Abstract
Study Design:
Historically controlled clinical trial.
Objectives:
Patients presenting for correction of adolescent idiopathic scoliosis (AIS) by posterior spinal fusion may benefit from structured clinical pathways. We studied the effects of implementing a published clinical pathway for the perioperative care of patients with AIS that required intraoperative use of methadone at our institution.
Methods:
We performed a historically controlled clinical trial of patients undergoing posterior spinal fusion for AIS by comparing a retrospectively collected control group of 25 patients with a prospective experimental group of 14 patients receiving methadone, gabapentin, propofol, and remifentanil as part of a new clinical pathway.
Results:
Use of the pathway decreased average pain scores evaluated by the Numeric Rating Scale in the 24 hours following surgery (4.8 [4-6] to 3.4 [2-4], P = .03 [−2.6 to −0.2; t = −2.3]) and postoperative opioid consumption by 76% (41 [29-51] mg to 10 [4-17] mg, P < .001 [−45 to −15; Welch’s t = 4.9]) during the same period. Improved analgesia and reduced reliance on opioids facilitated other postoperative elements of the clinical pathway and shortened the average hospital length of stay by 1 day (4 [3-6] days to 3 [3-5] days, P = .001 [−2 to −1; U = 67, Z = −3.3]).
Conclusions:
Multimodal analgesia and a clinical pathway add value in the perioperative care of patients undergoing posterior spinal fusion for AIS by improving analgesia and shortening hospitalization. The prospective arm of the trial was registered at clinicaltrials.gov under NCT02481570.
Introduction
Adolescent idiopathic scoliosis (AIS) is the most common pediatric spinal disorder in North America. 1 Surgical correction of AIS presents many challenges, including risk of injury to the spinal cord, use of anesthetic regimens that allow for monitoring of the spinal cord, management of significant intraoperative blood loss, and control of pain from complex tissue trauma.
Approaches to pain control for patients with AIS are varied. Intraoperative management of pain should allow for a wake-up test. Postoperative considerations include adequate pain control with no respiratory depression, minimal sedation, and no untoward gastrointestinal symptoms. A multimodal approach to pain control is often employed, using opioids such as remifentanil or sufentanil and N-methyl-
A 2016 study by Gornitzky et al 6 described the development of a clinical pathway focused on improving the treatment of pain while reducing hospital recovery time of these patients. They noted improved mean daily pain scores during the first 3 postoperative days as well as faster time to discontinuation of patient-controlled analgesia pain management, urinary catheter removal, and discharge to home. They also saw a decrease in opioid consumption and need for treatment of related side effects.
We designed a historically controlled study to evaluate if this clinical pathway could offer a pragmatic approach to standardizing care for AIS patients and yield similar benefits at our tertiary care center. In addition, this pathway offered the opportunity to introduce methadone into perioperative care. We hypothesized that use of the pathway would shorten emergence times, improve postoperative analgesia, and decrease hospital length of stay. We compared a prospective group of 14 patients scheduled for AIS-related posterior spinal fusion on the clinical pathway to a retrospectively collected group of 25 patients previously cared for by the same surgical staff, but in the absence of a standardized approach.
Methods
Cohorts
This study was approved by the University of Florida Institutional Review Board under IRB201500120 for the prospective cohort and under IRB201600071 for the historical control group. The prospective arm of the trial was registered at clinicaltrials.gov under NCT02481570. The study adhered to the TIDieR checklist. Fifteen adolescent patients who presented to our facility between 2015 and 2017 for surgical correction of idiopathic scoliosis involving multilevel posterior spinal fusion gave written consent for the prospective arm of the study. The patients were ASA Physical Status Class I or II. Patients undergoing current treatment with opioids, α-2 agonists, anticonvulsants, or antidepressants were excluded from the study. One patient was excluded from the experimental group prior to surgery because features of his presentation suggested a neuromuscular cause of his scoliosis.
The historical controls were 25 adolescents who underwent idiopathic scoliosis correction at the same facility between 2013 and 2015 under the care of the same surgeon.
Pathways
For patients in the historical control group, there was no specific protocol for premedication, intraoperative anesthesia, or routine postoperative care aimed at pain control and mobilization.
