Abstract
Study Design
Retrospective propensity-matched cohort study.
Objectives
Aspirin’s role in bone remodeling after spinal fusion is unclear. We evaluated the relationship between aspirin use and pseudoarthrosis after ACDF surgery.
Methods
Data was collected from TriNetX, a large research platform including 67 participating healthcare organizations, to identify single- and multi-level ACDF cohorts stratified by no aspirin, aspirin 81 mg and 325 mg daily use. The primary outcome was diagnosis of pseudoarthrosis. Propensity score matching (1:1 ratio) was performed for demographic/clinical variables, followed by risk analyses to determine odds of outcome at 6-months, 1-year, and 2-years. Reoperation rate (additional spinal fusion) was assessed for cohorts with significant results.
Results
There were significantly lower pseudoarthrosis rates in the 81 mg aspirin group (n = 2504) for single-level ACDF at 6-months, 1-year, and 2-years, and a significantly lower reoperation rate at 2-years, compared to patients not taking aspirin. There were no differences in pseudoarthrosis for single-level ACDF with 325 mg dosing (n = 799) and for multi-level ACDF with 81 mg (n = 2287) or 325 mg (n = 678) dosing.
Conclusions
Patients taking 81 mg aspirin had lower pseudoarthrosis rates at 6-months, 1-year, and 2-years and fewer reoperations at 2-years compared with single-level ACDF patients not taking aspirin. No significant associations between aspirin use and pseudoarthrosis were observed for 325 mg dosing or for multi-level ACDF. These results suggest the hypothesis that 81 mg or 325 mg aspirin does not increase the risk of pseudoarthrosis after ACDF. However, changes to clinical practice are not recommended until prospective clinical studies are completed.
Introduction
Aspirin is a unique nonsteroidal anti-inflammatory drug (NSAID) that irreversibly inhibits cyclooxygenase-1 (COX-1) and COX-2. After cervical disc arthroplasty, NSAIDs are often administered to prevent unwanted arthrodesis and heterotopic ossification.1,2 On the other hand after spinal fusion, NSAIDs are often resumed judiciously, due to their presumed negative impact on successful bony fusion. 3 However, their influence on bone healing and arthrodesis is not well understood, and reports in the literature are conflicting on this question. It has been reported that NSAIDs are not associated with increased risk of nonunion in some studies,4,5 but others report significantly higher rates of pseudoarthrosis.6,7
Compared with other NSAIDs, aspirin has distinctive pharmacological properties. First, aspirin is an irreversible antiplatelet agent used for thrombosis prevention and for improving tissue microcirculation.8,9 Second, aspirin induces more potent inhibition of COX-1 than COX-2 at lower doses (eg, 75-100 mg/day), and this property has been shown to result in preferential decrease in osteoclast activity and upregulation of osteogenic pathways. 10 In addition, aspirin reduces the production of prostaglandins, such as PGE2, for which an association with increased bone mass has been shown.11,12 As a result, low doses of aspirin may produce an anabolic effect on bone remodeling, leading to pathways favoring bone production. Presently, the association between aspirin and bone formation/healing remains understudied. There is no clinical study to date which has investigated the association of aspirin use and pseudoarthrosis after spinal fusion surgery.
Over the past 60 years, life expectancy in the United States has increased to nearly 80 years. 13 In 2019, approximately one in three adults aged 40 and older, and nearly 50% of adults aged 70 or older, reported aspirin use for atherosclerotic cardiovascular disease prevention. 14 The American Heart Association predicts that the prevalence of cardiovascular disease and stroke will increase over the next 30 years to affect more than 184 million adults by the year 2050. 15 Furthermore, the mean age of patients who seek treatment for spinal disorders 16 and the incidence of cervical spine surgery are expected to increase in parallel. 17 Therefore, further research is needed to assess the relationship between aspirin use and outcomes after spinal fusion surgery, including bone fusion and reoperation.
In this study, we investigated (1) whether aspirin is associated with pseudoarthrosis following single and multi-level anterior cervical discectomy and fusion (ACDF), and (2) whether there was a dose-dependent effect on outcomes. Based on current basic scientific understanding of aspirin’s role in bone remodeling and physiology, we hypothesized that aspirin use does not increase rates of pseudoarthrosis following ACDF surgery.
Materials and Methods
Study Design
This study is a retrospective propensity-matched cohort analysis using data from health care organizations in the United States enrolled in TriNetX, a global federated health research platform which aggregates de-identified electronic health record (EHR) data compliant with the Health Insurance Portability and Accountability Act (HIPAA). TriNetX includes diagnostic, procedural, medication, and laboratory codes and values from a large consortium of participating health care centers, including our own institution. 18 The database was queried using International Classification of Disease 10th revision codes (ICD-10), Current Procedural Terminology (CPT), and RxNorm Concept Unique Identifier codes (RxNorm).
Cohort Selection
Data was collected from TriNetX. At the time of data collection, there were 67 HCOs in the network including our center, with data available from over 106 million patients. An initial query was performed which identified 88 488 total patients who underwent anterior cervical discectomy and fusion (ACDF) using CPT code 22551. To identify adult patients who underwent single-level ACDF, procedural codes were used for inclusion and exclusion criteria, and parameters were set for age ≥ 18 years. Procedural codes for anterior cervical interbody arthrodesis below C2 (CPT 22551) and anterior instrumentation by anterior vertebral body plating (CPT 22845) were included. Codes for additional levels of anterior interbody arthrodesis below C2 (CPT 22552), posterior cervical arthrodesis below C2 (CPT 22600), additional levels of posterior cervical arthrodesis below C2 (CPT 22614), and posterior instrumentation (CPT 22840) were excluded. For patients who underwent multi-level ACDF, procedural codes for anterior cervical interbody arthrodesis below C2 (CPT 22551), additional levels of anterior interbody arthrodesis below C2 (CPT 22552), and anterior instrumentation (CPT 22845) were included. Codes for posterior cervical arthrodesis below C2 (CPT 22600), additional levels of posterior cervical arthrodesis below C2 (CPT 22614), and posterior instrumentation (CPT 22840) were excluded.
