Abstract
Study Design:
Systematic review and meta-analysis.
Objectives:
Cervical spinal cord compression (SCC) due to degenerative changes of the spine is a frequent finding on magnetic resonance imaging (MRI). While most people remain asymptomatic, a proportion develop symptoms of degenerative cervical myelopathy (DCM). DCM is an often-progressive neurological disease that can cause quadriplegia. The epidemiology of SCC and DCM is poorly understood. We sought to estimate the prevalence of degenerative cervical SCC and DCM from cross-sectional cohorts undergoing MRI.
Methods:
We conducted a systematic review and meta-analysis of MRI reports on human subjects older than 16 years with degenerative SCC. A predetermined search strategy was used to identify relevant literature on MEDLINE. Title and abstract screenings were followed by full text screening. Data was extracted and analyzed by fixed or random-effects models.
Results:
The present search returned 1506 publications. Following our exclusion criteria, 19 studies were included. Subgroup analysis of 3786 individuals estimated the prevalence of asymptomatic SCC in a healthy population as 24.2% with a significantly higher prevalence of SCC in older populations compared with younger populations and American/European populations compared with Asian populations. Subgroup analysis of 1202 individuals estimated the prevalence of DCM in a healthy population as 2.3%.
Conclusions:
We present the first estimates of the prevalence of asymptomatic SCC and DCM. Studies investigating the epidemiology of SCC are heterogeneous in methodology and results. These data indicate the need for more studies into the epidemiology of SCC and DCM performed with consistent methodologies.
Keywords
Introduction
Cervical spinal cord compression (SCC) due to degeneration of the cervical spine is a frequent finding on magnetic resonance imaging (MRI). 1 -3 Degenerative changes include spondylosis, degenerative disc disease, ligamental hypertrophy, and ossification of the posterior longitudinal ligament. 4 SCC mainly occurs during later stages of life and in most cases remains asymptomatic. 5 -7 Nevertheless, a subset of individuals will develop symptoms, causing a condition that has recently been termed degenerative cervical myelopathy (DCM). 4,8
DCM is a heterogeneous disease with symptoms ranging from mild sensory or motor disturbances to loss of bladder control and tetraplegia. 9,10 It has a severe impact on quality of life with SF-36 (Short Form–36) scores comparable to or worse than many other chronic diseases. 11 Moderate/severe and worsening forms of DCM require surgical decompression. 12 -15 Surgery is able to arrest disease progression and provide limited functional improvement. 14,16 The average age of patients undergoing surgical decompression is in the mid-50s. 16 However, DCM patients often face long delays in diagnosis and treatment, which in turn lead to increased disability. 17
Delayed diagnosis and treatment may be a consequence of a lack of awareness by the general public and health professionals, lack of resources in the form of access to necessary imaging, the absence of dedicated patient pathways, and inadequate access to relevant health professionals. A better understanding of the epidemiology of asymptomatic SCC and DCM is necessary to adequately address the existing problems, optimize recognition and treatment, and consequently reduce disability.
SCC is a necessary but not sufficient prerequisite for the development of DCM. There is no direct relationship between the degree of SCC and the severity of DCM symptoms. 18 Indeed, reported rates of progression from asymptomatic SCC to DCM of at least 1.8% per year indicate that SCC usually does not cause DCM. 5 -7 The prevalence of SCC has been investigated in multiple studies using cervical MRI scans (Figure 1), but no systematic review or meta-analysis of these studies has been undertaken so far. A previous narrative analysis suggested a prevalence of SCC of 4.9% to 13% in the general population, but this study only assessed 5 studies. 18 Present information with regard to the prevalence of DCM largely derives from surgical cohort studies, which underestimate the true prevalence since only a subset of DCM patients undergo surgery. 19

Search strategy used in MEDLINE.
The objectives of this study were to estimate the pooled prevalence of SCC and DCM in healthy adult populations to arrive at a more reliable epidemiological estimate. We also aimed to highlight any shortcomings in the existing data. We aimed to identify cross-sectional studies reporting the prevalence of SCC or DCM. This systematic review is the first quantitative analysis of pooled studies investigating SCC or DCM.
Methods
Search Strategy and Study Selection
The following search strategy, summarized in Figure 1, was used to identify original research investigating the frequency of SCC or DCM on MEDLINE. Index articles 1,2,20 -22 were screened for keywords, including relevant MeSH terms (Figure 1), and experience from existing reviews adapted. 23 -26 The search was conducted on May 15, 2018. References were exported and subsequently managed using Microsoft Excel. The present review was not registered and the protocol was not made available.
