Abstract
Study design:
This is a retrospective chart review.
Objectives:
To identify the incidence of, and variables correlated with, femoral ring allograft (FRA) fracture following anterior lumbar interbody fusion (ALIF).
Methods:
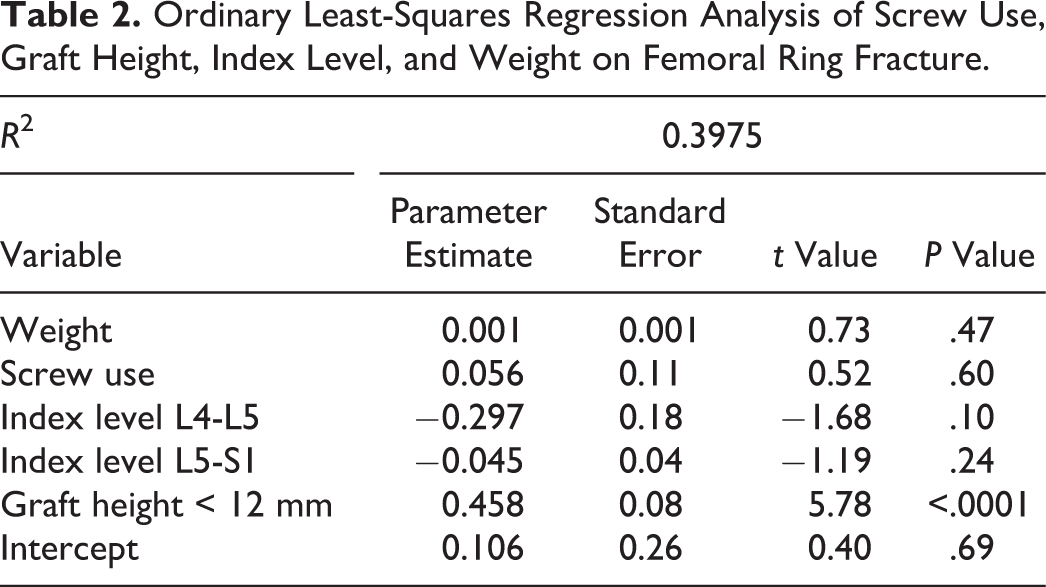
All patients who underwent ALIF using FRAs at an academic institution over 10 years were included. Postoperative radiographs were reviewed by both the primary and senior authors; fracture and no-fracture groups were created for comparison. Patient and surgical characteristics were extracted from electronic medical records. Frequency data comparisons were performed using contingency table analysis; comparisons of means were analyzed for continuous variables. A multivariate linear regression model was developed using screw use, graft height <12 mm, index level, and weight as variables.
Results:
A total of 76 FRAs in 59 patients were identified, 13 (17%) of which fractured. Age, sex, smoking status, use of buttress screws, weight, index level, and presence of spondylolisthesis were not correlated with incidence of fracture (P > .05). There was a significant correlation between the height of FRA and incidence of fracture; 2% (1/52) of grafts ≥12 mm and 50% (12/24) of grafts <12 mm fractured (P < .0001). Using ordinary least-squares regression, this result was independent of patient weight, use of screws, and index level. Of 10 patients, 9 did not require revision surgery to achieve fusion.
Conclusions:
Graft height was the only variable correlated with incidence of FRA fracture. Graft height <12 mm is an independent risk factor for FRA fracture in patients undergoing ALIF, and their use should be avoided in ALIF procedures.