These cohorts were further stratified by aspirin use. The medication code for aspirin (RxNorm 1191) was used to define medication exposure. This code was incorporated as an exclusion criterion for cohorts not taking aspirin. To identify cohorts taking aspirin, this code was incorporated into the inclusion criteria and stratified by dosing parameters for 81 mg and 325 mg use daily. To capture active medication use, aspirin exposure was further specified such that its use must have been documented within 3-months both prior to and following surgery.
Variable Selection
Demographic variables were generated for each cohort of patients. Age at index surgery as well as male and female sex were selected for analysis. Using ICD-10 codes, diagnoses of osteoporosis (ICD-10 M81), tobacco use (ICD-10 Z72.0), systemic steroid use (ICD-10 Z79.52) chronic kidney disease (ICD-10 N18) were selected. Verified ICD-10 codes for variables in the 5-item modified Frailty Index (mFI-5) were also selected, 19 including hypertension (ICD-10 I10), diabetes (ICD-10 E08-E13), chronic obstructive pulmonary disease (ICD-10 J44), and congestive heart failure (ICD-10 I50.9). Additional variables selected included commonly used NSAIDs such as ibuprofen (RxNorm 5640), naproxen (RxNorm 7258), and celecoxib (RxNorm 140587). Laboratory values in TriNetX were curated by the database using harmonized related codes, to generate a curated TriNetx (TNX Curated) code. Low-density lipoprotein (LDL) 20 was also selected as a variable (TNX Curated 9002), as well as body mass index (BMI) and BMI ≥ 30 kg/m2 (TNX Curated 9083).
Statistical Analysis
Within TriNetX, cohorts were matched using the selected variables as described above in a 1:1 ratio using a greedy nearest-neighbor algorithm that utilizes logistic regression to estimate similar-sized cohorts, with a caliper of 0.1 pooled standard deviations. The variables were selected to included known risk factors for pseudoarthrosis of the spine, 21 mFI-5, and LDL based on a recent study. 20 Baseline characteristics of each cohort as well as the standardized mean differences (SMD) were recorded before and after propensity score matching (well-matched covariates were defined by SMD < 0.1).22-25
The primary outcome was pseudoarthrosis at 6-months, 1-year, and 2-years after surgery. Pseudoarthrosis was defined by ICD-10 code M96.0 recorded in the patient’s electronic medical record at each time point. Pseudoarthrosis is typically diagnosed by surgeons and radiologists when lack of bony fusion is observed between treated vertebral levels based on structural imaging, such as CT and X-ray. Patients taking aspirin 81 mg daily who underwent single-level ACDF were compared with patients who underwent single-level ACDF and were not taking aspirin. Similar analyses were performed for use of aspirin 325 mg daily, as well as comparative cohorts for patients who underwent multi-level ACDF at each time point. Our study design is illustrated in Figure 1. Study Design. *Patients Queried From 67 Health Care Organizations From the United States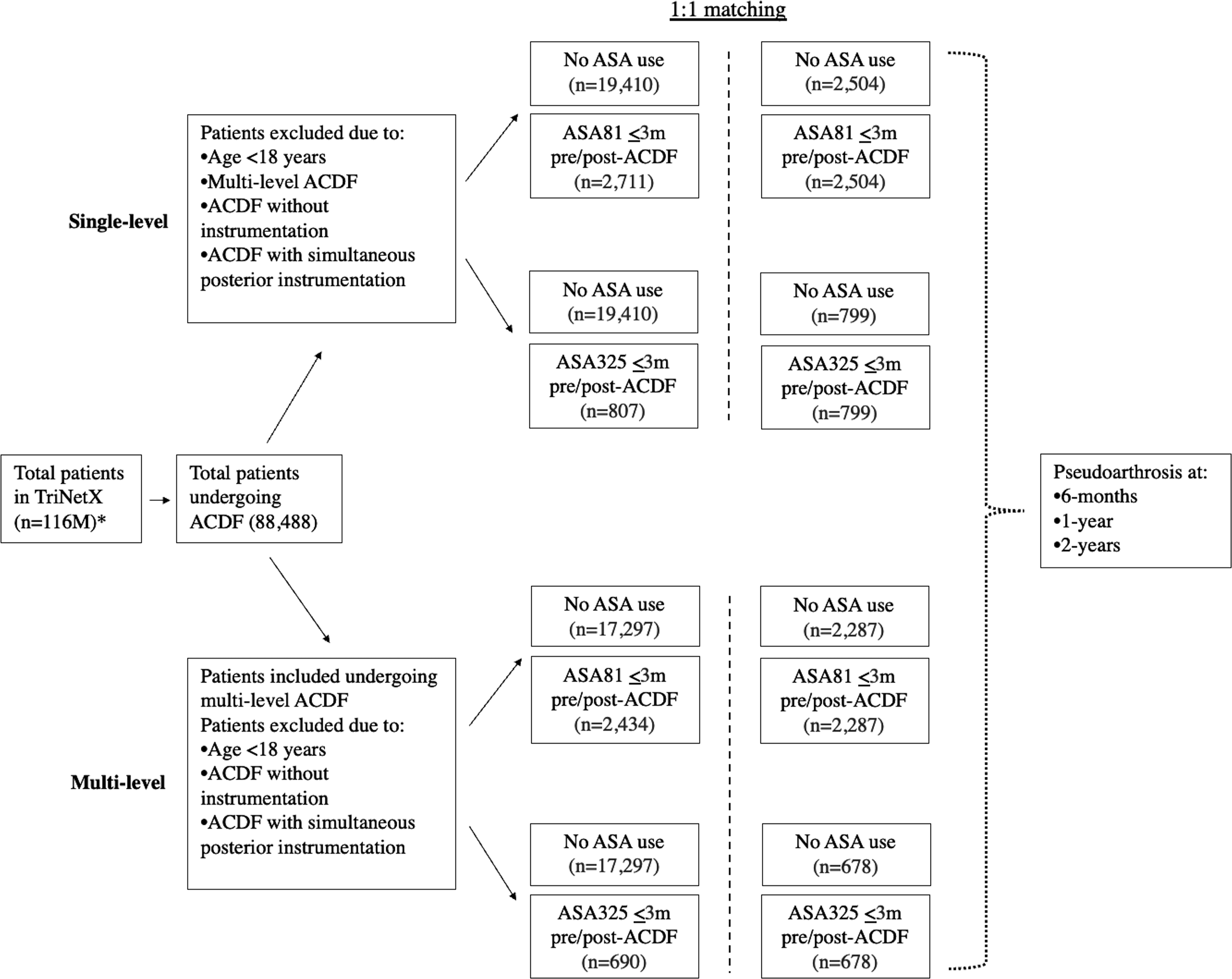
For any cohort(s) found to have a significant difference in rate of pseudoarthrosis, rates of reoperation with additional (anterior or posterior) spinal fusion were also evaluated at 6-months, 1-year, and 2-years after surgery. Reoperation with spinal fusion was defined by revision anterior cervical fusion or posterior cervical fusion. 26 For each analysis, odd ratios with 95% confidence intervals (CI) were reported, and P-values < 0.05 were considered statistically significant. TriNetX calculates P-values using t-tests for continuous variables and z-tests for categorical variables. Correction for multiple comparisons was not performed as each outcome timepoint was pertinent for this study.
