Abstract
Study Design:
Systematic literature review.
Objective:
Our primary objective was to compare reported fusion rates after anterior cervical discectomy and fusion (ACDF) using structural allograft versus polyetheretherketone (PEEK) interbody devices in patients with cervical spine degeneration. Our secondary objectives were to compare differences in rates of subsidence and reoperation and in patient-reported outcomes between the 2 groups.
Methods:
Through a systematic review of the English-language literature using various databases, we identified 4702 articles. After we applied inclusion and exclusion criteria, 14 articles (7 randomized controlled trials, 4 prospective studies, and 3 retrospective studies) reporting fusion rates of structural allograft or PEEK interbody devices were eligible for our analysis. No randomized controlled trials compared outcomes of structural allograft versus PEEK interbody devices. Extracted data included authors, study years, study designs, sample sizes, patient ages, duration of follow-up, types of interbody devices used, fusion rates, definition of fusion, reoperation rates, subsidence rates, and patient-reported outcomes.
Results:
Fusion rates were 82% to 100% for allograft and 88% to 98% for PEEK interbody devices. The reported data were insufficient to perform meta-analysis. Structural allograft had the highest reported rate of reoperation (14%), and PEEK interbody devices had the highest reported subsidence rate (18%). Patient-reported outcomes improved in both groups. There was insufficient high-quality evidence to compare the associations of various PEEK modifications with fusion rates.
Conclusion:
Fusion rates were similar between structural allograft and PEEK interbody devices when used for ACDF for cervical spine degeneration. Currently, there is insufficient high-quality evidence to assess associations of PEEK modifications with fusion rates.
Level of Evidence:
II.
Keywords
Introduction
Anterior cervical discectomy and fusion (ACDF) is a common surgical treatment for cervical spine degeneration and associated cervical nerve root or spinal cord compression. 1,2 Indications for ACDF include cervical radiculopathy or myelopathy secondary to degenerative disc disease, disc herniation, spondylosis, and spinal stenosis. Interbody devices used in ACDF facilitate fusion, correct kyphosis, and restore foraminal height. 3,4 When ACDF was initially described by Cloward 5 in 1958, structural autograft bone was used for interbody support. However, during the past several decades, various alternatives to structural autograft have gained popularity, including structural cadaveric allograft, polyetheretherketone (PEEK) interbody devices, and metal interbody devices. 6
A 2017 survey of AOSpine members found that PEEK interbody devices were commonly used world-wide and structural allografts were commonly used in North America. 7 PEEK has multiple theoretical advantages, including biocompatibility, radiolucency, favorable elasticity, and ease of procurement and storage. 8 -10 The major drawback of PEEK is that it is bio-inert and limits host bone integration. 11 Various strategies have been proposed to improve the biologic integration of PEEK, including augmenting PEEK devices with graft extenders, such as demineralized bone matrix or cellular products, as well as modifications to the PEEK device. 12,13 Proposed modifications include surface enhancements, such as increased surface porosity or titanium coating, and impregnation of PEEK with bioactive materials such as hydroxyapatite. 14 The effects of these modifications on fusion success and clinical outcomes are unclear.
Structural allograft, including cortical allograft, corticocancellous or composite grafts, and dense cancellous grafts, is an effective alternative to autograft to achieve bony fusion in ACDF. 15,16 Among surgeons who use allograft, cortical and corticocancellous composite grafts are more popular because they provide additional structural support and resistance against subsidence, 17 whereas dense cancellous grafts have been reported to have high rates of resorption. 18
Our primary objective was to compare reported fusion rates after ACDF using structural allograft versus PEEK interbody devices in patients with cervical spine degeneration. Our secondary objectives were to compare differences in the rates of subsidence and reoperation and in patient-reported outcomes between the 2 groups.
Methods
Our study used publicly available information and thus was not subject to review by our institutional review board.
Search Strategy
We designed our literature search in accordance with the Preferred Reporting Items of Systematic Reviews and Meta-Analyses guidelines. 19 With the assistance of a clinical informationist, we performed a comprehensive literature search using PubMed, Embase, The Cochrane Library, Web of Science, Scopus, ClinicalTrials.gov, and CINAHL Plus databases in October 2018 (see Appendix 1 in the online version of the article). We identified 4702 articles (Figure 1).
