Abstract
Study Design:
Retrospective cohort study.
Objectives:
Sarcopenia is a risk factor for medical complications following spine surgery. However, the role of sarcopenia as a risk factor for proximal junctional disease (PJD) remains undefined. This study evaluates whether sarcopenia is an independent predictor of proximal junctional kyphosis (PJK) and proximal junctional failure (PJF) following adult spinal deformity (ASD) surgery.
Methods:
ASD patients who underwent thoracic spine to pelvis fusion with 2-year clinical and radiographic follow-up were reviewed for development of PJK and PJD. Average psoas cross-sectional area on preoperative axial computed tomography or magnetic resonance imaging at L4 was recorded. Previously described PJD risk factors were assessed for each patient, and multivariate linear regression was performed to identify independent risk factors for PJK and PJF. Disease-specific thresholds were calculated for sarcopenia based on psoas cross-sectional area.
Results:
Of 32 patients, PJK and PJF occurred in 20 (62.5%) and 12 (37.5%), respectively. Multivariate analysis demonstrated psoas cross-sectional area to be the most powerful independent predictor of PJK (P = .02) and PJF (P = .009). Setting ASD disease–specific psoas cross-sectional area thresholds of <12 cm2 in men and <8 cm2 in women resulted in a PJF rate of 69.2% for patients below these thresholds, relative to 15.8% for those above the thresholds.
Conclusions:
Sarcopenia is an independent, modifiable predictor of PJK and PJF, and is easily assessed on standard preoperative computed tomography or magnetic resonance imaging. Surgeons should include sarcopenia in preoperative risk assessment and consider added measures to avoid PJF in sarcopenic patients.
Keywords
Introduction
Adult spinal deformity (ASD) is associated with significant pain and disability, particularly in patients with positive sagittal imbalance. 1 Surgical restoration of sagittal balance is associated with dramatic improvements in health-related quality of life 2 ; however, correction of large sagittal plane deformity places patients at high risk for postoperative development of proximal junctional disease (PJD). 3
PJD following ASD surgery ranges from asymptomatic proximal junctional kyphosis (PJK) to catastrophic proximal junctional failure (PJF; Figure 1) associated with severe pain and disability, neurologic injury, loss of sagittal correction, and the need for revision surgery.3,4 A variety of preventive techniques have been proposed, yet PJD remains a persistent problem following ASD surgery. 4 There are several modes of failure leading to PJD, including vertebral compression fracture and posterior ligamentous disruption. A significant challenge in preventing PJD is that its etiology is not fully understood and is likely multifactorial. 4 Previously reported PJD risk factors include older age, osteopenia, obesity, and a large magnitude of both preoperative sagittal imbalance and intraoperative correction.5,6 It is not clear that each risk factor contributes equally to each mode of failure. Certainly, a better understanding of PJD etiology will allow for more effective and individualized prevention strategies.
