Abstract
Study Design:
Retrospective cohort study.
Objective:
To determine the rates of perioperative complications in patients undergoing anterior cervical discectomy and fusion (ACDF) with allograft versus synthetic cage.
Methods:
A large national administrative health care database was queried for ACDF procedures performed between 2007 and 2014 using ICD-9 (International Statistical Classification of Diseases, 9th revision) and CPT (Current Procedural Terminology) codes. Cases that utilized structural allograft and synthetic cages were identified via CPT codes. Gender, age, frequency of obesity, cigarette use, diabetes, and number of levels fused were compared between the 2 cohorts using χ2 test. Complications within 90 days were identified via ICD-9 codes and compared between the 2 cohorts. Revision rates within 2 years were noted.
Results:
A total of 10 648 ACDF cases using synthetic cages and 7135 ACDFs using structural allograft were identified. The demographics between the 2 cohorts were similar. Overall complication rate was 8.71% in the synthetic cage group compared with 7.76% in the structural allograft group (P < .01). Use of synthetic cage was associated with higher rate of respiratory complications, 0.57% compared with 0.31% in the structural allograft cohort (P = .03), while use of structural allograft was associated with a higher rate of dysphagia, 0.64% compared with 0.33% (P < .01). Revision rate at 2 years was 0.50% and 0.56% in the synthetic cage and allograft groups, respectively (P = .03).
Conclusions:
This data suggests that synthetic cages are associated with a marginally higher overall rate of complications with similar revision rates.
Keywords
Introduction
Anterior cervical discectomy and fusion (ACDF) has been the gold standard for the treatment of patients with severe cervical spondylosis resistant to conservative management. 1,2 Originally described in the 1950s, the surgery involves an anterior approach to the cervical spine, followed by discectomy, and an interbody graft insertion. 1,3 Over the past 6 decades, the technique and available technologies for ACDFs have rapidly evolved.
Initial description of ACDFs did not include anterior instrumentation; today an anterior plate and screws are commonly used as adjuncts for stabilization in ACDF surgery. The other aspect that has seen rapid evolution is the availability of graft options for ACDFs. Classically, iliac crest bone graft (ICBG) was used as a dowel or wedge. This served as a structural autograft. The advantage of ICBG autograft is that it is the only structural graft option that provides the 3 key characteristics of a graft: osteoinductivity, osteoconductivity, and osteogenic potential. 4 -6 The downside of ICBG is significant donor site morbidity. Between 9% and 40% of patients have been reported to have significant donor site pain, and up to 10% will have continued pain 1 year after surgery. 7 -12
Advances in biologics have been aimed at achieving similar benefits to ICBG while avoiding the associated morbidity of autograft harvest. Allograft and synthetic cages are 2 common adjuncts used in ACDFs in place of autograft. Allografts have the advantage of avoiding donor site morbidity and provide a high degree of osteoconductivity with variable amounts of osteoinductivity. Another option for structural grafts are synthetic cages, composed of polyetheretherketone (PEEK), titanium, or carbon fiber. These cages offer an osteoconductive scaffold and can be packed with nonstructural graft in order to achieve fusion. The concern with synthetic cages is cost, the potential for subsidence which has been cited as high as 29%, as well as high rates of dysphagia. 13
While both allograft and synthetic cages are commonly used structural graft options in the setting of ACDF, there is a paucity of evidence comparing the complication rates between these 2 graft options. The aim of this study is to use a large national sample to evaluate the complication rates between structural allograft and synthetics cages when used in ACDF.
Methods
Orthopedic subset within the Humana database (PearlDiver Technolgies Inc, Warsaw, IN) was queried for patients undergoing ACDF between 2007 and 2014 using International Statistical Classification of Diseases, 9th Revision (ICD-9), and Current Procedural Terminology (CPT) codes. The Pearl Diver database is de-identified and does not require the approval of an institutional review board for use. Patients undergoing ACDF procedure were selected using CPT codes 22 551 (anterior interbody fusion, discectomy, decompression) or 22 554 (anterior cervical fusion) and 63 075 (anterior cervical discectomy). Cases that utilized structural allograft and synthetic cages were identified via CPT codes (22 851 and 20 931, respectively). Gender, age, frequency of obesity, cigarette use, diabetes, and number of levels fused were compared between the 2 cohorts using χ2 test. These variables were chosen because they have been previously identified as correlating with frequency of complications.