We performed additional sensitivity analyses evaluating different independent variables to test the robustness of our results. Given aspirin’s role as both an antiplatelet agent and an NSAID, clopidogrel and ibuprofen were selected to test whether comparable medications would produce similar results. For these analyses, aspirin was included as a variable in propensity matching.
Results
Propensity Score Matching of Cohorts
Comparison of Patients Undergoing Single-Level ACDF Taking Aspirin 81 mg Daily Versus Those Not Taking Aspirin, Before and After Propensity Score Matching
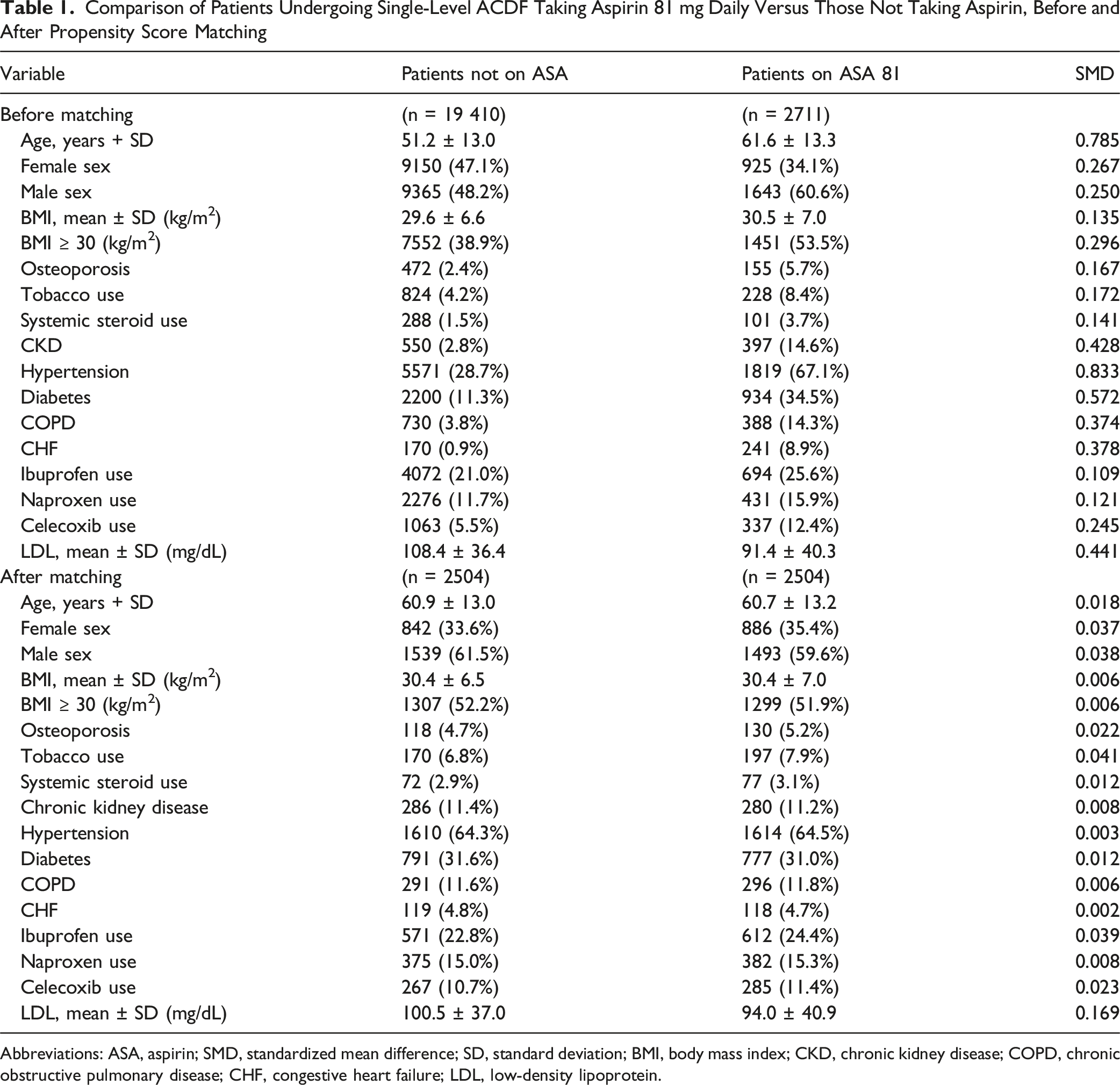
Abbreviations: ASA, aspirin; SMD, standardized mean difference; SD, standard deviation; BMI, body mass index; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CHF, congestive heart failure; LDL, low-density lipoprotein.
