced or became less recognizable. Consequently, accurate distinction of remaining ICBG material is impeded. In order to avoid bias as result of difference in traceability of grafts, mineralized bone and graft tissue were analyzed as one. This method was considered legitimate as low amounts of mineralized tissue were found at the 1 month evaluation point. Furthermore, redundant graft structures that are not properly incorporated in the fusion mass are disregarded in µFE analysis as these loose structures do not contribute to the stiffness of the construct.
Smit et al previously used µCT to analyze the 3D bone architecture within spinal cages up to 3 years after surgery and found trabecular bone changes comparable to those found for growing bone, i.e. development of a coarse homogenous bone architecture within the spinal cage as maturation progresses. 31 Signs of remodeling of the new bone construct in the present study were quantified using µCT analysis and compared to the change in morphological parameters of unaffected cancellous bone remote from the operative site. Specifically for the P-15 L group, construct stiffness increased over time even though the bone volume did not increase further after 3 months. This indicates that extensive bone remodeling was still proceeding up to 6 months after surgery.
In conclusion, this study highlights the promising performance of the peptide enhanced bone graft substitute P-15 L for lumbar interbody fusion indications. Clinical trials should be contemplated to evaluate the clinical outcome and cost benefit over time for P-15 L as this study demonstrates superior short term results based on observed trabecular bone development of the new bone as compared to iliac crest autologous bone.
Since P-15 L exhibits similar fusion results as iliac crest autologous bone, the results of this study suggest that the introduction of P-15 L into the clinic as a stand-alone alternative graft material will obviate the necessity for harvesting iliac crest bone reducing the risk of donor site morbidity and limited availability. Moreover, P-15 L provides consistent material properties and does not require extensive preparation with cells or other biological agents prior to clinical use thereby reducing costs and mitigating risks associated with dosing which may be linked to adverse events including osteolysis and ectopic bone formation. These benefits may have short and long-term cost advantages which would require further evaluation within clinical studies.
Key Points
– Bone graft substitute P-15 L is safe to use for spinal interbody fusion indications
– Bone graft substitute P-15 L is equivalent to iliac crest autologous bone grafting
– New bone formation and remodeling is expedited by P-15 L grafting compared to iliac crest autologous bone grafting
Footnotes
Acknowledgments
The authors would like to thank the AccelLAB Inc. team for their expertise in the execution of the animal protocol.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Cerapedics, Inc.
