Abstract
Study Design:
Level 4 retrospective case series.
Objectives:
Surgical site infection (SSI) is one of the main complications of instrumented spinal fusion. The aim of our study was to evaluate infection recurrence (same bacteria) or reinfection (different bacteria) in posterior spinal fusion in children.
Methods:
A retrospective study was conducted to evaluate patients who were successfully treated for SSI after instrumented spinal fusion due to deformity, with irrigation and debridement (I&D) procedures, followed by antibiotic therapy, with a follow-up of at least 2 years.
Results:
Overall, 29 patients with a mean age of 14 + 3 years were evaluated. Preoperative diagnosis was nonidiopathic scoliosis in 23, idiopathic scoliosis in 5, and Scheuermann’s disease in 1 patient. The etiology was Gram-positive cocci (40.9%), Gram-negative bacilli (27.2%), and polymicrobial infection (31.8%). A mean of 1.5 (1-3) I&D procedures were performed. Intravenous antibiotic treatment was given for a mean of 15.8 (4-86) days, followed by oral treatment for a mean of 335.1 (0-1095) days. Mean follow-up was 5 + 2 years (2 to 14 + 7 years) during which 28 patients were cured (96.6%) and 1 patient developed reinfection (3.4%). This reinfection was treated with oral clindamycin for 6 months. After the infection persisted, the decision was to remove the implants.
Conclusions:
In this series of 29 pediatric patients who underwent instrumented spinal fusion due to deformity, we reported one case of reinfection (3.4%). Given that 96.6% of infections were resolved, we suggest treatment with I&D procedures with retention of implants to treat acute SSI.
Keywords
Introduction
Spinal deformities in children often require complex surgeries, including spinal fusion with instrumentation. Surgical site infection (SSI) is one of the main complications of this type of surgery. 1
In the guidelines of the Centers for Disease Control and Prevention (CDC), acute superficial SSI is defined as an infection affecting the skin and subcutaneous tissue, while a deep SSI affects deep tissues such as fascial and muscle layers, both recurring within 90 postoperative days. 2 According to a multicenter study, 67% of the SSIs are identified within 30 days and 90% within 6 months after surgery. 3 Ramo et al found that 43% of the infections occur 3 months and 27% 12 months after surgery. 4
Incidence reports of SSI after instrumented spinal fusion varies according to the underlying disease. Different incidence rates have been reported in multiple reports available in the literature. SSI rate in adolescent idiopathic scoliosis has the lowest incidence from 0.9% to 3% in different studies. 5,6 SSI rate for congenital scoliosis is 6.1%, and for syndromic scoliosis it is 6.9%. 3 The highest infection rate is found in neuromuscular scoliosis, associated with, for example, cerebral palsy, with a reported range from 4% to 14%, 7,8 or myelomeningocele, with a range from 8% to 24%. 9,10
Treatment of SSI depends on the type of infection. Acute SSI is typically treated with aggressive irrigation and debridement (I&D) procedures, primary closure with deep and/or superficial drains, and antibiotics with implant retention. The initial success of this treatment is variable. Although implant removal may be an option to reduce the bacterial burden and to remove the biofilm produced by certain bacteria, it would threaten stability of the spine. Therefore, it is not the treatment of choice in acute infections. 11
Late-onset SSI are generally treated similarly to acute infections, and removal of the implant may be indicated once a solid fusion mass has been confirmed. 12 At our center, the patient is considered to be cured from infection after completing appropriate antibiotic therapy for at least 6 to 12 months in the absence of clinical signs of infection and laboratory parameters including white blood cells (WBCs), erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) within the normal ranges. To our knowledge, there are scarce reports in published literature on long-term infection recurrence after successful treatment of spinal SSI.
The aim of our study was to evaluate infection recurrence or reinfections, defined as infection recurrence by the same microorganism and reinfection by a different microorganism, after successful treatment of acute SSI in children that underwent spinal instrumented fusion.
Material and Methods
A retrospective descriptive study was conducted to evaluate patients who underwent surgery for spine deformity, either scoliosis or kyphoscoliosis, between January 2004 and January 2016 who experienced acute SSI. Inclusion criteria were patients younger than 21 years; with 6 or more levels fused; with a simple or double approach; thoracic, lumbar, sacral, and/or pelvic instrumentation; 1- or 2-stage surgery; diagnosed with an acute SSI; treated with surgical I&D procedures; culture sample taken; appropriate antibiotic therapy; considered cured according to the above-mentioned definition; and with at least 2 years of follow-up after successful treatment of the acute infection.