A comprehensive perioperative clinical pathway modeled after the rapid recovery pathway 4 developed at the Children’s Hospital of Philadelphia (CHOP; Table 1) was adopted for the prospective experimental group. Preoperatively, patients received oral gabapentin and acetaminophen. Premedication with anxiolytics was at the discretion of the attending anesthesiologist. Intraoperative anesthesia was provided via total intravenous anesthesia with a combination of propofol and remifentanil infusions and a single dose of intravenous methadone. The initial dose of methadone (0.4 mg/kg lean body weight [LBW]) was chosen based on a literature review and pharmacokinetic simulation; the dose was subsequently reduced to 0.3 mg/kg LBW and then 0.25 mg/kg LBW to facilitate emergence and a potential intraoperative wake-up test. Timing and doses of remifentanil, propofol, and methadone were entered into an optimization algorithm that employed pharmacokinetic and pharmacodynamic model estimations to balance adequate analgesia with rapid emergence. 7 -9 Each patient was extubated according to standard clinical criteria and after following commands. Emergence time was calculated as the difference between the time of extubation and the time at which anesthetic agents were discontinued. The postoperative phase of care for these patients occurred according to the schedule displayed in Table 1.
Clinical Pathway.
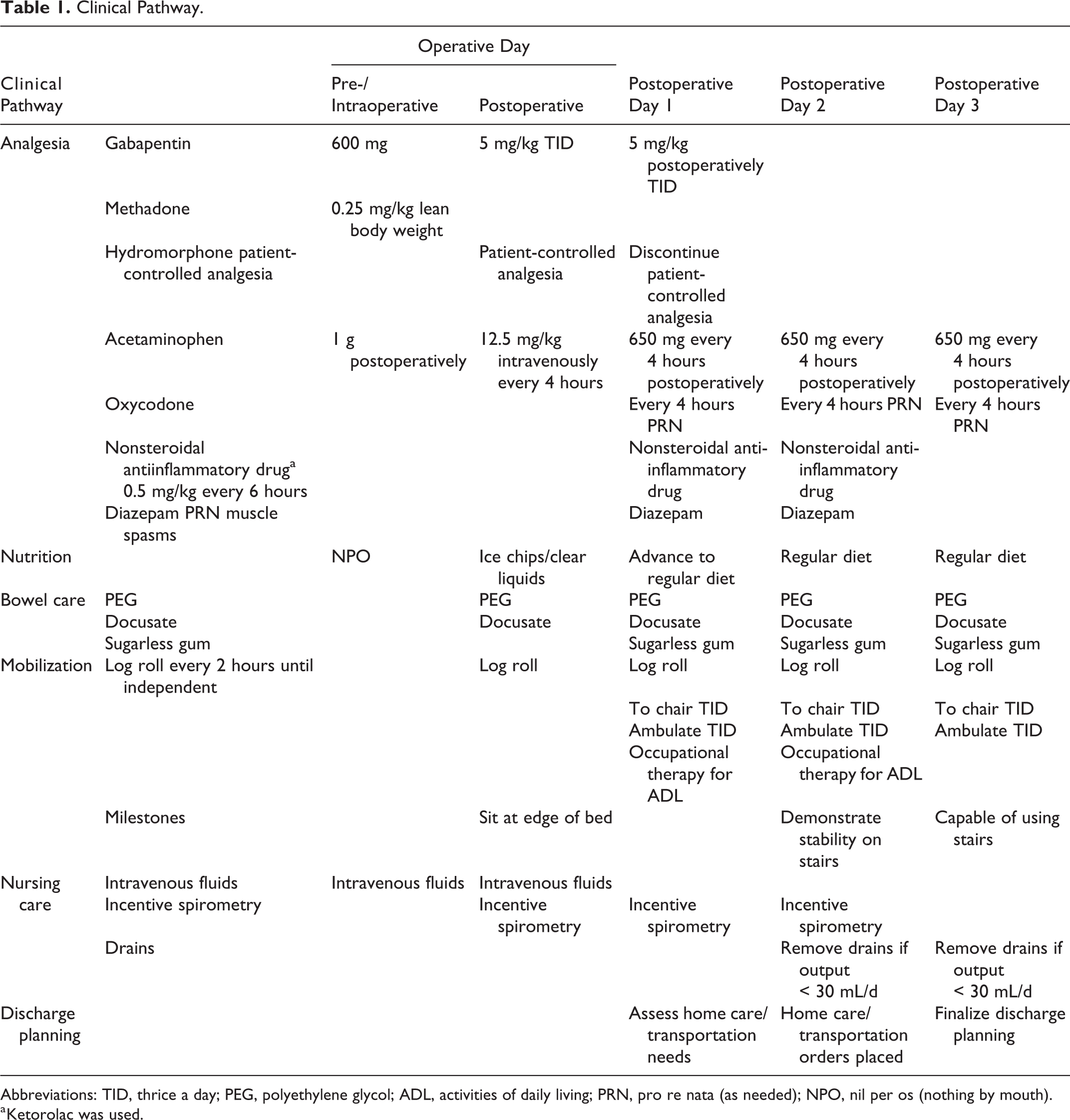
Abbreviations: TID, thrice a day; PEG, polyethylene glycol; ADL, activities of daily living; PRN, pro re nata (as needed); NPO, nil per os (nothing by mouth).
a Ketorolac was used.