The following complications were identified via ICD-9 codes: cardiac, respiratory, deep venous thrombosis or pulmonary embolism, central nervous system, hematoma/seroma, accidental puncture of nerve or blood vessel, wound complications, postoperative infection, hoarseness, and dysphagia. Complications within 90 days of surgery were collected in this study. Data was also collected on revision surgery within 1 and 2 years.
In addition to the above, data outputs included the number of ACDF procedures per year, patient age at the time of surgery (broken down into 5-year intervals from 0 to 95 and older), patient gender, geographical regions within the United States (West, East, South, and Northeast), and the surgical location (inpatient, outpatient, or ambulatory center). Data was reported as total numbers as well as percentages in the given year or region. For statistical analysis, the χ2 test was used to calculate the difference between the various scaffolds in costs, demographics, and annual trends. A logistic regression model was used to evaluate whether graft type was an independent predictor of any complication. Variables the model adjusted for included age, gender, Charlson Comorbidity Index, as well as number of levels fused.
Results
A total of 10 648 ACDF cases using synthetic cages and 7135 ACDFs using structural allograft were identified in the Humana database between 2007 and 2014. The demographics in terms of age and gender of the structural allograft cohort and the synthetic cage cohorts are similar (Table 1). There was no statistically significant difference in obesity and tobacco use between the 2 groups (Table 2). Diabetes was marginally more prevalent in the synthetic cage cohort (25.76% vs 23.91%, P < .01).
Comparison of Demographics Between the Synthetic Cage and Structural Allograft Cohorts.
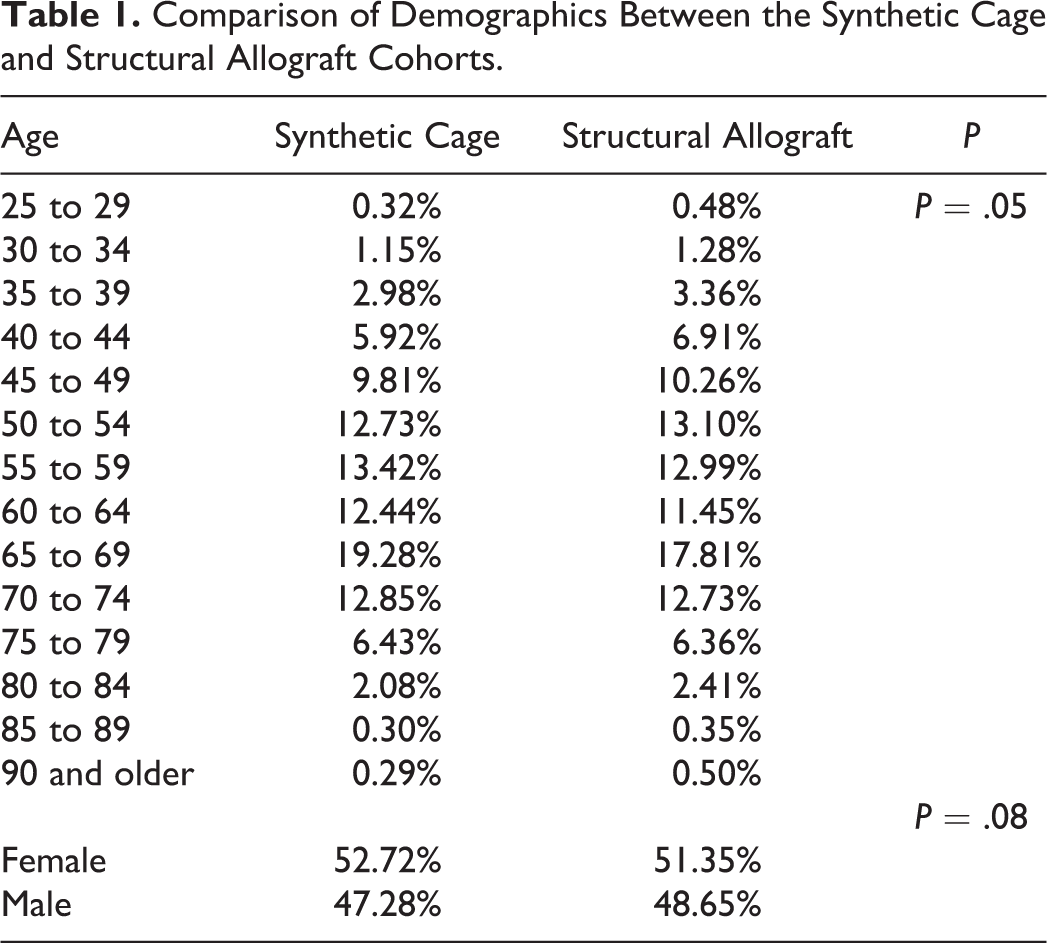
Rates of Obesity, Tobacco Use, Diabetes, and Number of Levels Fused in the Synthetic Cage and Allograft Cohorts.
