Abstract
Study Design:
Proteomic analysis of human intervertebral discs.
Objectives:
To compare the characters of scoliotic discs and discs from magnetic resonance imaging (MRI)–normal voluntary organ donors controls used in disc research employing proteomics and establish “true controls” that can be utilized for future intervertebral disc (IVD) research.
Methods:
Eight MRI-normal discs from 8 brain-dead voluntary organ donors (ND) and 8 scoliotic discs (SD) from 3 patients who underwent anterior surgery for adolescent idiopathic scoliosis were subjected to tandem mass spectrometry, and further analysis was performed.
Results:
Mass spectrometry identified a total of 235 proteins in ND and 438 proteins in the SD group. Proteins involved in extracellular matrix integrity (Versican, keratins KRT6A, KRT14, KRT5, and KRT 13A1, A-kinase anchor protein 13, coagulation factor XIII A chain, proteoglycan 4) and proteins involved in transcription and DNA repair (Von Willebrand factor A domain-containing 3B, eukaryotic initiation factor 2B, histone H4, leukocyte cell–derived chemotaxin 2) were found to be downregulated in SD. Inflammatory proteins (C3, C1S), and oxidative stress response proteins (peroxiredoxin-2,6, catalase, myeloperoxidase, apolipoprotein E) were found to be upregulated in SD. These changes were reflected at the pathway level also.
Conclusion:
Findings of our study confirm that scoliotic discs have an abundance of inflammatory, oxidative stress response proteins, which are either absent or downregulated in the ND group indicating that scoliotic discs are not pathologically inert. Furthermore, this study has established MRI-normal discs from voluntary organ donors as the “true” control for molecular studies in IVD research.
Keywords
Introduction
Disc degeneration (DD) is attributed to 40% of low back pain (LBP), 1 which is the single most common cause of disability worldwide. 2 Also, DD is the most researched subject in spinal disorders. Despite being extensively researched, the intricate mechanisms involved in homeostasis, the etiology, and pathomechanisms involved in disc degeneration are still unclear. The most important reason for this elusiveness has been, lack of appropriate controls for making a precise comparison against degenerated discs. The inclusion of appropriate controls representing the original living status of intervertebral disc (IVD) will shed more light on the molecular mechanisms underlying the development of DD. Current research on disc degeneration uses heterogeneous controls for comparison. The commonly employed controls include scoliotic discs, traumatic discs, 3 discs from cadavers, 4 or discs from voluntary organ donors. 5 Also, results from animal studies have been extrapolated to humans due to a lack of appropriate human controls. However, the results from these studies have led to poor reproducibility and contributed to confusion rather than clarity. Hence, there is a need for defining appropriate controls for inclusion in any anatomical and molecular studies aiming at understanding the etiology of DD. In this study, we sought to compare the protein profiles of magnetic resonance imaging (MRI)–normal IVD harvested from brain-dead voluntary organ donors against the most commonly used control discs obtained from scoliosis patients by employing proteomics strategy and establish “true controls” that can be utilized for future IVD research.
Materials and Methods
Sample Collection
The study was conducted after getting proper ethical clearance and approval from the institutional review board. Eight L4-L5 discs from 8 brain-dead voluntary organ donors screened for devoid of low back pain formed the normal discs group (ND), whereas scoliotic disc group (SD) consists of 8 discs from 3 adolescent idiopathic scoliosis (AIS) patients who underwent anterior deformity correction procedure (Figure 1). All the samples were collected in aseptic operating room conditions, washed with 1X phosphate buffered saline (PBS) for the removal of bloodstains and snap-frozen in liquid nitrogen before storing at −80°C.
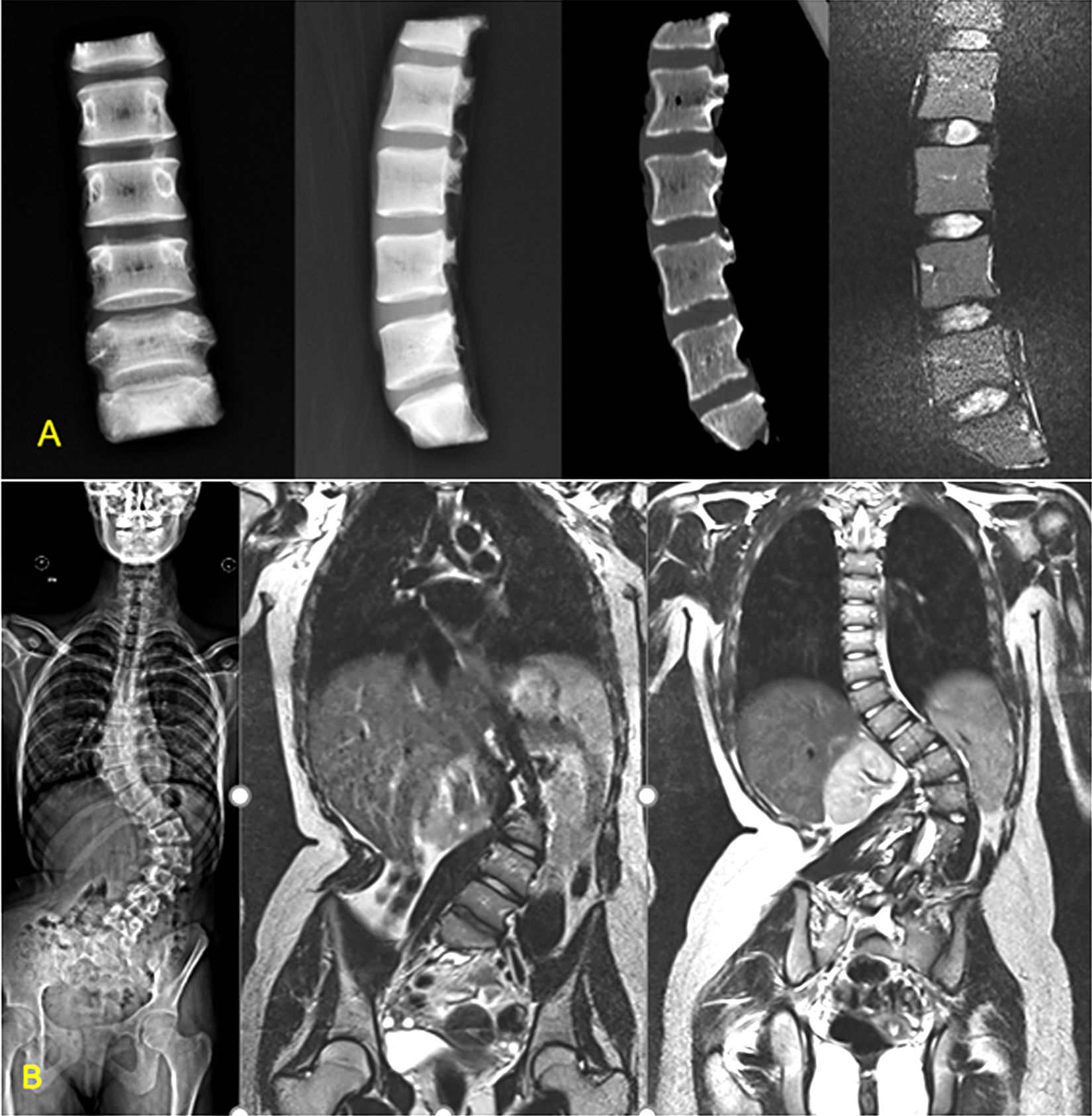
Imaging of the study groups. (A) Normal group: Brain-dead voluntary organ donors. Imaging shows no degenerative changes. (B) Scoliosis group: Adolescent idiopathic scoliosis—magnetic resonance imaging showing normal discs.
