Abstract
Bone graft substitutes have been used routinely for spine fusion for decades, yet clinical evidence establishing comparative data remains sparse. With recent scrutiny paid to the outcomes, complications, and costs associated with osteobiologics, a need to improve available data guiding efficacious use exists. We review the currently available clinical literature, studying the outcomes of various biologics in posterolateral lumbar spine fusion, and establish the need for a multicenter, independent osteobiologics registry.
Keywords
Lumbar spine fusions are performed at a rate of 250,000 per year in the United States alone, 1 and in each case, surgeons are faced with the critical decisions regarding choice of bone graft. In an arena that once demonstrated near-exclusive use of iliac crest bone graft in 1990s, surgeons are now split between the use of growth factors, allograft, demineralized bone matrix (DBM), and ceramics, to name just a few. Despite this transformation to the use of bone graft substitutes to avoid the complications associated with autograft harvest, the comparative and high-quality evidence that researchers and practitioners must rely on is sparse. Although bone regenerative technology research has been conducted for over 4 decades, there has yet to be established an accepted, cost-effective, and efficacious algorithm for patients undergoing lumbar spinal fusion.
Outcomes from the use of bone graft substitutes in bone healing are almost nonexistent except for a few commercially available products. The chief reason for this is the expense involved in designing and implementing clinical studies. Though select osteobiologics have been studied in detail, such as bone morphogenetic protein, the resources to obtain similar evidence for other products are often unavailable. Furthermore, even the available data vary substantially in quality, design, outcome measures, and consequently, conclusions. Finally, many products are regulated as “minimally manipulated human allograft” by the Food and Drug Administration (FDA), which does not require a randomized controlled trial or premarket approval for clinical use. Because a large amount data outside of the peer-reviewed literature are often pushed as industry-driven marketing tools, surgeons must actively seek high-quality evidence to make appropriate choices for patient care. Due to the large number of osteobiologics on the market, there exists a need for a focused study group to collect, analyze, and report data.
To that end, complications have been reported from the off-label and misuse of bone graft substitutes. Because products are commonly used off-label without formal data, invariably, there is an incomplete understanding of mechanism, tissue response, and proper application before patients are exposed. The most recent fallout of the clinical studies involving bone morphogenetic protein has led to increased scrutiny of data presentation, potential conflicts of interest, and patient safety. 2 As it stands now, the void of an independent organization to study outcomes, costs, and technique makes it difficult to navigate through mere allegations and true fact.
This article will review the clinical evidence on bone graft substitutes in lumbar posterolateral spine fusion and present the need for a collaborative effort to improve the available data.
Study Design
Medline, Embase, and Cochrane Central Register of Controlled Trials through July 2010 were searched for publications from 1980 to 2011 with a combination of the keyword “spine fusion” and one other of the following: “bone graft substitute,” “allograft,” “ceramic,” “demineralized bone matrix,” “autograft,” “local bone,” “bone marrow aspirate,” “growth factor,” “bone morphogenetic protein,” and “stem cells.” Query results were reviewed by two independent observers, and articles for inclusion of this study were selected using the following criteria:
Clinical studies, level of evidence grade I to IV and agreed upon according to Journal of Bone and Joint Surgery (American Edition) guidelines by two independent reviewers
Sample size of >10 patients
Instrumented posterolateral lumbar fusion only (one- to three-level; no interbody)
Minimum of 1-year radiographic follow-up
Diagnosis of a degenerative condition of the lumbar spine (spondylolisthesis, degenerative disc disease)
Adult patients with a minimum age of 18 years old
Reported fusion rates
Fusion rate was defined by plain radiographs and/or computed tomography (CT) imaging depending on the method reported in the respective study. Approximately 80% of the publications utilized plain radiographs with anteroposterior/lateral/flexion/extension views as the primary means to evaluate bone fusion. CT scan was often used in cases of disagreement between reviewers for each publication, as determined by the respective authors. CT scan has been historically more stringent in the assessment of solid bony fusion and generally has led to lower fusion rates than that assessed with plain radiographs. However, because of recent concerns with cancer risk from the radiation exposure from CT technology, 3 more recent clinical studies have focused on the use of routine plain radiographs to assess bony growth. For the purposes of this article, because studies that utilized CT scan were far fewer than plain radiographs and included each biologic, all radiographic data were compiled together.
