Abstract
Background:
During shortages, elastomeric half-mask respirators (EHMRs) are an alternative to reusing N95 filtering facepiece respirators but require between-use disinfection. The objectives of this study were to (a) measure microbial reductions on EHMR surfaces under laboratory conditions by a standardized procedure using wipes impregnated with health care disinfectants and to (b) measure microbial reductions on EHMRs disinfected by volunteer health care providers.
Method:
We inoculated EHMR (Honeywell model RU8500) surfaces with Pseudomonas aeruginosa, Bacillus atrophaeus spores, and bacteriophages MS2 and Φ6, and disinfected them using two wipes with hydrogen peroxide (HP), alcohols, and quaternary ammonium compounds (QACs). Then, we randomized 54 volunteer subjects into three groups (Group 1: two wipes with instructions, Group 2: five wipes with instructions, Group 3: no instructions or set number of wipes) and used 0.5% HP wipes without precleaning on EHMRs inoculated with Raoultella terrigena and MS2.
Findings:
The laboratory study demonstrated that all organisms achieved at least 4 log10 median reductions (HP>QAC/alcohol>QAC>QAC/saline). Pseudomonas was highly susceptible to HP and QAC/alcohol and Φ6 to all disinfectants. MS2 reduction was highest using HP and lowest using QAC/saline. Bacillus was least susceptible. The volunteer study showed a 3 to 4 log10 average reductions of bacteria and virus; Raoultella reductions were greater than MS2, with variability within and between subjects. Conclusions: HP disinfectant wipes used in laboratory and by volunteers reduce bacteria and viruses on EHMRs by 3 to 4 log10 on average.
Implications for Practice:
Commercially available hospital disinfectant wipes reduce bacteria and viruses on EHMRs and can fill the need for between-use disinfection. HP and combination QAC/alcohol have the greatest efficacy under our test conditions.
Background
To prevent health care personnel (HCP) exposure to respiratory pathogens during patient care, the Centers for Disease Control and Prevention (CDC) recommends single-use N95 filtering facepiece respirators (FFRs; Centers for Disease Control and Prevention, 2022), the most widespread respiratory protection in health care (National Personal Protective Equipment Technology Laboratory, 2022). However, hospitals that only supply FFRs are vulnerable to shortages during crises such as the COVID-19 pandemic, (De Perio et al., 2020; Dugdale & Walensky, 2020) which forced many to assemble ad hoc emergency measures such as FFR reuse (Grossman et al., 2020). Reused FFRs may be contaminated by pathogens during patient care. FFR disinfection is challenging (Cheng et al., 2020); testing must demonstrate a reduction in microbes without a reduction in filtration efficiency (Centers for Disease Control and Prevention, 2020; Lindsley et al., 2015; National Personal Protective Equipment Technology Laboratory, 2019).
One FFR alternative is the elastomeric half-mask respirator (EHMR), a synthetic or rubber facepiece secured by straps, with a port or ports to attach replaceable filters, cartridges, or canisters to address respiratory hazards. EHMR are presumed not to lose fit capabilities and can be reused indefinitely if they maintain integrity, can be cleaned and disinfected, and replaceable filtration media are available. EHMR disinfection studies (3M Personal Safety Division, 2021; Bessesen et al., 2015; National Academies of Sciences, Engineering, and Medicine, 2019; Subhash et al., 2014) have used different disinfectants, test microbes, disinfection protocols, and sampling methods. Protocols that target pathogens found in hospitals are needed (Bowdle et al., 2021). Several brands of wipes impregnated with different health care disinfectants are available, and there is existing evidence of efficacy that suggests they are promising candidates for routine EHMR disinfection. Therefore, the objectives of this study were to: (a) measure the reduction of microbes on EHMR surfaces under laboratory conditions by a standardized procedure using wipes impregnated with common health care disinfectants, and (b) measure the reduction of microbes on EHMRs by disinfection without precleaning, performed by volunteer health care providers.
Implications for Occupational Health Practice
This study found that commercially available hospital disinfectant wipes reduce bacteria and viruses on EHMRs. HP and combination QAC/alcohol showed the greatest efficacy under our test conditions; it is unclear from this study whether instructions provided to individuals on how to disinfect a respirator affected the reduction of microbes. In the future, evaluation of higher concentrations may be needed to optimize reduction of all classes of organisms in health care environments and must demonstrate compatibility with the performance of the components of the EHMR.