Comparison of Patients Undergoing Single-Level ACDF Taking Aspirin 325 mg Daily Versus Those Not Taking Aspirin, Before and After Propensity Score Matching
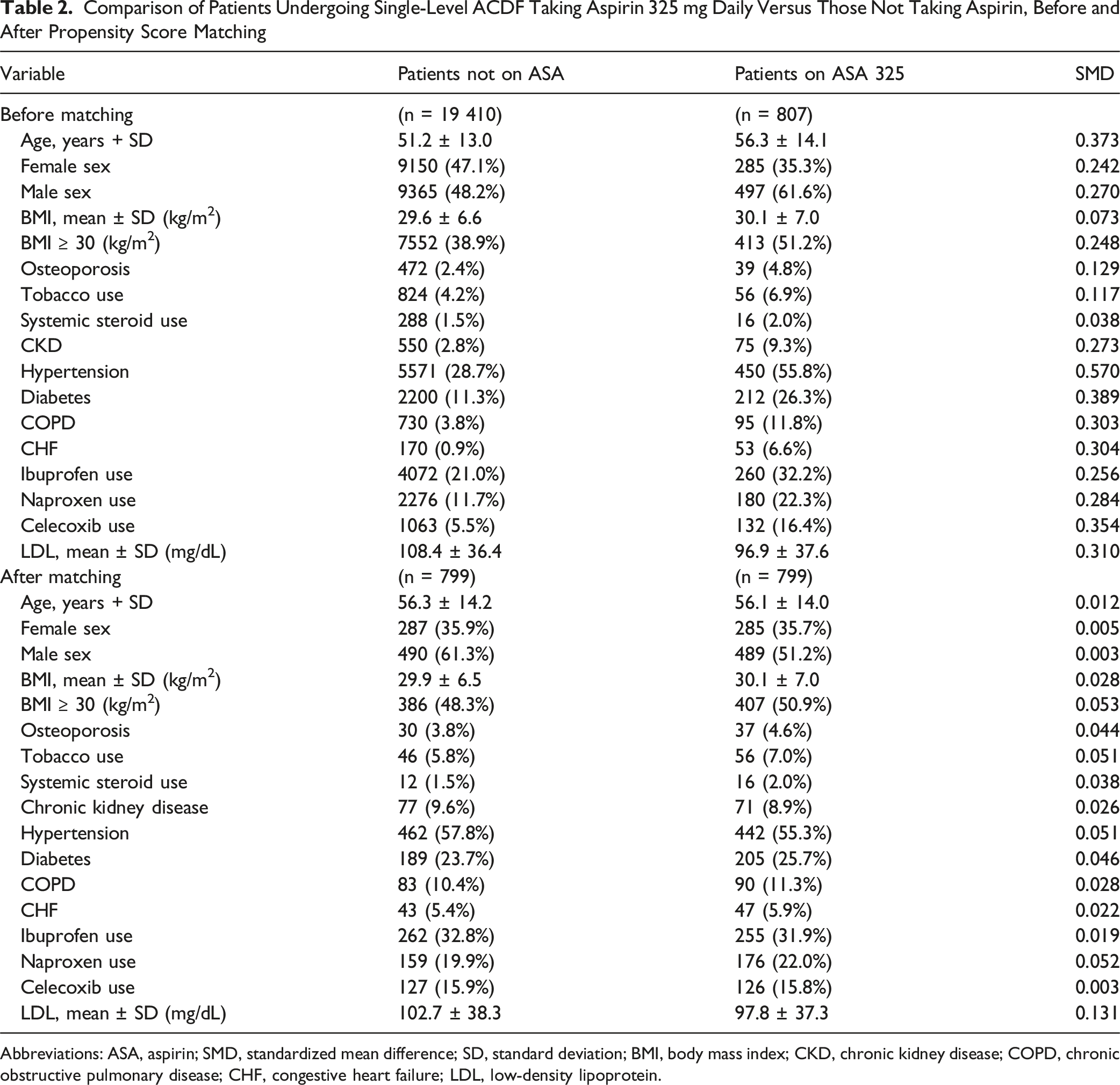
Abbreviations: ASA, aspirin; SMD, standardized mean difference; SD, standard deviation; BMI, body mass index; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CHF, congestive heart failure; LDL, low-density lipoprotein.
Comparison of Undergoing Multi-Level ACDF Taking Aspirin 81 mg Daily Versus Those Not Taking Aspirin, Before and After Propensity Score Matching
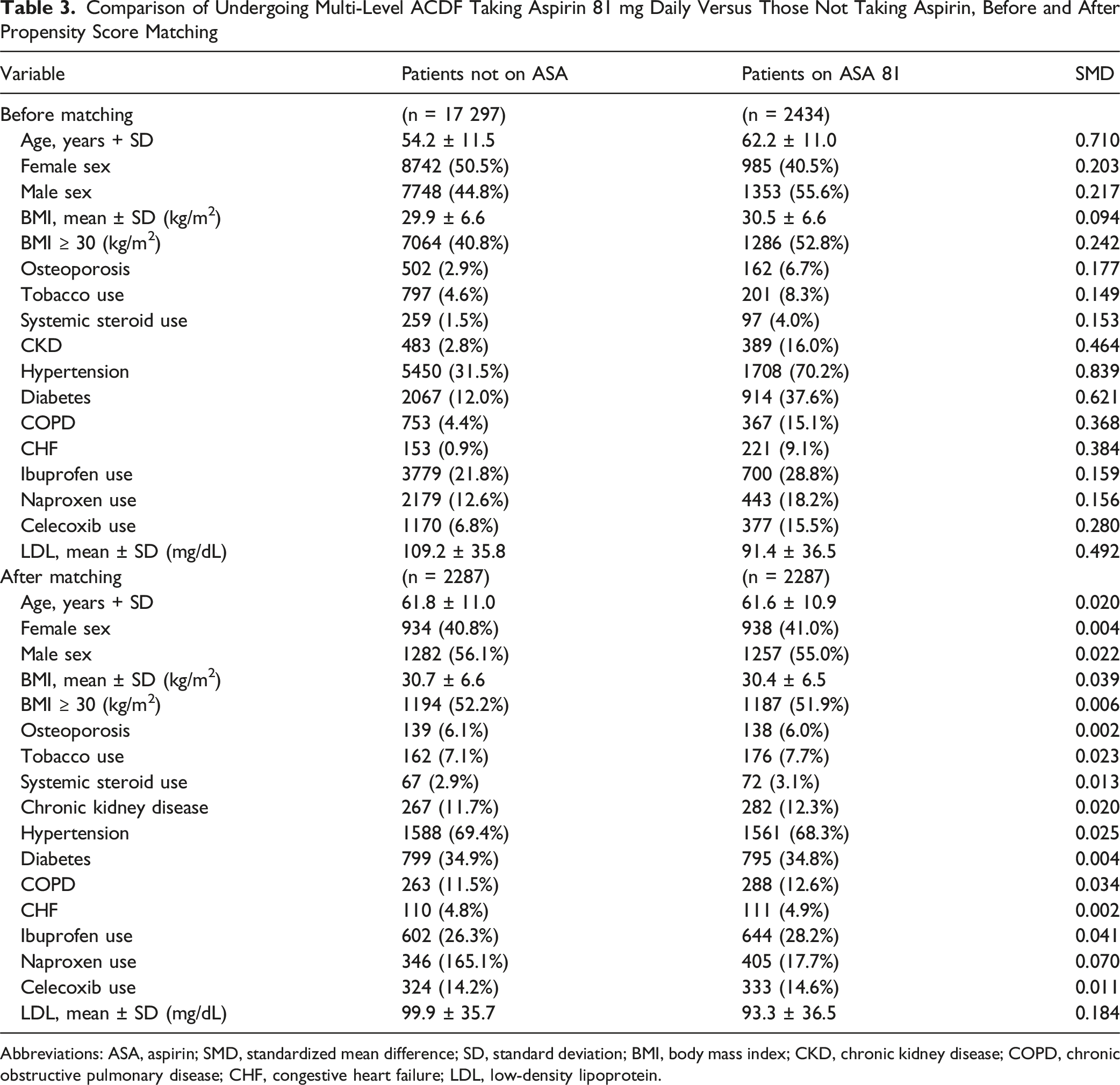
