Abstract
Study design
Systematic Review of the Literature.
Objective
The purpose of this study was to perform a systematic review describing fusion rates for anterior cervical discectomy and fusion (ACDF) using autograft vs various interbody devices augmented with different osteobiologic materials.
Methods
A systematic review limited to the English language was performed in Medline, Embase and Cochrane library using Medical Subject Heading (MeSH) terms. Studies that evaluated fusion after ACDF using autografts and osteobiologics combined with PEEK, carbon fibre, or metal cages were searched for. Articles in full text that met the criteria were included in the review. The main outcomes evaluated were the time taken to merge, the definition of the fusion assessment, and the modality of the fusion assessment. The risk of bias of each article was assessed by the MINORS score or ROB 2.0 depending on the randomisation process.
Results
The total number of references reviewed was six hundred and eighty-two. After applying the inclusion criteria, 54 were selected for the retrieval of the full text. Eight studies were selected and included for final analysis in this study. Fusion rates were reported between 83.3% and 100% for autograft groups compared to 46.5% and 100% for various interbody device/osteobiological combinations. The overall quality of the evidence in all radiographic fusion studies was considered insufficient due to a serious risk of bias.
Conclusion
Mechanical interbody devices augmented with osteobiologics performed similarly to autografts in terms of reliability and efficacy. Their time to fusion and fusion rate were comparable to autografts at the end of the final follow-up.
Introduction
Anterior cervical discectomy and fusion is 1 of the most successful surgical procedures for the treatment of degenerative cervical disc disease.1,2 The anterior approach to ACDF was first described by Cloward 3 and Robinson and Smith 4 in the 1950s. In both of these procedures, fusion was achieved with an autologous bone graft obtained from the iliac crest. Many studies demonstrated successful clinical and radiological outcomes following ACDF using an iliac crest graft with fusion rates up to 100%. Iliac crest bone grafts have been considered almost perfect because they are relatively easy to harvest, cost-free, and meet all the requirements of the ideal graft: osteogenesis, osteoinduction, and osteoconduction. Furthermore, there is no possibility of foreign tissue reaction or allergy at the recipient site. In addition, it is easier to radiologically assess the fusion rate because there are less artefacts on the imaging. Follow-up computerized tomography can be a powerful tool for assessment of fusion but only if visualisation of the endplate – graft interface is not obscured by artefacts produced by the foreign material. This concern is somewhat mitigated by the use of nonmetal interbody spacers. 5 However, the disadvantages and complications associated with the harvesting of bone from the iliac crest are not uncommon. Although there are disadvantages associated with autograft usage, especially donor site morbidity, it is still considered the gold standard due to reliably high fusion rates.6,7 Other potential complications associated with autografts include superficial infections, herniated abdominal contents, neurovascular injuries, seromas, minor haematomas, and iliac wing fractures.8,9
To overcome these complications associated with autografts, alternative materials have been developed and are currently being used. These include mechanical interbody spacers (made of PEEK, carbon fiber or metal) or allografts (corticocancellous or composite). The choice of structural graft material to be used typically depends on the surgeon’s preference and local availability. There is worldwide variation among surgeons when selecting the graft material for ACDF. According to a survey conducted in 2017, 64.1% of spine surgeons around the world preferred mechanical interbody fusion devices, while 20% preferred the autologous iliac crest as a structural component used in ACDF. 10 This survey among spine surgeons observes that there are 2 different schools of practice when it comes to performing ACDF.