Introduction
Anterior lumbar interbody fusion (ALIF) is a well-established method of achieving spinal arthrodesis. It is used for treatment of degenerative disc disease, lumbar scoliosis, and spondylolisthesis. 1 -3 The advantage of ALIF, as compared with posterior fusion, is that it facilitates restoration of lumbar lordosis and sagittal balance by restoring the height of the collapsed disc space via implantation of grafts and cages that are larger than those possible through posterior procedures. Used in conjunction with a posterior procedure, ALIF also facilitates successful arthrodesis by establishing greater stability at the lumbo-sacral junction, which has a propensity for pseudoarthrosis. 4 -9 When used for degenerative disc disease and associated stenosis, ALIF increases intervertebral height, indirectly enlarging the space of the neural foramina in the lumbar spine. 10 -12
A variety of implants and materials have been used as interbody devices for ALIF procedures. 13 Femoral ring allograft (FRA) is an effective and relatively cost-efficient choice for anterior interbody graft. 13 -15 It is often preferred over synthetic implants because of its osteoconductive properties. Acting as an interbody spacer, the allograft must resist compressive dynamic loads in excess of 8800 N and compressive static loads as high as 3400 N. 16 -19 Whereas some of these loads are shared in front-back constructs, the loads experienced by anterior allograft implants are substantial. 2,3,20
There are reports of FRA failures in both in vivo and in vitro settings, 14,19 which could potentially lead to collapse of the disc space and subsequent nonunion or malunion. Despite this observed phenomenon, the factors that influence FRA fracture incidence in ALIF procedures are unknown. The purpose of this study was to determine graft, patient, and surgical variables correlated with the incidence of fracture of FRA used for ALIF procedures. The working hypothesis was that the size of the patient, as approximated by weight, height, and body mass index (BMI), would be significantly correlated with a higher incidence of fractured grafts.
Methods
Institutional review board approval was obtained for this single-center, multisurgeon retrospective review of patient charts and corresponding images. Case logs of 2 fellowship-trained academic spine surgeons were queried using CPT code 22 851 (Application of biomechanical device to vertebral defect or interspace) between September 2006 and September 2016. Patient charts identified in this initial query were reviewed to confirm application of a FRA for an ALIF procedure. This process ultimately yielded 59 cases for detailed review of medical records and postoperative images that were included in the final analysis.
All patients had a direct anterior approach to the lumbar spine for anterior fusion and insertion of the FRA, followed by posterior fusion and instrumentation using a pedicle screw system. All patients had follow-up X-rays at 6 weeks, 3 months, 6 months, and 1 year. Computed tomography (CT) scans were ordered for patients who reported increased pain in order to look for graft or hardware failure or misplacement.
Patient demographic, perioperative, and postoperative information were reviewed. Age, sex, height, weight, and need for additional surgery at the index level were recorded from the patient’s electronic medical record. The use of anterior buttress screws, the height of the allograft, the presence of spondylolisthesis at index level, and the level of fusion were recorded from the patients’ operative reports.
Postoperative radiographs and CT scans were reviewed by both a senior staff spine surgeon and an orthopaedic resident. Each graft evaluated was then grouped into a “fracture” group or a “no fracture” group based on review of the images. These 2 groups were then compared to identify characteristics correlated with the incidence of fracture of the FRA used for ALIF.
Statistical analysis was performed using SAS (Cary, NC) and Microsoft Excel (Redmond, WA). Mean values and SDs were calculated for each group. P values were calculated using the Student t-test for age, height, weight, BMI, and graft height. Preliminary review of the data indicated that there was an obvious difference in incidence of fracture between those with grafts <12 mm or ≥12 mm in height. Thus, P values were calculated for sex, spondylolisthesis at index level, index level (L3-L4, L4-L5, L5-S1), screw use, and graft height less than 12 mm using contingency table analysis and the Fisher exact test (Table 1). Significance was set at the .05 level. A multivariate linear regression model was developed using screw use, graft height less than 12 mm, index level, and weight to control for potential confounding (Table 2).