Statistical Analyses
All data was collected retrospectively via review of patient electronic medical records to avoid bias and to more closely mirror data available from the control group. Intraoperative opioid doses were converted to fentanyl infusion equivalents using American Pain Society guidelines 10 based on the following ratios: remifentanil (0.8:1), sufentanil (0.2:1), fentanyl (1:1), and methadone (100:1). Postoperative opioid doses were converted to intravenous morphine equivalents based on the following ratios: oxycodone (2.5:1), hydromorphone (0.25:1), morphine (1:1), and methadone (1:1). 11 Methadone was included in the intraoperative opioid dose. On transport to the pediatric intensive care unit, the nurse collected pain scores per standard protocol.
Comparisons between the experimental group (combined dose) and the historical control group were done with a 2-tailed t test. Comparisons among high-dose (0.3 or 0.4 mg/kg) and low-dose (0.25 mg/kg) methadone groups and the historical controls were performed with analysis of variance (ANOVA; F test). If data violated the normality assumption, nonparametric tests (Mann-Whitney U test for 2 groups and ANOVA on ranks [H test] for 3 groups) were performed. If data violated the equal variance assumption, Welch’s correction was performed. Pain scores over time were compared with 2-way repeated-measures ANOVA. Because of unequal variances, opioid consumption was compared between the experimental group and historical controls by way of multiple t tests with Welch’s correction for unequal variances and Bonferroni correction for multiple comparisons. P values <.05 were considered statistically significant. A power analysis was based on a study by Gottschalk et al, 12 which showed a greater than 50% reduction in morphine use on postoperative day 1 for patients treated intraoperatively with methadone compared with those treated with sufentanil. With alpha set to 0.05, beta to 0.2, and a power of 0.8, we found that a study population of 11 experimental and 11 control patients was sufficient to perform statistical tests. A study of 15 experimental patients was developed to ensure adequate subjects were enrolled.
Results
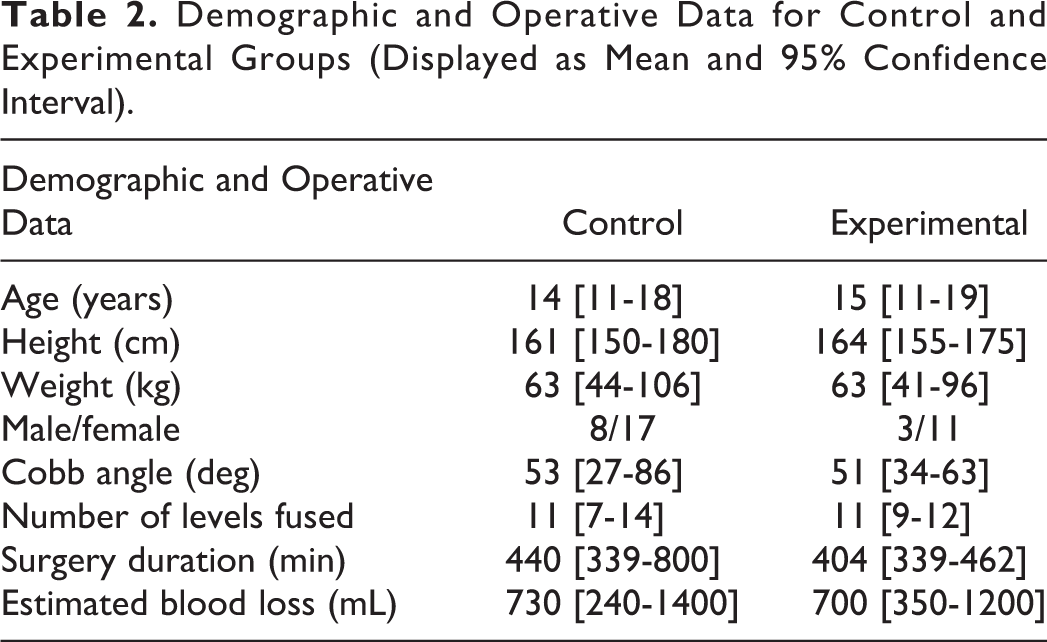
Both study groups were similar in patient demographics and details of the severity of scoliosis and its correction (Table 2). There was a higher percentage of males in the control group, likely a result of random sampling.