Protein Extraction and Prefractionation
Total protein was extracted from 200 mg of nucleus pulposus (NP) component of human intervertebral discs by radio immunoprecipitation assay buffer (RIPA) followed by 2% sodium dodecyl sulfate (SDS) buffer.6,7 Both membrane and cellular protein fractions were extracted and subjected to precipitation using protein sample-methanol-chloroform-water in the ratio (1:4:1:3). Purified proteins were quantified using bicinchoninic acid (BCA) assay before prefractionation. About 100 µg of purified total proteins were loaded onto 10% SDS–polyacrylamide gel electrophoresis (SDS-PAGE) for the separation based on molecular weight.
In-Gel Tryptic Digestion
Prefractionated protein samples resolved in the form of small bands were cut and chopped finely into 1.5-mm gel pieces. Then, they were subjected to reduction using 25 mM dithiothreitol (DTT) to convert all cysteine disulfide bond into cysteine-free sulfhydryl groups, followed by alkylation using 55 mM iodoacetamide (IAA). 5 Alkylated pieces were washed with 100 mM ammonium bicarbonate before digesting the proteins with 600 ng of trypsin per protein band. The digestion was set at 37 °C for 16 hours. Digested tryptic peptides were extracted from gel bands using 0.1% trifluoro acetic acid (TFA) and acetonitrile (ACN) on sonication (15-second pulsation on and off for 5 cycles). Extracted peptides were purified using Agilent c18 stage columns as per manufacturer’s instruction and subjected to ESI-LC-MS/MS (electrospray ionization–liquid chromatography–tandem mass spectrometry) analysis.
Tandem Mass Spectrometry
Purified trypsin-digested peptides were reconstituted with 0.1% trifluoroacetic acid/formic acid (TFA/FA) in 10% ACN before injecting into Orbitrap Velos Pro Mass Spectrometer at a concentration of 1000 femtomoles (fmol) per duplicate run as described earlier. 8 The raw files (.raw/.msf) obtained from the machine were searched against annotated UNIPROT consortium for identification of total proteins.
Data Processing
The MS/MS raw data acquired from Orbitrap Velos Pro Mass Spectrometer were searched against universal human proteome databases for the global identification of proteins with the help of Proteome Discoverer v 1.4 available with Mascot and SEQUEST search engines. The peptide spectrum matches (PSMs) from SequestHT and Mascot were postprocessed using the Percolator algorithm. The peptides with rank one and having a q-value <0.01 were considered for protein identification.
Bioinformatics Analysis
Total proteins were analyzed to remove the duplicate, unreviewed entries for further analysis. Spectral counts obtained by LC/MS-MS were further normalized by the normalized spectral abundance factor (NSAF) method. 9 Comparative proteome analysis was performed by FunRich, version: FunRich 3.0, a stand-alone software used for functional analysis, to understand the shared homology of proteins between groups. 10 Gene ontology was performed using Panther GO-slim classification tool version 14.0 11 and David Database version 6.7 12 to catalog proteins based on their molecular, biological, and cellular functions. Pathway enrichment analysis was performed using ShinyGO version 0.61 13 database with interface access to KEGG pathways along with Reactome pathway browser 14 version 3.6 to understand the pathomechanisms.
Statistical Analysis
Differences in protein expression between ND and SD groups were analyzed using SPSS software version 25. Based on the normality assessment, independent t tests for 2 variables and Mann-Whitney U tests were used in case of normality assumption violation. Two-tailed alpha was set at 0.05.
Data Availability
Raw data sets of this experiment have been deposited to the Proteome Xchange Consortium via the PRIDE 15 partner repository with identifier PXD018968 and project DOI 10.6019/PXD018968.
Results
Human IVDs from the groups (ND and SD) examined in this study comprised mixed gender populations, as listed in Table 1. The age (mean ± SD) of the subjects was 37.6 ± 16.9 years in the ND group and 19.25 ± 8.10 in the SD group.
Demographic Details of the Samples Considered for This Study.
Abbreviations: ND, normal disc; SD, scoliotic disc.
Mass Spectrometry Identified More Number of Proteins in Scoliotic Discs
Tandem mass spectrometric analysis of ND and SD identified a total of 1813 proteins in ND and 2297 proteins in SD (Figure 2). Total proteins were verified with their peptide sequences using the UNIPROT database, and uncharacterized/putative proteins were removed (Supplementary File 1). Applying stringent statistical criteria (cutoff PSM ≥5 and the presence of a protein in at least 2 [25%] samples) identified a total of 235 proteins in ND and 438 in the SD group.
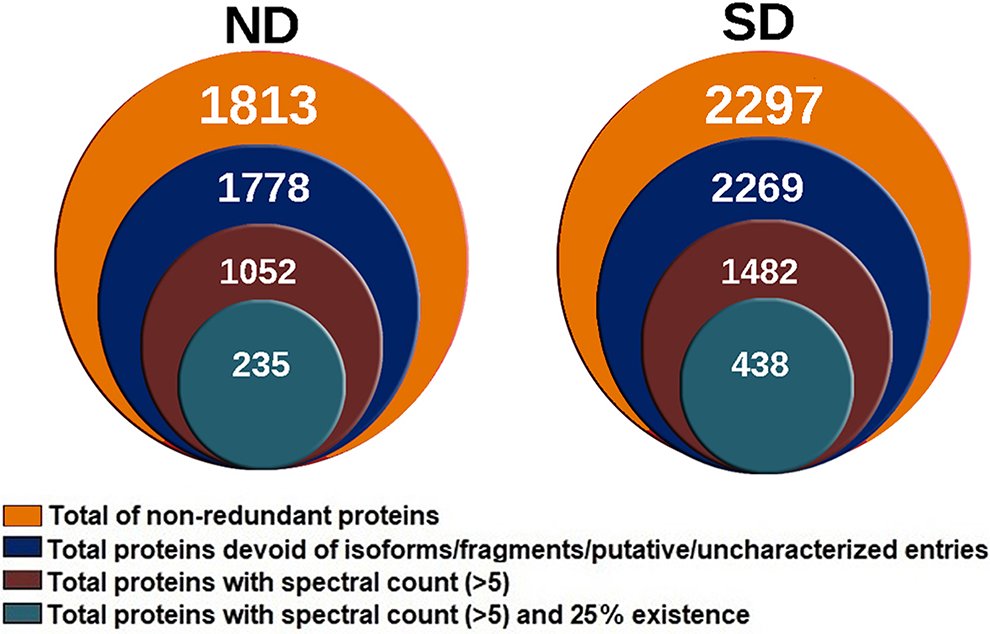
Stacked Venn diagrams showing the total number of proteins at various levels of stringency. The innermost circle shows total proteins selected at a high stringency cutoff considered for the entire study. At high stringency, the normal disc (ND) group has 235 proteins, and the scoliotic group (SD) group has 438 proteins in total.