For studies utilizing more than one type of bone graft substitute, groups were categorized in accordance with the authors’ original objective and hypothesis. For example, if the authors’ intent was to study the efficacy of a ceramic, then ceramic/local autograft or ceramic/BMA was placed in the ceramic group. Studies involving bone marrow aspirate (BMA) that was processed and concentrated were classified into a BMA group, whereas nonconcentrated BMA groups were categorized into those of the accompanying bone graft substitute. In the bone morphogenetic protein group, only those studies utilizing FDA-approved products for routine use (rhBMP-2 and the product INFUSE™) were included.
Results
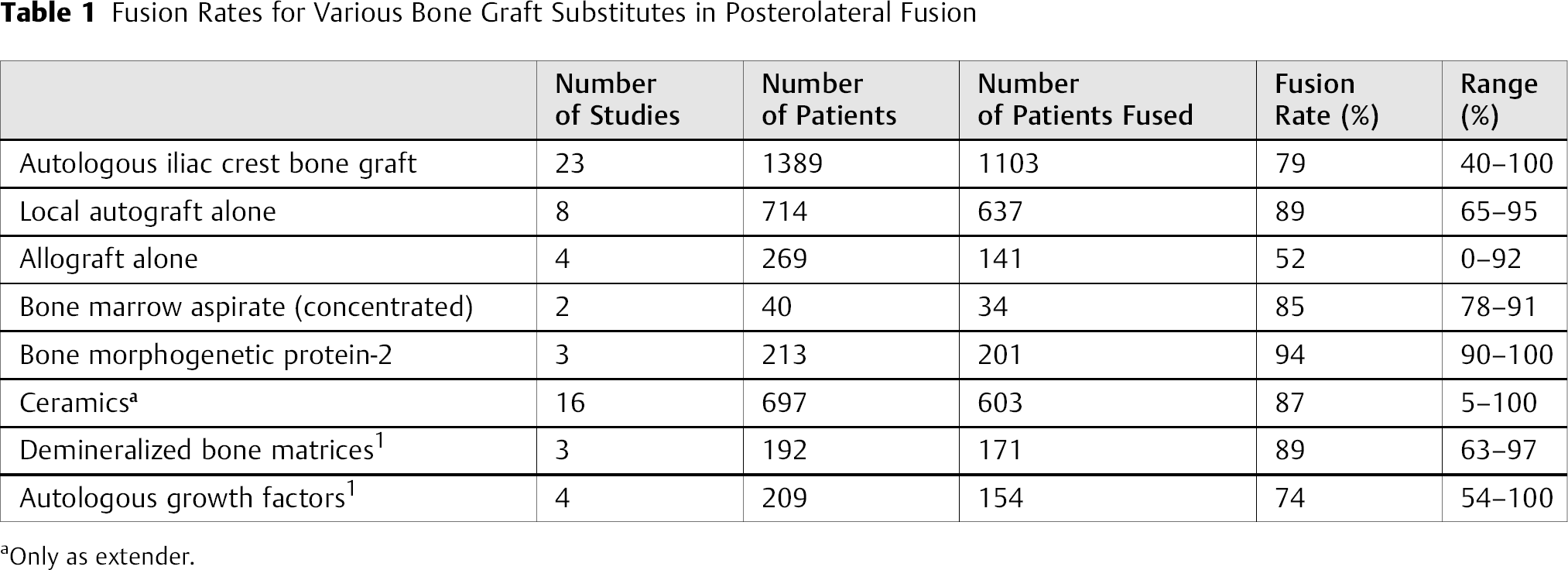
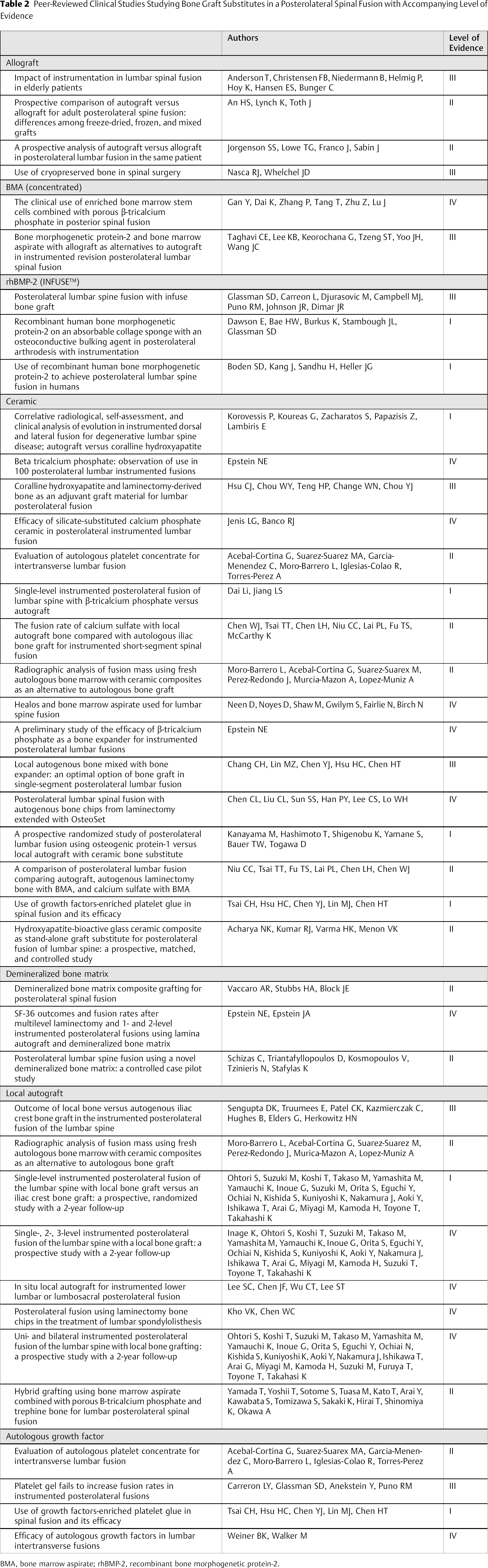
A total of 38 studies met the inclusion criteria. Of these studies, four provided data on allograft alone, 4 , 5 , 6 , 7 two on concentrated BMA, 8 , 9 16 on ceramics, 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 , 18 , 19 , 20 , 21 , 22 , 23 , 24 , 25 three on DBMs, 26 , 27 , 28 eight on local autograft alone, 17 , 29 , 30 , 31 , 32 , 33 , 34 three with recombinant bone morphogenetic protein-2 (rhBMP-2; INFUSE™), 35 , 36 , 37 and four on autologous growth factors 14 , 25 , 38 , 39 (Table 1). Articles were subsequently classified based on level of evidence: 8 level I, 10 level II, 8 level III, 12 level IV (Table 2). A group with the use of autologous iliac crest bone graft was performed in 23 of these studies (as a respective control group), and the collective fusion rate calculated.
Fusion Rates for Various Bone Graft Substitutes in Posterolateral Fusion