Abstracts and titles of all references were screened (SS) and reviews and case reports were excluded. BD independently assessed title and abstract screening by random selection of 160 abstracts confirming homogeneity. Articles published before 1985 were also excluded because MRI was not in routine clinical use before this point. Articles were included if the title/abstract suggested a clinical study or series reporting on MRI of the cervical spine that may provide information on SCC. A low threshold was used for inclusion in full text screening. Full text articles were screened independently (SS and MS). Articles were included if they met the following criteria: (
Data Extraction
An extraction tool was piloted and iteratively developed using a random subset of included articles (BMD and SS). Data items included demographic data such as gender, age and study location, sample size, frequency of SCC, the presence of T2 hyperintensity and the presence of symptoms. DCM was defined as the presence of at least one sign/symptom of DCM 27 in the presence of degenerative SCC. The inclusion criteria and the definition of SCC used in each study was also collected, along with MRI protocol data—specifically the imaging techniques used, the magnetic field strength of the MRI scanner, slice thickness, and the number of reviewers who analyzed the collected data (Supplementary Table S1). SS extracted data by identifying each data element in the full text of all included articles.
Quality and Bias Assessment
SS and MS assessed the risk of bias within studies using 6 of the 10 criteria developed by Hoy et al 28 that were determined to be of relevance (Supplementary Figure S1). Studies found to have high bias (scoring 5 or 6) were excluded from analysis. The risk of bias across studies was not assessed. SS and MS assessed the quality of each study using a set of 9 criteria (Supplementary Figure S2), based on the Joanna Briggs Institute Prevalence Critical Appraisal tool. 29 These criteria evaluated sample selection and representativeness, as well as statistical methods; each study was assigned a quality score between 0 and 9.