Abbreviations: ASA, aspirin; SMD, standardized mean difference; SD, standard deviation; BMI, body mass index; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CHF, congestive heart failure; LDL, low-density lipoprotein.
Comparison of Patients Undergoing Multi-Level ACDF Taking Aspirin 325 mg Daily Versus Those Not Taking Aspirin, Before and After Propensity Score Matching
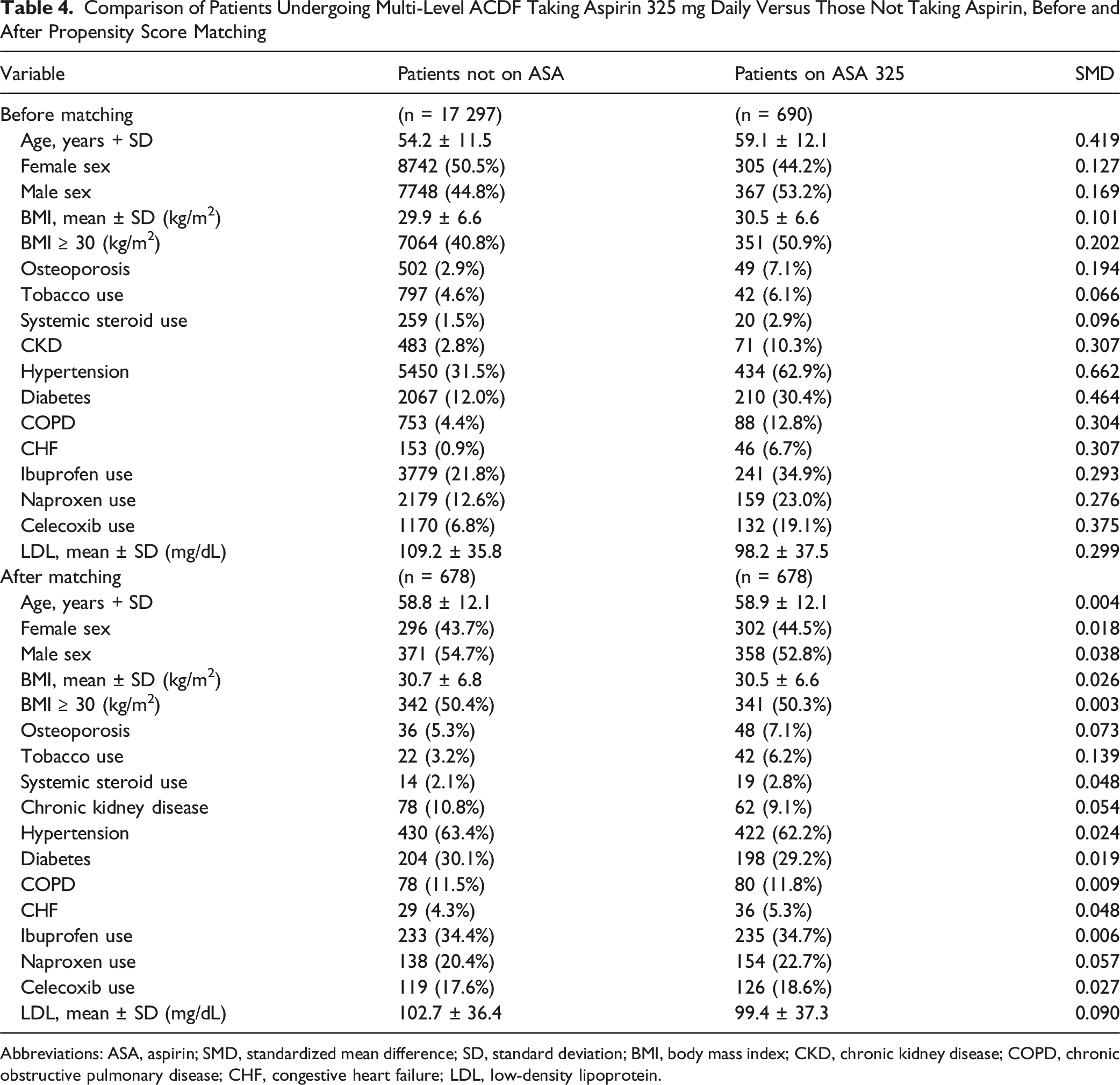
Abbreviations: ASA, aspirin; SMD, standardized mean difference; SD, standard deviation; BMI, body mass index; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CHF, congestive heart failure; LDL, low-density lipoprotein.