Demographic and Operative Data for Control and Experimental Groups (Displayed as Mean and 95% Confidence Interval).
As expected, the anesthetic regimens of the historical control group were much more heterogeneous than those of the experimental group (Table 3). In the historical control group, no patients received preoperative acetaminophen and only three received preoperative gabapentin. Nearly one-half (11 of 25) of the control patients received ketamine infusions, and 2 received infusions of dexmedetomidine. The added anesthetic and analgesic effect of ketamine or dexmedetomidine, combined with the absence of methadone, likely accounts for the lower overall intraoperative opioid use in the historical control group. The intraoperative opioids used were remifentanil, fentanyl, sufentanil, morphine, and hydromorphone in 8, 14, 17, 5, and 10 historical controls, respectively. There was no statistically significant difference in the use of propofol between the historical control group and the experimental group.
Intraoperative Data, Including Surgical Duration and Anesthetic Infusion Rates (Mean and 95% Confidence Interval).
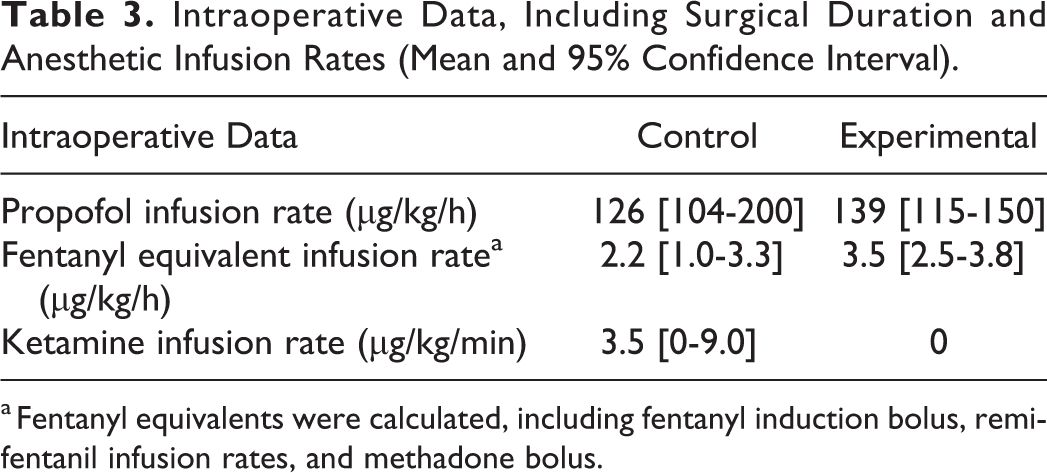
a Fentanyl equivalents were calculated, including fentanyl induction bolus, remifentanil infusion rates, and methadone bolus.
For comparisons of emergence and postoperative opioid consumption to the historic control group, the experimental group was analyzed as a whole (Table 4) and separated into 2 groups based on the methadone dose (Table 5). Emergence times for patients receiving >0.25 mg/kg methadone (n = 5) were prolonged compared to emergence times in patients receiving 0.25 mg/kg methadone. One of the patients (>0.25 mg/kg methadone) could not be extubated in the operating room and was extubated on arrival in the intensive care unit. A methadone dose >0.25 mg/kg nearly eliminated the need for supplemental opioids in the first 24 hours after surgery. Pain scores and opioid consumption were reduced in all patients who received methadone when compared with historical controls. A methadone dose of 0.25 mg/kg resulted in a consistently rapid emergence within 7 minutes of discontinuing propofol and remifentanil.
Clinical Results for the Control and Combined (High and Low Methadone Dose) Experimental Group.