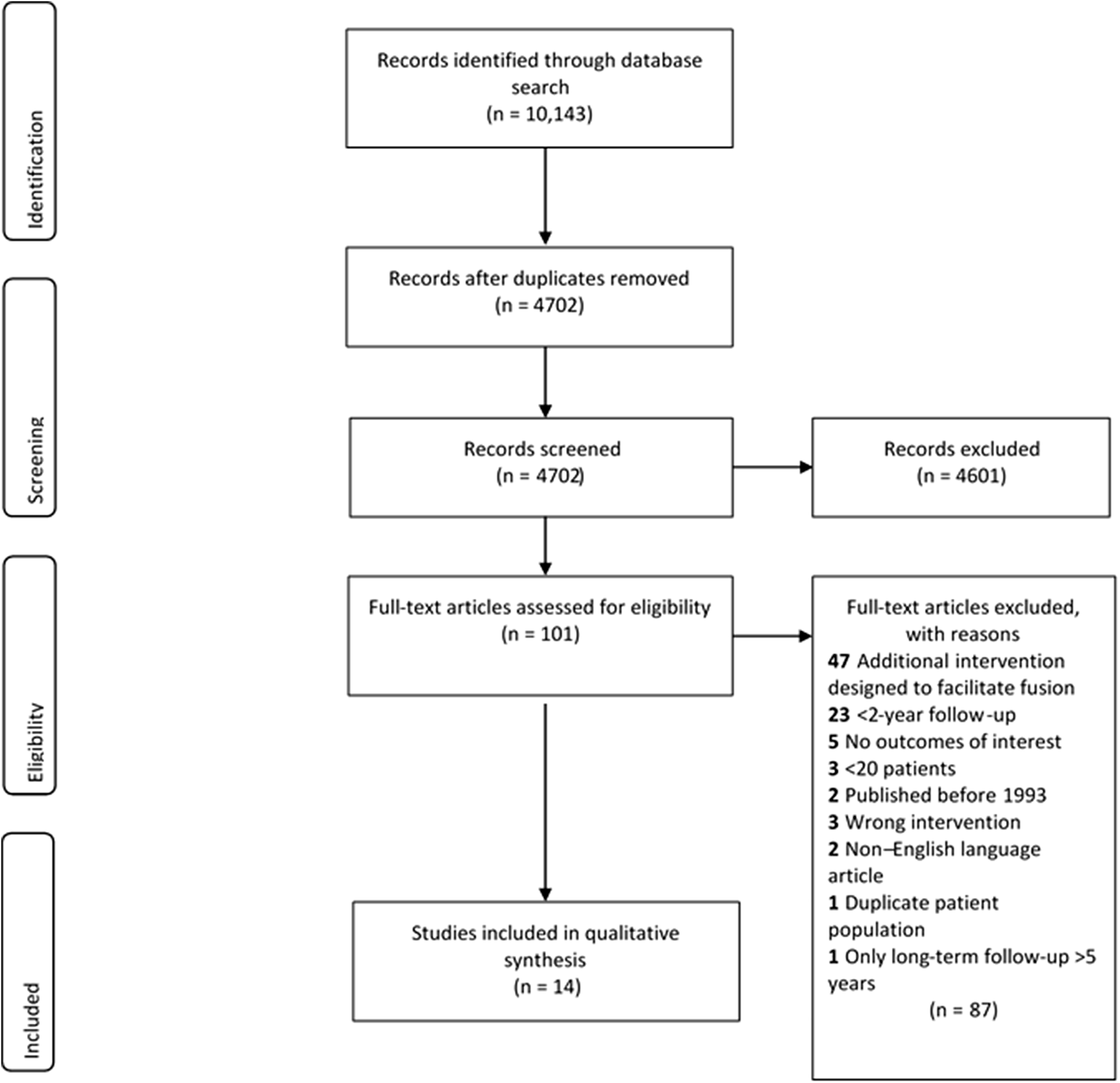
Literature search performed using PubMed, Embase, The Cochrane Library, Web of Science, Scopus, ClinicalTrials.gov, and CINAHL Plus databases in October 2018. The purpose was to compare the fusion rates of structural allograft versus polyetheretherketone interbody devices in patients who underwent anterior cervical discectomy and fusion for cervical spine degeneration.