The GRADE (Grading of Recommendations, Assessment, Development and Evaluations) approach was used to score the quality of the prevalence estimates. 30 Each estimate was scored as very low-, low-, moderate-, or high-quality evidence. Because the included studies were observational, we initially scored the evidence as low quality and modified this based on the risk of bias, directness of evidence, consistency of results, and risk of publication bias.
Data Analysis
Data was analyzed in Microsoft Excel using a premade meta-analysis spreadsheet with the equation to calculate the Q statistic
Results
Study Selection
The selection process is summarized as a PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram in Figure 2. The initial search identified 1506 articles and abstract screening eliminated 1243 studies. Full text screening of the remaining 263 articles identified 29 eligible articles. A reference hand screen identified 2 more eligible studies leaving 31 articles providing distinguishable data on the frequency of SCC that were considered for meta-analysis. A high risk of bias resulted in 12 articles being excluded from further analysis. 33 -45 Consequently, 19 articles from 10 countries were included in the present analysis 1 -3,5,20,46 -59 (Table 1).
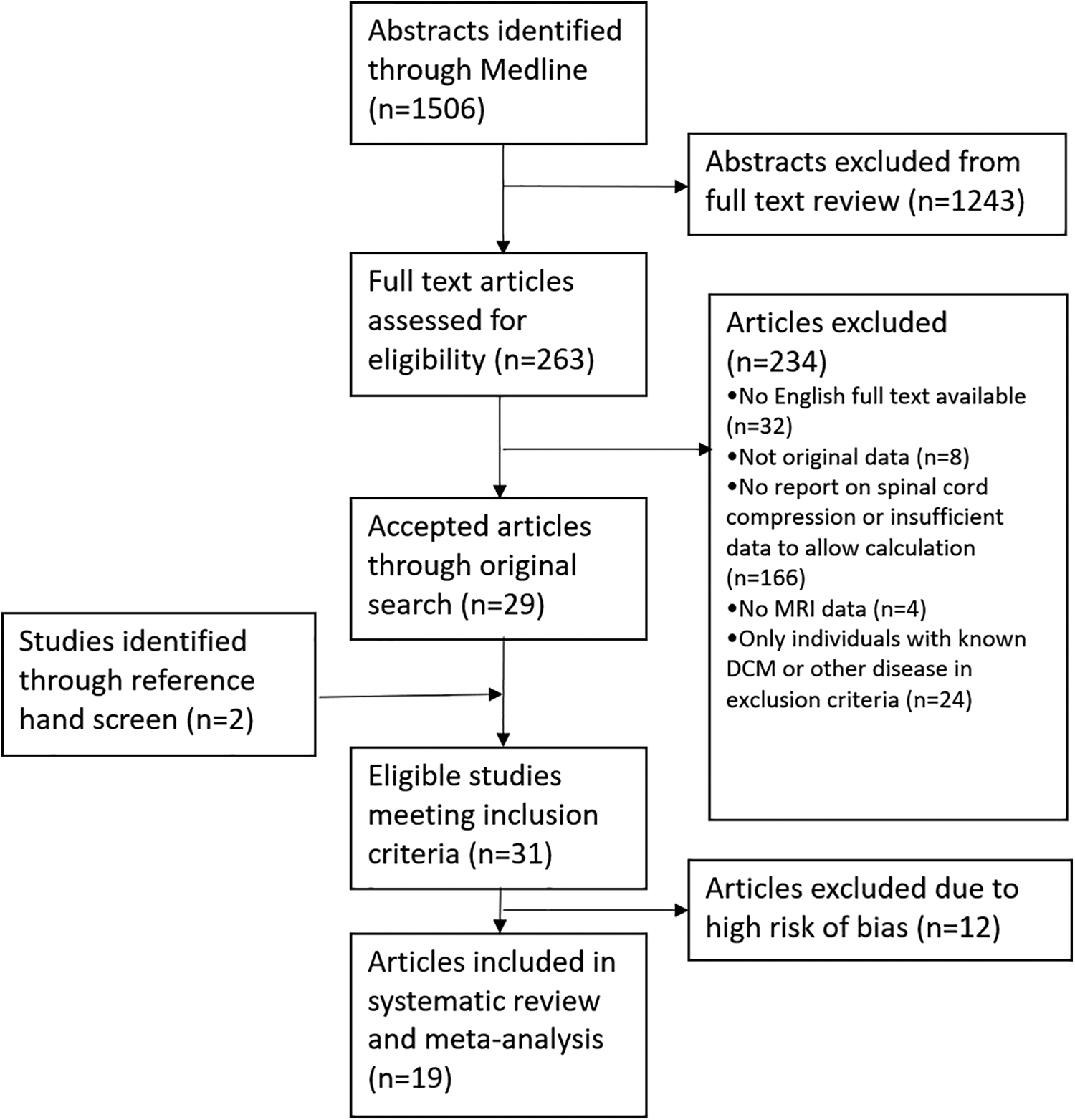
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram. A total of 1506 articles were screened for relevance, and 19 studies were included in this review. Thirteen studies that met the inclusion criteria were excluded due to a high risk of bias.
Details of the 19 Included Studies.a
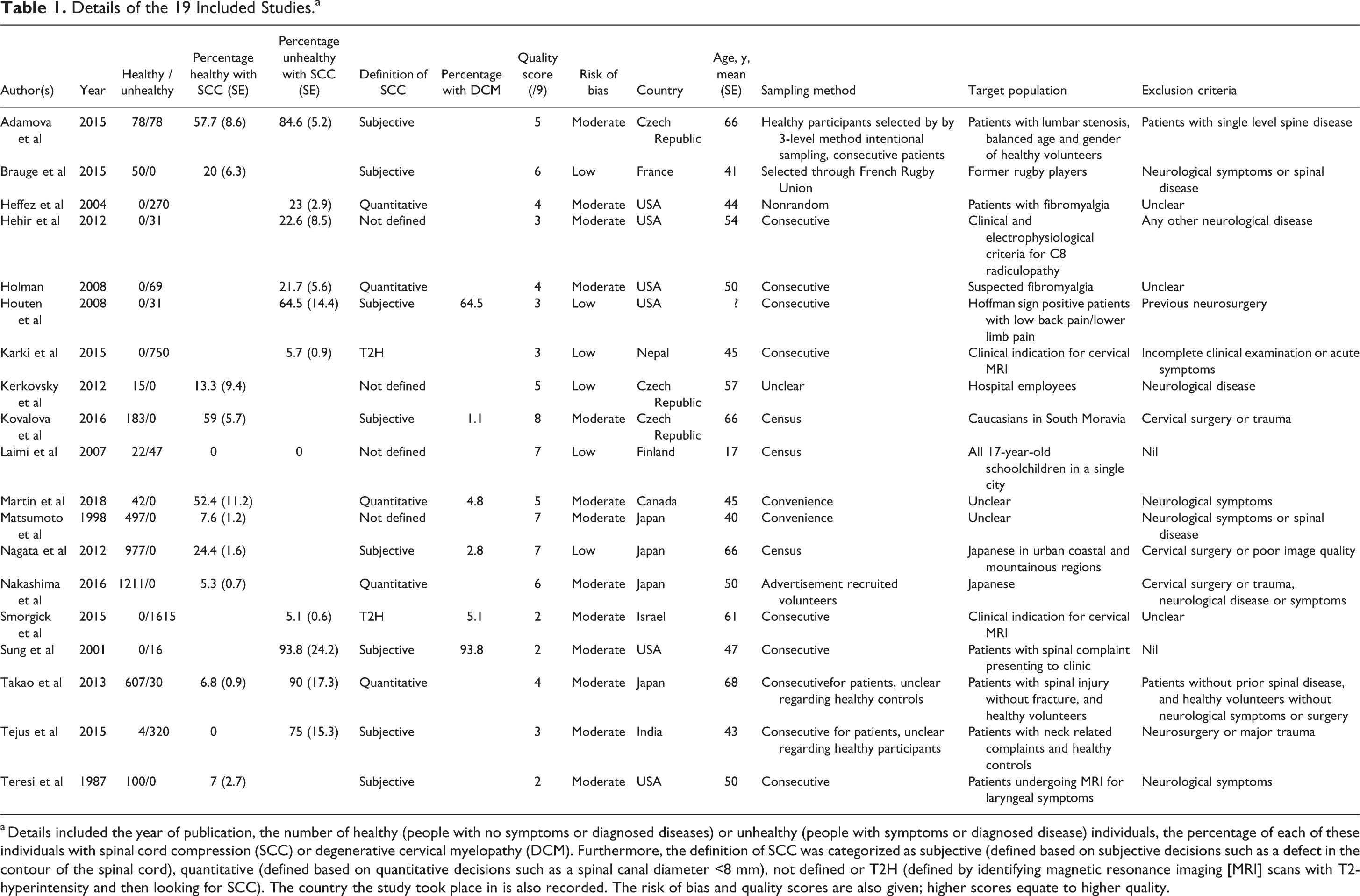