To enhance fusion rates, interbody devices are combined with a variety of nonstructural graft materials such as osteobiologic materials or bone graft substitutes. 11 The most common bone graft substitutes that are used in conjunction with these interbody devices are autografts, allografts, calcium sulphate, tricalcium phosphate followed by synthetic materials like such as bone morphogenic protein (BMP), demineralised bone matrix (DBM), ceramics and other synthetic materials.12-16 Fusion rates vary with each osteobiologic, as every combination with the interbody device is unique in itself, leading to variations in radiological outcomes. Although ACDF commonly requires short hospital admission and is well tolerated by most patients, surgical delays, complications, and increased length of stay can be influenced by identifiable risk factors such as preoperative altered functional status, high comorbidity burden, and chronic steroid use. 17 Furthermore, high creatinine values, anemia, hyponatremia, and leukocytosis were found to be associated with any complications of 30 days and unplanned readmissions after the ACDF procedure, but no long-term impact on the fusion rate associated with these risk factors was observed. 18
Given the numerous osteobiologics and interbody devices available for use in ACDF, there is a pressing need for a critical review of fusion rates across different combinations of these treatments. To date, there is no real objective rationale for the preference for 1 type of osteobiologic over another. Complicating the issue even further, there is currently no evidence on the cost effect between osteobiologics and autologous bone graft. Not all osteobiologics are available at every spinal point of care and, therefore, in many circumstances, the choice made by the surgeon is mandated by availability and cost.
The purpose of this present review is to analyse whether osteobiologics increase fusion in ACDF surgery performed with mechanical interbody devices and to compare its fusion rates with structural autograft.
Materials and Methods
Search Strategy
The literature search and review were carried out in accordance with the PRISMA guidelines. A comprehensive literature search was performed using the Medline, Embase, and Cochrane library to identify studies that evaluated fusion rates with the use of mechanical interbody spacers with ostebiologics and compared with that with autografts. The search was limited to the English language and was searched until October 2020.
Inclusion and Exclusion Criteria
Studies are included when mechanical interbody devices (PEEK, carbon fiber, metal cages) augmented with osteobiologics were used for ACDF and were compared to structural autografts. The studies excluded were case reports, ACDF surgery using allografts, patients with spinal cord injury, fracture, or infection, studies in skeletally immature patients, case series with fewer than 10 patients per group, animal, in vitro, and biomechanical studies.
Data Extraction
The following data were extracted. 1) study design, 2) patient characteristics, 3) interventions, 4) inclusion and exclusion criteria, 5) fusion rates, 6) modality of fusion evaluation, 7) time to fusion, 8) definition of fusion, 9) follow-up of each treatment group, 10) various clinical outcomes, 11) type of osteobiologics used, 12) adverse events, 13) level of incidence, funding, and conflict of interest. Fusion rates were compared at different intervals of final follow-up, as follow-up times were inconsistently reported in the included studies. All data extracted were examined for possible pooling.
Study Selection
Two independent reviewers (MS,CC) screened the title and abstracts for eligibility, and the third independent reviewer (VA) resolved the disagreements between the 2. Rayyan QCRI software was used for screening and selection.
Study Quality and Overall Strength
The risk of bias was assessed for each article using the MINORS score for nonrandomised control trials (non-RCT) and ROB 2.0 for randomised control trials (RCT). In the case of RCT, the initial strength of the evidence was considered high and could be downgraded based on the randomisation process, deviations from the intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result.
Data Analysis
The extracted data were summarised in tables and classified according to the graft tissue used. For the available continuous variables, the mean differences and variance between baseline and follow-up values were calculated, while the percentages (risk proportions) were determined for dichotomous variables by adding risks in case the patient experiences an event. The collected data were not pooled due to the heterogeneity of the studies.