Demographic and Injury-Related Characteristics of Femoral Ring Allograft Participants.a
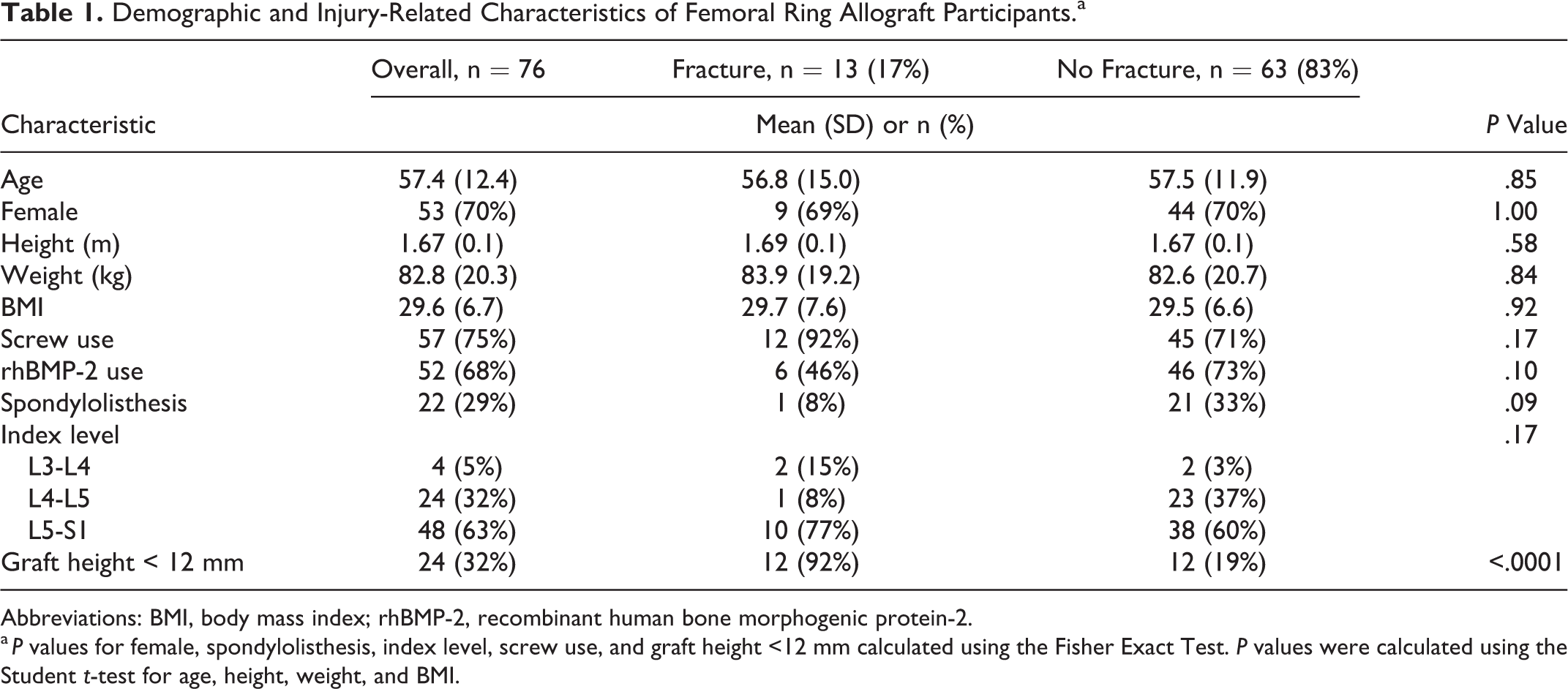
Abbreviations: BMI, body mass index; rhBMP-2, recombinant human bone morphogenic protein-2.
a P values for female, spondylolisthesis, index level, screw use, and graft height <12 mm calculated using the Fisher Exact Test. P values were calculated using the Student t-test for age, height, weight, and BMI.
Ordinary Least-Squares Regression Analysis of Screw Use, Graft Height, Index Level, and Weight on Femoral Ring Fracture.
Results
The identified cohort of 59 patients included 76 FRAs, 13 (17%) of which fractured. All patients had posterior instrumentation in conjunction with ALIF. The range of time to graft failure was 2 days to 224 days, with a mean and median time to failure of 78 and 43 days, respectively.
Age, sex, patient size, and use of screws were not correlated with incidence of fracture (Table 1). Our original hypothesis of patient size as a factor influencing incidence of fracture was not supported when analyzed with weight and BMI as continuous variables, nor when analyzed with weight and BMI as frequency distributions based on quartiles (Figure 1).