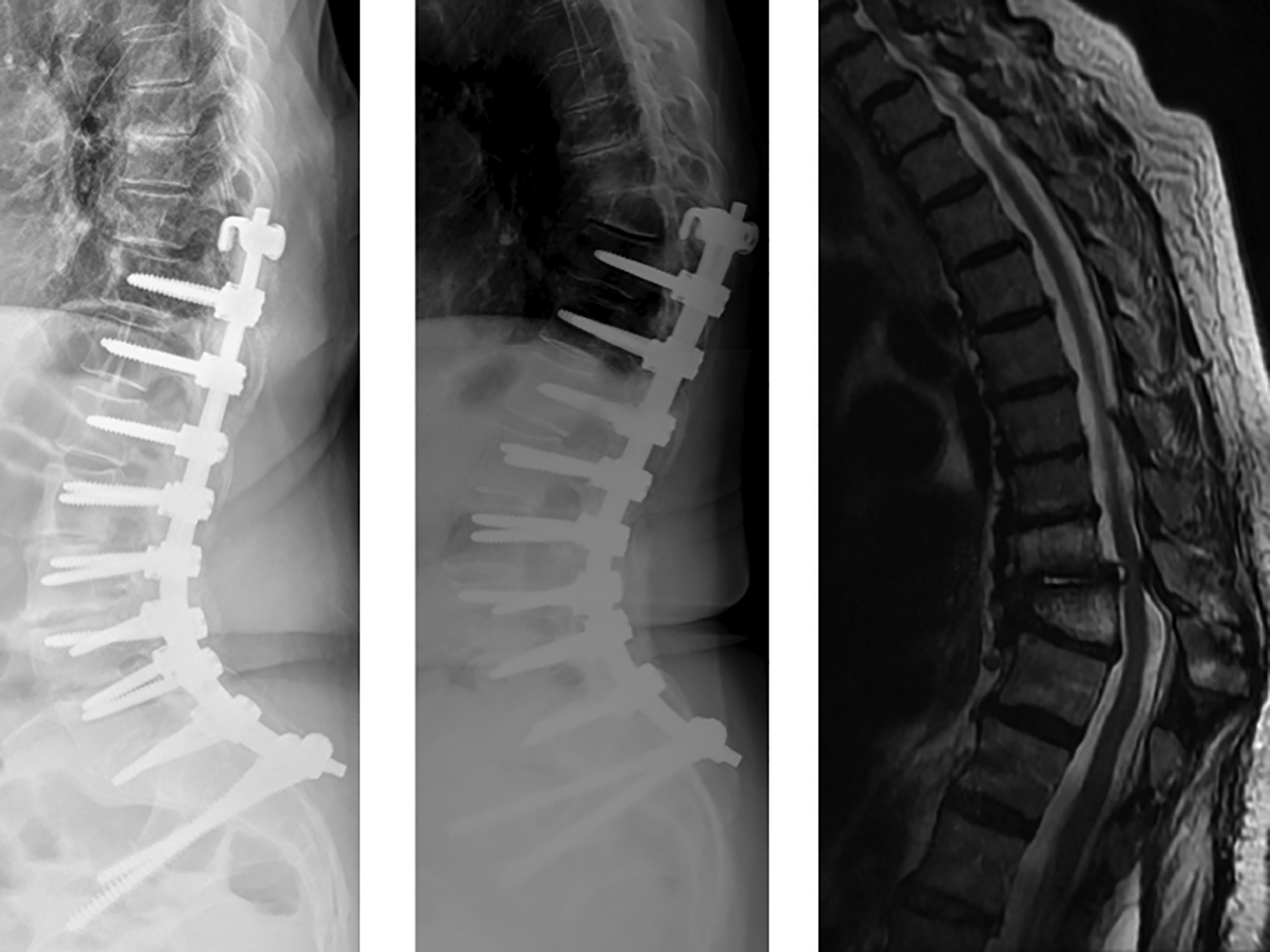
Radiograph of a patient who initially underwent T10-pelvis posterior spinal fusion for adult degenerative scoliosis (left panel). At 2-year follow-up (middle panel), the patient had developed proximal junctional failure at T9 with magnetic resonance imaging demonstrating severe stenosis (right panel) and neurologic deficits necessitating revision surgery.
Sarcopenia, defined as deficient skeletal muscle mass relative to total body mass, is a risk factor for poor outcomes and increased mortality in a diverse range of medical and surgical populations.7-10 For example, sarcopenia has been associated with dropped head syndrome, 11 degenerative scoliosis, 12 and has been correlated with cervical spine malalignment after laminoplasty, 13 indicating that muscle mass plays an important role in the maintenance of normal sagittal balance. Importantly, sarcopenia is a modifiable risk factor, with treatment, including resistance exercise and diet modification.14,15 Despite being a known risk factor for poor surgical outcomes,7-10 limited data exists on the relationship between sarcopenia and PJD. 16 As a better understanding of modifiable PJD risk factors may lead to improved surgical outcomes, the aim of this study was to determine whether sarcopenia is an independent risk factor for PJD in patients undergoing ASD surgery.
Materials and Methods
Patient Population
We hypothesized that sarcopenia (Figure 2) would be a significant, independent risk factor for development of PJD. Institutional review board approval was obtained, and this study was conducted following STROBE (Strengthening the Reporting of Observational studies in Epidemiology) guidelines. Retrospective review was performed of prospectively collected data from a single-institution clinical registry of thoracolumbar ASD patients. All patients included in the registry had an underlying diagnosis of adult degenerative scoliosis, and all patients provided consent for registry inclusion. Criteria for registry inclusion were age >18 years and at least one of the following preoperative measures of thoracolumbar deformity: sagittal vertical axis (SVA) >5 cm, coronal Cobb angle >20°, pelvic incidence–lumbar lordosis (PI-LL) mismatch >20°, or pelvic tilt (PT) >20°. All ASD patients who underwent posterolateral fusion from the thoracic spine to the pelvis, with or without anterior fusion and with a minimum of 2-year clinical and radiographic follow-up were included.