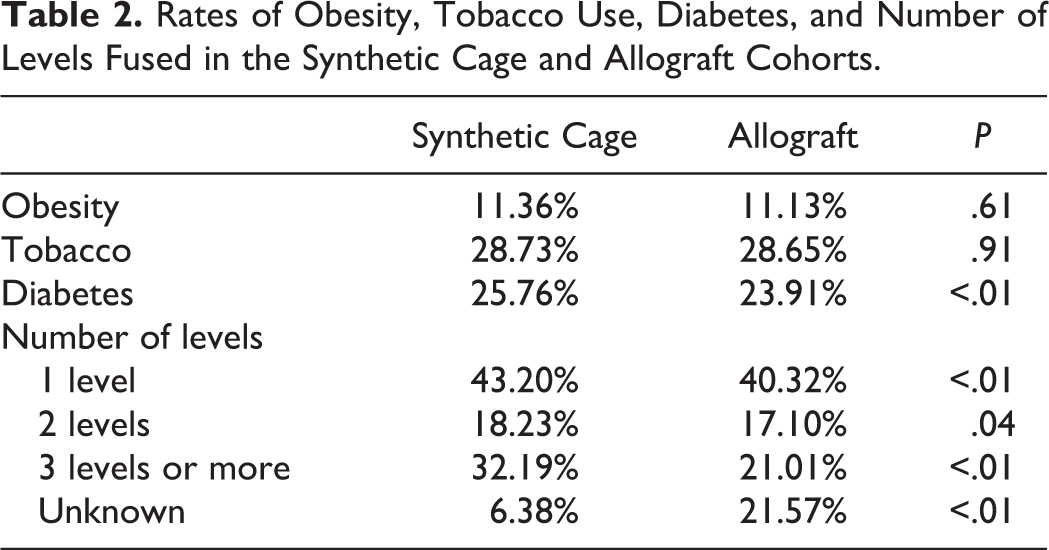
A higher proportion of cases using allograft had missing data regarding number of levels fused. Synthetic cages were more frequently used in cases requiring 3 or more levels (32% vs 21%, P < .01). While synthetic cages had higher percentages in each category of number of levels fused, the 3 or more levels category displayed the largest difference between the 2 cohorts. Peak age group for both structural allograft and synthetic cage is 65 to 69 years, with 19.28% of the synthetic cage cohort and 17.81% of the allograft cohort fitting within this age group. The gender distribution of the 2 cohorts was also similar, with women comprising 52.7% of the synthetic cage group and 51.4% of the structural allograft group (P = .07).
Overall complication rate was 8.71% in the synthetic cage group compared with 7.76% in the structural allograft group. The majority of the specific complications including cardiac complications, venous thromboembolic events, central nervous system–related complications, postoperative hematoma/seroma, accidental laceration of nerve or blood vessel, wound infections, postoperative infection, and hoarseness showed no statistically significant difference between the 2 cohorts (Table 3). Use of synthetic cage was associated with higher rate of respiratory complications, 0.57% compared with 0.31% in the structural allograft cohort (P = .03), while use of structural allograft was associated with a higher rate of dysphagia, 0.64% compared with 0.33% (P < .01).