Methods
Laboratory Study
Microbes
Sets of three EHMRs (Honeywell model RU8500) were inoculated with organisms chosen as surrogates representative of different classes of health care-acquired infections (HAIs), including gram-negative bacteria, spore-forming bacteria, and viruses. Although respirator use protects against respiratory pathogen exposure, common HAI organisms are still present during respiratory disease outbreaks. Therefore, surrogate organisms in this study were chosen for two reasons: (a) to represent respiratory pathogens that might be expelled onto a HCP’s respirator by a patient during care and (b) to represent HAIs that are commonly present in the health care environment and can pose a risk of transfer to respirators via contaminated hands and gloves during handling. In laboratory studies, P. aeruginosa, itself a cause of HAIs, was chosen to also represent gram-negative HAIs such as Klebsiella and E. coli. Bacillus atrophaeus was chosen to represent the gram-positive spore-forming pathogen Clostridioides difficile. Two bacteriophages served as surrogates for human viruses. Φ6 was a surrogate for enveloped respiratory viruses such as SARS-CoV-2 and MS2, a non-enveloped virus, was chosen as a conservative surrogate to represent human viruses that are more resistant to disinfection and environmental stressors. In the volunteer study, Raoultella terrigena was chosen as a surrogate that represented gram-negative HAIs while posing minimal risk to study participants handling respirators.
There is variation in methods for respirator disinfection testing across studies; methods in this study were adapted from Lawrence et al. (Lawrence et al., 2017). EHMRs were dried at room temperature for 30 min before disinfection. Microbes were suspended in trypticase soy broth, (target concentration 107–1010 plaque forming units/milliliter [PFU/mL] for viruses and 108 colony forming units/milliliter [CFU/mL} for bacteria). Four sites were inoculated with 50 µL of suspension on each site: nose 2 cm directly above the vent, center back of the outward-facing side of the strap, and top edge of the right and left filter covers. These sites were chosen as locations where respiratory droplets from a coughing or sneezing patient might land (nose, filter covers), and where a person would be likely to touch the respirator during wearing or handling (filter covers, strap). The 50 µL was placed on each of the sites in ten 5 µL drops to simulate the deposition of droplets on the respirator surface. The suspension was inoculated in droplets on discrete locations on the respirator to simulate the expected deposition of respiratory secretions from a cough or sneeze, which would most likely not uniformly coat the respirator surface. EHMRs were dried at room temperature for 30 min before disinfection to simulate microbes sitting on the respirator surface for the length of a patient care encounter.
Disinfecting Wipes
Wipes were EPA-approved hospital disinfectants with combinations of hydrogen peroxide (HP), alcohols, and quaternary ammonium compounds (QAC): 0.5% HP (Oxivir® TB Wipes; Virox Technologies Inc.), 0.76% didecyldimethylammonium chloride+7.5% ethanol+15% isopropanol (CaviWipes1; Metrex Research), 0.4% benzalkonium chloride (The Big Ones; Allegro Industries), and 0.9% sodium chloride+benzalkonium chloride (Hygéa Sterile saline Wipes; Professional Disposables International, Inc.). Respirators were disinfected by the same person, using a standard procedure with five wipes. They sat at room temperature for manufacturer-recommended contact time (1 min for 0.5% HP and 0.76% didecyldimethylammonium chloride+ethanol+isopropanol). There was no manufacturer-provided contact time for sodium chloride+benzalkonium chloride, so 2 min was used based on contact time for 0.4% benzalkonium chloride.
Recovery and Assay
Sites were sampled post-disinfection by polyester swab premoistened with neutralizing buffer. Swabs were suspended in a neutralizing buffer, vortexed for 30 s, and plated on trypticase soy agar for P. aeruginosa and B. atrophaeus. Φ6 and MS2 were enumerated by the single agar layer method with hosts P. syringae and Escherichia coli (ATCC#15597). All were incubated overnight at 37°C.
Statistical Analysis
Results were expressed as log10 reduction, a standard measure for testing the effectiveness of disinfectants against microbes. Log reductions measure the remaining viable microbes on a respirator site relative to the starting concentration of viable microbes at that site. The reduction was expressed as log10(N1/N0), where N1 is the contaminant concentration on the respirator after disinfection and N0 is the concentration in the inoculum applied to the respirator before disinfection. The detection limits of experiments were the maximum observable reduction of microbes in an experiment, calculated as log10(1/N0) for each organism from each EHMR. The use of the detection limit makes it possible to quantify the theoretical log reduction from an experiment where no viable microbes are remaining on a surface. This reflects the fact that the measurable reduction of a microbe by a disinfectant is always relative to the starting quantity. For example, if 500,000 viable organisms (5.7 log10) are inoculated at a site, the smallest measurable number remaining at that site is one organism. Therefore, the maximum log10 reduction that can be experimentally measured is 5.7 log10. If there are no microbes remaining, then the reduction achieved is ≥5.7 log10. This allows the calculation of a predicted number of surviving microbes on a respirator from any starting concentration. The chi-square tests were used to compare the number of times organisms were recovered across sites, disinfectants, and organism types. Log-rank tests were used to compare the reductions. All analyses were performed using R.