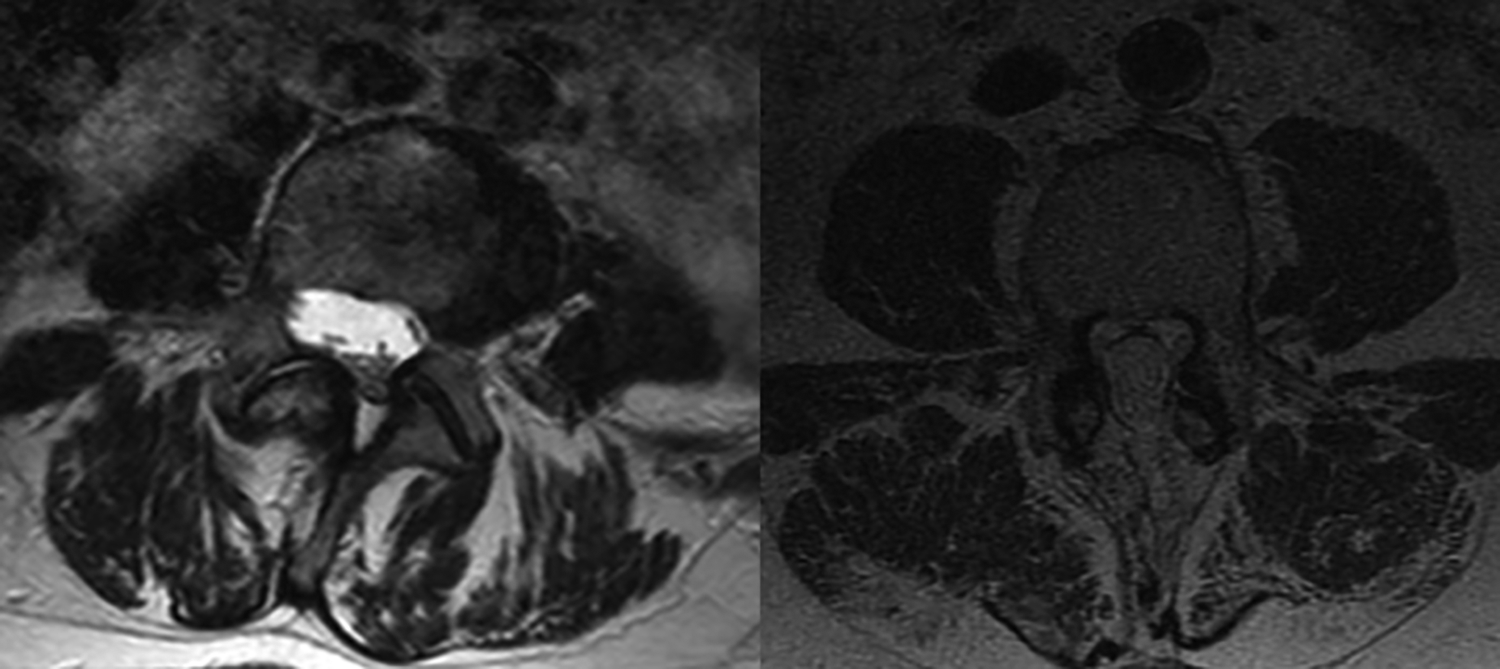
Example images of patients with and without sarcopenia. Left panel shows axial T2 magnetic resonance imaging (MRI) at the level of L4 pedicles in a 63-year-old woman with psoas cross-sectional area of 682 mm2. She went on to develop hardware failure and proximal junctional disease (PJD), necessitating revision surgery. Right panel shows axial T2 MRI in a 69-year-old man with psoas cross-sectional area of 1996 mm2; he did not develop PJD at final follow-up.
Data Collection
PJK was defined as proximal junctional angle (PJA) or sagittal Cobb angle between the inferior endplate of the upper instrumented vertebra (UIV) and superior endplate of UIV + 2 of at least 10°, and an increase from preoperative PJA of greater than 10°. 3 PJF was defined as hardware failure (screw pullout or rod fracture), vertebral body fracture at UIV or UIV + 1, or need for revision surgery specifically due to PJD. Radiographic and clinical assessments were made preoperatively and at 3, 6, 9, 12, and 24 months postoperatively. Specifically, PJK was defined as the maximum increase in the PJA between the preoperative lateral x-ray and any postoperative lateral x-ray during the first 2 years following surgery.
Muscle mass measurement for identification of sarcopenia was performed by obtaining the average cross-sectional area of the 2 psoas muscles at the level of the L4 pedicle, as previously described. 8 Measurements were obtained from preoperative computed tomography (CT) or magnetic resonance imaging (MRI) scan using the freehand region of interest tool (Figure 3) in our PACS (picture archiving and communication system) system (Intellispace PACS Enterprise, Philips). A total of 21 patients had undergone preoperative CT scans of the lumbar spine or abdomen/pelvis. In these patients, axial CT Hounsfield units of trabecular bone at the L4 level were measured as an index of bone density, as previously described.17,18 However, bone density was not included in the final multivariable model given insufficient number of patients.