a Details included the year of publication, the number of healthy (people with no symptoms or diagnosed diseases) or unhealthy (people with symptoms or diagnosed disease) individuals, the percentage of each of these individuals with spinal cord compression (SCC) or degenerative cervical myelopathy (DCM). Furthermore, the definition of SCC was categorized as subjective (defined based on subjective decisions such as a defect in the contour of the spinal cord), quantitative (defined based on quantitative decisions such as a spinal canal diameter <8 mm), not defined or T2H (defined by identifying magnetic resonance imaging [MRI] scans with T2-hyperintensity and then looking for SCC). The country the study took place in is also recorded. The risk of bias and quality scores are also given; higher scores equate to higher quality.
Study Characteristics
Of the 19 studies, 11 provided information on healthy individuals, 6 provided data on individuals with symptoms not obviously related to the spinal cord and 5 investigated patients diagnosed with diseases other than DCM. These 19 studies involved a total of 6755 individuals, of which 936 (13.86%) had SCC. To assess heterogeneity between studies, Cochran Q and I2 statistics were calculated. This yielded an I2 value of 97, indicating high heterogeneity. Pooled analysis across all studies was therefore not appropriate. Instead a subgroup analysis was conducted to investigate potential sources of heterogeneity. This revealed that in particular the location of the study, the age of the population imaged, and the presence or absence symptoms influenced the prevalence of SCC. The definition of SCC, the study year, the slice thickness used on MRI and the tesla power of the MRI did not significantly affect rates of SCC. Insufficient studies provided a breakdown on the rate of SCC in men and women to allow gender comparisons. ´
Prevalence of SCC in Healthy and Symptomatic Populations
The first analysis separated studies based on the pretest probability of SCC (Figure 3). This was defined as
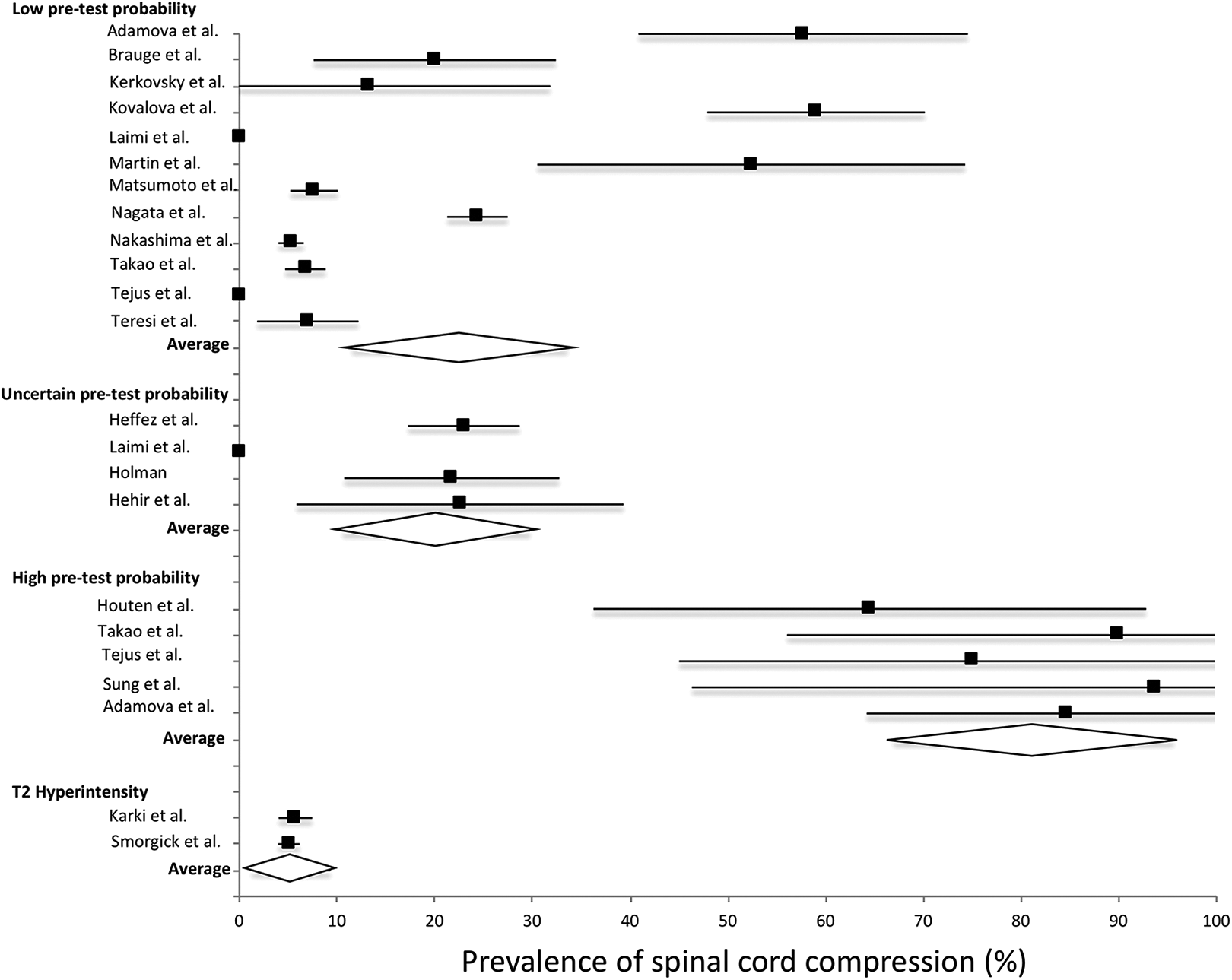