Inclusion and Exclusion Criteria
We included studies that reported results of ACDF using structural allograft, PEEK interbody devices, or modified PEEK interbody devices; analyzed patients aged ≥18 years; included results of >20 patients; were published in 1993 or later; were written in English; were randomized controlled trials or retrospective, prospective, or observational cohort studies; and reported 2-year radiographic follow-up. (For comparative studies in which only 1 of the study groups met the eligibility criteria, we limited our data abstraction and analysis to that group.)
We excluded studies that reported on ACDF using zero-profile or stand-alone devices; ACDF performed without anterior cervical plating; cervical corpectomy; circumferential (360° or 540°) fusion; interventions to facilitate fusion in addition to the interbody device (eg, bone morphogenic protein, stem cells, autograft, platelets); ACDF for nondegenerative conditions; ACDF for adjacent-segment disease (adjacent to previous ACDF); or revision ACDF for nonunion or other conditions. We also excluded animal studies, cadaveric studies, case reports, correspondence, letters to the editor, technical notes, abstracts, and poster presentations. Level of evidence of the articles included in this systematic review was assessed using the Oxford Centre for Evidence-Based Medicine. 20
Study Selection
Two independent reviewers screened the studies for eligibility. A third reviewer served as an arbitrator when there was disagreement between the 2 primary reviewers. Each article’s title and abstract were reviewed. Articles that could not be excluded on the basis of this review were retrieved for full-text review (n = 101). Covidence Systematic Review Software (Veritas Health Innovation, Melbourne, Victoria, Australia) was used to facilitate the screening and selection process. Fourteen articles (7 randomized controlled trials, 4 prospective studies, and 3 retrospective studies) met our eligibility criteria and were included in the review (Table 1).