Comparative analysis of proteins revealed that 157 (30.23%) proteins were found to be common between ND and SD groups (Figure 3). Out of 157 proteins, 35 (22.29%) proteins were core matrisomal proteins, 19 (12.10%) were matrisomal associated proteins and remaining 103 (65.06%) were comprised of keratins, immune regulators, proteases and proteins coding for basal metabolisms such as such keratinization, cornification, homeostasis, protein processing, and maturation. About 78 (15.31%) proteins were found only in the ND group, and 281 (54.45%) were found only in the SD group.
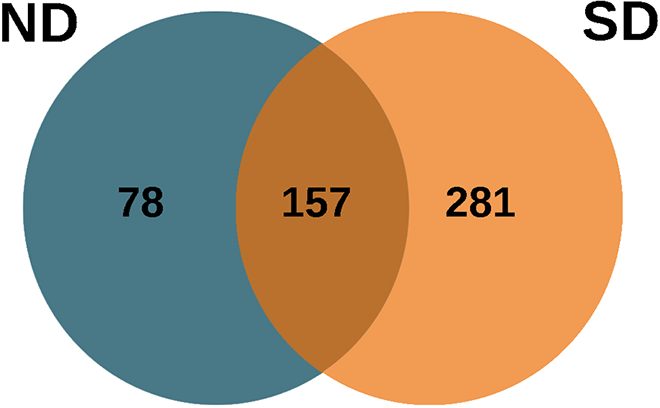
Venn diagram showing the number of unique and common proteins identified in normal disc (ND) and scoliotic disc (SD) groups. There were 157 proteins found common to both the groups with 78 unique in ND and 281 unique in SD groups. The diagram was generated with FunRich, version 3.0. FunRich 3.0 is a stand-alone software used for functional analysis.
Scoliotic Disc Proteins Exhibit Significant Alterations in Their Biological and Molecular Functions
To elucidate the possible mechanisms in ND and SD groups, differential expression analysis was conducted, revealing 60 proteins to be differentially regulated. Out of 157 proteins, 51 (32%) proteins were found to be upregulated, and (106) 68% proteins to be downregulated in SD (Figure 4A). About 60 differentially expressed proteins (DEPs) with log 2 FC ≥ ±1.0 (FC is fold change) were considered for the analysis (Figure 4B). Altogether, 13 proteins were found with significant p-value <0.05, as shown in Table 2. The proteins that were upregulated in SD above 2.0-fold include blood proteins such as ALB, HBB, and HB1, and complement protein (C3), confirming the regulation of inflammatory cascades. Proteins such as proteoglycan IV (PRG4), Von Willebrand factor A domain containing 3B (VWA3B), histone H4 (HIST1H4H), zinc finger protein (ZNF646), keratin 15, keratin 4 were found to be downregulated for 2.0 fold and above in SD.
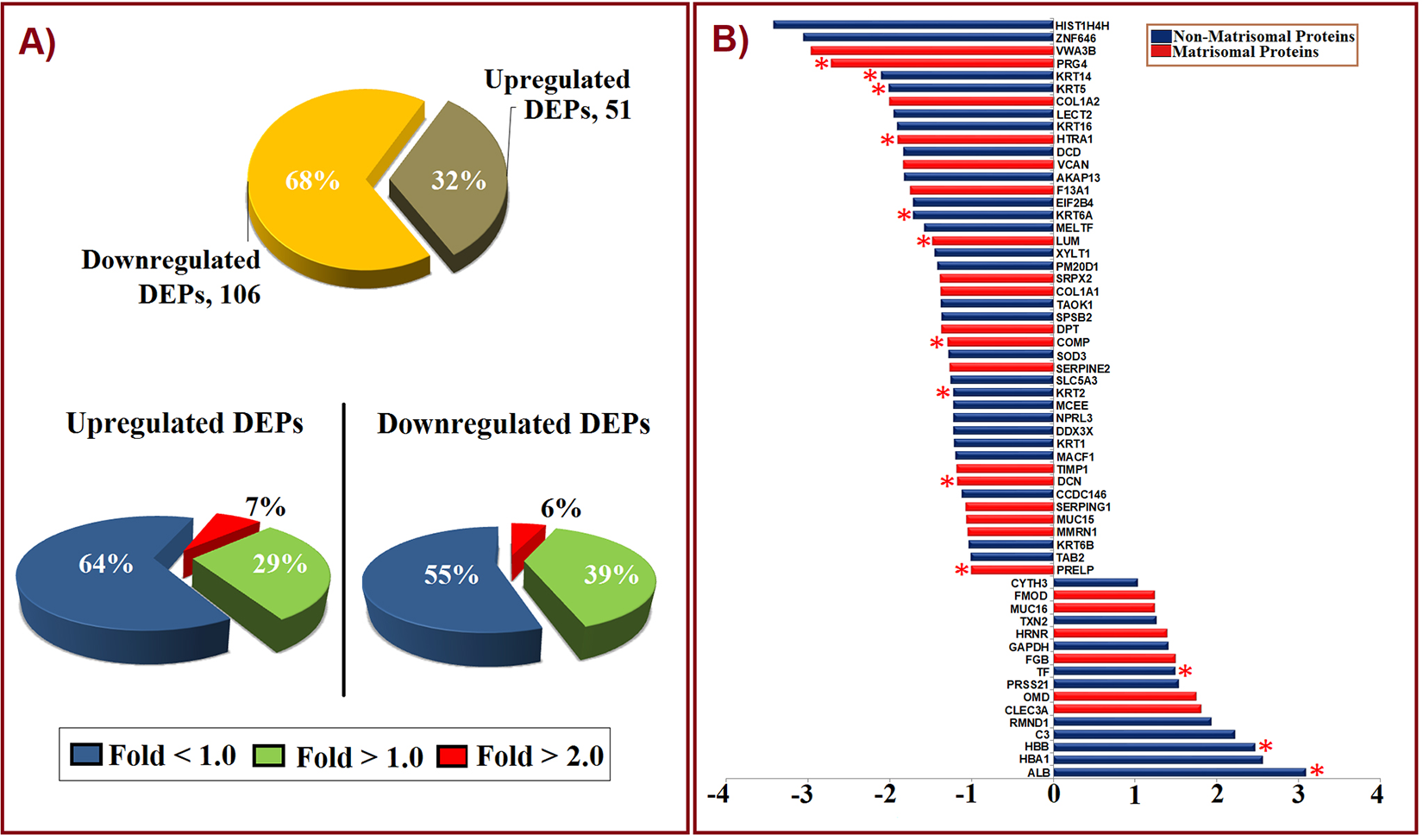
Characterization of proteins based on their regulation (up and down). (A) Pie chart describing the contribution of up and downregulated proteins in terms of fold change (log 2 FC). More proteins were downregulated (68%) in the scoliotic disc (SD) group when compared against the normal disc (ND) group, and upregulated proteins contributed to 32% of total 157 common proteins. Pie charts also depict the contribution of differentially expressed proteins (DEPs) between fold changes (>1.0 and <3.5) in both up- and downregulation. (B) Bar chart showing DEPs with log 2 FC ≥ ±1.0 (up- and downregulation) in SD when compared with ND group considered under this study. *Indicates statistical significance (P < .05), using t test.