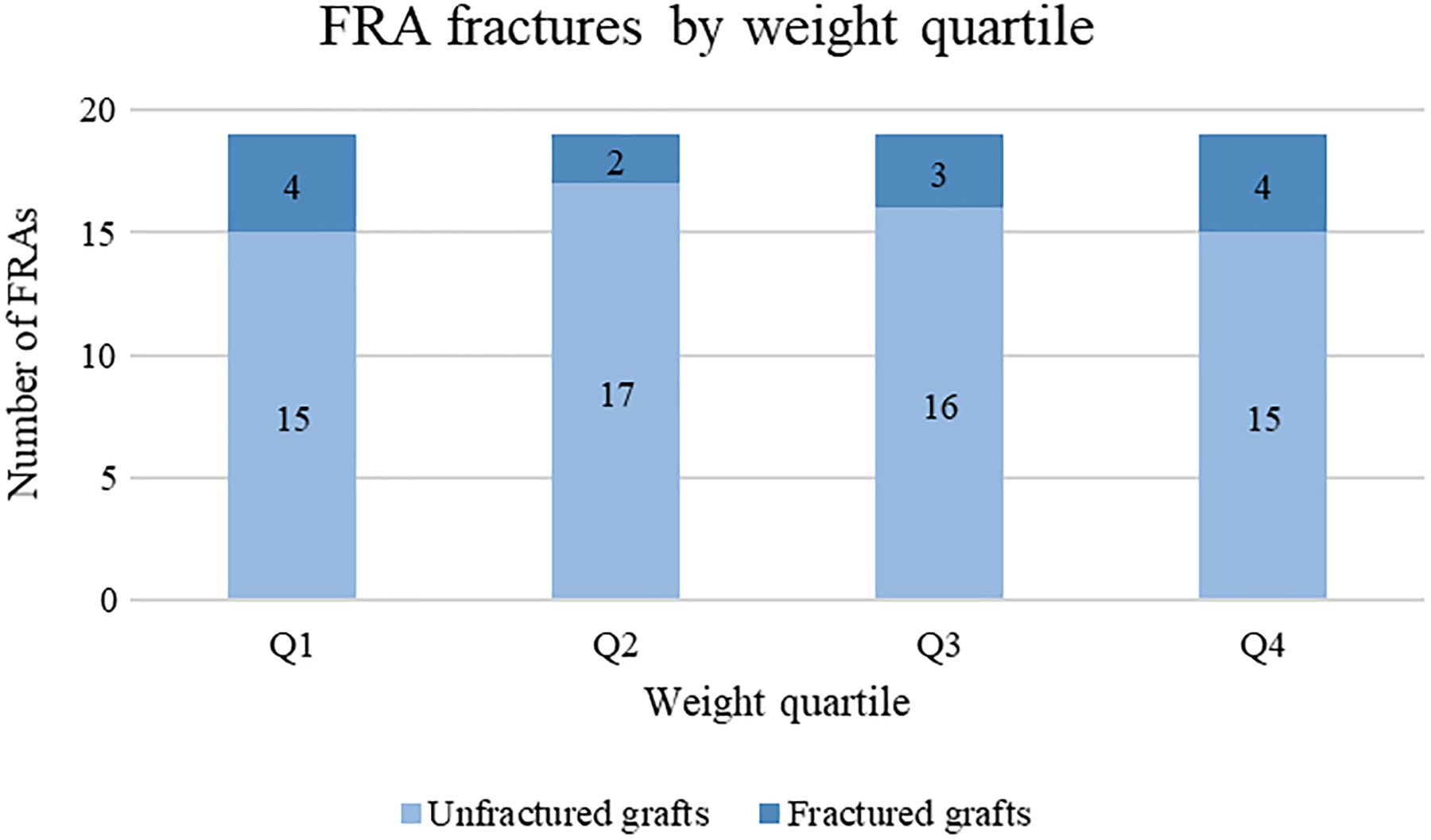
Numbers of femoral ring allografts (FRAs) fractured and unfractured by patient weight quartile.