Results
Total number of citations reviewed was six hundred and eighty two (Figure 1). One hundred forty-nine duplicates were removed and after applying the inclusion criteria, 54 articles were retrieved for full texts. Of these 54 full texts, 46 were excluded due to the absence of a comparator group (n = 19, comparator other than autograft (n = 9), no osteobiologic was used (n = 7), no radiological assessment of fusion (n = 5) and the comparator group using local autograft (n = 6). Eight studies were selected and included for the final analysis in this study (Table 1). Flow chart showing the literature search and article selection process. Study Characteristics and Risk of Bias Assessment for Studies of Fusion Rates Who Underwent Anterior Cervical Decompression and Fusion. Abbrevations – NR, Not reported; TCP, tricalcium phosphate; PEEK, Polyetheretherketone.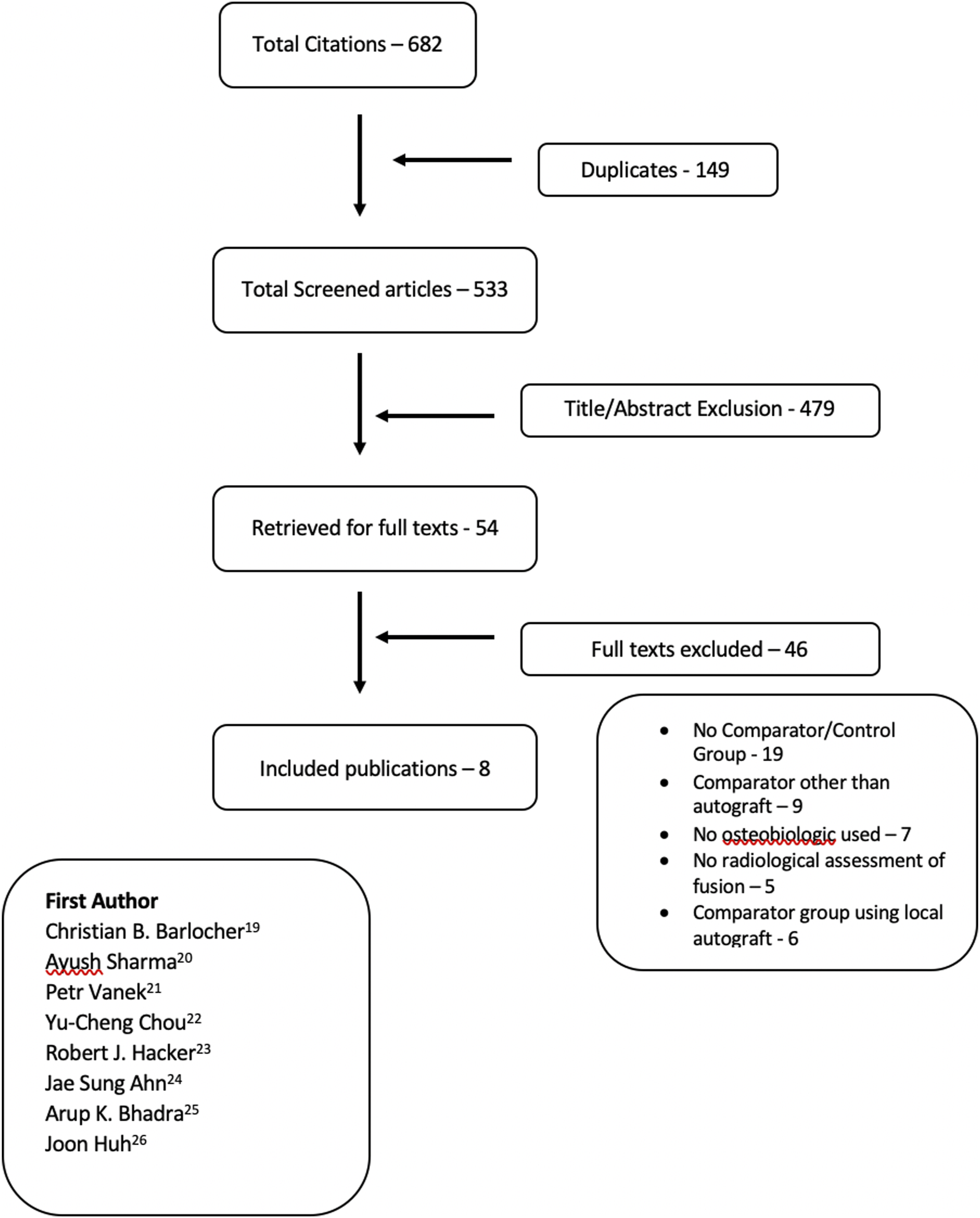
Fusion Rates of Mechanical Interbody Devices Augmented With Tricalcium Phosphate vs Autograft
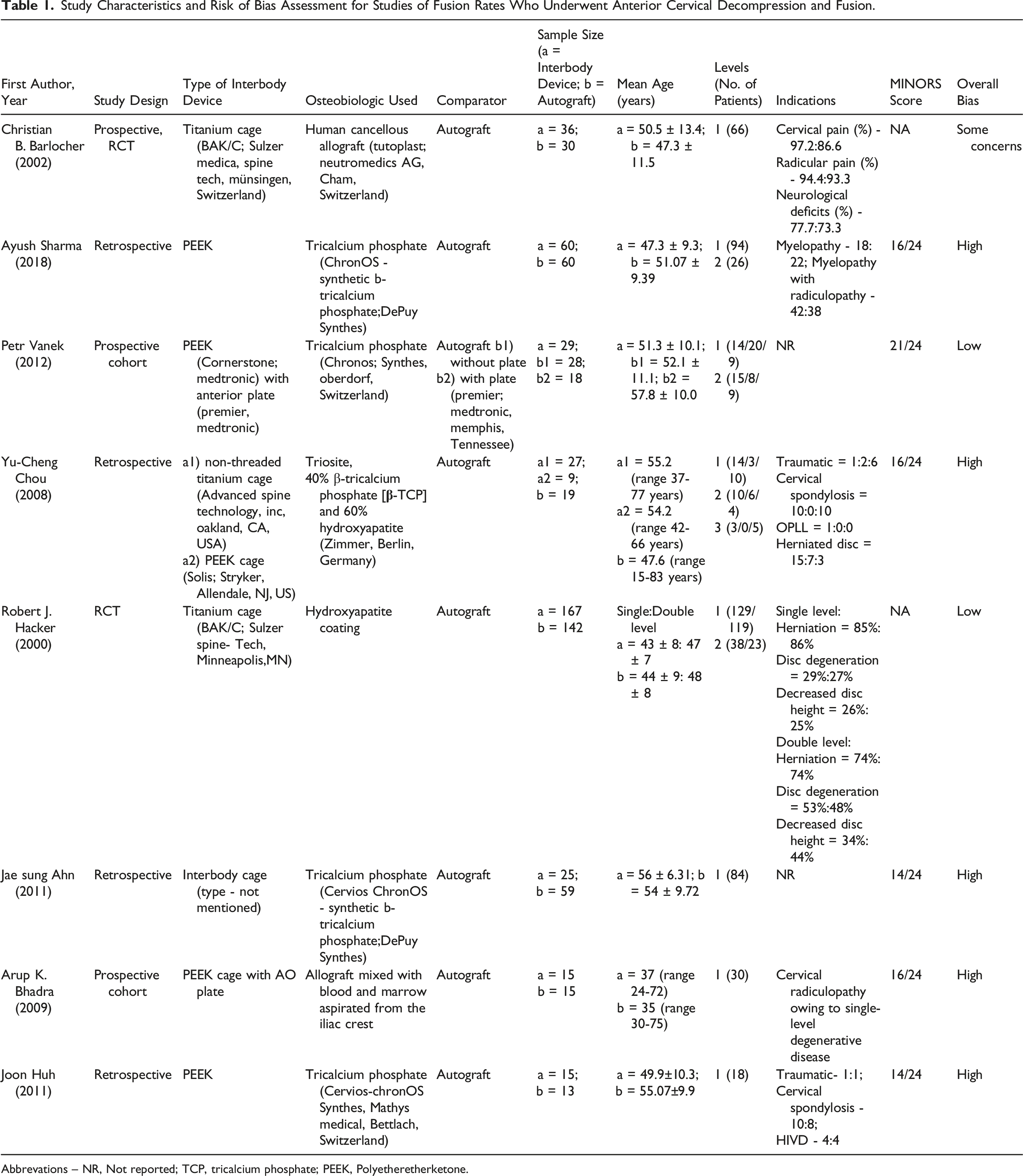