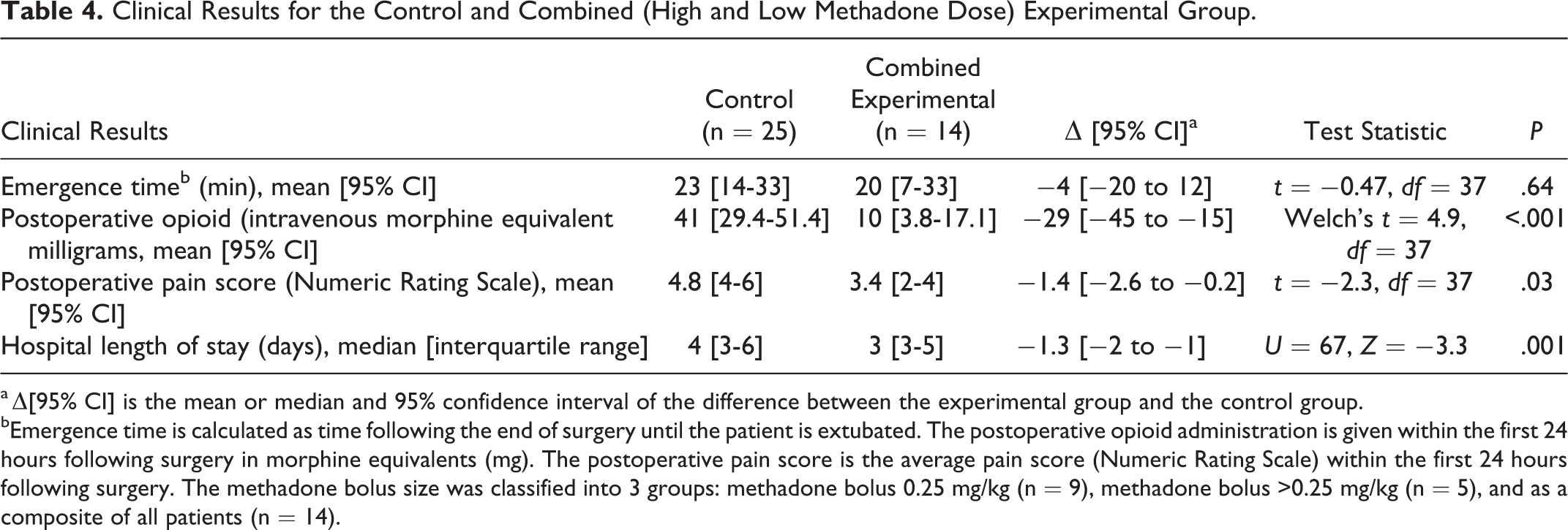
a Δ[95% CI] is the mean or median and 95% confidence interval of the difference between the experimental group and the control group.
bEmergence time is calculated as time following the end of surgery until the patient is extubated. The postoperative opioid administration is given within the first 24 hours following surgery in morphine equivalents (mg). The postoperative pain score is the average pain score (Numeric Rating Scale) within the first 24 hours following surgery. The methadone bolus size was classified into 3 groups: methadone bolus 0.25 mg/kg (n = 9), methadone bolus >0.25 mg/kg (n = 5), and as a composite of all patients (n = 14).
Clinical Results for the Control and High- and Low-Dose Methadone Experimental Groups.
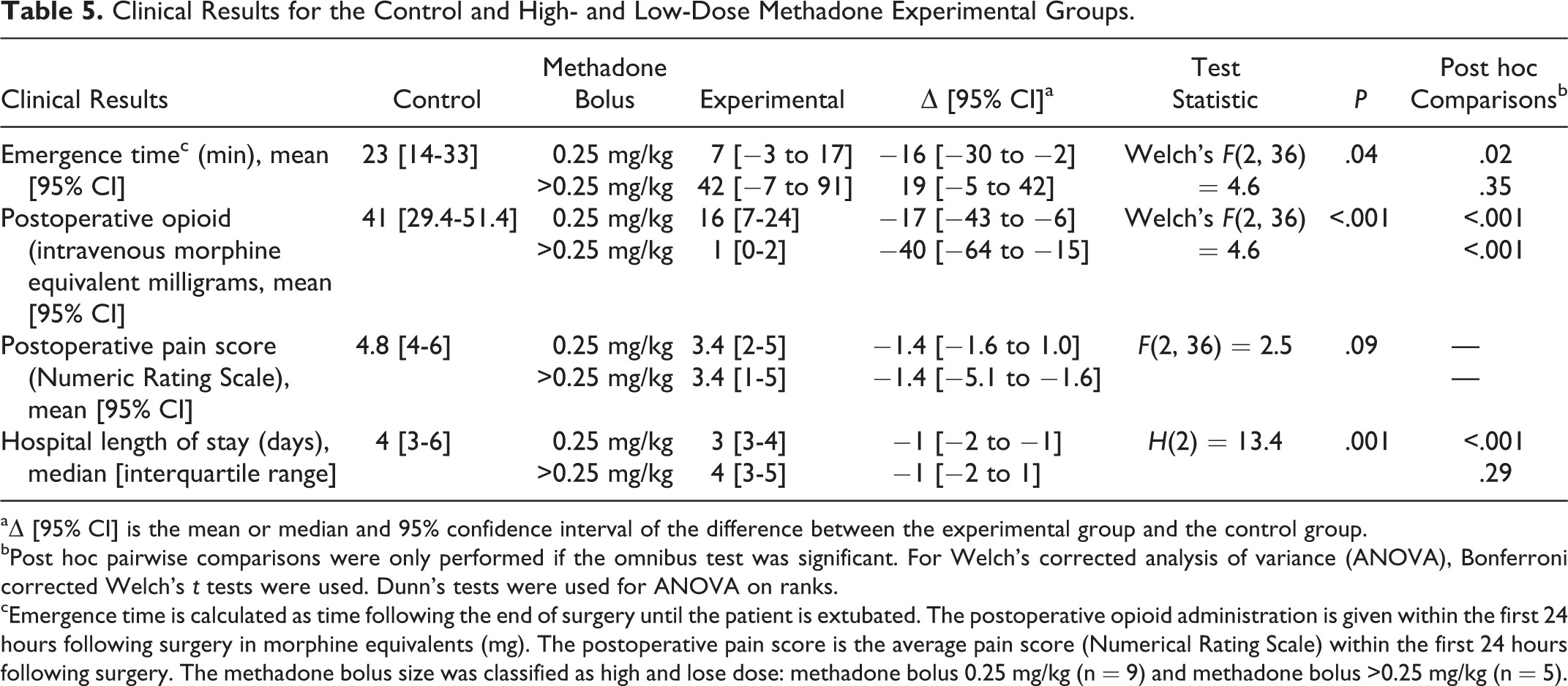
aΔ [95% CI] is the mean or median and 95% confidence interval of the difference between the experimental group and the control group.
bPost hoc pairwise comparisons were only performed if the omnibus test was significant. For Welch’s corrected analysis of variance (ANOVA), Bonferroni corrected Welch’s t tests were used. Dunn’s tests were used for ANOVA on ranks.