Of the 76 total grafts, 22 (29%) were placed at a level with spondylolisthesis (Table 1). Only 1 of the 22 (5%) fractured as compared with 12 of the 54 grafts placed at levels without spondylolisthesis (22%, P = .09; Table 1). Based on the placement of grafts, 2 of 4 grafts placed at L3-L4 (50%) fractured, 1 of 24 grafts placed at L4-L5 (4%) fractured, and 10 of 48 grafts placed at L5-S1 (21%) fractured (Table 1). The difference in index level of grafts was not statistically significant (P = .17).
There was an increase in incidence of graft fractures in grafts less than 12 mm in height (Table 1): 50% of grafts <12 mm fractured (12 of 24), as compared with 2% of grafts ≥12 mm (1 of 52, P < .0001). Using ordinary least-squares regression analysis, this result was found to be independent of patient weight, use of screws, and index vertebral level (Table 2).
Of the 13 levels, 12 (92%) included in the fracture cohort had an anterior buttress screw. A detailed review of radiographs in this cohort revealed that the graft fracture line extended from the area of contact with the screw in each of these 12 fractured grafts (Figures 2 and 3). However, 71% of the nonfractured cohort also had screws inserted, so we could not demonstrate that the use of screws led to an increased incidence of graft fracture (P = .17). Within our entire study population, only 19 levels (25%) did not have an anterior buttress screw placed. Only 1 of these levels (5%) had a fractured graft.
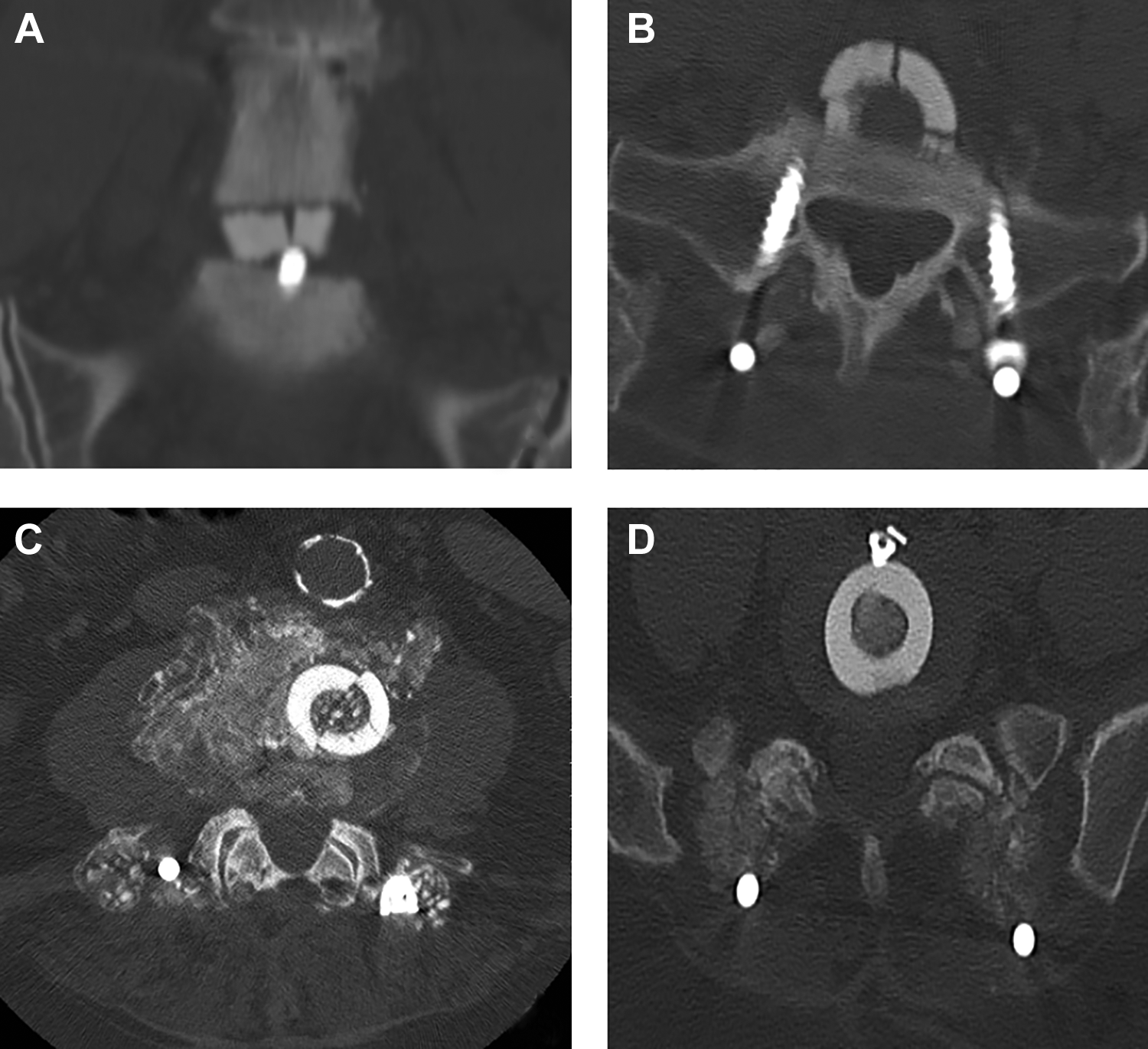
Computed tomography scan examples of patients with femoral ring allografts for anterior lumbar interbody fusion. Fracture of graft at L5-S1 with the fracture line touching the contact point of the graft and screw (A and B). Fracture of the graft at L4-5 without an anterior buttress screw (C). Femoral ring graft at L5-S1 with anterior buttress screw and no fracture (D).
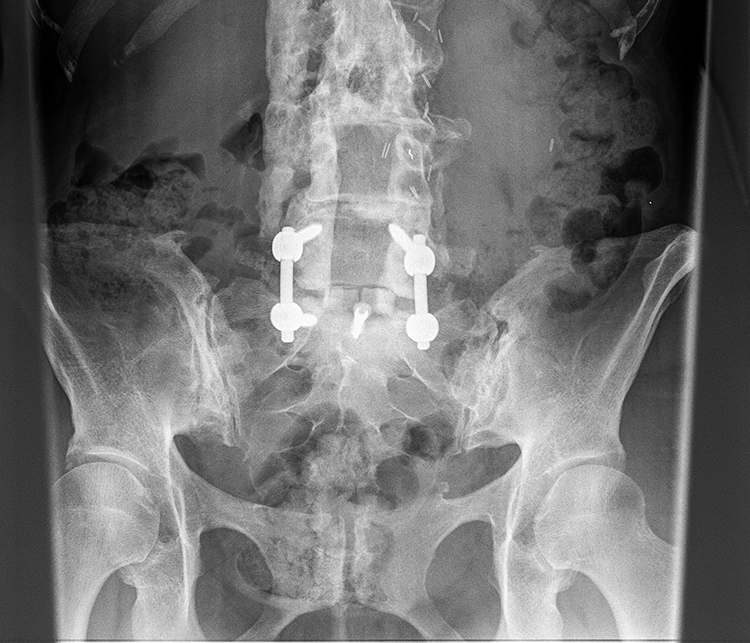
X-ray example of patient with a fractured femoral ring allograft and fracture line that touches the contact point of the graft and screw.
Recombinant human bone morphogenic protein-2 (rhBMP-2) was used in 52 of the 76 levels where FRAs were placed. rhBMP-2 was used in 46 of the 63 nonfractured levels (73%) and 6 of the 13 fractured levels (46%, P = .10; Table 1).
Only 1 of 10 patients required surgery because of their fractured FRA. This patient had a fracture of the graft as well as loss of lordosis. A 15° PEEK cage was used to replace the fractured FRA and restore lumbar lordosis.