Only as extender.
Peer-Reviewed Clinical Studies Studying Bone Graft Substitutes in a Posterolateral Spinal Fusion with Accompanying Level of Evidence
BMA, bone marrow aspirate; rhBMP-2, recombinant bone morphogenetic protein-2.
Iliac Crest Bone Graft
Iliac crest bone graft was harvested in a control group in 23 studies in a total of 1389 patients. A total of 79% (range 40 to 100%) were deemed to be successfully fused by the respective authors. Surgical technique, volume, and patient population varied with each study; however, in many studies, the details regarding graft harvest were not included. These data are consistent with historical data that report fusion rates with the use of iliac crest bone graft in posterolateral lumbar fusion from 50 to 90%. 29 , 40
Local Bone Graft
Although some studies have reported unacceptable fusion rates in clinical trials in the past, 40 , 41 the use of local bone graft from laminectomy and facetectomy sites during decompression has grown in popularity in recent years. Improvements in technique of local bone preparation, such as soft tissue dissection, morselization, and bone preservation, have led to many surgeons’ use in the lumbar spine. In vitro data have suggested that osteoblast content is greater in cancellous bone from laminar bone than iliac crest. 42 The one significant disadvantage in the use of local bone graft is the limited supply that depends upon the spinal levels decompressed. In eight studies with a total of 714 patients with local bone graft alone (without bone marrow aspirate), a total of 89% (range 65 to 95%) successfully fused based upon plain radiographic imaging.