A total of 5 cohorts (n = 362) compared the tricalcium phosphate fusion rates used with PEEK or titanium and compared with autograft. Most of the participants included were male (M: F = 213: 149). Ayush Sharma et al (2018) in the retrospective cohort compared PEEK with TCP and autograft. Fusion rates were determined by flexion-extension radiographs at 3,6,12 months and CT at 1 year. At the end of 1 year, they did not observe significant differences in the fusion rate between the PEEK and autograft groups (93.3% vs 96.6%). In general, the fusion rates were better for single-level ACDF (95.74%) compared to two-level ACDF (76%). Petr Vanek et al (2012) in the prospective study observed 100% fusion rates determined by CT at the end of 2 years of follow-up comparing PEEK with TCP and autograft. They did not observe differences in terms of fusion with or without additional plate application. Joon Huh et al (2011) had similar observations in their retrospective study comparing PEEK with TCP and autograft at the end of 1 year with no significant differences (93.3% vs 100%) as assessed by CT scan. Chou et al (2008) in the retrospective study compared the fusion rates of titanium cage and PEEK with Triosite (40% TCP and 60% hydroxyapatite) with autograft. They concluded that the radiographic results were better in the PEEK and autograft groups where 100% fusion was achieved at the end of 12 months, but the titanium group with triosite achieved only 46.51% fusion determined by x-rays at the end of 12 months. In a study by Ahn et al (2011) comparing TCP and autograft, the type of interbody device was not mentioned. Fusion rates were evaluated by flexion-extension radiographs at the end of 1 year, and the autograft group performing better with 100% fusion compared to TCP with 88%.
Fusion Rates of Mechanical Interbody Device Augmented With Human Cancellous Allograft vs Autograft
Two cohorts with a sample size of 96 compared PEEK and titanium cages augmented with allograft to autograft. Barlocher et al (2002), in their prospective trial, observed that titanium-implanted patients and allograft-implanted patients had significantly better fusion rates at 6 months compared to the autograft group (86.1% vs 65.3%; P-value <.05). However, there were no major differences in terms of fusion at the end of 1 year (97.2% vs 93.3%). Bhadra et al (2009), compared PEEK cage augmented with allograft mixed with blood and marrow aspirated from the iliac crest and concluded that there were no differences in radiographic fusion rates between the groups at the end of 1 year (both at 100%).