Discussion
We evaluated correlations between the incidence of FRA fracture when used for ALIF procedures and intrinsic graft characteristics, patient characteristics, and surgical factors. It was hypothesized that increases in patient weight would increase the incidence of FRA fracture, but this hypothesis was not supported in our study cohort. Instead, graft height, an intrinsic property of the graft itself, was an isolated risk factor for fracture. The difference in fracture rate between grafts <12 mm in height and grafts ≥12 mm in height was 25-fold.
Previous studies have shown that FRAs are more cost-efficient interbody devices as compared with engineered synthetic cages. 14,21,22 Other studies have shown lower fusion rates, higher graft subsidence, and a natural history of graft resorption when FRAs are used for ALIF; none have shown a difference in clinical outcomes between FRAs and engineered synthetic cages. 14,23,24 Other intrinsic characteristics of the graft that could potentially influence rate of fracture include cortical thickness, cortical density, and age of the donor. These factors have not been studied in vivo. Unfortunately, this information was not available for our study. Hart et al 19 performed a biomechanical study evaluating FRA characteristics and load to failure using a cadaveric model. Using FRAs 20 mm in height, cortical wall thickness, but not ring diameter or age of the cadaveric donor, was significantly correlated with increased load to failure. Their model demonstrated that a difference in thickness of approximately 2 mm accounted for a nearly 5× increase in load to failure. 19 Because the study by Hart et al kept the FRA height constant, we cannot extrapolate what effect a change in height would have had on their graft fracture rate. It is possible that shorter grafts might be more prone to failure than taller grafts because of crack propagation that can extend through the entire height of the graft. However, we do not have a clear explanation as to the mechanism by which a shorter graft would be subjected to a higher fracture rate. It can be surmised that the decreased cortical surface area of a FRA, because of decreased cortical thickness as shown in the study by Hart et al or decreased cortical height as shown in this study cohort, results in higher load per millimeter cubed experienced by the graft 2 and, thus, less overall load tolerated until failure. Alternatively, it is possible that grafts with less height might be more prone to failure than grafts with more height because of crack propagation that can extend through the entire height of the graft.
Although it can be theorized that spondylolisthesis would increase the incidence of graft fracture through increased instability, this hypothesis was not found to be true. In fact, there was an observed trend toward a protective effect of spondylolisthesis on the incidence of fracture in the FRA (P = .09). However, the study was underpowered to statistically support this finding. Similarly, it has been suggested that the use of rhBMP-2 may structurally weaken FRAs in standalone ALIF procedures and, thus, lead to failure. 25 Although all our ALIFs were secured with posterior instrumentation, this hypothesis was also unsupported. We found a trend toward a protective effect of rhBMP-2 use (P = .10). Another trend noted was that more grafts failed at the L5-S1 level than at the L4-5 level, although this result was also not supported statistically.
Analysis of the fracture group’s radiographic images showed that nearly all fracture lines extended from the location where the anterior buttress screw contacted the graft. This may suggest that the anterior buttress screw creates a focal area of increased contact force on an already stressed graft, or an area of notching on the graft caused by the screw, potentially contributing to the graft’s failure. However, further study with increased numbers is necessary to better evaluate these variables in vivo, or a detailed biomechanical study should be done to test these hypotheses in vitro. This study demonstrated that the height of the FRA, rather than characteristics of the patients into whom they are placed, is correlated with incidence of fracture. Although graft height for ALIF is often determined by the anatomy of the patient and the distractibility of the disc space, this data suggests that the surgeon should make a concerted effort to place a 12 mm or larger graft in the interbody space when using FRA for ALIF. Additional variables such as force required for disc distraction and graft placement or changes in disc height and segmental lordosis could help determine additional risk factors for graft fracture and are ripe for further study. Another limitation of this study is that its results cannot be easily extrapolated to synthetic grafts because the material used in these implants is anisotropic and noncrystalline in nature.
Although radiographic findings in patients with a fractured graft can be quite concerning, we found no absolute reason to revise the construct because there was no neurological damage, and in time, patients fused and became asymptomatic.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