Risk Analysis
Odds of Pseudoarthrosis After Propensity Score Matching in Patients Stratified by Aspirin Use
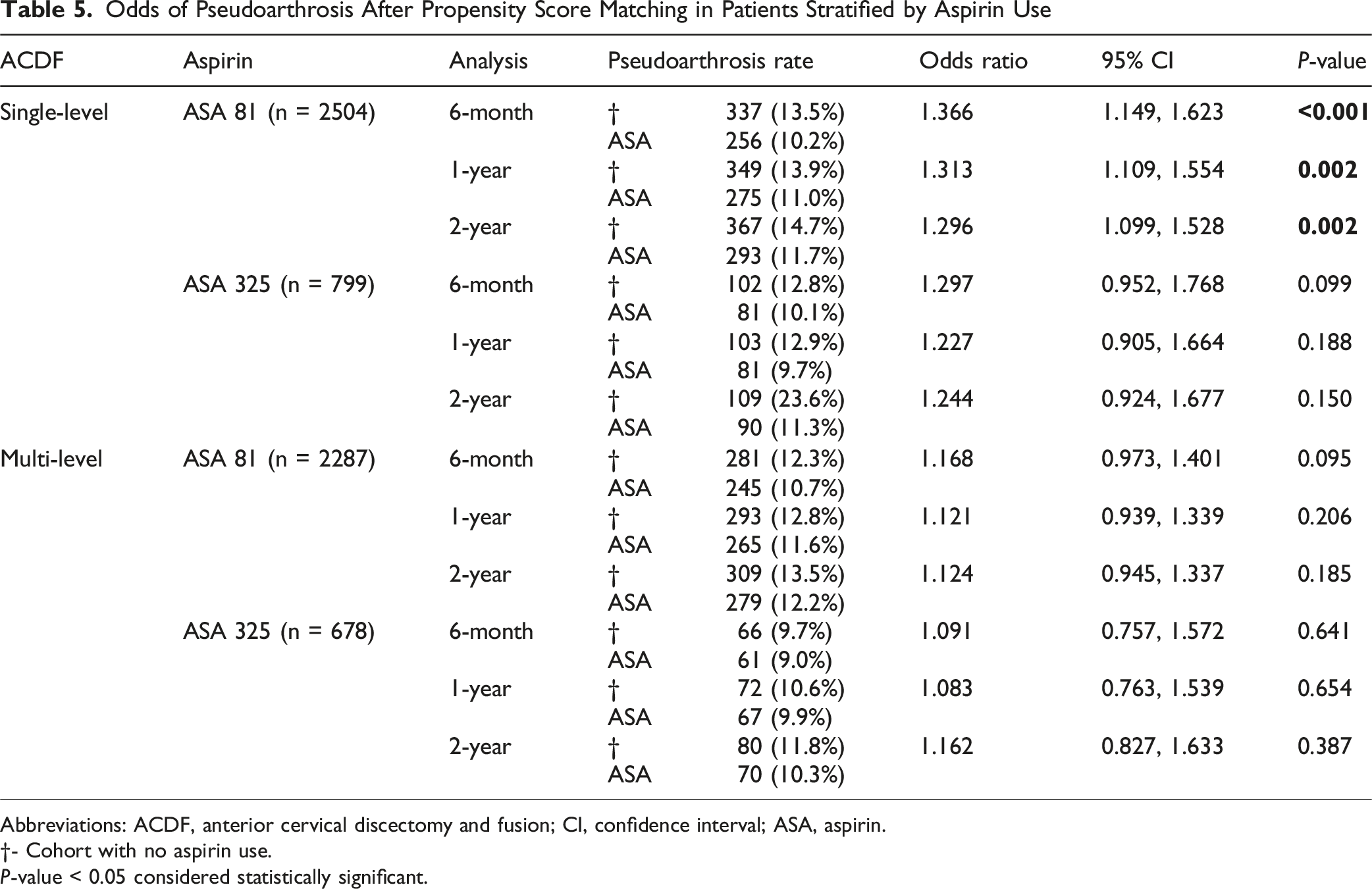
Abbreviations: ACDF, anterior cervical discectomy and fusion; CI, confidence interval; ASA, aspirin.
†- Cohort with no aspirin use.
P-value < 0.05 considered statistically significant.
Odds of Reoperation After Propensity Score Matching in Patients Undergoing Single-Level ACDF Stratified by Aspirin 81 mg Use
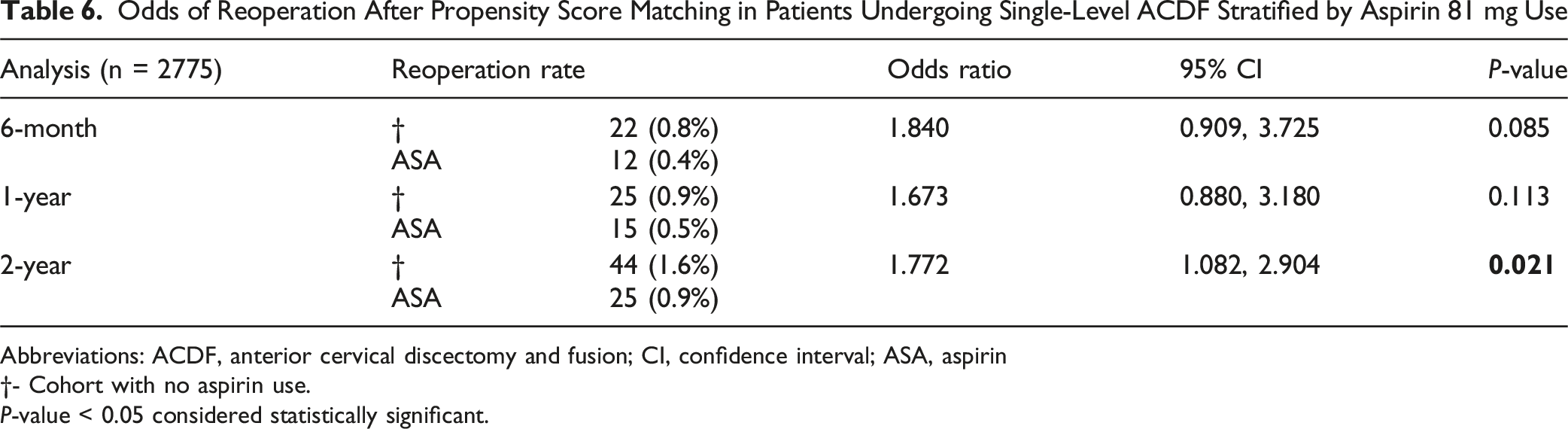
Abbreviations: ACDF, anterior cervical discectomy and fusion; CI, confidence interval; ASA, aspirin
†- Cohort with no aspirin use.