Fusion Rates of Mechanical Interbody Device Augmented With Hydroxyapatite vs Autograft
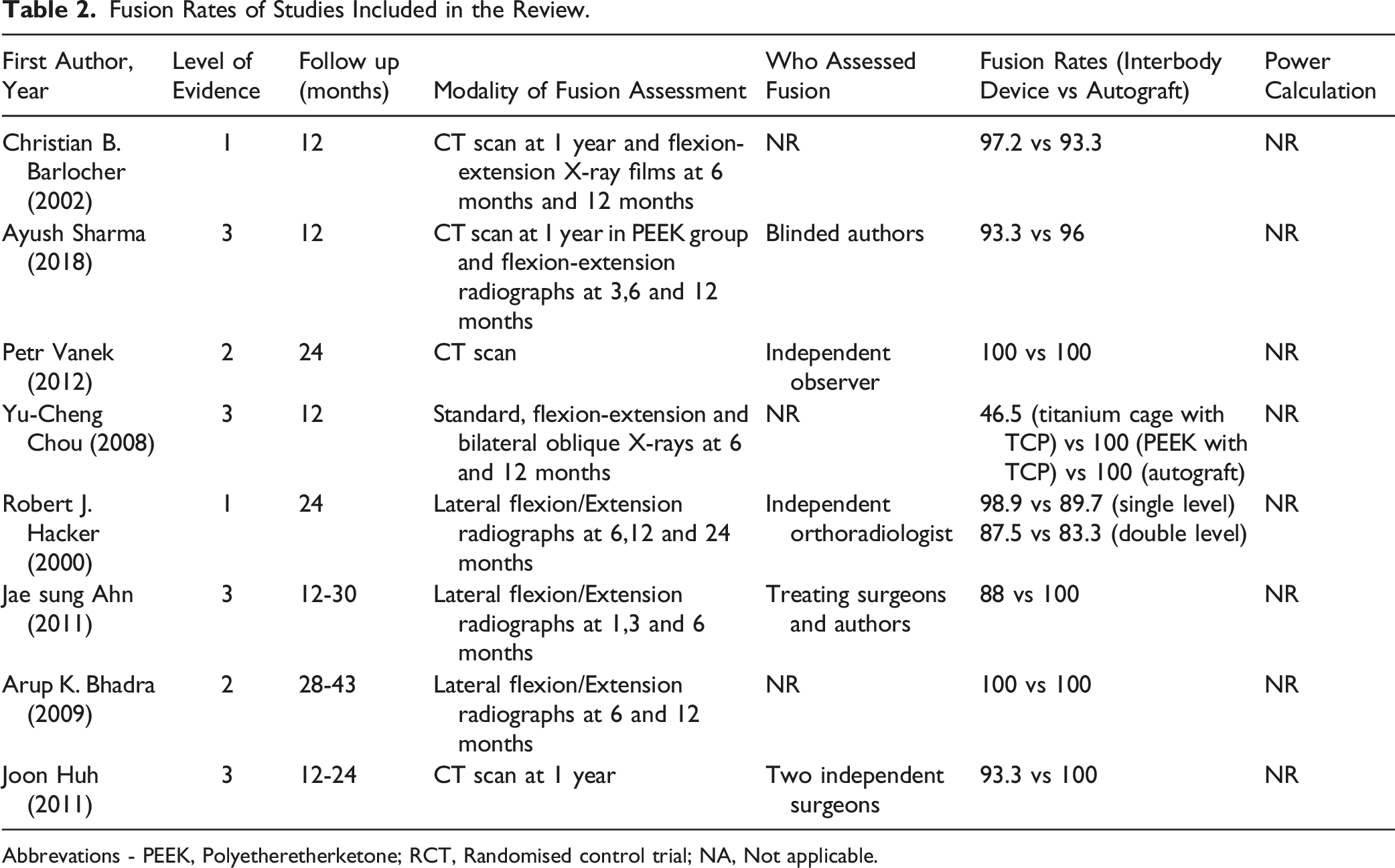
Fusion Rates of Studies Included in the Review.
Abbrevations - PEEK, Polyetheretherketone; RCT, Randomised control trial; NA, Not applicable.
Assessment of Bias
Non-RCT articles were evaluated for risk of bias using the MINORS score, and RCTs were evaluated using the ROB 2.0 method. The overall risk of bias was high in 5 cohorts and low in a prospective cohort by Petr Vanek et al. The total score ranged between 14 and 21 for non-RCTs. When the RCT by Christian Barlocher was evaluated for risk of bias, there were some concerns, whereas the risk was low in the RCT by Robert J Hacker.