cEmergence time is calculated as time following the end of surgery until the patient is extubated. The postoperative opioid administration is given within the first 24 hours following surgery in morphine equivalents (mg). The postoperative pain score is the average pain score (Numerical Rating Scale) within the first 24 hours following surgery. The methadone bolus size was classified as high and lose dose: methadone bolus 0.25 mg/kg (n = 9) and methadone bolus >0.25 mg/kg (n = 5).
All patients treated with the clinical pathway demonstrated improved postoperative analgesia (Figure 1). Numeric Rating Scale (NRS) pain scores during the first 24 postoperative hours were reduced by 30% (t = −2.3, df = 37, P = .03, mean difference = −1.4, 95% CI −2.6 to −0.2). This trend of significantly decreased NRS scores was apparent through the first postoperative day. The decrease in NRS scores observed in postoperative days 2 and 3 were not statistically significant. The improved postoperative analgesia for the patients treated on the clinical pathway correlates with observed decreases in supplemental opioid pain medication at postoperative day 0 (t = 4.85, df = 35, P < .001, mean difference = −30.0, 95% CI −45.2 to −14.7) and at postoperative day 2 (t = 2.74, df = 29, P = .041, mean difference = −13.6, 95% CI −26.5 to −0.6). Although not statistically significant at all measured intervals, the decrease in opiate use ranged from 25% to 66%.