Studies of Fusion Rates in Patients Who Underwent Anterior Cervical Discectomy and Fusion for Cervical Spine Degeneration.
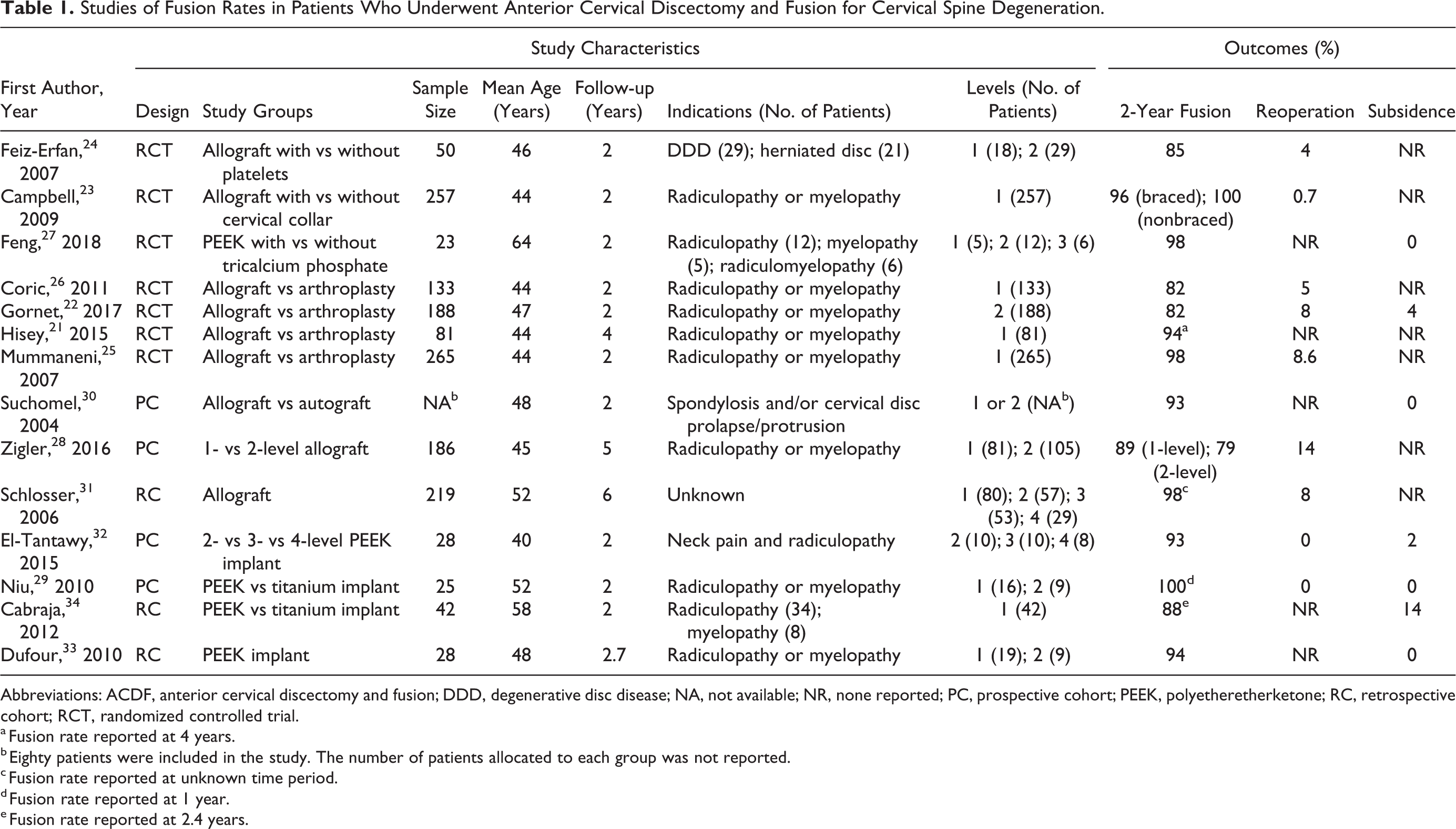
Abbreviations: ACDF, anterior cervical discectomy and fusion; DDD, degenerative disc disease; NA, not available; NR, none reported; PC, prospective cohort; PEEK, polyetheretherketone; RC, retrospective cohort; RCT, randomized controlled trial.
a Fusion rate reported at 4 years.
b Eighty patients were included in the study. The number of patients allocated to each group was not reported.
c Fusion rate reported at unknown time period.
d Fusion rate reported at 1 year.
e Fusion rate reported at 2.4 years.
Data Extraction and Analysis
Using a standardized data extraction form, we extracted the following information: authors, study years, study designs, sample sizes, patient ages, duration of follow-up, types of interbody device used, fusion rates, definitions of fusion, reoperation rates, subsidence rates, and patient-reported outcomes (ie, visual analogue scale [VAS] for pain, Neck Disability Index [NDI], Patient-Reported Outcomes Measurement Information System, and Odom’s criteria). Descriptive statistics were calculated and reported as appropriate. There were insufficient data to perform a meta-analysis.
Results
Fusion Rates
In 11 studies, fusion was assessed by an independent or blinded physician. 21 -31 Criteria for fusion success differed slightly among studies, although motion (commonly defined as <2° or <4° change in angulation between the spinous processes) on flexion-extension radiographs is the commonly used method to determine fusion success (Table 2).
Definitions of Fusion in Studies of Structural Allograft and PEEK Interbody Devices in Patients With Cervical Spine Degeneration Who Underwent Anterior Cervical Discectomy and Fusion.