In our institution, the procedure used in a patient with SSI is the revision of the surgical wound to the deep plane, debridement of the necrotic tissues, the irrigation with abundant volumes of physiological solution (3 to 6 liters), sampling for culture, placement of superficial/deep drainages, primary wound closure, and IV (intravenous) antibiotics treatment adjusted to antibiogram with control of clinical and laboratory parameters. According to clinical and laboratory evolution, serial I&D approach is conducted, or the change to oral antibiotics and discharge from hospital. Periodic follow-up is performed carrying out laboratory tests with parameters of WBCs, CRP, and ESR. In case of signs or symptoms of relapse of infection, complementary magnetic resonance imaging and computed tomography studies are performed, in which in case of evidencing a solid arthrodesis, revision surgery is proposed to remove surgical implants with debridement and antibiotic therapy. If solid arthrodesis is not evident, options such as the use of a VAC (vacuum-assisted closure) system or the removal of implants and the placement of an orthosis are considered (Figure 1).
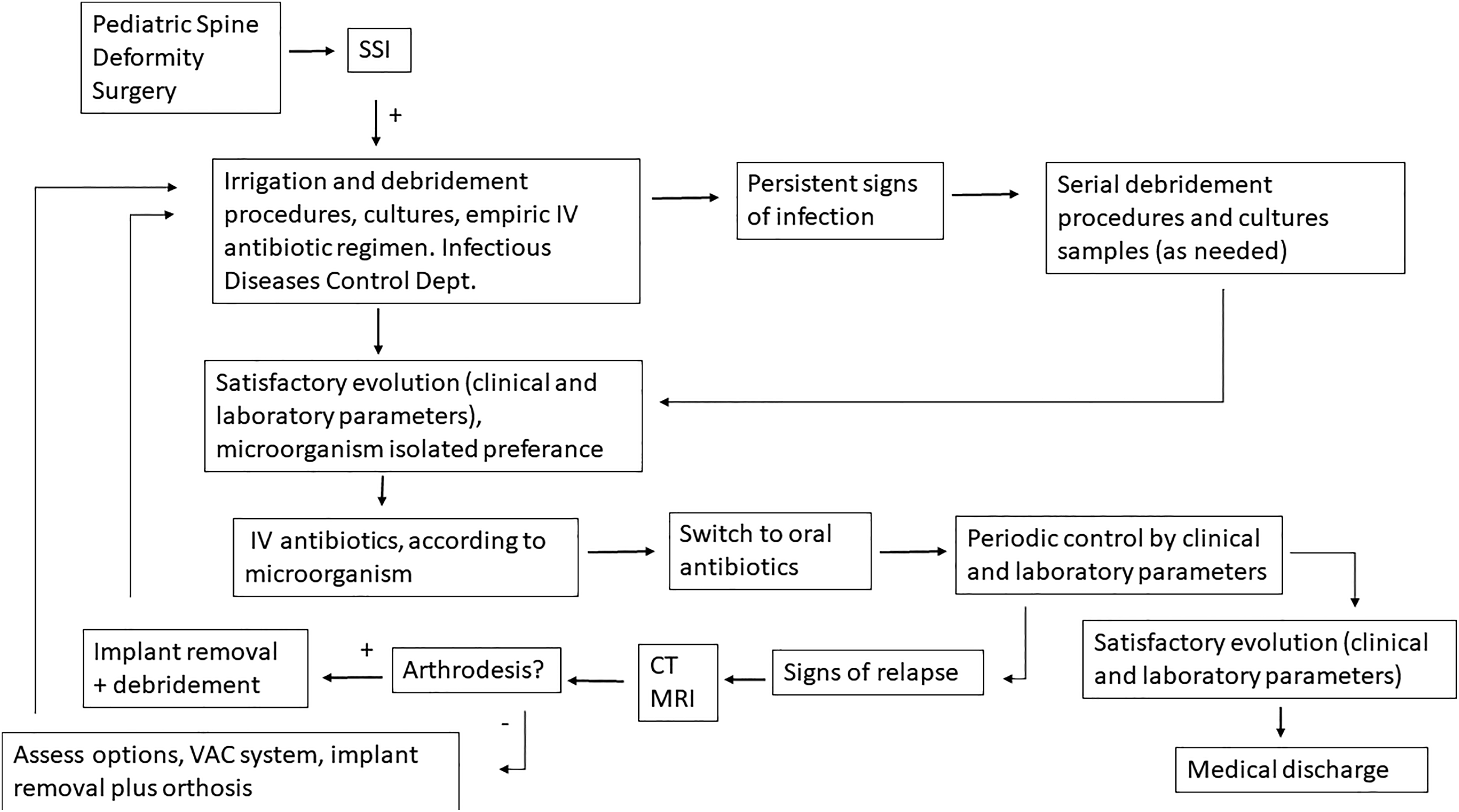
Algorithm used to treat surgical site infection. SSI, surgical site infection; Dept, department; CT, computed tomography; MRI, magnetic resonance imaging; VAC, vacuum-assisted closure.