Allograft
Allograft, or processed cadaveric bone, confers advantages over autogenous bone graft in an abundant supply of material and the avoidance of complications associated with graft harvest. Preserved through either frozen or freeze-dried processes, antigenicity is reduced, leading to increased graft incorporation. Although the risk of disease transmission exists with the use of allograft, standards in screening donor tissue have reduced the risk of HIV disease transmission to no greater than 1 in 1.5 million. 43 Because allograft-based products are classified as minimally manipulated human cellular tissue products by the FDA, little data are required for a product to reach the marketplace. Allografts are thought to only offer osteoconductive activity.
A total of 269 patients treated with allograft alone in a posterolateral lumbar fusion in four studies were identified. Formulations were described as chips, powder, and “matchsticks.” A collective fusion rate of 52% was calculated (range 0 to 92%).
Demineralized Bone Matrix
DBM is derived from the acid extraction of the mineralized phase of allograft bone. Allogeneic bone is crushed to a particle size of 74 to 420 μm, followed by demineralization in 0.5 N HCl mEq/g for 3 hours. 44 In vitro studies have demonstrated osteoinductivity with survival retention of growth factors after this process. However, there is wide variability in the way DBMs are processed including sterilization methods, associated carrier, and percent of human tissue present. Commercial preparations include a variety of associated substances such as glycerol, hyaluronic acid, gelatin, and calcium sulfate powder. 45 , 46
Preclinical studies have demonstrated a wide variability in the performance of various DBMs in the formation of bone and osteogenic gene expression. 45 , 46 , 47 Much of this inconsistency can be attributed to the different ways that DBM is constructed. Furthermore, the quality of donor tissue is also thought to be directly related to in vivo performance. Finally, translational studies have repeatedly demonstrated the wide variability in this osteoinductivity not only among different products, but also with different lots of the same product. 48
Despite the fact that over 50 DBM products are commercially available for use in the lumbar spine, only three clinical studies in 192 patients report its use in the posterolateral lumbar spine. In each of these studies, DBM was used as an extender to local bone graft. No studies have been identified that reported outcomes from the use of DBM alone in this setting. An 89% (range 62 to 95%) fusion rate was calculated in these studies. Of the three studies that met the inclusion criteria, two were level III evidence and one was level IV.
Bone Marrow Aspirate
Autologous bone marrow aspirate (BMA) has been a topic of research for many years. Harvested through a minimally invasive procedure, BMA provides a population of osteoprogenitor cells and critical growth factors that help cell differentiation, leading to bone healing. In vitro studies have demonstrated high colony-forming unit count from BMA obtained from either vertebral body or iliac crest. 49 Because BMA alone lacks localized structural support, it is often used clinically with an appropriate carrier such as a ceramic.
There is a substantial variability in the processing of BMA, notably with concentrated and nonconcentrated techniques. With a concentrated protocol, bone marrow aspirate is harvested in small volumes and processed through centrifugation, cell separation, and/or retention steps to provide a higher concentration of osteoblast progenitor cells. The proportion of osteogenic progenitor cells in bone marrow has been reported between 1 in 5000 to 100,000 cells. Cell retention techniques can increase this population up to fourfold, theoretically leading to higher performance.
Two studies utilizing a concentrated BMA protocol enrolled 40 patients who underwent a posterolateral lumbar spine fusion. A ceramic carrier (tricalcium phosphate) was used in 22 patients and allograft in 18 patients. An 85% fusion rate was achieved (range 78 to 91%).