Discussion
The purpose of this review was to determine whether osteobiologics enhance fusion when combined with mechanical interbody devices and to compare their fusion rates with those of standard structural autografts. In general, fusion rates were reported to be high in both comparative arms, allowing us to comment that mechanical interbody devices filled with biomaterials such as tricalcium phosphate, human cancellous allograft, and hydroxyapatite performed similarly in terms of radiological results compared to structural autografts. A total of 8 studies were included in the final analysis, with fusion rates ranging from 46.5% to 100%. Four out of 8 were retrospective studies, 2 were prospective cohorts, and 2 were randomised control trials. Due to the heterogeneity of the data, a quantitative reporting meta-analysis could not be performed. The overall quality of the evidence for radiographic fusion in all studies was insufficient. Most non-RCT studies demonstrated a high risk of bias due to a wrong patient enrolment method, lack of follow-up, and improper statistical analysis.
The fusion rates at the end of 1 year were high for human cancellous allograft (97.2% - 100%), tricalcium phosphate (88% - 100%), hydroxyapatite (98.9%) and were comparable to structural autograft (89.7% - 100%). However, Yu-Cheng Chou et al reported that PEEK cages had higher fusion rates (100%) compared to titanium cages (46.5%) when combined with TCP. The lower fusion rates could be attributed to the sample size of the titanium group, which had a large number of multilevel ACDF procedures, which has previously been observed to have lower fusion rates compared to single-level ACDF procedures. This shows that future randomised control trials are needed that directly compare PEEK and titanium combined with various osteobiologics are needed.
Since the introduction of surgical fusion in 1911, many methods have been used to assess it. From static radiographs to dynamic magnetic resonance imaging. Dynamic radiographs improve the accuracy of fusion assessment when compared to static radiographs but still have few limitations such as two-dimensional assessment, disagreement on allowable motion, etc. With the advent of fine-cut imaging and 3D reconstruction, CT is widely used for fusion assessment. However, there are significant concerns about the high radiation exposure due to CT. 27 The gold standard for the assessment of fusion is described by Rhee et al as <1-mm of interspinous motion on flexion-extension radiographs, with computed tomography (CT) follow-up if fusion status was indeterminate regardless of graft or instrumentation type. 28 In this review, CT alone is observed to be used to assess fusion in 2 of 8 studies, while 6/8 preferred dynamic radiographs as a modality to assess fusion rates at long-term follow-up and was defined by the presence of bony trabeculation throughout the graft.
The structural autograft has been widely used for the ACDF procedure for many decades and is considered the ‘gold standard’ to achieve consistent fusion. 29 However, a literature search showed that the number of articles on the use of structural tricortical iliac crest graft for ACDF has decreased in the last decade. There is a paradigm change, where the number of publications on mechanical interbody devices and other osteobiologics in ACDF has increased in the recent past. This change can be attributed in part to the preferences of surgeons and patients. Osteobiologics are the key to successful spinal fusion procedures, including ACDF, and the variability in their use reflects the uncertainty with respect to their effectiveness and potential risks. 30 Patient satisfaction is a factor that is almost neglected when it comes to ACDF graft choices. One such study examined patient satisfaction after ACDF surgery and found that 95.4% of patients who underwent revision ACDF surgery preferred synthetic bone graft substitute over structural autograft. Ninety-one percent of the patients rated the incision at the graft site as more painful than at the index anterior cervical surgical site. 31 The authors were also against the traditional recommendation that autograft is the ‘gold standard’ for ACDF. The statement that autograft should not be considered as the “gold standard” but merely 1 of several options for ACDF in this modern era of graft substitutes merits further discussion among peers.
Historically, iliac crest is considered to be the best source of autologous bone graft. In an effort to avoid morbidity, surgeons have explored various other sites of autologous graft harvest which included ribs and cervical lamina.32,33 However in any of these methods, the need for second surgical incision could not be avoided. Hence, local autograft obtained after removing osteophytes was used to fill the interbody spacer in order to avoid complications of second surgical site. But the main concern with this method is the volume of local graft obtained. In a study by O. Neill et al, only 20% of patients had enough volume of local graft which could be filled completely inside the interbody spacer. 34 In majority of the patients, volume of local graft was not enough to fill the interbody spacer and had to be supplemented by demineralised bone matrix.