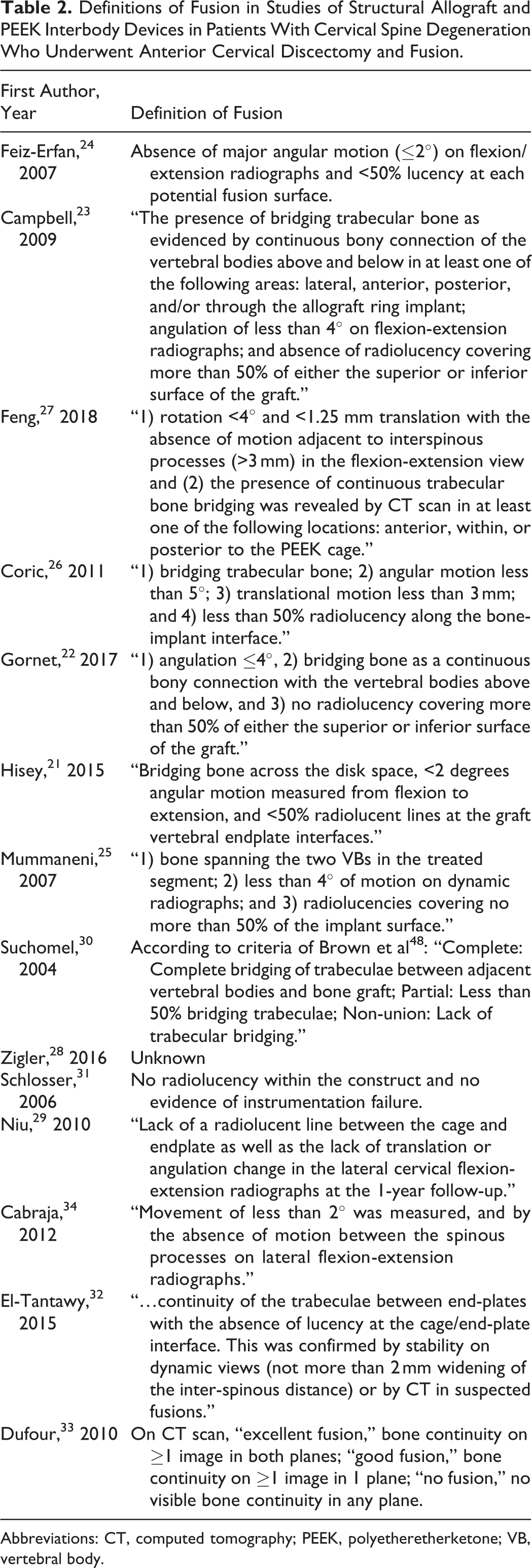
Abbreviations: CT, computed tomography; PEEK, polyetheretherketone; VB, vertebral body.
Structural Allograft
Nine studies (1354 patients) reported fusion data with the use of structural allograft: 5 used corticocancellous allograft, 21,24,26,28,31 2 used unspecified cortical allograft, 22,25 1 used fibular allograft, 30 and 1 did not report the type of allograft used. 23 The fusion rates among these 9 studies were 82% to 100%. Among the 5 studies that used corticocancellous allograft, fusion rates at 2 years or more were 82% to 97% (Table 1).
A prospective comparison of 1-level and 2-level ACDF using corticocancellous allograft reported fusion rates of 89% and 79%, respectively, at 2 years, and 93% and 86%, respectively, at 5 years. 28 The reported fusion rates for unspecified cortical grafts at 2 years were 82% 22 to 97%. 25 In a prospective study, Suchomel et al 30 assessed fusion rates of fibular allograft versus structural autograft and found that the fibular allograft group had fusion rates of 85.5% at 1 year and 93.4% at 2 years.
PEEK Interbody Devices
Five studies using PEEK interbody devices (146 patients) were included in our final analysis, including 1 randomized controlled trial, 27 2 prospective studies, 29,32 and 2 retrospective studies. 33,34 Rates of fusion for PEEK interbody devices in these 5 studies were 88% to 98%.
In a prospective study by El-Tantawy, 32 10 patients underwent 2-level fusion, 10 underwent 3-level fusion, and 8 underwent 4-level fusion with stand-alone PEEK cage. Fusion was achieved at a mean of 14.9 weeks for 2-level, 15.2 weeks for 3-level, and 16.1 weeks for 4-level fusion, with no significant differences. The authors reported a fusion rate of 93% at 2-year follow-up (Table 2). Dufour et al 33 reported a fusion rate of 94% for 28 PEEK patients at a mean follow-up of 32 months.
Subsidence Rates
Structural Allograft
Among the studies of structural allograft, only 2 reported subsidence rates. Gornet et al 22 reported the highest subsidence rate (4.3%, 6/138 patients) at 2-year follow-up. In the study by Suchomel et al, 30 none of the patients developed subsidence at 2-year follow-up.
PEEK Interbody Devices
High subsidence rates were reported in the study of PEEK interbody devices by El-Tantawy, 32 occurring at 7 fusion levels in 5 patients (18%); however, no patient underwent revision surgery. Cabraja et al 34 reported a subsidence rate of 14% (6/42), with no reoperations. The remaining articles discussing PEEK fusion rates did not report any subsidence among their patients. 27,29,33
Reoperation Rates
Structural Allograft
In studies using structural allograft interbody, the incidence of reoperation for all reasons was 0.7% (1/149 patients) to 13.9% (26/186 patients) (Table 1). 23,28 Overall, the rates of reoperation for pseudarthrosis with structural allograft interbody were 1.8% to 6.4%. 28,31
PEEK Interbody Devices
Among the 5 studies involving PEEK devices, 2 reported no reoperations 29,32 and 3 did not monitor for reoperation 27,33,34 (Table 1).