Ceramics
Ceramic carriers are derived from a process called “sintering,” which uses high temperatures to extract individual crystals that fused together at crystal grain boundaries. 44 These products are composed of collagen, tricalcium phosphate, calcium phosphate, calcium sulfate, and/or hydroxyapatite, which are all synthetically based. Because each of these components has different binding, biodegradability, and adhesion characteristics, there is variability seen among carriers depending on composition. Although harboring no osteoinductivity, these carriers can provide an osteoconductive matrix with improved biomechanical properties compared with other conventional carriers.
In eight studies utilizing ceramics as a bone graft extender in our review, a fusion rate of 85% was observed (range 5 to 100%). In each of the studies reporting the use of ceramics in posterolateral lumbar fusion, an osteoinductive autologous source of cells was used (local autograft and/or BMA).
Bone Morphogenetic Protein
First described by Dr. Marshall Urist in 1965, 50 bone morphogenetic protein-2 received FDA approval for the application in the anterior lumbar spine in 2002. Since then, an estimated 85% of the clinical use of rhBMP-2 has been off-label, 51 often in the posterolateral lumbar spine. The complication rates with the use of rhBMP-2 have been well-publicized 2 , 52 , 53 , 54 , 55 , 56 , 57 , 58 , 59 , 60 , 61 ; however, it appears that the incidence of untoward effects is less when used in the posterolateral lumbar compared with other areas.
Bone morphogenetic protein activates the bone healing cascade through the Smad complex protein pathway that leads to the activation of important genes such as runx and osterix. Although there are over 20 proteins identified in the BMP family, BMP-2 has been the most widely studied in the induction of bone formation. 62 BMPs have other important roles in differentiation of a wide variety of cells such as chondrogenic, cardiac, neural, and epidermal induction. 63 The exact mechanisms behind the reported complications has not been elucidated; however, investigators have postulated angiogenic, inflammatory, and osteoclastic pathways. 64 , 65 , 66
Three studies were identified utilizing the INFUSE™ product containing rhBMP-2 and an absorbable collagen sponge in a posterolateral lumbar spine fusion. A fusion rate of 94% was achieved in this group.
Platelet Concentrates
Platelet concentrate products are derived from platelet-rich plasma (PRP), which involves concentrating whole blood through a centrifugation process. The result is a pellet rich in growth factors such as platelet-derived growth factor, transforming growth factor-β, vascular endothelial growth factor, insulin growth factor, and epidermal growth factor. Furthermore, mesenchymal stem cells are also present, which provide an opportunity for differentiation into primary osteoblasts.
However, variability in processing methods, classification systems, and terminology has led to wide inconsistency in the results of its use in many orthopedic conditions, including bone healing. For example, reported platelet concentration counts between different purification systems are drastically different. 67 This variability is increased, considering that the method of platelet counting (manual versus automated) has not been standardized. 68 Finally, It is thought that inhibitory factors, both identified and unknown, are also present in PRP that may modulate the host response after implantation.
Two studies were identified that reported the use of PRP in spine fusion. Surprisingly, a low rate of fusion was observed at 60%. In fact, in these studies, the authors concluded that the addition of PRP inhibited successful lumbar spine fusion. This fusion rate was significantly lower than iliac crest bone graft alone group in one study. 38
Discussion
Despite the widespread use of bone graft substitutes in posterolateral lumbar spine fusion on a daily basis worldwide, the clinical evidence that helps guide decision making and reduce complication rates is sparse. Even many of the available studies have substantial methodological flaws, including the lack of a control group. Historical data across 3 decades are difficult to compare because of the improvement of instrumentation, variations of surgical technique, and inherent differences in processing methods of products such as that of BMA, DBM, and platelet concentrates. The landscape for the use of these products has and will continue to rapidly change as increased scrutiny is pointed toward the elimination of complications, cost-reduction strategies, and the regulatory climate around bone graft substitutes. Given the number and range of biologics on the market, comparative clinical effectiveness research is important to improve treatments and outcomes for patients electing for spine surgery.
The burden on surgeon education to date has been largely left to the opinions of industry representatives making claims about product safety and efficacy. In addition, the large number of products is not easily addressed by independent research from isolated sources. Finally, the constant addition of 510k-approved and “minimally manipulated human allograft” graft products to the market occurs at a rate that exceeds that of independent research.