Comparison of Complication Rates Between the Synthetic Cage and Structural Allograft Cohorts.a
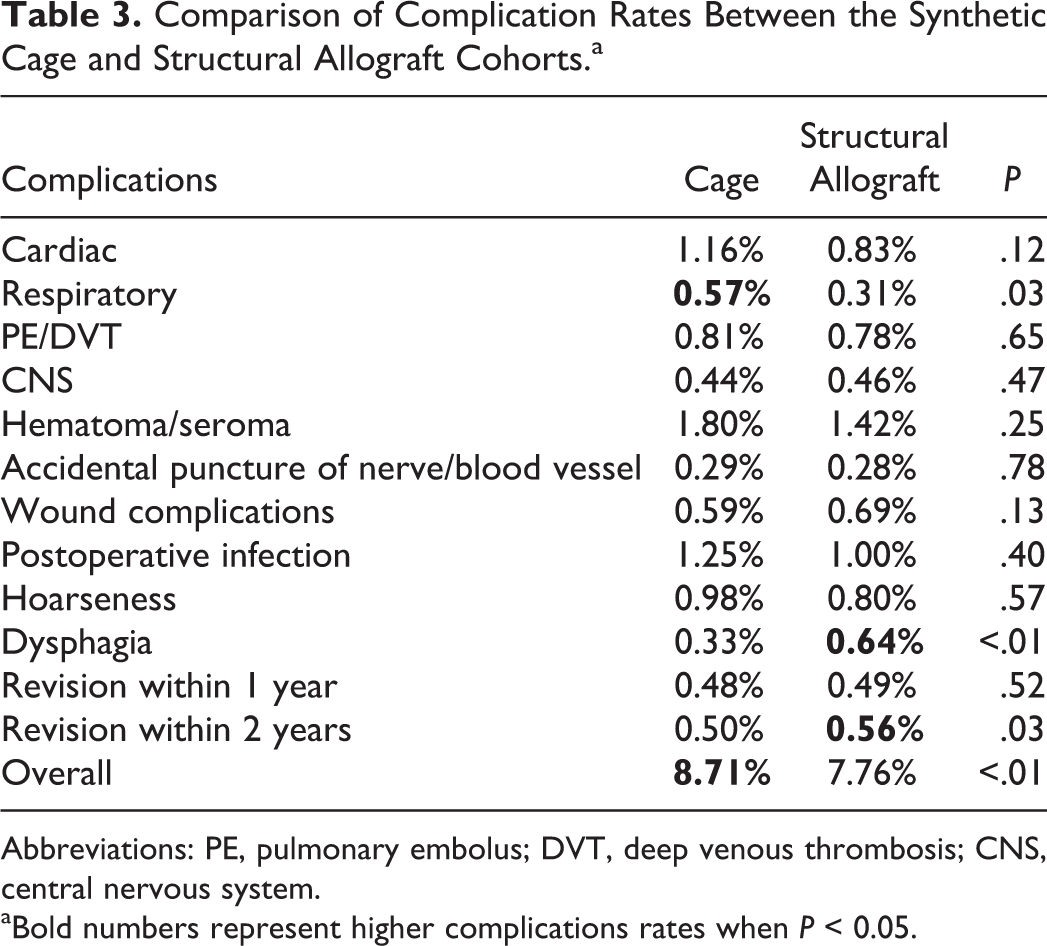
Abbreviations: PE, pulmonary embolus; DVT, deep venous thrombosis; CNS, central nervous system. aBold numbers represent higher complications rates when P < 0.05.
The analysis also investigated rate of revision within 1 and 2 years. There was no statistically significant different between revisions within 1 year. The structural allograft cohort had a statistically significantly higher rate of revision at 2 years, with 0.56% of cases being revised compared with 0.50% in the synthetic cage group. The logistic regression (Table 4) showed that graft type was not an independent predictor of developing a postoperative complication (odds ratio = 1.08, P = .21).