Patients with a history of previous spine surgery as well as those with myelomeningocele, tumors, or previous infections were excluded from the study.
The study variables included were age, sex, underlying disease, time from infection onset to resolution, number of I&D procedures required for cure, isolated microorganism, antibiotic therapy, and outcomes. A standardized data abstraction sheet was made to fulfill with the required data. Only one author extracted all clinical data from the clinical records.
Results
Overall, 29 clinical charts were evaluated. Eighteen patients were female and 11 male with a mean age of 14 + 3 years (range, 6 + 7 to 20 + 1 years) at the time of surgery. Preoperative diagnosis was neuromuscular scoliosis in 14, syndromic scoliosis in 6, idiopathic scoliosis in 5, congenital scoliosis in 3, and Scheuermann’s disease in 1 patient. All patients presented with acute postoperative deep SSI according to the CDC criteria.
A positive culture was observed in 22 out of 29 cases (75.8%). The most frequently found microorganism, Staphylococcus aureus, was isolated in 9 patients (40.9%), 6 of which were methicillin-resistant S aureus (MRSA, 66.6%) and 3 methicillin-susceptible S aureus (MSSA, 33.33%), followed by Gram-negative bacilli in 6 patients (27.2%): Escherichia coli in 2 cases, and Klebsiella, Enterobacter cloacae, Proteus mirabilis, and Acinetobacter in one each. In 7 patients (31.8%), 2 microorganisms were isolated, including MRSA, Enterobacter, Pseudomona, Serratia, and Proteus.
Mean time from surgery to diagnosis of SSI was 23.7 days (5-90 days). In all cases treatment consisted of surgical I&D with the sample taken for culture and histopathological studies. Aggressive debridement of the devitalized tissue was performed, with high-volume irrigation with normal saline, and closure of the layers, leaving 2 drains in place in the surgical field. In none of the patients the implant was removed during the acute episode. Postoperatively, empirical antibiotic therapy was given. In 23 cases (79.3%) one surgical debridement was performed, 4 cases (13.7%) required a second debridement, and in 2 patients (6.8%) 3 I&Ds were performed, with a mean of 1.5 I&D per patient. Mean duration of IV antibiotic treatment, prescribed by the Department of Infectious Diseases based on an antibiogram, was 15.8 days (4-86 days), followed by oral maintenance therapy for a mean of 335.1 days (0-1095 days). Treatment was discontinued when ESR and CRP levels were within normal ranges. It is important to highlight that in cases who did not receive maintenance therapy, cultures were completely negative and postoperative outcome was good both clinically and according to laboratory results.
Mean follow-up was 5 + 2 years (2 years to 14 + 7 years) during which all 28 patients were considered cured without signs of infection recurrence. In this series, only one patient (3.4%) developed a reinfection. This 6-year-old patient had syndromic scoliosis that was treated with instrumented posterior spinal fusion of T4-L2 in a 2-stage surgery. On day 9 after the second surgery, surgical debridement was performed and a multi-resistant Acinetobacter was isolated. Antibiogram-based IV antibiotic therapy (vancomycin + meropenem) was given for 3 months followed by 10 months of oral treatment with good outcome, for which reason it was discontinued. In the fourth year of follow-up the patient showed signs of infection. Fine-needle biopsy was performed and culture results were positive for MSSA. Oral clindamycin was initiated for 6 months, but failed to resolve the recurrent infection. As a result, the decision was made to perform revision surgery to remove the spinal implant, leading to infection resolution. The patient was seen at regular visits for 5 years for follow-up and there were no signs of infection recurrence (Table 1).
Clinical characteristics of patients diagnosed with SSI following instrumented spinal fusion due to deformity, cultures and treatment performed.