Patient-Reported Outcomes
Eleven studies reported patient-reported outcomes, with VAS and NDI being the most frequently used measures. 21 -29,32,34
Structural Allograft
Seven studies described patient-reported outcomes, with an overall significant improvement in clinical outcomes assessed by VAS and NDI scores. 21 -26,28 Feiz-Erfan et al 24 reported subjective improvements overall. In VAS scores ranging from 3 to 30, the mean VAS score at baseline for all patients was 15.5; at 2-year follow-up the mean score had improved to 7.0. 24 Similarly, Coric et al 26 showed a >20% improvement in NDI scores at 2-year follow-up. The methods used for assessing patient-reported outcomes were heterogeneous, limiting quantitative analysis of association of allograft interbody with clinical outcomes.
PEEK Interbody Devices
Overall outcomes improved among the 4 studies that reported patient-reported outcomes. 27,29,32,34 The success rate of surgery as measured by Odom’s criteria was 64% in the study by Cabraja et al. 34 In the randomized controlled trial comparing an empty PEEK cage with PEEK packed with β-tricalcium phosphate, patients in both groups showed improvements in VAS, NDI, and Japanese Orthopaedic Association scores. 27
Discussion
We conducted a systematic review of the literature to compare fusion rates of structural allograft versus PEEK interbody devices in patients with cervical spine degeneration who underwent ACDF. Of the 14 studies included in our review, the overall fusion rates were 82% to 100% 21 -26,28,30,31 for structural allograft and 88% to 98% 27,29,32 -34 for PEEK interbody devices. The lack of randomized controlled trials comparing these 2 interbody devices, in addition to the variability in methodology to assess fusion, sample sizes, and other methods among studies, precluded meta-analysis.
Because fusion is a key determinant of success after ACDF, 35 a standard for determining fusion rate is needed in cervical spine fusion studies. Several widely varying criteria for determining radiographic fusion were reported (Table 2). The most common components of the definitions included <50% radiolucency along the bone-implant surface and a specified degree of angular motion on dynamic radiographs (<2° or <4°). Most studies classified fusion as a binary variable; however, some studies classified fusion as “complete,” “partial,” and “nonunion” 30 or “excellent,” “good,” and “no fusion.” 33 Most studies used multiple observers to determine radiographic fusion, and none of the 14 studies reported intraobserver differences in determination of fusion. In 2008, it was presumed that a consensus definition of cervical fusion was beginning to form. 36 In a recently published systematic review by Oshina et al, 37 10 criteria were described for fusion assessment in ACDF. Most commonly, the surgeon’s subjective assessment of bridging trabecular bone between endplates and the absence of a radiolucent gap were used to assess fusion. The problem of heterogeneity in fusion assessment is commonly described in systematic reviews and meta-analyses reporting fusion rates in ACDF. 38 -40 A universally accepted criterion for fusion assessment is needed to standardize studies and allow for comparison. 36
Patient-reported outcomes were reported in only 10 of the 14 studies, indicating that patient-reported outcomes are often not the focus of studies that describe interbody devices, even among those with 2-year follow-up. In the studies that described patient-reported outcomes, the results were not always directly comparable, further illustrating the need for reporting and standardizing outcomes in cervical spine surgery studies. A recent review by Nayak et al 41 also acknowledged that the numerous patient-reported outcome measures and the variability in minimal clinically important differences between these measures limit comparisons of clinical outcomes in spine surgery research.