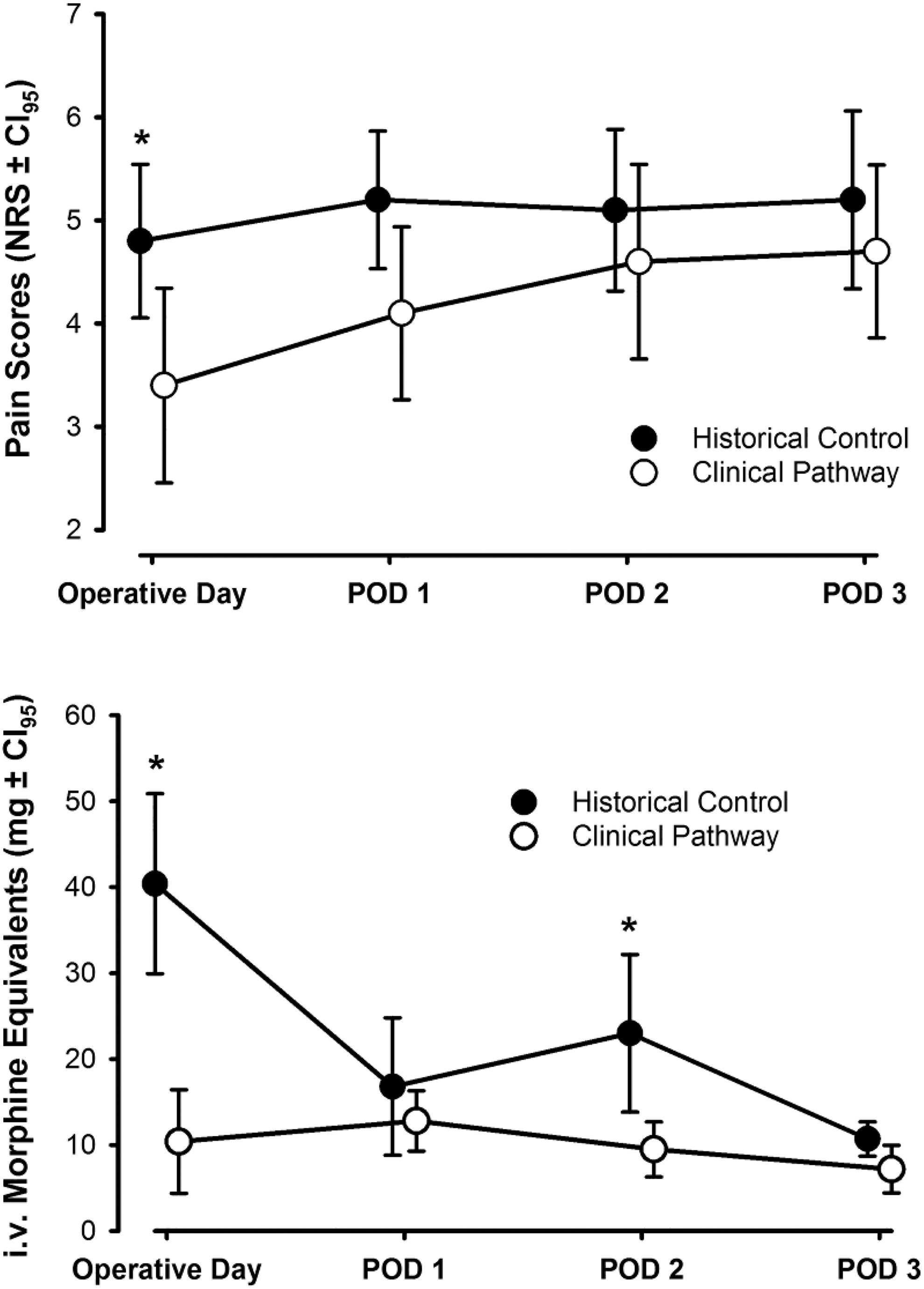
Pain scores and opioid consumption in the postoperative period. Data is depicted as means and 95% confidence intervals. Pain scores were lower in the group treated on the clinical pathway, although the difference was not clinically or statistically significant beyond the day of surgery. Opioid consumption was significantly lower in patients treated on the clinical pathway compared with historical controls. *P < .05 compared with clinical pathway.
The expectations by patients and staff for milestones in the pathway as well as the improved analgesia despite aggressive structured mobilization facilitated recovery. The length of stay was decreased significantly in patients treated with the new clinical pathway (Tables 4 and 5).
Three of the patients in the control group received preoperative gabapentin and 8 received it postoperatively. In those who received gabapentin treatment, mean morphine equivalent usage was 22 (0-44) mg, whereas those who did not receive postoperative gabapentin used more morphine equivalents (65 [19-75] mg). In those patients from the control group receiving postoperative gabapentin, their usage of supplemental opioid pain medications was similar to that in the experimental group receiving 0.25 mg/kg methadone and gabapentin.
Discussion
The comprehensive clinical pathway for patients undergoing posterior spinal fusion for correction of AIS improved care compared with historical controls at our institution. The pathway resulted in decreased preoperative and intraoperative variability, decreased postoperative use of adjunctive opioid pain medication, improved patient comfort, and decreased hospital length of stay. The clinical pathway allowed the successful introduction of methadone into perioperative care.