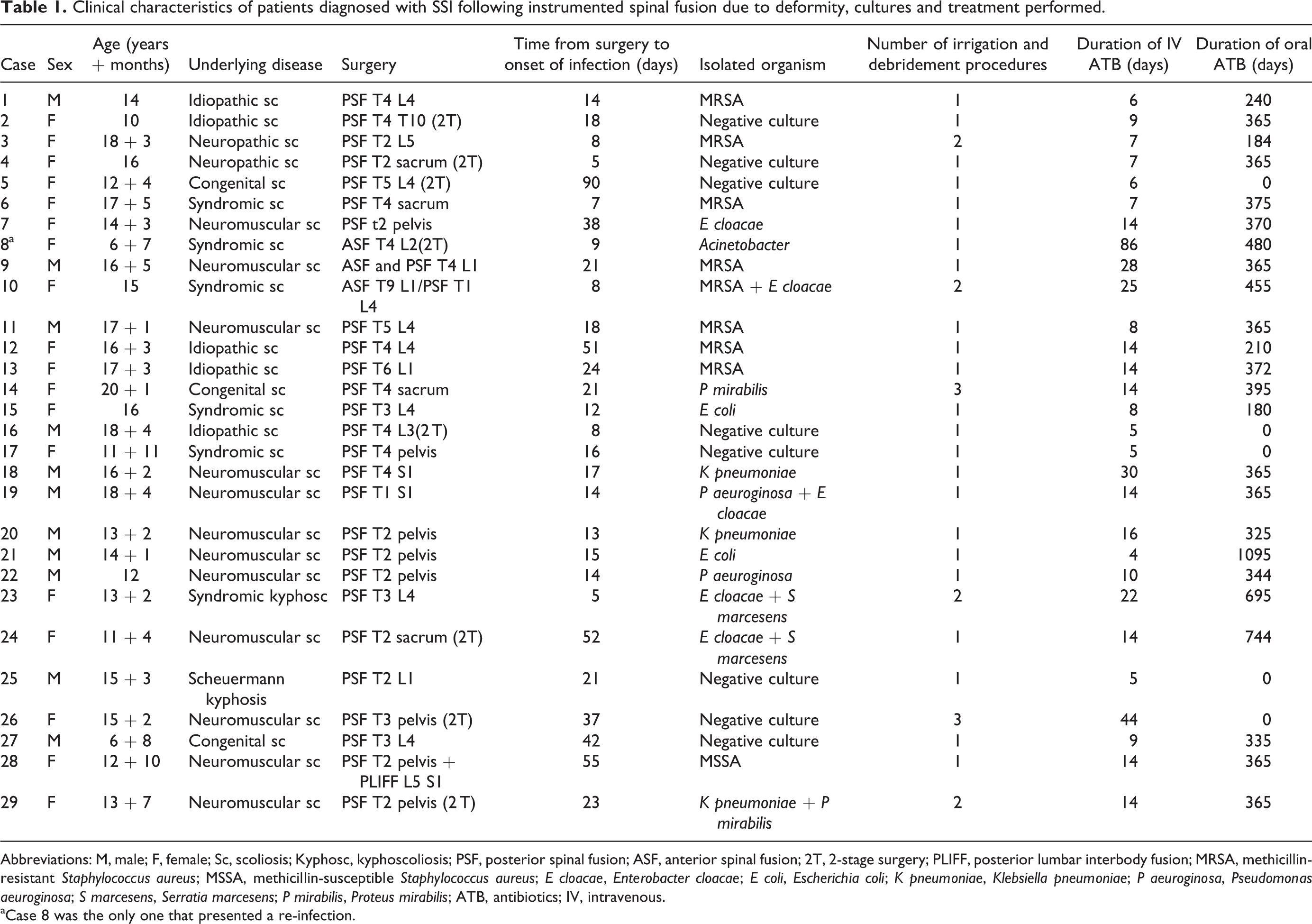
Abbreviations: M, male; F, female; Sc, scoliosis; Kyphosc, kyphoscoliosis; PSF, posterior spinal fusion; ASF, anterior spinal fusion; 2T, 2-stage surgery; PLIFF, posterior lumbar interbody fusion; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-susceptible Staphylococcus aureus; E cloacae, Enterobacter cloacae; E coli, Escherichia coli; K pneumoniae, Klebsiella pneumoniae; P aeuroginosa, Pseudomonas aeuroginosa; S marcesens, Serratia marcesens; P mirabilis, Proteus mirabilis; ATB, antibiotics; IV, intravenous.
aCase 8 was the only one that presented a re-infection.