We required minimum radiographic follow-up of 2 years for inclusion in our review (Table 3). The 1 high-quality study excluded because of a lack of 2-year radiographic follow-up was the PIERCE-PEEK Study (Prospective International Multicenter Evaluation of Radiological and Clinical Effects of Stand-Alone PEEK Intervertebral Spacers for ACDF) by Suess et al. 9 The authors followed a cohort of 356 patients who underwent ACDF with PEEK interbody devices without any osteopromotive fillers and without additional anterior instrumentation. Complete radiographic fusion occurred for 43% of patients at 6 months, 73% of patients at 12 months, and 83% of patients at 18 months. The authors recommended against using PEEK interbody devices alone without fillers because of delayed fusion. It is unclear whether delayed fusion was the result of a lack of osteopromotive fillers or a lack of anterior instrumentation to provide additional stabilization to support fusion.
Power Calculation, Sample Size, and Reporting of Patients Lost to Follow-up.
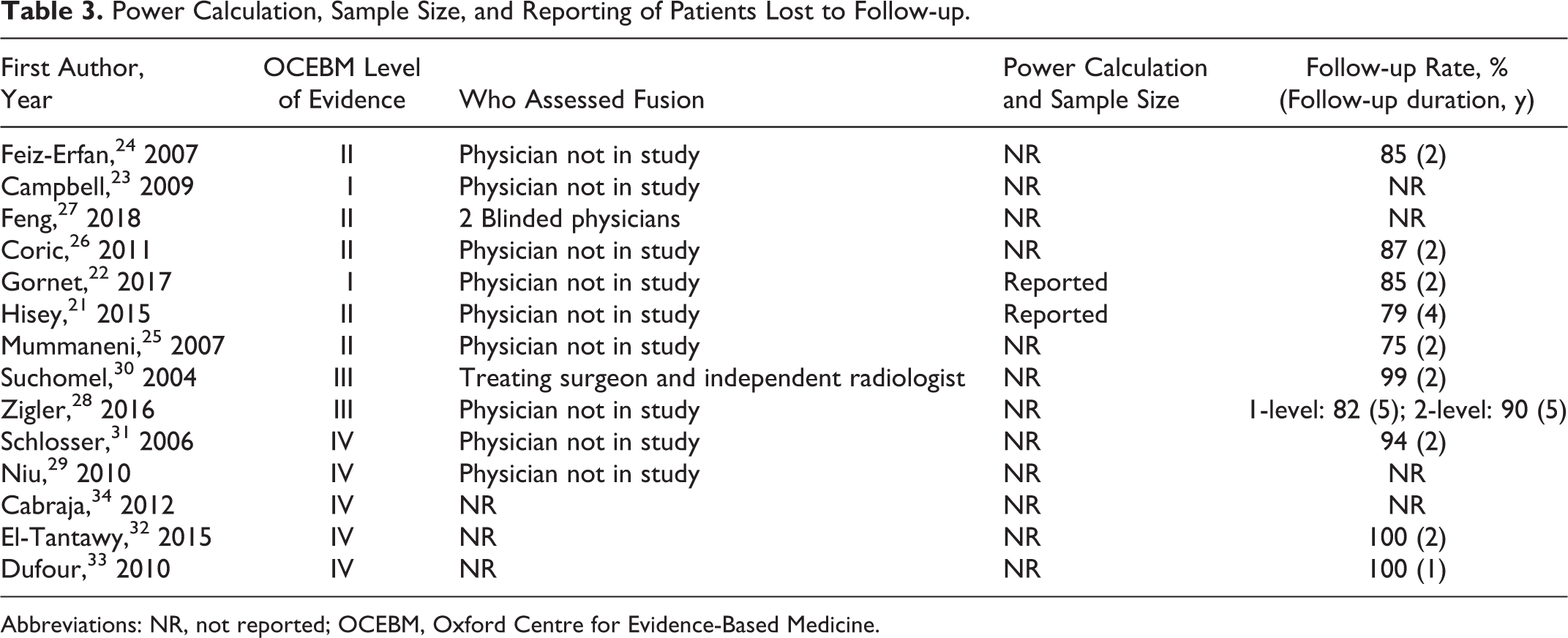
Abbreviations: NR, not reported; OCEBM, Oxford Centre for Evidence-Based Medicine.
The use of bone graft extenders or biologic materials in addition to PEEK cages has been reported and advocated to improve fusion success. 42 However, the reported fusion rates with materials such as hydroxyapatite are approximately 85%, which is similar to the rate achieved with structural allograft or PEEK alone. 38 Future studies examining the influence of interbody devices on fusion rates should adjust for use of bone graft extender and fusion augmentation materials.