P-value < 0.05 considered statistically significant.
Sensitivity Analysis
Risk analyses for propensity matched cohorts using the independent variables clopidogrel and ibuprofen are presented in Supplemental Tables 1 and 2, respectively. Patients taking either medication did not demonstrate significant differences in rates of pseudoarthrosis at 6-months, 1-year, or 2-years.
Discussion
Aspirin use was not associated with increased rates of reported pseudoarthrosis, regardless of aspirin dosage and whether patients underwent single- or multi-level ACDF. Prior to propensity score matching, patients taking aspirin were significantly older with more co-morbidities compared with patients not taking aspirin, with some demographic and clinical characteristics carrying higher differences based on Cohen’s effect categories. 27 After matching, daily use of aspirin 81 mg was associated with a significantly decreased rate of reported pseudoarthrosis following single-level ACDF at 6-months, 1-year, and 2-years. In addition, patients taking 81 mg aspirin had significantly decreased rates of reoperation with additional spinal fusion at 2-years following surgery. There were no significant differences observed in patients taking aspirin 325 mg daily who underwent single-level ACDF compared with patients who were not taking aspirin, and there were no significant differences observed with either aspirin dose for multi-level ACDF.
Due to their theoretical negative effects on bone fusion, NSAID use is often halted or restarted in delayed fashion after spinal fusion. 28 The literature continues to be conflicted on whether these medications influence pseudoarthrosis rates post-operatively.4,6,29,30 Therefore, the optimal time point to restart NSAIDs, if at all, remains uncertain after spinal fusion surgery. Compared with other NSAIDs, aspirin possesses pharmacological properties which are distinct. “Low” doses of aspirin, ie, 81 mg daily, are commonly used in thrombosis prevention, such as preventing ischemic stroke after myocardial infarction.31,32 Increasingly, low-dose aspirin is also being used for venous thromboembolism prevention following surgery. 33 At lower doses (eg, 75-100 mg/day), aspirin concentrations in plasma are usually <100 μg/mL, while at higher doses (eg, 1000 mg/day), plasma concentrations can range from 150-300 μg/mL.10,34 At lower concentrations, aspirin preferentially inhibits COX-1, while at higher doses of aspirin favors inhibition of COX-2. 10
Inhibition of COX-1 has been shown to decrease osteoclast activity and promote osteogenic pathways. 10 In animal models, COX-1 knockout mice show improved bone density, suggesting that COX-1 may negatively regulate bone formation, while COX-2 knockout mice show reduced bone density, suggesting an anabolic role of COX-2. 35 The dose-dependent selective inhibition of COX-1 vs COX-2 may also explain aspirin’s more pronounced effects on pseudoarthrosis rates at lower doses (81 mg vs 325 mg). Furthermore, aspirin has been shown to improve tissue microcirculatory perfusion in animal models, which may contribute to improved bone healing. 9 However, these mechanisms, as well as the pathways associated with the downstream differential effects of COX-1 and COX-2 inhibition, require further investigation.
At the low dose of aspirin (81 mg daily), significantly decreased rates of reported pseudoarthrosis was found in this study. This may suggest a potential anabolic (ie, bone-producing) effect after single-level ACDF. However, this effect was not seen in the multi-level ACDF cohort. It has been shown that pseudoarthrosis and reoperation rates increase with increasing number of ACDF levels performed.36-40 Biomechanically, multi-level anterior cervical constructs have more graft-endplate interfaces requiring bony fusion, and they are exposed to higher stresses particularly at the caudal level.41,42 This has correlated with slower fusion and higher rates of needing revision surgery for non-union.42,43 Furthermore, after surgery, prostaglandins such as PGE2 (produced by COX-1 and COX-2 enzymes), are upregulated as part of the inflammatory response that influence both osteoblast and osteoclast activities.44,45 Low-to-moderate doses of PGE2 generally increase bone mass in animal models,11,12 while higher doses or sustained PGE2 can be catabolic to bone tissue.46,47 However, these mechanisms remain speculative and it may be that the biomechanics of multi-level surgery alone may account for decreased rates of arthrodesis, as stated above. More research is needed to better elucidate the potential catabolic effect of multi-level surgery on pseudoarthrosis.
In our study, there were no differences in reoperation at 6-months or 1-year, but patients taking 81 mg aspirin had significantly lower rates of additional anterior cervical fusion or posterior cervical fusion at 2-years following single-level ACDF surgery. Prior studies have demonstrated that while pseudoarthrosis at 1-year after ACDF surgery may not be predictive of long-term non-union, pseudoarthrosis at 2-years is a poor prognostic factor for clinical worsening and need for additional fusion surgery.48,49 Our results raise the hypothesis of a potentially clinically significant effect of 81 mg aspirin after single-level ACDF based on both pseudoarthrosis and reoperation rates. However, one limitation of this hypothesis is that the specific level(s) of re-operation are not available within TriNetX, and therefore it cannot be excluded that some of the reoperation cases were performed for adjacent segment degeneration in the setting successful bony fusion.