The introduction and adoption of clinical pathways can decrease complications and length of stay for many different illnesses and surgical cases. 6,13 The intraoperative care of patients undergoing AIS correction was highly variable prior to pathway implementation, adversely affecting patient comfort and ability to engage in postoperative therapy, likely increasing the variability in hospital length of stay.
This pathway was constructed to maximize treatment of postoperative pain around the concept of “preemptive analgesia”
12,14
and an opioid-sparing, multimodal approach that combines comprehensive preoperative, intraoperative, and postoperative care. Methadone was selected for its prolonged effect on µ opioid receptors and its N-methyl-
Our experience with this pathway parallels that of its initial roll-out at CHOP. 6 In particular, we corroborated their findings of clinically meaningful improvements in postoperative pain during the first 24 hours as well as reduced opioid consumption during that same time frame. There was an overall trend toward decreased pain scores and opioid use throughout all time periods compared with the historical control. Unlike the initial roll-out at CHOP, we did not require a 6-month training period for staff, attesting to the well-integrated nature of this pathway. Nonetheless, we observed a similarly shortened hospital length of stay. Our findings add to the available information gleaned by the CHOP study because methadone did not significantly increase the emergence time at the 0.25 mg/kg dose, but rather made emergence time shorter. We also showed a trend toward improved pain control with the >0.25 mg/kg dose at the expense of prolonged emergence. Additionally, we were able to show that the clinical pathway does improve care in an institution with a much smaller AIS surgical population.
Our findings are consistent with those of Gottschalk et al 12 with regard to the methadone-induced decrease in postoperative NRS pain scores and decreased use of supplemental narcotic pain medication. However, they reported the greatest reduction in narcotic use in the 48- to 72-hour postoperative period. We observed significant differences in pain scores only within the first 24 hours. We hypothesize that the gains in pain control on postoperative days 2 and 3 were offset by the aggressive structured mobilization prescribed by the clinical pathway. There was a trend toward decreased pain scores in the experimental group throughout all time points. Postoperative opioid use also decreased throughout the measured intervals but only significantly so within the first 24 hours and on the second postoperative day. Of note, our use of gabapentin stopped on postoperative day 1. We are unsure if continuation would result in any additional benefit.
Our study has limitations. First, we reduced the methadone dose during the course of our study. Long, variable emergence times, likely due to the synergy between methadone and gabapentin 15 , prompted the dose reductions. Only the first patient received 0.4 mg/kg. The next four patients received 0.3 mg/kg before we settled on 0.25 mg/kg for the remaining patients. As desired, at the 0.25 mg/kg dose, the emergence time was significantly shorter than at the higher doses of methadone or in the control group. When the patients treated on the clinical pathway were separated into 2 groups (0.25 mg/kg vs >0.25 mg/kg methadone), the 0.25 mg/kg methadone group did show greater need for supplemental opioids, but there was no reduction in patient comfort. Our study size (n = 39) for 2 groups was similar to a previous spinal correction study using methadone. 13 Though there appears to be a distinctive statistical difference in emergence time and postoperative opioid consumption for the 2 experimental study populations, the low group subject number deemphasizes the findings. A second limitation is that our study relied on medical record documentation for all postoperative data. We chose this approach for pragmatic reasons and because it reflects real-world practice. Furthermore, any missing information or bias should have affected the historical control group and the clinical pathway groups similarly, as these were consecutive patients operated on by the same team across a 4-year span. A third limitation in our study is the gender distribution between the 2 groups. It is widely accepted that there is a female predominance within the population of people with progressive scoliosis; however, our control group was made up of a relatively larger percentage of males. This was unexpected and likely due to random sampling of a small population. It has been reported that males emerge from propofol anesthesia more slowly than females, 16 but the significance of the difference of emergence times between the 2 groups due to the gender disproportions is unknown. Furthermore, pain may be perceived differently between sexes.
In summary, a comprehensive clinical pathway featuring a single dose of methadone with a standardized intraoperative anesthetic regimen combined with preoperative “preemptive analgesia” and postoperative standardized treatment reduced patient pain ratings, use of adjunctive pain medication, and hospital length of stay. No obstacles to the implementation of the pathway were identified.
Footnotes
Acknowledgments
The authors thank Dr Terrie Vasilopoulos, PhD, for her statistical support and Corey Astrom, ELS, for her editorial expertise.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by departmental resources from the University of Florida Departments of Anesthesiology and Orthopaedics.