Output of Logistic Regression Model With Any Complication as the Dependent Variable.a
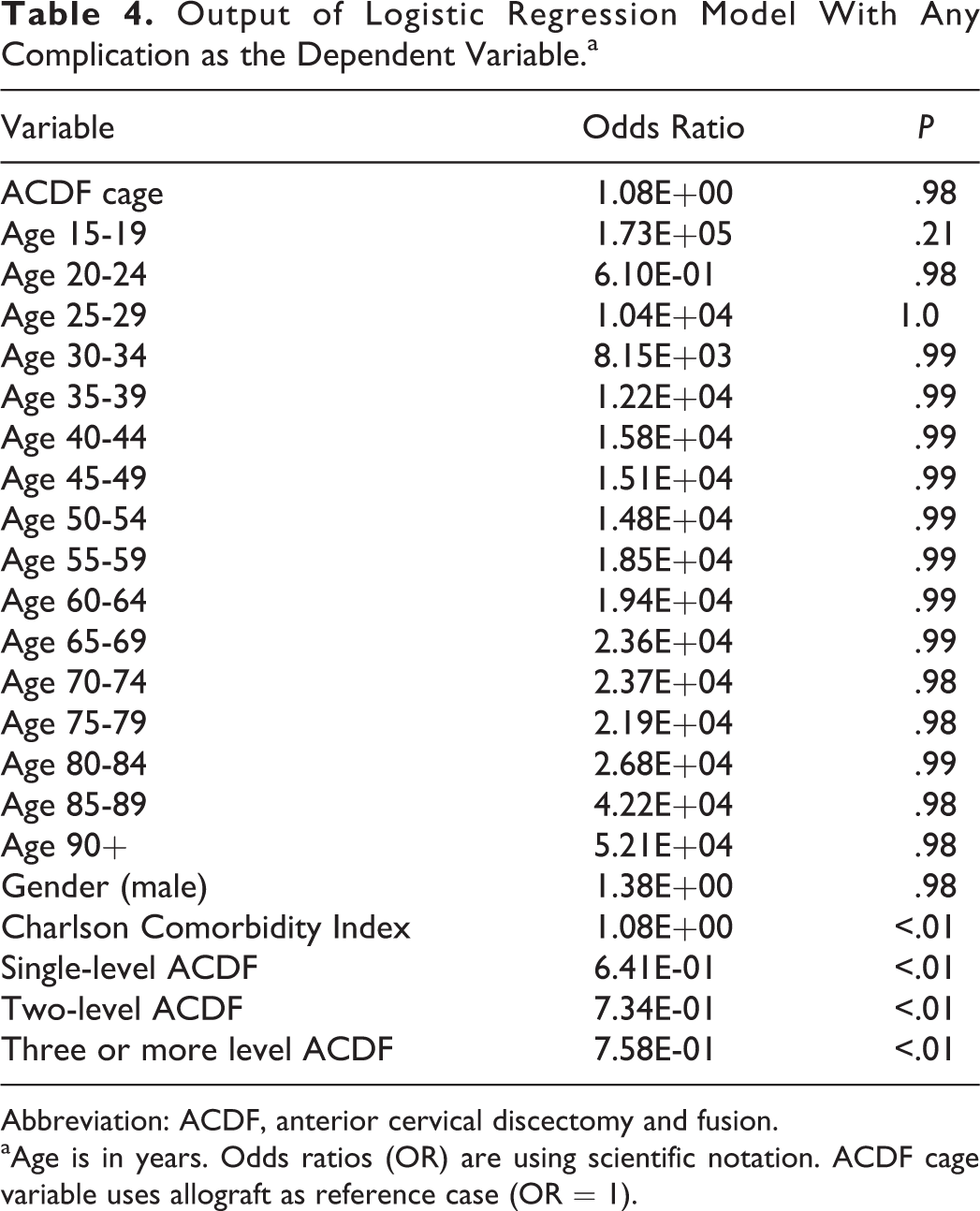
Abbreviation: ACDF, anterior cervical discectomy and fusion.
aAge is in years. Odds ratios (OR) are using scientific notation. ACDF cage variable uses allograft as reference case (OR = 1).
Discussion
Structural allograft and synthetic cages are 2 viable graft options for ACDF surgery that avoid the significant morbidity and chronic pain associated with the harvest of ICBG. 7 Each option is associated with a unique profile of pros and cons. Synthetic cages provide a structurally stable scaffold that can be packed with nonstructural graft. Synthetic cages have been shown to have good results in terms of fusion when compared with autograft and allografts. This comes with the downside of cost, and with some studies showing higher rates of dysphagia. 14,15 Hacker et al demonstrated higher fusion rates with ACDF using titanium cage in 1- and 2-level cases compared with uninstrumented allograft and autograft. 16 In this study, the fusion rates were 98% versus 90% for titanium age and allograft/autograft, respectively.
The use of an anterior plate is a pivotal factor to consider when evaluating data on the outcomes of grafts in ACDF surgery. The introduction of anterior plates has substantially increased the fusion rates and decreased subsidence for allografts to a level comparable to that of autograft. 17 -20 While the Hacker data suggests that titanium cages may have a higher fusion rates compared with uninstruments allograft, it is unclear whether that would hold true when instrumented allografts are used.