Differentially Expressed Proteins (DEPs) That Are Statistically Significant With log 2 FC > ±1.0.
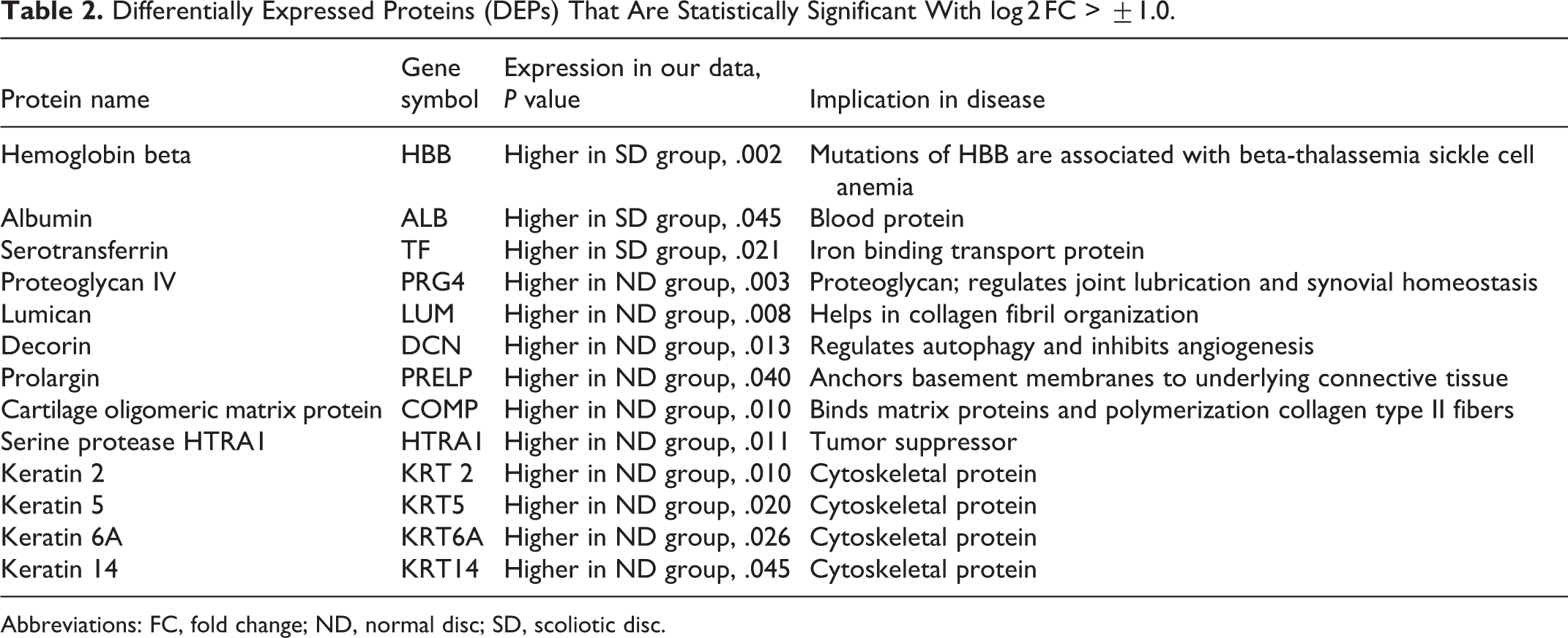
Abbreviations: FC, fold change; ND, normal disc; SD, scoliotic disc.
On their functional characterization of the biological process using David database, the upregulated DEPs in SD group were involved in hydrogen peroxide catabolic process, cellular oxidant detoxification, response to hypoxia, positive regulation of reactive oxygen species and complement activation along with other basal metabolic processes (Figure 5). Whereas, the downregulated DEPs were involved in positive regulation of NF-κB transcription activity, negative regulation of endopeptidase activity, and negative regulation of beta-amyloid activity enunciating normal metabolism involving cell survival, proliferation, and differentiation.
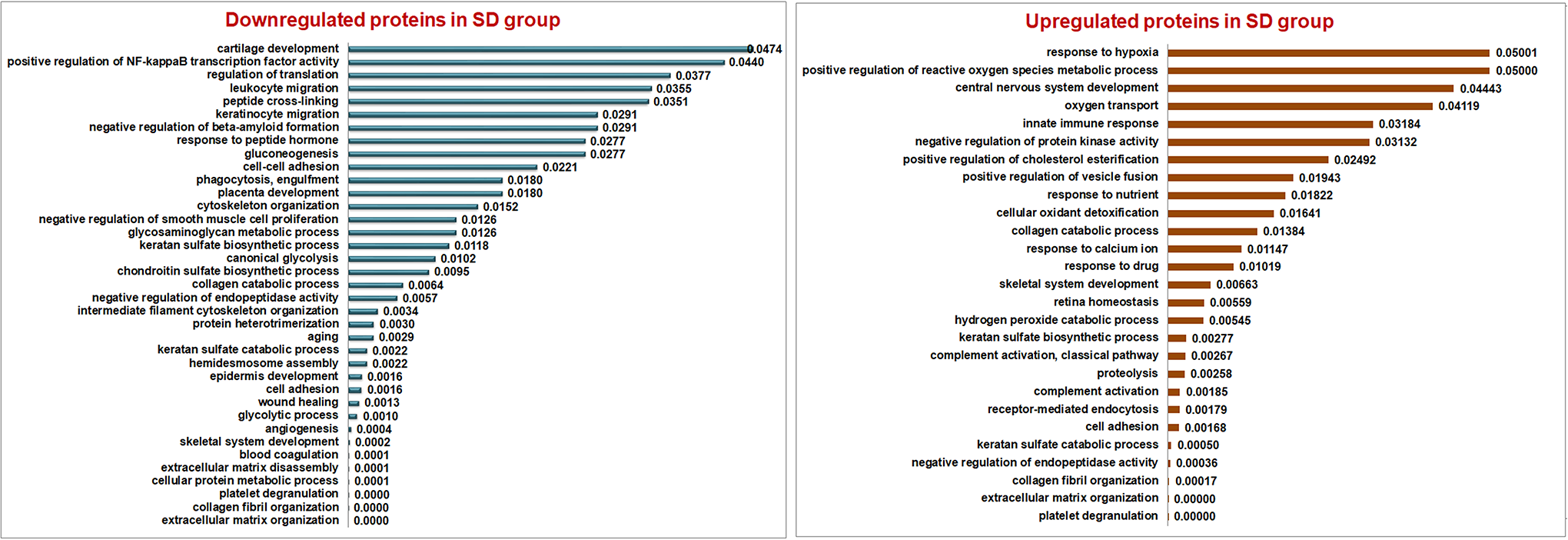
Biological process involved in both upregulated (A) and downregulated (B) differentially expressed proteins (DEPs) in the scoliotic disc (SD) group, using David database version 6.8 that were significantly enriched (P < .05). Downregulated proteins in the SD group were found to be actively involved in the basal metabolic process such as cartilage development, regulation of translation, cell-cell adhesion, cytoskeletal organization, collagen catabolic process and extracellular matrix organization. Upregulated proteins in SD were found involved in stress-induced inflammatory processes such as response to hypoxia, cellular oxidant detoxification, hydrogen peroxide catabolic process and complement activation.