In clinical practice, the timing and decision to resume aspirin may vary. Often, once deemed reasonable from a hemostasis standpoint, aspirin therapy is typically resumed for patients with cardiovascular indications or risk factors. In a separate study, patients taking antiplatelet agents after ACDF surgery demonstrated no differences in frequency of blood transfusions, myocardial infarctions, cerebrovascular accidents, and length of stay compared with patients not taking antiplatelet agents. 50 Despite this, some surgeons may still elect to hold aspirin indefinitely if there is no clear indication for continuing the medication. Our results suggest that resuming aspirin may be reasonable strictly from a bony fusion standpoint, in that 325 mg aspirin was not significantly associated with pseudoarthrosis and 81 mg aspirin was associated with lower rates of pseudoarthrosis and reoperation with additional spinal fusion following single-level ACDF. However, these results should be interpreted with caution, and it should be noted the odds ratios of the results are modest. The potential benefits of starting low-dose aspirin after ACDF surgery in patients not previously taking the medication are not clear, and we do not advocate for this based on the results of this study. Changes to clinical practice are therefore not recommended until prospective clinical studies are completed to better characterize the effects of aspirin on bone healing after spinal fusion and to assess its potential dose-dependent properties on clinical outcomes.
This study had several additional limitations. The data used from TriNetX includes patients from participating health care centers only, and there is potential for missing patient data. Details are unknown whether participating health care centers were primarily academic centers, which may limit generalizability. The potential for misclassification bias exists in the database’s reliance on electronic medical records utilizing procedural, diagnostic, laboratory, and medication codes. Subgroup analyses were not performed for multiple reasons. First, due to the large number of potential confounding variables as it pertains to pseudoarthrosis, identifying a subset of patients who may benefit most from aspirin use after ACDF would be exploratory. Second, patients taking aspirin represent a largely different cohort due to baseline differences in demographics and medical comorbidities as seen in the pre-matching SMDs. Rather, our results were further scrutinized with additional analyses of a complementary outcome measure (rate of reoperation) and sensitivity analyses of similar medications. Variables including graft material, implant type, individual surgeon experience are not reported. The number of levels fused in the “multi-level” group is not specified in the TriNetX database. As these variables are unknown, they may have influenced the results. In addition, other medications may have been prescribed which could affect bone healing.
Active aspirin use was defined by documented medication use within 3-months both prior to and following surgery. This window was selected to account for the limitations inherent to the TriNetX database, as it is restricted to recorded medication terms. In other words, 3-months is felt to represent as adequate time window in which patients present to the clinic or the hospital where their medication use is documented. Nonetheless, this may introduce bias and although a narrower time window could have been considered, 3-months was selected based on the rationale above, as supported by prior literature evaluating effects of active medication use on surgical outcomes.20,51-53 Additionally, true medication adherence is not verifiable, and aspirin may have been withheld for some patients in the immediate peri-operative period and possibly beyond, which could influence results.
The TriNetX database is limited to the use of ICD coding to capture the diagnosis of pseudoarthrosis, which therefore carries risk of underreporting. In addition, radiographic validation of pseudoarthrosis is not available in this large multi-center study, and the criteria used by each provider to designate this code (ie, failure of bony fusion) may have been inconsistent and non-standardized. While this may limit the applicability of this diagnosis, the sample sizes are likely large enough to minimize the influence of any large discrepancies in criteria for diagnosis. In addition, reoperation with additional spinal fusion was evaluated in this study as a complementary outcome measure to further assess significant results. As noted above, it is possible that some patients underwent additional fusion surgery for reasons other than non-union, and these results should therefore be interpreted with caution.
Variables used in propensity score matching may have also been under-coded, including smoking status and osteoporosis, for instance. The sample sizes of patients taking aspirin 325 mg daily were smaller compared with patients taking aspirin 81 mg daily, which may influence the results. Confidence intervals for some comparisons were wide, suggesting limited precision due to relatively smaller sample sizes, particularly in the aspirin 325 mg group.
Conclusions
Patients taking aspirin 81 mg daily who underwent single-level ACDF had significantly decreased rates of reported pseudoarthrosis at 6-months, 1-year, and 2-years, and lower rate of reoperation with spinal fusion at 2-years, compared to patients not taking aspirin. Patients taking aspirin 325 mg daily who underwent single-level ACDF, as well as patients taking either aspirin 81 mg or 325 mg daily who underwent multi-level ACDF did not demonstrate significantly different rates of pseudoarthrosis. Resuming low-dose aspirin after ACDF surgery may be reasonable from an arthrodesis standpoint. However, these results are hypothesis-generating and we do not advocate their implementation into standard clinical practice. Prospective clinical studies are needed to further evaluate the potential dose-dependent effects of aspirin on bone remodeling and spinal fusion.
Supplemental Material
Supplemental Material - Does Aspirin Use Influence Rates of Pseudoarthrosis After Anterior Cervical Discectomy and Fusion?
Supplemental Material for Does Aspirin Use Influence Rates of Pseudoarthrosis After Anterior Cervical Discectomy and Fusion? by Kevin T. Kim, Ziam Khan, Joseph Stains and Timothy Chryssikos in Global Spine Journal
Footnotes
Acknowledgments
Dr Chixiang Chen for his guidance on statistics in this study.
Ethics Considerations
This study was approved by the local Institutional Review Board with consent exemption under 45 CFR 46.101(b).
Author Contributions
Kevin T. Kim: Conception and design, Acquisition and analysis/interpretation of data, Drafting and critical revision of the manuscript.
Ziam Khan: Acquisition of data, Drafting of manuscript.
Joseph Stains: Conception and design, Analysis/interpretation of data, Drafting and critical revision of the manuscript.
Timothy Chryssikos: Conception and design, Analysis/interpretation of data, Drafting and critical revision of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data available upon request, within the limitations of the TriNetX database.
Supplemental Material
Supplemental material for this article is available online.