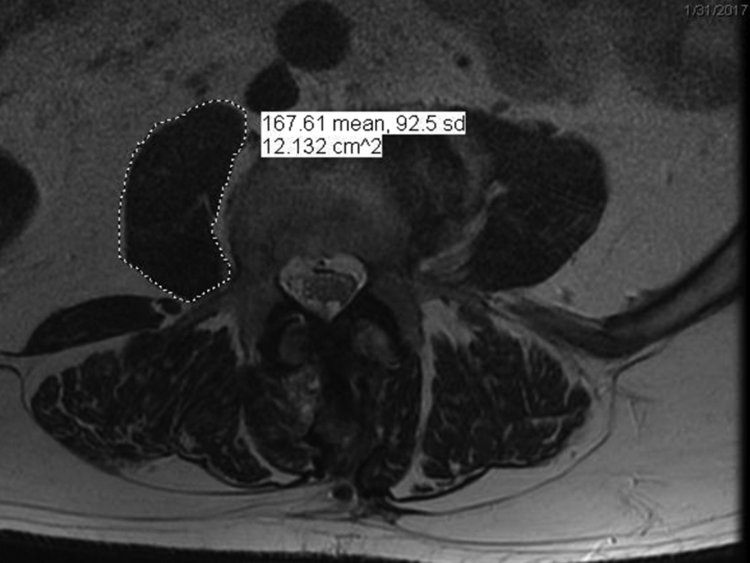
Measurement of psoas cross-sectional area on axial magnetic resonance imaging using region of interest tool.
Covariates previously described as risk factors for PJD, including age, gender, body mass index (BMI), UIV in the upper versus lower thoracic spine, combined anterior/posterior fusion, 3-column osteotomy, revision surgery, preoperative SVA, and change in SVA were collected.
Statistical Analysis
Primary outcome measures included degrees of PJK and the rate of PJF. Categorical variables were compared using 2-tailed Fisher’s exact test. Multivariate linear regression was performed to identify independent risk factors for PJK, and a separate multivariate linear regression was performed to identify independent risk factors for PJF. To determine whether psoas cross-sectional area should be indexed to patient size or height, multivariate regression analyses were repeated using psoas cross-sectional area corrected for patient height and for L4 vertebral body area ratios. Results of the repeated analyses were then compared using goodness of fit to the analyses performed using unadjusted psoas cross-sectional areas. Odds ratios for developing PJK and PJF were calculated based on psoas cross-sectional areas for both men and women, and subsequently used to determine disease specific thresholds for sarcopenia. Microsoft Excel (Microsoft Corp) and Wizard (version 1.9.42) were used for statistical calculations with level of significance defined as P = .05.
Results
Patients and Baseline Demographics
Of 52 patients in the ASD registry, 39 underwent instrumented posterolateral spinal fusion from the thoracic spine to the pelvis. Of the 39 patients, 7 were excluded for insufficient clinical or radiographic follow-up. The remaining 32 patients were included in our study. Patient demographics, baseline radiographic characteristics, and surgical variables are summarized in Table 1; with the exception of psoas cross-sectional area, no significant difference was observed between cohorts with regard to baseline characteristics, radiographic parameters, or surgical variables. All patients in the cohort without PJD, as well as all patients in the cohort with PJF, had undergone proximal augmentation with hooks at their UIV. Three patients in the PJK cohort did not have proximal augmentation constructs; all other patients in the PJK cohort had hook augmentation at their UIV.
Baseline Patient Characteristics and Surgical Variables.