Subgroup analysis of spinal cord compression (SCC) prevalence based on the pretest probability of SCC. The low pretest group contained healthy individuals with no known symptoms or disease. The uncertain pretest probability group contained individuals with symptoms or diseases not thought to be directly related to degenerative cervical myelopathy (DCM). These included individuals suffering from dizziness, headaches, radiculopathy, or fibromyalgia. The high pretest probability group included individuals suffering from symptoms or signs of DCM (and hence SCC would be expected) or possible related diseases such as lumbar spinal stenosis and traumatic spinal cord injury without major fracture. This high pretest probability subgroup had a significantly higher prevalence of SCC than any other subgroup.
The
The
The
The
The
Prevalence of SCC and Age
The high heterogeneity and highly different characteristics in the
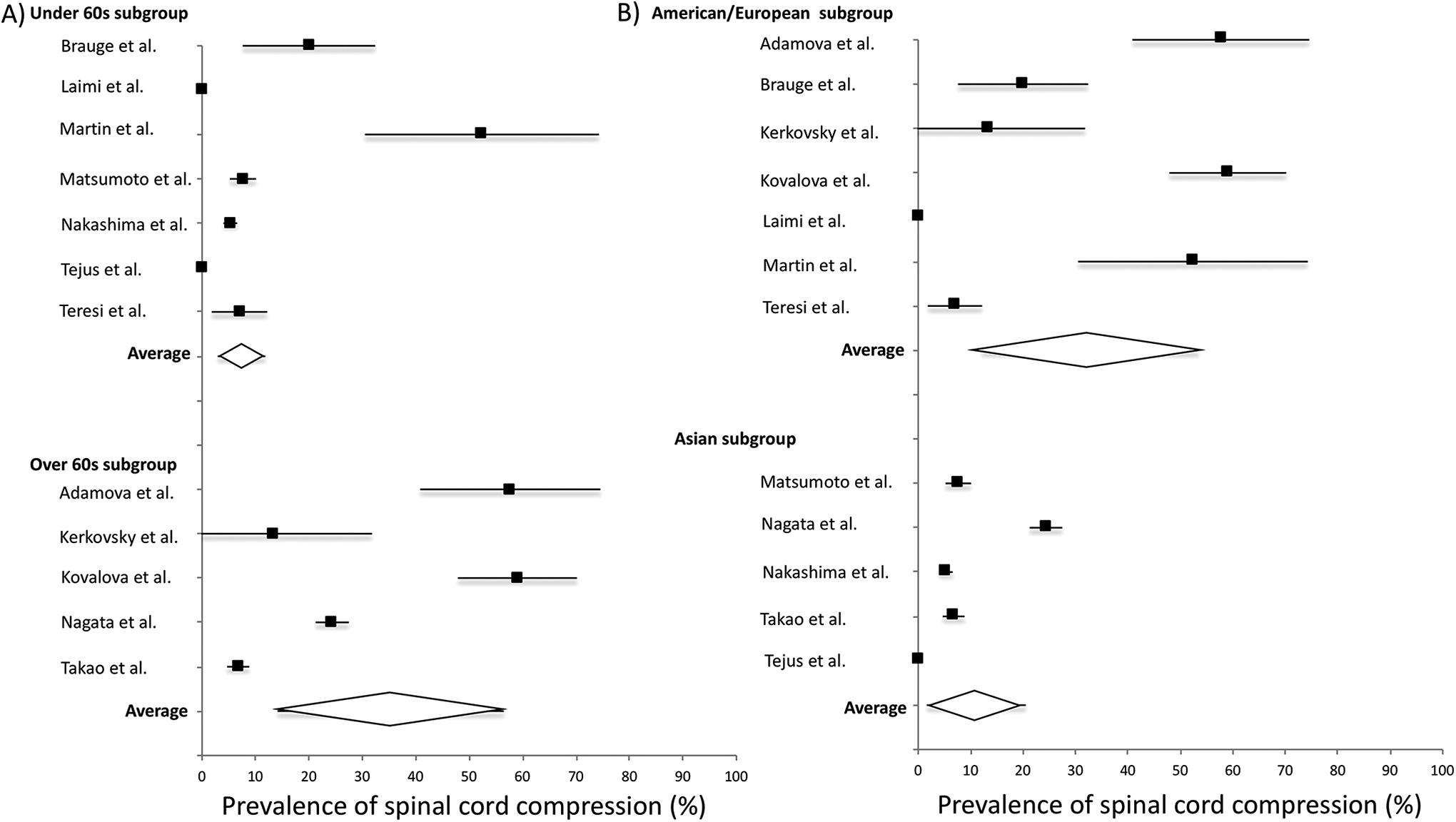
Subgroup analysis of the prevalence of spinal cord compression (SCC) in healthy participants separated by age and geographical location. (A) The studies included in the low pretest probability group in Figure 2 were divided based on a mean age of below or above 60 years. The over 60s subgroup had a significantly higher prevalence of SCC than the under 60s subgroup. (B) The studies included in the low pretest probability group in Figure 2 were divided based on the continent the study participants lived in. American/European participants had a significantly higher prevalence of SCCC compared with Asian populations.
Prevalence of SCC in European/American Populations
To assess the possible role of geographical location in SCC variation, we performed a new subgroup analysis on all the
Insufficient data existed for subgroup analysis based on gender. Subgroup analysis for the remaining subgroups identified no difference between subgroups.
Estimation of the Prevalence of DCM