The field of spine surgery deserves a comprehensive, controlled, multicenter, and independently managed osteobiologics registry that would collect data on patients in a standardized fashion. Utilizing such a tool would allow for the study of many different products in a homogenous patient population with standardized follow-up points. With this prospective study design, the conclusions derived would be much powerful than a systematic review. An effective method for collecting postmarket data are the registry model. Herndon et al describe the stakeholders and the deficiencies in the current approach to health care technology assessment and in doing so define the role for clinical databases/registries in postmarket surveillance and health care technology assessment. 69 Registries are a means of prospectively collecting data that enables continued assessment of effectiveness, especially in regards to new technology and treatments. 11 Similarly, Malchau et al described a stepwise algorithm of introducing new technology, where the final step is administering registry-based studies. 70
The Osteobiologics Collaborative, an independent study group, will assemble uniquely qualified clinicians and clinician-scientists to address this void through the Resources for Medical Education and Collaboration (RMEC) Osteobiologics Registry. To reach this goal, the following will be implemented:
A study group of clinical investigators and researchers to commit to the collection, analysis, and dissemination of data surrounding the application of osteobiologics in spine surgery;
Organized meetings to develop, enable, and promote research surrounding the application of osteobiologics in spine surgery;
Collection of multicenter data surrounding the application of osteobiologics in spine surgery;
Resources and pathways for dissemination of data and evidence to clinicians that effectively communicate safety and efficacy considerations in the application of osteobiologics.
Conclusions
Bone graft substitutes and extenders are utilized daily in spine surgery, yet the evidence surrounding commercially available products that guides surgeon choice is sparse. Independent efforts to study and report comparative results with different spinal biologics are necessary in a cost-conscious, outcomes-driven health care world. A spine fusion registry promoted by The Osteobiologics Collaborative, through the Resources for Medical Education and Collaboration, is one step toward answering important questions to improve the delivery of patient care with spinal disorders.
This effort will aim to improve patient care and outcomes, reduce complications, track cost data, and identify solutions for osteobiologics in spine surgery. For more information, please contact: Caitlyn McCullough, Executive Director, Resources for Medical Education and Collaboration, 1 Mercado Street, Ste 202, Durango, CO 81301 (phone: 970–375–3649; e-mail:
Funding
Funding was made possible in part (grant #11-278) from the Orthopaedic Research and Education Foundation (OREF). The views expressed in written conference materials, the views expressed by speakers and in publications or written conference materials do not necessarily reflect official policies of OREF.
Disclosures
W. K. Hsu, Consulting: Stryker, Pioneer, Medtronic, Zimmer, Graftys; Research Support: Baxter, Pioneer
M. S. Nickoli, None
J. C. Wang, Royalties: Medtronics, Stryker, Seaspine, Osprey, Aesculap, Biomet, Amedica, Zimmer, Synthes; Stock Ownership: Fziomed; Private Investments: Promethean Spine, Paradigm Spine, Benevenue, NexGen, K2 Medical, Pioneer, Amedica, Vertiflex, Electrocore, Surgitech, Axiomed; Board of Directors: North American Spine Society, Cervical Spine Research Society, AO Spine/AO Foundation; Scientific Advisory Board: VG Innovations, Corespine, Expanding Orthopaedics, Syndicom, Osprey, Amedica, Bone Biologics, Curative Biosciences, PearlDiver, Inc., Pioneer, Seaspine
J. R. Lieberman, Research Support: Amgen, Inc., NIH; Advisory Board, Amgen, Inc.
H. S. An, Consulting: Advanced Biologics, Inc., Pioneer, Inc.; Research support: Baxter, Inc.
S. T. Yoon, None
J. A. Youssef, Consultant: NuVasive, Integra; Royalty: NuVasive, Aesculap/B. Braun, Osprey, Amedica; Research Support: DePuy, Stryker, NuVasive, BioSurface Engineering Technologies, Globus Medical, Advanced Technologies in Regenerative Medicine, Axial Biotech, Vertiflex; Stock Options: Amedica, Pioneer, Vertiflex, Benvenue Medical, Inc., Paradigm Spine, Promethean Surgical Devices, Spinal Ventures
D. S. Brodke, None
C. M. McCullough, None