Allografts are osteoconductive, provide a variable amount of osteoinductivity, but lack the osteogenic potential of autografts. 21 The pros include availability and lack of donor site morbidity, and come at the expense of cost, variation in osteoinductive potential between both manufacturers based on sterilization techniques and donors, as well the potential risk of disease transmission and host rejection. The risk of disease transmission is exceedingly low. Buck et al estimated that the risk of receiving an allograft from an HIV-positive donor is 1 in 1.67 million. 22 Fusion rates of plated allografts appear to be comparable to autograft. 19,20
This study set out to investigate the complication profiles between structural allograft and synthetic cages in the setting of ACDF. The Humana database was used for this purpose. The database contains over 22 million patient records, spanning from 2007 to 2016, with capabilities to track patients longitudinally. The significant size and broad scope of the database afford a great degree of extrinsic validity to studies that use this data. The presented analysis suggests that synthetic cages are associated with a higher rate of overall complications, 8.71% versus 7.76%. The majority of this difference is due to a higher rate of respiratory complications with synthetic cages. Structural allograft, on the other hand, is associated with more dysphagia, and a statistically significantly higher rate of revision within 2 years by 0.06%.
While the presented analysis finds a statistically significant difference in complication and revision rates on univariate analysis, the 0.95% and 0.06% differences, respectively, are likely considered clinically insignificant by most. Furthermore, regression analysis correcting for age, gender, comorbidity burden, and number of levels fused found that structural graft choice was not an independent predictor of complications. This data suggests that other factors such as cost should be weighed more heavily than complication rates when choosing between synthetic cage and allograft for structural graft in the setting of an ACDF.
It is important to note that the synthetic cage cohort shows a higher rate of diabetes and higher proportion of 3 or more level fusion. In this setting, a comparable revision rate may be seen as being in favor of the synthetic cage cohort. These results are similar to a previous cohort that compared the outcomes of 37 titanium cages to that of 94 allografts and found no difference in complication rates. 23 The revision rate in this study appears to be much lower than the revision rates reported for both cervical disc arthroplasty and ACDF in studies comparing the 2 procedures. 24,25 This suggests that reporting of revision rates in this administrative database is likely of lower fidelity compared with the prospective randomized clinical trials used to compare ACDF and cervical disc arthroplasty.
The limitations of this study must be acknowledged. This study was performed using a large administrative database. While this provides the largest sample compiled on this topic, the clinical resolution of the data is limited. The outcomes captured are limited to those that have been appropriately coded for within the database. Clinical details regarding the use of an anterior plate, adjunct use of biologics such as bone morphogenetic protein, duration of surgery, and blood loss are unavailable and have the potential to bias the results of the aforementioned factors if they have an unequal distribution between the 2 cohorts. Furthermore, the baseline demographics of the 2 cohorts are not identical, with age distribution being slightly different between the 2 cohorts (P = .05), which is another source of potential bias within the study. Clinical details of revision surgery, including whether revisions were performed at index level and reason for revision, are also unfortunately unavailable.
The multitude of available options for structural and nonstructural grafts in ACDF surgery has made for complicated decision making regarding the optimal graft choice. While this study provides insight into the complication rates with the use of allograft versus synthetic cage, it is far from identifying the optimal graft choice for ACDF surgery. The large number of permutations of possible structural and nonstructural graft combinations further complicates the logistics of the ideal study to answer this question. Additional large studies are necessary to further elucidate which graft combination provides the ultimate combination of fusion rates, low complications, and low cost.
Footnotes
Authors’ Note
Portions of this work were presented in the form of a podium presentation at the North American Spine Society 2017 in Boston, MA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by AOSpine and departmental funds. AOSpine is a clinical division of the AO Foundation—an independent medically guided nonprofit organization. The AOSpine Knowledge Forums are pathology focused working groups acting on behalf of AOSpine in their domain of scientific expertise. Each forum consists of a steering committee of up to 10 international spine experts who meet on a regular basis to discuss research, assess the best evidence for current practices, and formulate clinical trials to advance spine care worldwide. Study support is provided directly through AOSpine’s Research department.