Across the 19 studies, 6 provided data on the presence or absence of DCM. A total 2864 individuals were assessed for DCM symptoms or signs of whom 148 were diagnosed with DCM. The heterogeneity in methodology and target population precluded meta-analysis. The calculated I2 value was 90.3. Three of these studies recruited presumed healthy participants and three recruited individuals with signs of DCM who had not been diagnosed with any other disease. Sub-group analysis was performed to separate these groups into
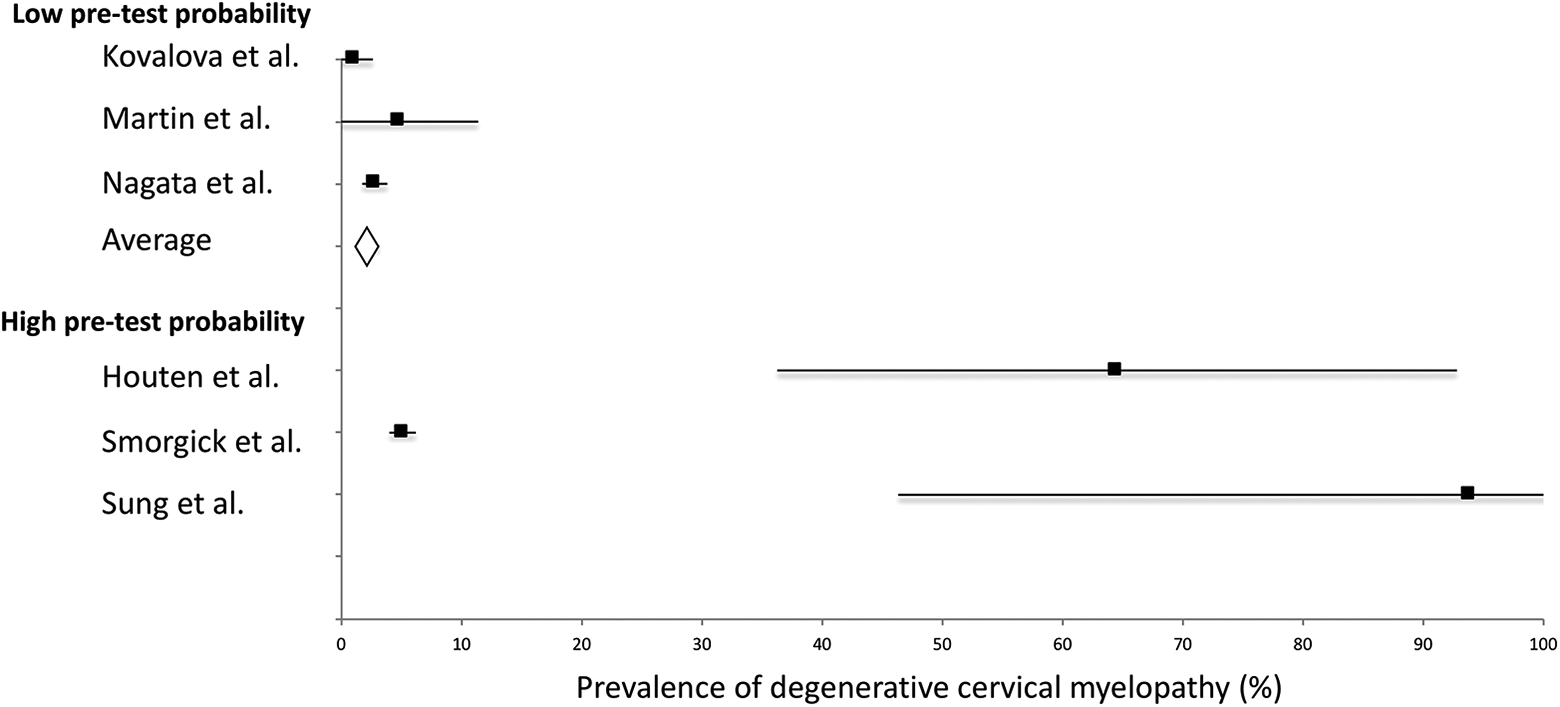
Subgroup analysis of the prevalence of degenerative cervical myelopathy (DCM) based on the pretest probability of DCM. The low pretest probability subgroup included individuals who entered their respective studies aware of no symptoms. The high pretest probability subgroup involved individuals who were recruited to the study because of their symptoms.
Risk of Bias Within Studies
The risk of bias in the included studies was low or moderate (Table 1). Thirteen studies had a moderate risk of bias and 6 studies had a low risk of bias.
Discussion
We present the first pooled estimates of the prevalence of SCC from the published literature. Overall, the included studies showed a wide range of prevalence, and therefore only sub-group meta-analysis was deemed possible. The pooled estimate of SCC in the healthy population was 24.2%. However, studies were characterized by high heterogeneity. Using author defined factors to investigate the potential sources of heterogeneity, we identified increasing age (>60 years: 35.3%), European/North American (39.7%) populations or populations with potential symptoms of myelopathy more likely to identify SCC (81.3%). No other investigated factors created subgroups with significantly different rates of SCC.
The investigation of populations with compatible symptoms of myelopathy would be expected to significantly increase the probability of identifying SCC. Nevertheless, the calculated prevalence of over 80%, and the low observed heterogeneity are striking, especially when considering the fact that the symptoms of myelopathy are numerous and overlap with many other related and also unrelated conditions. 26 In a series of patients presenting with nontraumatic tetraparesis only 24% had DCM, the leading differential, but this is far from 86%. 60