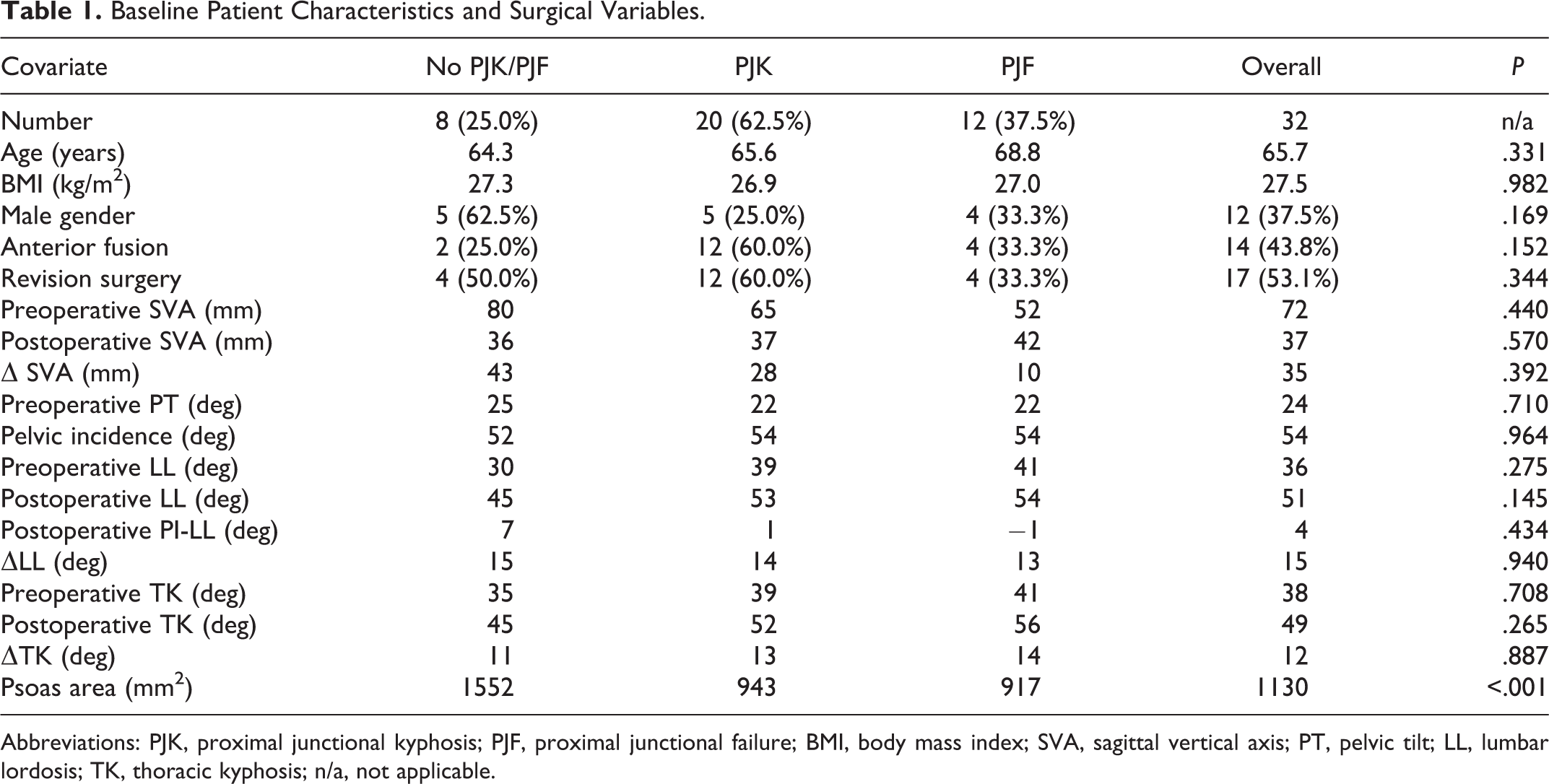
Abbreviations: PJK, proximal junctional kyphosis; PJF, proximal junctional failure; BMI, body mass index; SVA, sagittal vertical axis; PT, pelvic tilt; LL, lumbar lordosis; TK, thoracic kyphosis; n/a, not applicable.
Table 2 lists descriptions of the index procedures performed and associated junctional complications. Among all patients, mean preoperative SVA was 72 mm, and mean postoperative SVA was 37 mm. Lumbar lordosis increased from a mean of 36° preoperatively to 51° postoperatively. Of the 32 patients, PJK and PJF occurred in 20 (62.5%) and 12 (37.5%), respectively. The mean magnitude of PJK was 16°. The rate of PJF was 33.3% in men and 40% in women (P = .73). The mode of PJF was construct failure in 7 patients, vertebral body fracture in 12 patients, and posterior osseo-ligamentous failure in 5 patients, with many patients having multiple simultaneous modes of failure. The mean cross-sectional psoas area was 1130 mm2 (range: 518-1996 mm2), with mean of 1545 mm2 in men and 880 mm2 in women.
Procedure Type and Junctional Complications.

Abbreviations: F, female; M, male; PJK, proximal junctional kyphosis; PSF, posterior spinal fusion; ALIF, anterior lumbar interbody fusion; PSO, pedicle subtraction osteotomy; VCR, vertebral column resection.
Multivariable Analysis, Disease-Specific Sarcopenia Thresholds, and Bone Density
Table 3 lists results of multivariable analysis for PJK risk factors. Of risk factors included in the analysis, only psoas cross-sectional area was significantly associated with development of PJK. Results of multivariable analysis for PJF risk factors are shown in Table 4; of the included risk factors, only revision surgery (P = .04) and psoas cross-sectional area (P = .009) were significantly associated with development of PJF. Each 1 cm2 decrease in psoas cross-sectional area was associated with an increase of 2.8° of PJK (95% CI 0.7°-4.9°) and a 1.1% increase in the risk of PJF (95% CI 0.3%-1.9%). The R2 value for the PJF regression using psoas area alone was 0.49, as compared with 0.44 after adjusting psoas cross-sectional area for patient height; R2 was 0.41 after adjusting psoas cross-sectional area for L4 vertebral body area. Thus, we concluded that correcting psoas area for patient size did not contribute significant additional predictive capacity to the model.