Pathway enrichment analysis using the Reactome pathway browser version 3.6 detected more than 800 pathways with more than 100 pathways having significant P value (P < .05) (Supplementary File 2). Notable examples, namely, extracellular matrix (ECM) proteoglycans, ECM organization, integrin cell surface interactions, along with complement cascades and coagulation cascades involving proteins such as clusterin (CLU), complement C1S (C1S), complement C3 (C3), fibrinogen, α1-antitrypsin (A1AT), protein C inhibitor (PCI), and other SERPIN family-related proteins (Figure 6).
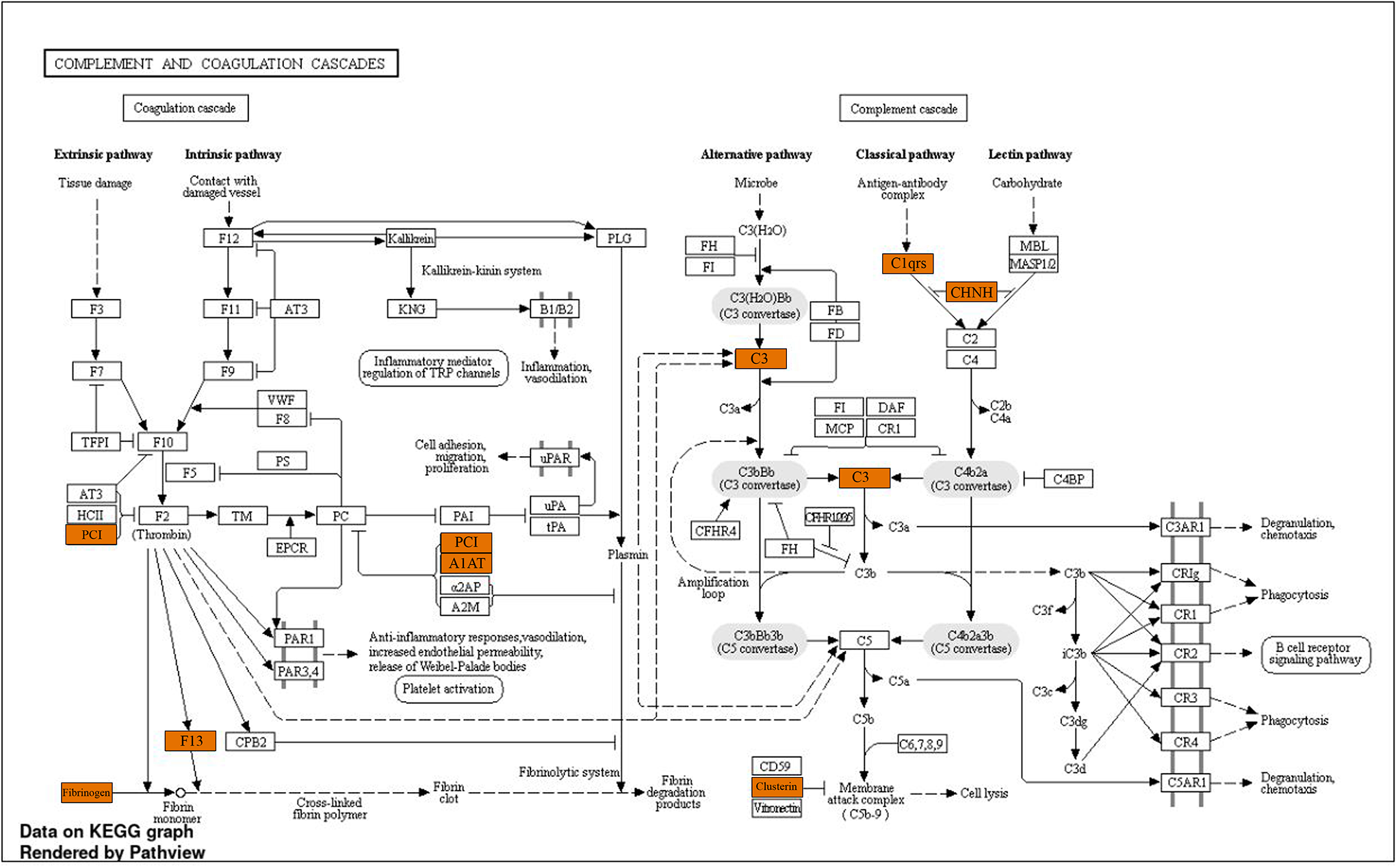
Pathway view created for complement and coagulation cascades using the KEGG database in ShinyGo version 6.0 with colored boxes indicating proteins present in our study.
Specific Proteins Expressed in Normal Disc and Scoliotic Disc Differ Strikingly
Characterization of specific proteins based on gene ontology to understand their functionality by Panther GO-slim classification tool version 14.0 revealed interesting events in ND and SD groups, as shown in Figure 7.
Proteins involved in molecular functions, namely, catalytic activity (41%), transporter activity (12%), and structural molecule activity (6%) were found to be predominant in ND (Figure 7A). Whereas the proteins involved in the above functions were significantly reduced and proteins involved in regulation activities like binding (44%) molecular function regulator activity (6%), and transcription regulation activity (2%) were found to be activated in SD. Under protein class, cytoskeletal proteins were found to be comparatively decreased in SD (6%), which was 23% in the ND group; whereas defense-immunity proteins were strikingly abundant in SD group (8%) in comparison with the ND group (2%) (Figure 7B).