The high prevalence of DCM in the presence of myelopathic symptoms raises the following question: Why is DCM often missed as a diagnosis. A potential explanation is the underuse of MRI in patients presenting with symptoms that are compatible with DCM. This points toward the need for developing adequate screening procedures and triaging toward MRI assessment, such as in the case of acoustic neuroma. 61 The fact that healthy populations and populations with non-myelopathic symptoms had comparable levels of SCC was expected and suggests MRI use can be targeted effectively toward at-risk populations.
A separate
Excluding these symptomatic populations from further analysis, age and study location were found to influence the prevalence of SCC. The association of SCC with increasing age is expected as cervical spondylosis is the consequence of degenerative changes of the spine. 4 However, the association with location, and especially the fact that Asian populations had a much lower prevalence of SCC compared with American/European populations was somewhat unexpected and is to our knowledge the first time it has been reported in the literature. Interestingly, age was not a confounder in this association. Among the cohort with an average age of greater than 60 years, 2 studies were carried out in Europe (Czech Republic), recording mean SCC rates of 57.7% and 59% and the other 2 studies in Japan, with mean SCC rates of 24.3% and 6.8%, indicating a genetic difference between study populations. It is unclear why American/European populations have a higher prevalence of SCC; indeed Asian individuals are at greater risk of OPLL (ossification of the posterior longitudinal ligament), a condition that often leads to cervical myelopathy. 62 However, limitations of the currently available data prevent a more conclusive analysis.