Multivatiate Linear Regression of Proximal Junctional Kyphosis Risk Factors.
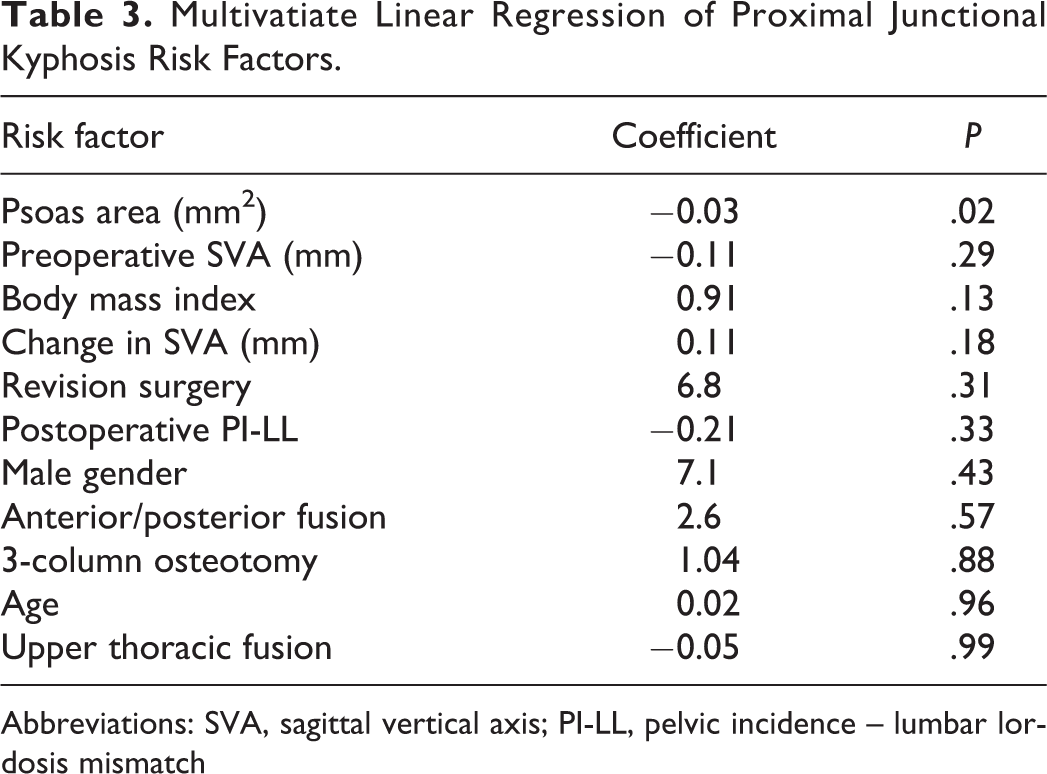
Abbreviations: SVA, sagittal vertical axis; PI-LL, pelvic incidence – lumbar lordosis mismatch
Multivariate Linear Regression of Proximal Junctional Failure Risk Factors.