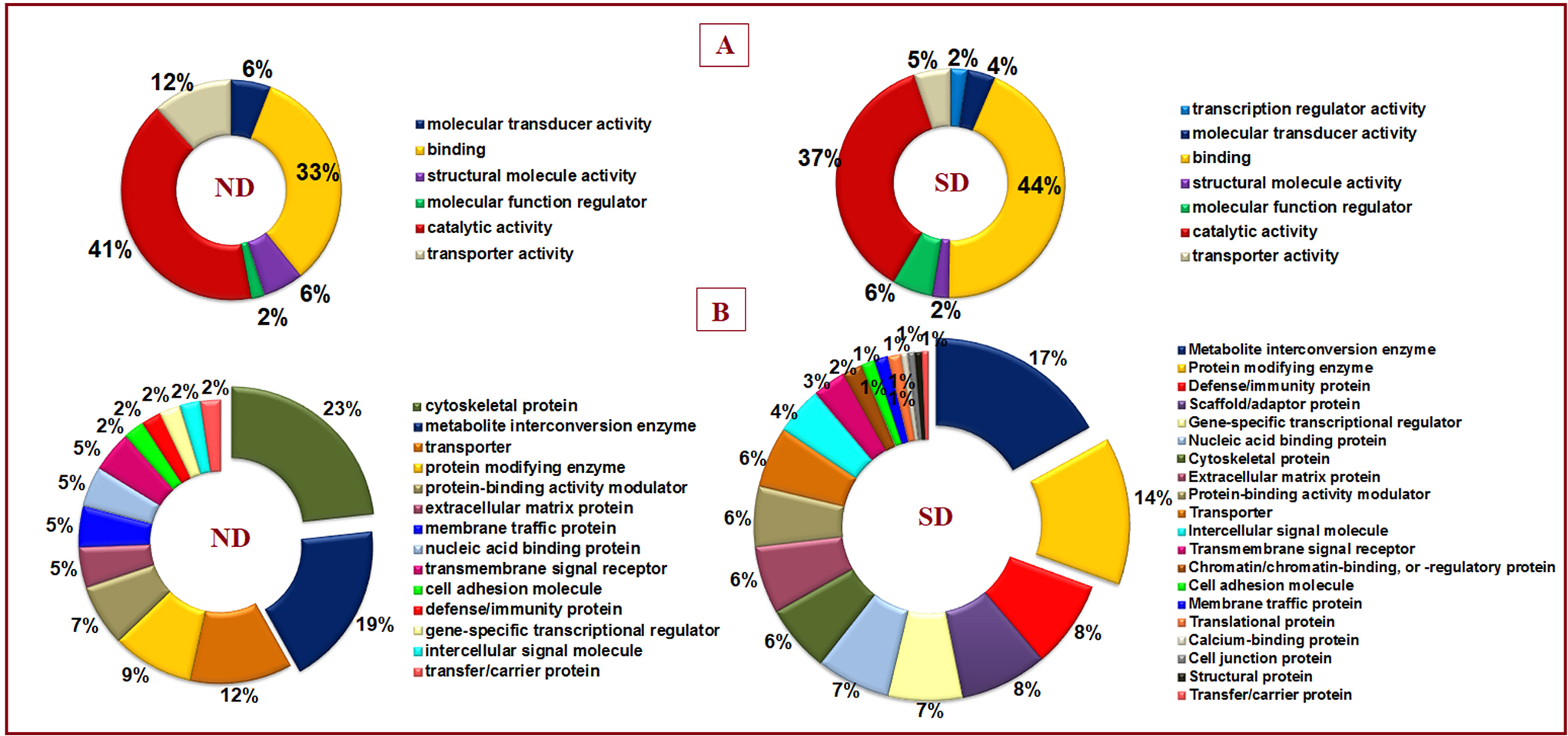
Gene ontological classification of normal disc (ND) and scoliotic disc (SD) groups derived using Panther GoSlim classification tool version 15.0. (A) Molecular function and (B) protein class to understand the class and function of unique proteins. Under molecular function, the majority of proteins were found to be involved in binding and catalytic activity with varied abundances in each group. Under protein class of ND group, proteins belonging to cytoskeletal, metabolite interconversion enzyme, and transporter classes are seen more. But in the SD group, the majority of proteins are seen grouped under metabolite interconversion, protein modifying and defense/immunity protein classes.
Classification based on biological process derived using ShinyGo version 6.0, reveals unlike events between ND and SD groups. In NDs, most cytoskeletal proteins are associated with keratinization, cornification, and vesicle-mediated transport. Other proteins codes for basal metabolism like cell communication, adhesion, and cell differentiation. In SD group, proteins such as peroxiredoxin-2 (PRDX2), peroxiredoxin-6 (PRDX6), catalase (CAT), apolipoprotein-E (APOE), myeloperoxidase (MPO), glutathione S-transferase P1 (GSTP1), hemoglobin A2 (HBA2), hemoglobin subunit delta (HBD), haptoglobin (HP) that influence redox homeostasis and reactive oxygen species were found to be shared among significant process responsible for detoxification, and other catabolic processes such as neutrophil degranulation and neutrophil activation (Figure 8).
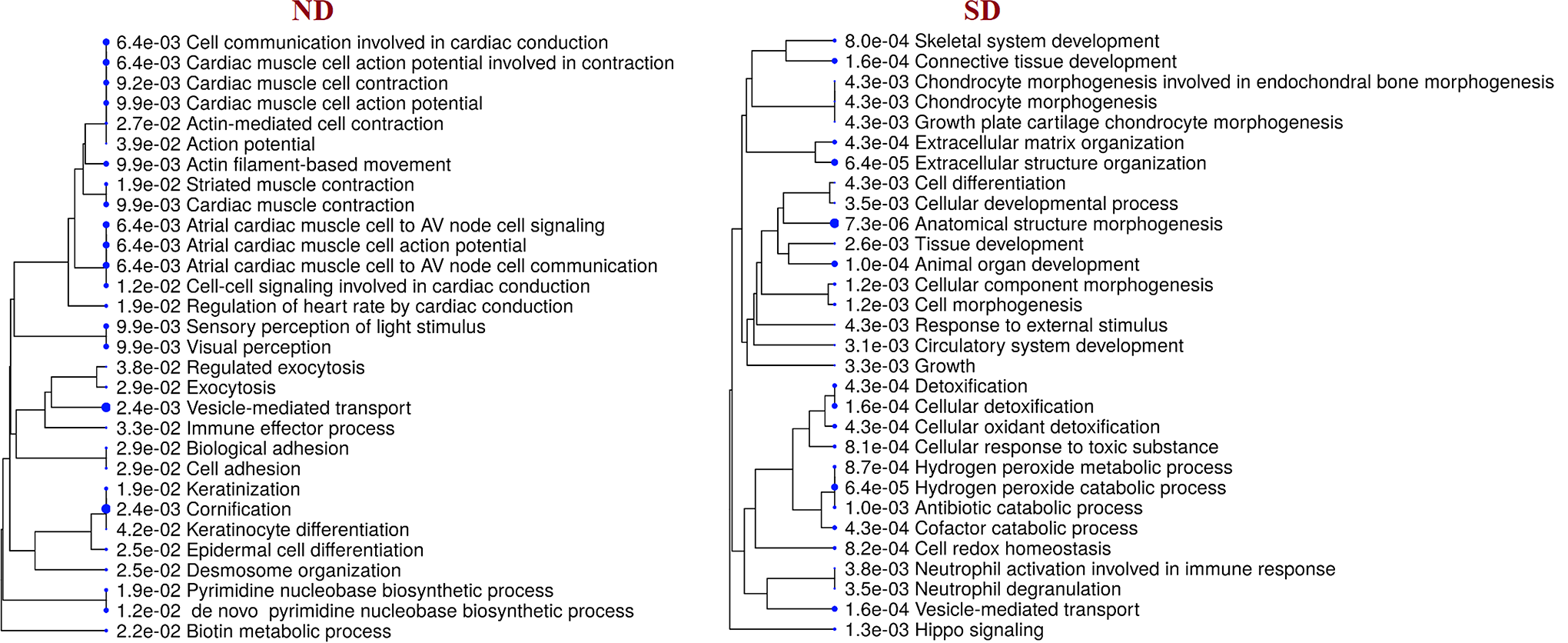
A hierarchical clustering tree demonstrates the correlation among significant biological process identified for unique proteins of normal disc (ND) and scoliotic disc (SD). The biological process with many shared genes is clustered together. Bigger dots indicate more significant P value. In the ND group, cornification, vesicle-mediated transport, action potential, actin filament–based movement are all enriched. In the SD group, anatomical structure organization, detoxification, cellular detoxification, hydrogen peroxide catabolic process, and neutrophil degranulation are enriched.