A number of studies included in the present analysis identified patients with DCM. Using a fixed-effects model in 3 presumed healthy groups, we found a point prevalence of DCM of 2.3%; roughly 10% of individuals with SCC. The prevalence of DCM identified in the present study is therefore orders of magnitude higher than the operating prevalence of 1.6 per 100 000 previously estimated. 19 There are 2 interpretations of this discrepancy. First, that the prevalence reported in these 3 studies is erroneously high. However, the 3 studies on which this calculation was based received high-quality scores in our assessment and displayed low heterogeneity. The alternate possibility is that DCM is underdiagnosed and/or rarely treated surgically.
The notion that DCM is underdiagnosed is supported by a high incidence (18%) of unrecognized DCM in a small series of patients presenting with a neck of femur fracture. 63 Moreover, in the longest observational study of asymptomatic SCC by Bednarik et al, 6 the likelihood of developing DCM increased over time, with 8% developing DCM after 1 year and 22% with median follow-up of 44 months. 6,7 The high incidence of asymptomatic SCC reported in cross-sectional studies also indicates that the prevalence of DCM is higher than previously estimated.
Not all individuals with DCM require surgery. Recent international guidelines recommend surgery for moderate to severe, or progressive forms of DCM. Surgery can be offered to mild DCM, but structured nonoperative management is also an option. 12 However, our recent retrospective cohort study of patients at a single tertiary hospital found that prior to publication of the guidelines, only half of DCM patients who met the criteria for surgical decompression received surgery in routine practice. 17
There are several limitations to our study. A major limitation was the low number of studies identified; this is especially pertinent for estimating the prevalence of DCM, which was based on 6 articles included in the present analysis. We attempted to increase the number of studies by including populations that were not symptom free. It is possible that relevant information was missed as only a single database was searched for relevant studies (MEDLINE). However, hand-search of references of included articles yielded only 2 articles, suggesting our search strategy identified the majority of relevant studies. Moreover, as far as relevant original research is concerned, MEDLINE indexes and abstracts the ∼4000 journals, which constitute the core clinical literature of biomedicine. Indeed, MEDLINE alone achieves over 90% coverage in an analysis of over 120 systematic reviews. 64
A further problem in the literature is that there are relatively few large studies (>100 participants) containing original data. While our statistical analysis incorporated study size into its weighting of different studies, this problem with the literature is unavoidable.
A likely source of heterogeneity amongst studies was the varying definition of SCC used. Eight studies used subjective definitions, 5 studies used quantitative definitions, 2 based their definition on T2 hyperintensity, and in 4 studies the definition of SCC was unclear. Even within each subgroup highly different definitions were used; as a result, subgroup analysis could not identify significant differences. Furthermore, many of the studies included were of low quality. The average quality of included studies was 4.5/9, with a range of 2 to 8. While we attempted to rectify this by excluding the studies with a high risk of bias (which were also often low quality), poor and variable methodology may still have contributed to the heterogeneity we observed. The limitations discussed above mean that interpretation of the results should be taken with caution.
Conclusion
This study reports the first pooled estimates of the prevalence of SCC on MRI. We found that approximately 20% of healthy individuals (or individuals with unrelated symptoms) had SCC, and this rose to 86% in populations with myelopathic features. Increasing age and the study location also positively influenced prevalence. The pooled, point prevalence of DCM was 2.3%. However, these findings are based on limited data and further rigorous investigation is required.
Supplemental Material
Supplemental Material, supplementary_figures_sys_review - The Prevalence of Asymptomatic and Symptomatic Spinal Cord Compression on Magnetic Resonance Imaging: A Systematic Review and Meta-analysis
Supplemental Material, supplementary_figures_sys_review for The Prevalence of Asymptomatic and Symptomatic Spinal Cord Compression on Magnetic Resonance Imaging: A Systematic Review and Meta-analysis by Sam S. Smith, Max E. Stewart, Benjamin M. Davies and Mark R. N. Kotter in Global Spine Journal
Footnotes
Author Contributions
SS and BD designed the study. SS and MS collected data. SS performed statistical analysis. SS, BD and MK wrote and reviewed the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research in the senior author’s laboratory is supported by a core support grant from the Wellcome Trust and MRC to the Wellcome Trust-Medical Research Council Cambridge Stem Cell Institute. MRNK is supported by a National Institute for Health Research Clinician Scientist Award, CS-2015-15-023, supported by the National Institute for Health Research. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health and Social Care.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