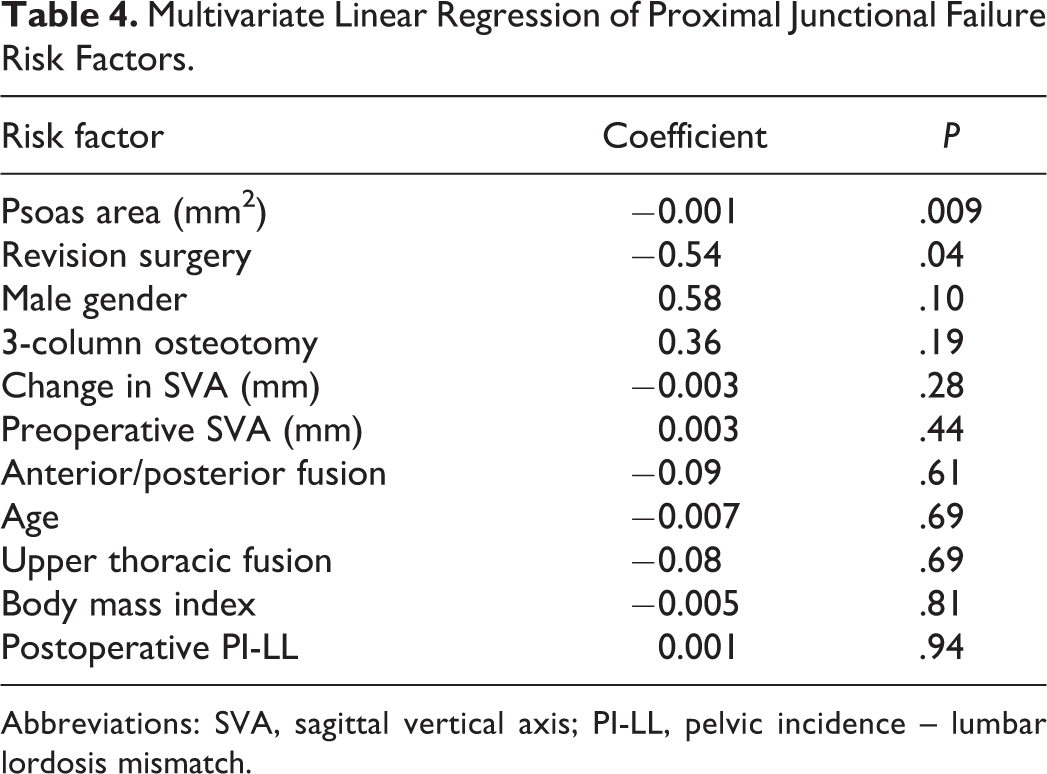
Abbreviations: SVA, sagittal vertical axis; PI-LL, pelvic incidence – lumbar lordosis mismatch.
Thresholds of 12 cm2 in men and 8 cm2 in women were found to maximize odds ratios of developing PJK. The rate of PJF was 69% in patients with psoas cross-sectional area below these thresholds and 16% in patients above these thresholds. Subanalysis of the 21 patients with preoperative CT scan demonstrated a low correlation (R2 = 0.02) between psoas cross-sectional area and bone density as measured by L4 Hounsfield units.
Discussion
In this 2-year follow-up study of 32 patients who underwent surgery for ASD, decreased psoas cross-sectional area was found to be significantly associated with development of both PJK and PJF after adjusting for previously described PJD risk factors. The association between psoas cross-sectional area and PJD was stronger than the association between PJD and other previously described PJD risk factors. Further analysis demonstrated that threshold psoas cross-sectional areas of 12 cm2 in men and 8 cm2 in women could predict development of PJD and may serve as reference values for defining sarcopenia in ASD patients.
To our knowledge, no studies have attempted to establish thresholds for defining sarcopenia in ASD, and only one other study has evaluated the relationship between sarcopenia and PJD. In their study of 49 ASD patients, Kim et al 16 found sarcopenia to be significantly associated with PJK but did not adjust for other known PJD risk factors, such as BMI. In addition to Kim et al, 16 there is evidence that skeletal muscle plays an important role as a dynamic stabilizer of spinal alignment. For example, dropped head syndrome is associated with sarcopenia as well as a variety of myopathies.11,19,20 PJK has also been linked to neuromuscular diseases such as diabetic neuropathy, prior stroke, and metabolic encephalopathy. 21 These previous studies, when combined with our findings, suggest that a complete risk assessment and prevention strategy for PJD needs to account for muscle strength and control.
Our study has important clinical implications. First, as psoas cross-sectional area is easily measured on standard preoperative imaging, surgeons should consider integrating psoas cross-sectional measurements into their preoperative workup. Patients identified as higher risk for PJK based on psoas cross-sectional area may subsequently be referred to physical therapy for preoperative strengthening, dieticians for nutritional optimization, or other specialists for appropriate management of comorbidities contributing to sarcopenia. In addition, preoperative identification of sarcopenia can help with preoperative planning, as these patients may benefit from augments such as tethers (Figure 4) to reduce their risk of PJD. 22
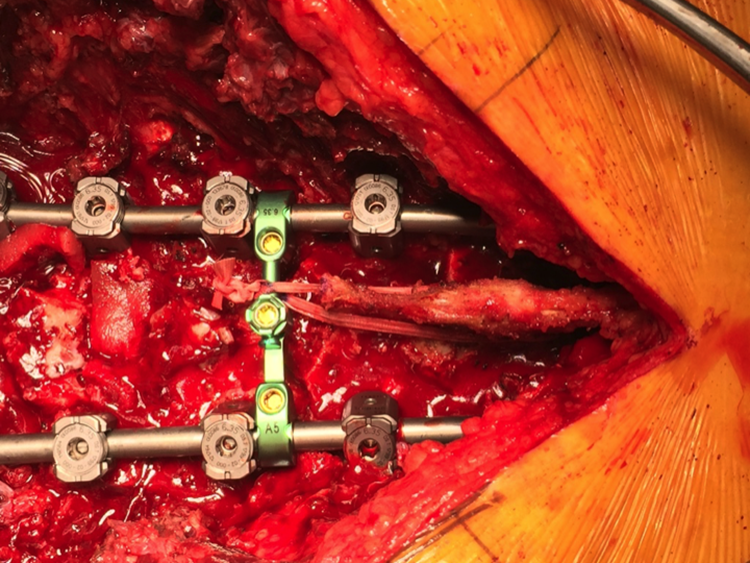
Intraoperative photo showing ligament augmentation with polyester fiber tape as a prophylaxis measure against proximal junctional kyphosis (PJK). The tape is passed through the spinous process of the upper instrumented vertebra and tensioned through a crosslink, tethering the junctional zone against flexion deformity forces.