Although ACDF is generally an effective procedure, it does carry a significant risk of morbidity. A recent review showed 13.2% to 19.3% overall morbidity rate, including but not limited to complications such as dysphagia, postoperative hematoma with and without epidural hematoma, exacerbation of myelopathy, symptomatic recurrent laryngeal nerve palsy, cerebrospinal fluid leak, wound infection, increased radiculopathy, Horner’s syndrome, respiratory insufficiency, esophageal perforation, and instrument failure. 35 As many of these complications could be caused by several factors, each individually lends itself to the dilemma between correlation and causation. There is a possibility that the biologics used in isolation and/or contingent on a patient’s specific comorbidities could have more influence on such morbidities than we currently suspect. Proving or disproving this would be very difficult given the multitude of factors that could potentially influence said morbidities and the inherent difficulty of producing a truly controlled experiment in the setting of surgery.
This review has its own set of limitations. The combination of synthetic cage filled with local bone chips from the osteophytes, a commonly practiced economic option, was not included in this search. Additionally, the effect of adding a plate to the fusion could not be separated. Yet, the most important limitation in this study was the heterogeneity of the included studies where meta-analysis could not be performed. Therefore, quantitative conclusions could not be drawn when all of these groups were compared. We believe that radiological results should not be the only comparative parameter when the ‘gold standard’ structural autograft is paired with other alternative biomaterials. Clinical outcomes play a major role in evaluating their long-term efficacy, a parameter that was not included in this systematic review.
Conclusion
In summary, we observed that there is very high incidence of bias in almost all studies. Although this systematic review had its own limitations, individual analysis allowed us to show with certainty that mechanical interbody devices augmented with osteobiologics performed similarly to autografts. The time to fusion may be longer for certain osteobiologics, but the fusion rate was comparable to autografts at the end of the final follow-up. Future studies should focus on minimising bias by performing a blinded assessment of fusion status, having adequate follow-up of 2 years, patient randomisation, and following a standardised protocol for evaluating radiological fusion. Surgeons performing the ACDF procedure may also consider patient satisfaction/preference when selecting grafts.
Supplemental Material
Supplemental Material - Do Osteobiologics Augment Fusion in Anterior Cervical Discectomy and Fusion Surgery Performed With Mechanical Interbody Devices (PEEK, Carbon Fiber, Metal Cages) and is the Fusion Rate Comparable to that With Autograft? – A Systematic Review
Supplemental Material for Do Osteobiologics Augment Fusion in Anterior Cervical Discectomy and Fusion Surgery Performed With Mechanical Interbody Devices (PEEK, Carbon Fiber, Metal Cages) and is the Fusion Rate Comparable to that With Autograft? – A Systematic Review by Viswanadha Arun-Kumar, Stipe Corluka, Zorica Buser, Yabin Wu, Mohammad El-Sharkawi, Charles André Carazzo, Nikhil Ponugoti, Jeffrey Wang and Hans Jörg Meisel in Global Spine Journal
Supplemental Material
Supplemental Material - Do Osteobiologics Augment Fusion in Anterior Cervical Discectomy and Fusion Surgery Performed With Mechanical Interbody Devices (PEEK, Carbon Fiber, Metal Cages) and is the Fusion Rate Comparable to that With Autograft? – A Systematic Review
Supplemental Material for Do Osteobiologics Augment Fusion in Anterior Cervical Discectomy and Fusion Surgery Performed With Mechanical Interbody Devices (PEEK, Carbon Fiber, Metal Cages) and is the Fusion Rate Comparable to that With Autograft? – A Systematic Review by Viswanadha Arun-Kumar, Stipe Corluka, Zorica Buser, Yabin Wu, Mohammad El-Sharkawi, Charles André Carazzo, Nikhil Ponugoti, Jeffrey Wang and Hans Jörg Meisel in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