Discussion
Disc degeneration is a highly researched subject in spinal pathologies. However, huge knowledge gaps exist with regard to its etiopathogenesis. Disc degeneration is often diagnosed late when patients present with symptoms. At this time, the irreversible pathological changes have already occurred, and surgical treatment remains the mainstay of treatment. Molecular studies on disc degeneration have utilized these disc samples with advanced degeneration acquired during the surgery. However, the availability of a disc sample with “no” degeneration or “early” degeneration has been sparse. This lack of availability of a true biologically inert control for comparative studies has plagued research on disc degeneration. Few authors have utilized discs from anterior scoliosis surgeries, fractures of spine, or cadavers as control discs in their studies. However, the validity of these studies remains questionable.
Scoliotic discs are the most commonly available and frequently used controls. Though both the scoliotic discs and discs from voluntary organ donors appear normal on MRI, the results from our study indicate substantial differences between scoliotic discs and control discs from voluntary organ donors. The differences were noted in terms of differential expression of common proteins, biological processes, molecular functions, pathways involved, and the presence or absence of unique proteins. Three findings were notable: (1) Proteins involved in basal metabolic processes, cellular functions, and structural proteins were downregulated in the SD group. (2) SD group had upregulation of inflammation cascade proteins and downregulation of its regulators. (3) SD group had upregulation of reactive oxygen species (ROS) and oxidative stress response proteins.
Downregulation of Structural and Repair Proteins in the SD Group
Many proteins that are involved in the maintenance of ECM and cellular process were downregulated in the SD group. Versican is a major proteoglycan and component of the ECM along with aggrecan. It is involved in cell adhesion, proliferation, migration, and plays a central role in ECM maintenance. It also binds hyaluronic acid and plays a role in cell adhesion. 16 Other proteoglycans, namely, lumican (LUM), decorin (DCN), and prolargin (PRELP), along with collagen 1A2, 1A1 were also downregulated in SD group indicating ECM disorganization.
Keratins KRT6A, KRT14, KRT5, and KRT 13A1 were downregulated in the SD group. Keratins play a role in enhancing mechanical properties by organizing filaments and also are involved in the wound healing process.17,18 A-kinase anchor protein 13(AKAP 13A) or Rho guanine nucleotide exchange factor 13 is the guanine nucleotide exchange factor (GEF) for the RhoA small GTPase protein.19,20 It activates Rho, a major regulator of the cell actin cytoskeleton. 21 Coagulation factor XIII A chain (F13A1) plays a role in stabilizing the fibrin clot. It also cross-links α-2-plasmin inhibitor, or fibronectin, to the alpha chains of fibrin. The defects of this gene have been implicated in bleeding diathesis and impaired wound healing.22,23 Proteoglycan 4 (PRG4) or lubricin have a unique role in cell-cell and cell-ECM interactions. 24 Decreased levels of lubricin have been noted in the synovial fluid of patients with rheumatoid arthritis and osteoarthritis. 25 Downregulation of keratins, AKAP 13A, F13A, PRG4, and cytoskeletal proteins indicates reduced structural integrity in scoliotic discs.
Another finding was the downregulation of proteins that are involved in transcription, translation, and DNA repair in the SD group. Von Willebrand factor A domain containing 3B (VWA3B) are intracellular proteins that are thought to function in transcription, DNA repair, ribosomal, and membrane transport. Von Willebrand factor also acts as a receptor for proteasome-mediated degradation for ubiquitin-like proteins.26-28 Eukaryotic initiation factor 2B (EIF2B) is a GTP exchange factor necessary for protein synthesis. Stress responses lead to increase protein synthesis during which stress response proteins are produced, these proteins activate the kinases, which leads to phosphorylation of the eIF, and this reduces the activity of EIF2B. Mutations in EIF2B leads to phosphorylation of eIF, which is reported in neurological diseases such as leukodystrophy with vanishing white matter in the brain (VWM) and ovarioleukodystrophy.29,30 Histone H4 (HIST1H4H) is a core component of nucleosome. Nucleosomes wrap and compact DNA into chromatin, limiting DNA accessibility to the cellular machinery, which requires DNA as a template. They play a vital role in transcription regulation, DNA repair, DNA replication, and chromosomal stability. 31 Leukocyte cell–derived chemotaxin 2 (LECT2) seems to be related to the cell cycle or repair process following damage to a variety of cells and stimulates the growth of chondrocytes and osteoblasts. 32 Zinc finger protein 646 (ZNF646) also has a possible role in transcriptional regulation. The downregulation of these proteins indicates a defective transcription and DNA repair, which could lead to premature cell death. 33
Upregulation of Inflammatory Cascade Proteins and Downregulation of Its Regulators in the SD Group
Many proteins of complement cascades and innate immune systems such as complement C1S (C1S), complement C3 (C3), IGKC, IGHG1, and other SERPIN family–related proteins were upregulated in SD group. C3 is the chief component of the complement system and plays a vital role in the innate immune response. Complement-mediated inflammation is important not only in immunological defense reactions but also in the clearance of damaged/apoptotic cells. 34 This process is ensured by an alternate pathway, where tissue damage activates the complement cascade, and through a constant spontaneous process known as tick over, C3 complement protein is produced. 35 Excessive complements have been implicated in inflammaging 5 and many disorders like autoimmune disorders, Alzheimer’s, disease, macular degeneration, and Crohn’s disease. 36 Clusterin (CLU) is an important regulator of complements. CLU prevents uncontrolled membrane attack complex activity and thus plays a significant role in controlling terminal complement-mediated damage to host cells and chronic inflammation. 37 The downregulation of CLU was noted in the SD group. Similarly, SERPING1 or plasma protease inhibitor of C1, was downregulated in SD group. SERPING1 exhibits anti-inflammatory role by inhibition of complement system proteases (C1r, C1s, mannose-binding protein-associated serine protease 2-MASP2) and the plasma kallikrein–kinin system proteases. 38 It also inhibits chymotrypsin. 39
The SERPINE1, Plasminogen activator inhibitor-1 protein, was downregulated in SD group. It is an inhibitor of serine protease and also plays a vital role in signal transduction, cell adhesion, and migration. 40 It also inhibits plasmin-mediated breakdown of the ECM. 41 In contrast, C-type lectin domain family 3 member A (CLEC3A), which was upregulated in the SD group, enhances tissue plasminogen activator–mediated plasminogen activation. 42 Serine protease 21 (PRSS21) is a member of the trypsin family that cleaves peptide bonds subsequent to a positively charged amino acid (lysine or arginine). 43 The presence of proteins involved in the coagulation cascade in SD group indicates a response to tissue injury. Exposed collagen as a result of injury could activate coagulation cascades. 44
The coexistence of complement and coagulation cascade mediates innate immunity with coagulation enzymes activating complement cascades and both functioning as a proteolytic cascade. 45 Serine proteinases, along with activators and inhibitors, are majorly responsible for their interlink between coagulation and complement systems, and the dysregulation in coagulation or complement activities would, in turn, activate inflammatory cascades. 46
In biological processes, positive regulation of NF-κB transcription factor was noted in our study. NF-κB is a transcription factor involved in inflammatory and immune responses with additional roles in cell survival, proliferation, and differentiation. NF-κB can stimulate the expression of the immunoglobulin κ light chain in B cells and play a fundamental role in lymphocyte development and activation, and it is essential for innate and adaptive immune responses.47,48 Neutrophil activation and degranulation were among the biological processes uniquely found in the SD group. Neutrophils are essential regulators of an inflammatory response that acts as the first line of defense mechanism against inflammatory tissue damage either through degranulation or respiratory bursts. 49 These above findings indicate a pro-inflammatory state in scoliotic discs.