Discussion
Numerous studies have described variables related to SSI associated with scoliosis surgery in children. These variables include infection rates according to underlying condition, microbiology, risk factors, progression rate of the deformity in case the implant is removed, and need for reimplantation, among others. 3,4,13 -15 Nevertheless, we identified few studies assessing the long-term effectiveness of cure of acute SSI treated with I&D and antibiogram-based antibiotics with retention of the implant. 11,16
Lamberet et al did not find infection recurrence in a study of 26 pediatric patients after a follow-up of 24 months. The patients were treated with surgical I&D procedures with 3 to 6 liters of normal saline with 0.05% chlorhexidine with retention of the implant and antibiogram-based antibiotic therapy for a mean time of 19 weeks with a switch to oral antibiotics after a mean of 17 days. 11 Li et al recommend IV antibiotics for at least 4 to 6 weeks after debridement with retention of the implant, followed by oral antibiotics for 2 to 6 months. 16 In our study, the surgical I&D procedures were performed with large volumes of normal saline (3-6 liters) without addition of any pharmacological agent. Antibiotic treatment was more prolonged, with a duration of IV administration of a mean of 15.8 days (4-86 days), followed by an oral maintenance regimen of a mean of 335.1 days (0-1095 days). Curation rate was slightly lower (96.6%), but follow-up was longer (5 + 2 years).
A study by Floccari and Milbrandt 17 describes a personal communication by Glotzbecker et al, which has not been published so far, evaluating the treatment of 101 acute (less than 3 months postoperatively) SSI. Overall, 79.2% of the patients were successfully treated without recurrence of the infection, 70% underwent serial I&D procedures with implant retention, 7.9% needed implant exchange, and 1 patient cleared infection after implant removal. 17 Contrary to these data, in our series the infection resolved in 96.6% within a mean follow-up of 5 + 2 years, while serial I&D procedures were only performed in 20.6% of the cases. However, Glotzbecker et al reported more patients (n = 101) than the present study (n = 29); so if one was to combine the datasets, the overall percentage of resolved infections would be approximately 83.1%. However, considering the costs of revision surgery and the potential of destabilizing the spine, surgical I&D procedures may be an effective alternative to treat these patients.
In our study no strict protocol was used for antibiotic therapy, resulting in patients with negative cultures who received oral antibiotics for 12 months and others who did not receive oral treatment at all, while long-term outcome was good in both. Additionally, treatment was relatively prolonged with a mean of 2 weeks of IV and 47 weeks of oral antibiotics administration. Currently, no studies providing scientific evidence level I-II are available. In a recently published systematic review, the recommendation based on expert opinion was to treat a deep SSI with a 4 to 6-week course of IV antibiotics switched to oral antibiotics only in case of clinical improvement. Oral antibiotics may be used very long-term (duration is not specified). The authors also recommended to remove the prosthetic material if possible. 18 We have found no studies that recommend when to discontinue antibiotic treatment.
From the infectious diseases point of view, in deep SSI removal of the implant is recommended, especially when the microbial agent is highly virulent or response to antibiotic treatment is not adequate. 19 In our study, however, we did not consider this an option at treatment initiation because of the risk of spinal instability.
One of the limitations of this study is its retrospective nature, which involves the availability and accuracy of the medical records. Nevertheless, at the end of the data collection process, we were able to obtain all the relevant information. Another limitation was the small size and heterogeneity of the sample. Additionally, as no antibiotic treatment protocol was used, different scenarios as described above were observed, albeit with good long-term results, that is, no late recurrence.
Conclusions
In this retrospective case series of 29 pediatric patients who underwent posterior spinal fusion of 6 levels or more due to spinal deformity, associated with different etiologies and a mean follow-up of 5 + 2 years, no infection recurrence was observed; however, one patient had a reinfection after an acute SSI. We suggest that conservative treatment of acute SSI with surgical I&D procedures followed by algorithm-based antibiotic therapy may be a viable alternative to revision surgery with implant removal in pediatric spinal deformity. However, randomized trials are needed to provide additional evidence and the optimal dosing or length of administration of oral antibiotics has yet to be determined.
Footnotes
Author Contributions
Design of study: CM, LP, RR, CAT, MN
Consulting references: CM, LP, IAFW, EG, EB
Participation in drafting manuscript: CM, LP, IAFW, MN, EG
Revising manuscript: CM, IAFW, CAT, RR, MN
Approval of the final version of the paper: CM, LP, RR, CAT, IAFW, EG, EB, MN
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the hospital institutional review board (IRB). Because of the retrospective observational nature of the study, the IRB waived the requirement for informed consent.