Although PEEK provides good biocompatibility and strength, it is unable to bond directly to bone and to osseointegrate. 11,43 Modifications to PEEK designed to address the issue of osseointegration have gained popularity. These modifications include hydroxyapatite-coated PEEK, porous PEEK, titanium plasma–coated PEEK, carbon fiber–reinforced PEEK, and polyetherketone. 44 Our study did not control for these variations because there is insufficient high-quality evidence comparing these modifications. We hypothesize that modifications to PEEK result in superior biomechanical properties and superior osseointegration compared with unmodified, legacy PEEK devices.
Although we excluded patients who underwent ACDF without anterior plating, the use of stand-alone interbody devices without anterior plating is common in Europe. Studies of autograft report that use of an anterior cervical plate improved fusion rates from 93.5% to 98%. 45 To our knowledge, no high-quality studies have compared the outcomes of plated versus unplated 1- and 2-level ACDF with modern implants, indicating an important area for future research.
In addition to the potential differences in fusion rates between structural allograft and PEEK interbody devices, these devices may differ greatly in cost. Although studies comparing costs between structural allograft and PEEK devices are limited, a 2015 study by Virk et al 16 estimated the costs per quality-adjusted life-year for ACDF using PEEK ($3328), allograft ($2492), and autograft ($2492). However, they did not account for regional variations in cost. In some regions, because of storage, regulatory, and cultural reasons, obtaining structural allograft can be difficult or cost prohibitive. Future studies focusing on cost differences among interbody devices are needed.
When pooling results from the studies we analyzed, the subsidence rate was higher in patients who had PEEK versus allograft devices; however, none of the studies included in this review directly compared subsidence rates of PEEK versus allograft. It is important to note that although structural allograft had lower reported subsidence rates, it is difficult to assess for subsidence on radiographs because of the resemblance of allograft to native bone on imaging. In contrast, PEEK cages have embedded radiopaque markers, which potentially allow for better visualization of subsidence. In a direct comparative study that did not meet criteria for inclusion in our review, no significant difference in subsidence rates was seen between allograft and PEEK interbody devices used in ACDF. 46 It is also important to consider that although subsidence after ACDF procedures has been well studied, the effects of subsidence on clinical outcomes and fusion rates after ACDF remain unclear. 47
Limitations are inherent in systematic reviews. In our study, heterogeneity of fusion assessment, stringent inclusion and exclusion criteria, and possible missed cases contribute to the limitations. The discrepancy in fusion assessment prevented us from performing a meta-analysis. In addition, the lack of level-I evidence with a direct comparison of structural allograft versus PEEK makes it difficult to form robust conclusions. Further high-quality evidence is required to provide evidence-based treatments for patients with cervical spine degeneration undergoing ACDF. A thorough search and review of the literature, as well as strict adherence to the Preferred Reporting Items of Systematic reviews and Meta-Analyses guidelines contributed to the strengths of this systematic review.
Conclusions
Similar fusion rates have been reported for structural allograft and PEEK interbody devices for ACDF in patients with cervical spine degeneration. No high-quality randomized controlled trials or high-quality cohort comparisons directly compared structural allograft versus PEEK interbody devices in ACDF. Future research is needed to assess the outcomes of various modified PEEK devices and bone graft substitutes and extenders.
Supplemental Material
Supplemental Material, GSJ883256_Appendix1 - Structural Allograft Versus PEEK Implants in Anterior Cervical Discectomy and Fusion: A Systematic Review
Supplemental Material, GSJ883256_Appendix1 for Structural Allograft Versus PEEK Implants in Anterior Cervical Discectomy and Fusion: A Systematic Review by Amit Jain, Majd Marrache, Andrew Harris, Varun Puvanesarajah, Brian J. Neuman, Zorica Buser, Jeffrey C. Wang, S. Tim Yoon, Hans Jörg Meisel and AOSpine Knowledge Forum Degenerative in Global Spine Journal
Footnotes
Authors’ Note
This study used publicly available information and thus was not subject to review from our institutional review board.
Acknowledgements
We thank Carrie L. Price, MLS, clinical informationist at the Welch Medical Library, for her assistance in developing the search strategy, and Yabin Wu from AOSpine International for his tireless effort and support of this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
The supplemental material is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