Upregulation of Reactive Oxygen Species and Oxidative Stress Response Proteins in the SD Group
Proteins involved in redox homeostasis like PRDX2, PRDX6, CAT, APOE, MPO, and GSTP1 were upregulated in SD group. Oxidative stress induces inflammation and vice versa. PRDX2 is a ubiquitous redox-active intracellular enzyme. Once released, the extracellular PRDX2 acts as a redox-dependent inflammatory mediator, triggering macrophages to produce and release tumor necrosis factor–α (TNF-α). 50 Ye et al, 51 in their proteomic analysis of normal and degenerated discs, documented the presence of PRDX2 in degenerated discs and hypothesized an oxidative injury to have a possible role in disc degeneration. PRDX6, is the only peroxiredoxin capable of reducing phospholipid hydroperoxides and also plays crucial roles in lung phospholipid metabolism, lipid peroxidation repair, and inflammatory signaling. 52 Myeloperoxidase is a potent enzyme with antibacterial activity and is implicated in autoimmune disorders. 53 Catalase is a crucial enzyme an plays a role in reducing ROS and counteracting TNF-α-mediated apoptosis. 54 Decreased catalase has been associated with ligamentum flavum hypertrophy. 55 ROS-mediated oxidative injury to NP cells resulting in premature cell death have been well documented in the literature.56-58 Rajasekaran et al 5 also documented the presence of peroxiredoxin, CAT, and MPO in degenerated discs but not in MRI-normal voluntary organ donor discs.
APOE is involved in cholesterol transport protein linked with atherosclerosis, neurodegenerative disorders like Alzheimer’s disease. 59 Recently, knock out mice models of APOE have documented a premature intervertebral disc degeneration.60,61 GSTP1 is an intracellular detoxification enzyme that catalyzes the conjugation of many hydrophobic and electrophilic compounds with reduced glutathione. 62 GSTP1 also acts as a critical ligand-binding protein with a role in regulating kinase pathways. 63 Oxidative stress activates the mitogen-activated protein kinase (MAPK) signaling pathway and induces apoptosis by a pathway involving c-Jun N-terminal kinase (JNK) and pro-apoptotic proteins. 64 GSTP1 interacts directly with JNK and regulates cell death. 65 These findings reflected in the evaluation of biological processes where positive regulation of ROS, cellular oxidant detoxification processes were found to be upregulated in the SD group. The disparity between production and assimilation of ROS through detoxification causes increased oxidative stress response in the host leading to inflammation. 66 The unique presence of oxidative stress response proteins in the SD group serves as evidence for inflammatory response leading to tissue damage.
The above findings indicate that though scoliotic discs were looking “normal” in MRI, they had many biological processes indicating inflammatory and oxidative stresses. Also, the decreased proteoglycans, proteins involved in transcription and DNA repair indicate impaired structural integrity and cellular functions in scoliotic discs.
Stress profilometry studies on scoliotic discs have demonstrated abnormally high stresses in scoliotic discs on comparison with nonscoliotic control discs. 67 Moreover, these stresses where asymmetrical, stresses in the concave annulus were higher than in the convex annulus. These abnormal stresses could alter the biology of the discs and could have been responsible for our findings. Brown et al 68 documented the fragmentation of small leucine-rich proteoglycans (SLRP) in scoliotic discs and degenerated discs alike, indicating a breakdown of the ECM in scoliotic discs. 68 Rajasekaran et al, 69 in their study using postcontrast MRI, found decreased diffusion and cell viability in scoliotic discs in comparison to controls. They also noted calcification, neovascularization, and matrix degeneration in scoliotic discs providing evidence for tissue injury and early degeneration. 69
Calcification process may lead to impairment of nutrient supply and metabolism of the disc and promote degeneration. A study by Hristova et al 70 demonstrated calcification in degenerate discs and scoliotic discs and postulated that mineralization in scoliosis discs reflect a premature degenerative process. Osteomodulin, a protein found in abundance in the SD group, is implicated in the biomineralization processes and functions by binding of osteoblasts via the alpha(V)beta3-integrin. It has been claimed to be a biomarker to monitor early changes in subchondral bone metabolism in osteoarthritis. 71
Significance and Limitation of the Study
Ours is the first study to evaluate the validity of scoliotic discs as controls for IVD research. Here we establish MRI-normal voluntary organ donors as the precise controls for IVD research and recommend it for future studies. Our study also questions the validity of other studies that have employed inappropriate controls. Additionally, our study identified many downregulated proteins in scoliotic discs, which play a role in maintaining the structural integrity of the disc. These proteins can be further probed for their role in the etiopathogenesis of adolescent idiopathic scoliosis. The limitation of the study was that no separate analysis of concave versus convex discs was done.
Conclusion
In a first of its kind study, we evaluated the controls used in IVD research for their preciseness. Our findings indicate that scoliotic discs have an abundance of inflammatory, immune response, oxidative stress, and antioxidant proteins and are not biologically inert. Discs from MRI-normal voluntary organ donors are in a state of homeostasis and are void of pathological processes. Hence, we establish it as the ideal control for IVD research and recommend its use in future studies. Also, our study questions the validity of earlier studies that have utilized other controls.
Supplemental Material
sj-xlsx-1-gsj-10.1177_2192568220959038 – Supplemental Material for Can Scoliotic Discs Be Controls for Molecular Studies in Intervertebral Disc Research? Insights From Proteomics
Supplemental Material, sj-xlsx-1-gsj-10.1177_2192568220959038 for Can Scoliotic Discs Be Controls for Molecular Studies in Intervertebral Disc Research? Insights From Proteomics by S. Rajasekaran, Chitraa Tangavel, K. S. Sri Vijay Anand, Dilip Chand Raja Soundararajan, Sharon Miracle Nayagam, R. Sunmathi, M. Raveendran, Ajoy Prasad Shetty, Rishi Mugesh Kanna and B. T. Pushpa in Global Spine Journal
Supplemental Material
sj-xls-2-gsj-10.1177_2192568220959038 – Supplemental Material for Can Scoliotic Discs Be Controls for Molecular Studies in Intervertebral Disc Research? Insights From Proteomics
Supplemental Material, sj-xls-2-gsj-10.1177_2192568220959038 for Can Scoliotic Discs Be Controls for Molecular Studies in Intervertebral Disc Research? Insights From Proteomics by S. Rajasekaran, Chitraa Tangavel, K. S. Sri Vijay Anand, Dilip Chand Raja Soundararajan, Sharon Miracle Nayagam, R. Sunmathi, M. Raveendran, Ajoy Prasad Shetty, Rishi Mugesh Kanna and B. T. Pushpa in Global Spine Journal
Footnotes
Acknowledgments
The authors thank Ms Alishya Maria Jose for her help in data collection and maintenance and Mr Balakrishnan for his support in preparing figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by Ganga Orthopaedic Research & Education Foundation, Coimbatore, Tamil Nadu, India.
ORCID iD
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