Strengths of our study include multivariable analyses, 2-year follow-up data, and use of well-defined and easily adopted radiographic parameters, which increase generalizability of our results. For example, we chose to use psoas cross-sectional area as an index of sarcopenia, as it is the most commonly used index of skeletal muscle mass in previous studies of the relationship between sarcopenia and clinical outcomes.8,9,23 Psoas cross-sectional area has been closely correlated with other assessments of sarcopenia, including measurement of muscle mass on dual-energy x-ray absorptiometry (DEXA), 24 bioimpedance analysis, 24 grip strength, 25 and serum markers of muscle atrophy. 26
This study has several limitations. Generalizability is limited by small sample size and single-institution data. However, while the study was limited by its sample size, the strong correlation of sarcopenia with PJD suggests that sarcopenia may be among the strongest predictors of PJD. Further studies with larger patient populations will be necessary to objectively compare effect sizes of various PJD risk factors. The small sample size may also explain the decrease in R2 value when adjusted psoas cross-sectional area was included in the regression model. We specifically used the R2 value to determine whether adjustment of psoas cross-sectional area measurements would allow the model to more precisely fit patients’ risk for developing PJD. With the finding that utilizing unadjusted versus adjusted psoas cross-sectional area had minimal impact on the model’s fit of the data points, we decided to not include adjusted psoas cross-sectional area in the final model. The rationale for not including adjusted cross-sectional area is that this would introduce extra variables and potential bias into the model. Interestingly, the R2 value decreased when the adjusted, rather than nonadjusted psoas cross-sectional area was included in the model; however, this may be due to addition of more data points in the model. These specific data points included L4 vertebral body cross-sectional area and patient height, which were used separately to adjust psoas cross-sectional area, and which themselves were susceptible to additional measurement errors and outliers. The effect of such measurement errors and outliers may be compounded by the relatively small sample size of the study population. Given the small sample size, it is unlikely that any meaningful conclusion can be drawn from the decrease in R2 when adjusted psoas cross-sectional area is included in the model.
In addition to sample size, additional limitations include the study’s retrospective design, which precluded our ability to demonstrate a causative link between sarcopenia and PJD. Furthermore, while sarcopenia may be treated through exercise and nutrition,14,15 it is unknown whether such interventions would decrease surgical complication rates. Our study is also limited by the lack of bone mineral density data on all patients, as osteopenia may contribute to PJK. 16 We attempted to use CT Hounsfield units of trabecular bone as a substitute for DEXA data, as this has been correlated with DEXA assessment of bone density and fracture risk.17,18 With CT data, we did not find a correlation between osteopenia and sarcopenia. However, prior studies have reported a link between the conditions. 27 While osteopenia and sarcopenia may both result from and be signs of overall frailty, there is evidence that the relationship is site specific; leg muscle strength has been shown to be more predictive of proximal femoral bone density than total body skeletal muscle mass. 28 This implies that the 2 conditions may have a direct relationship due to local mechanotransductive effects, and as such, addressing sarcopenia may reduce the risk of PJK attributed to reduced bone mineral density.
In conclusion, we found that decreased psoas cross-sectional area is significantly associated with risk of developing PJD and were able to define disease-specific thresholds for sarcopenia in ASD. This is significant, as sarcopenia is a potentially modifiable PJD risk factor. As such, preoperative identification of sarcopenia may inform both preoperative patient optimization, as well as use of adjuncts when preoperative planning. Future studies should assess disease specific sarcopenia thresholds in ASD, as well as outcomes following interventions for sarcopenia.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Klineberg has worked as a consultant for DePuy Synthes Spine, Stryker, and Medicrea, and Drs Klineberg and Javidan have received honoraria and a fellowship grant for University of California, Davis from AOSpine. Dr Javidan has worked as a consultant for NuVasive, Alphatec, and Stryker, holds stocks in Alphatec, and has served as a consultant for Medicrea.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Drs Klineberg and Javidan have a fellowship grant for University of California, Davis from AOSpine.