Volunteer Studies
Subjects
Subjects were nurses and nursing students from the Emory Healthcare system and Emory University School of Nursing. This study was reviewed and approved by the Emory University Institutional Review Board (STUDY00002519; See 45 C.F.R. part 46.114; 21 C.F.R. part 56.114). Subjects completed a demographic survey including their nursing role, time in role, education level, and previous experience wearing EHMRs and other respirators.
Microbes
For each participant, one “test” respirator was inoculated on the nose, strap, left filter cover, and right filter cover with 50 µL of tryptic soy broth suspension containing Raoultella terrigena (surrogate for gram-negative HAI microbes, target 107–108 CFU/mL) and MS2 (target 107–108 PFU/mL). The respirator was dried at room temperature for 30 min and given to the participant. One “control” respirator per day was matched to all test respirators run that day. The control was inoculated identically to the test respirators, dried for 30 min, and sampled in the same manner. All sampling was done by the same investigator. EHMRs were used only once per day; at the end of the day they were disassembled, cleaned, and disinfected (Bessesen et al., 2015).
Disinfection
Based on the laboratory study, 0.5% HP wipes (Oxivir®) were used because they demonstrated the largest log10 reductions. Subjects were randomized into three cohorts: naive, two-wipe, and five-wipe. The naïve cohort was not given instructions on a number of wipes or how to wipe. They were told to disinfect the respirator, in the same manner they would if they had just removed it after patient care, and to remove the splash guards to disinfect the outside of the filter covers. For the other cohorts, there were two sets of step-by-step instructions: one using two wipes and one using five. Instructions were piloted iteratively for clarity with five people unfamiliar with the project. Subjects were filmed. A facilitator observed whether two- and five-wipe subjects followed instructions, noted departures on a checklist, and informed them in real time if they missed a step. To represent a scenario where an HCP might be disinfecting a respirator after one patient care encounter ends and they are moving on straight to the next patient, no precleaning of the respirator was done, only wipe disinfection.
Recovery and Assay
Inoculated sites were swabbed with a polyester sponge (3M Corp) premoistened with neutralizing buffer (BD Life Sciences). The sponge and 25 mL of phosphate-buffered saline+0.1% Tween were stomached in a bag for 2 min. The liquid was centrifuged at 2500×g for 15 min and supernatant assayed for MS2 by the double agar layer method. The pellet was resuspended in phosphate-buffered saline, assayed for R. terrigena by spread plating on a MacConkey agar, and incubated overnight at 37°C. The reduction was expressed as log10(N test /N control ), where N test is the concentration (R. terrigena CFU/mL, MS2 PFU/mL) recovered from the test respirator and N control is the concentration recovered from the control respirator.
Statistical Analysis
Overall, time per person was analyzed using a one-way analysis of variance (ANOVA). Multilevel modeling was used to analyze time spent disinfecting specific sites on the respirator, with fixed effects estimated for site and cohort and random effects estimated for subjects to account for potential correlations between repeated measures collected per person. Because the detection limit was frequently reached for both organisms, a two-stage approach was used to separately analyze each. First, a mixed effects logistic regression model was used to investigate site and cohort as predictors of reaching the detection limit. Next, site and cohort were investigated as predictors of the log10 reductions for observations that did not reach the detection limit using a linear mixed effects model. Each model included random intercepts per participant. All analyses were performed using R (R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org) with figures generated using R and GraphPad Prism (GraphPad, San Diego, CA, USA).
Results
Laboratory Study
Each organism-disinfectant pair underwent 3 trials where 4 surfaces were sampled (N=12 surfaces per pair; Table 1). Organisms were more frequently recovered from filter covers (31 of 48 and 64.6% of surfaces) than nose and strap (N=20, 41.7%), but recovery of organisms did not significantly differ across sites, X 2(3)=5.73, p=.13. The proportion of surfaces with any organism remaining was greatest for QAC/saline and least for HP and QAC/alcohol. Recovery also differed by organism, X2(3)=44.9, p<.001, with B. atrophaeus and MS2 recovered more frequently than Φ6 and P. aeruginosa.
Number and Percent of Surfaces a With Organisms Recovered After Use of Various Disinfectants in Laboratory Study
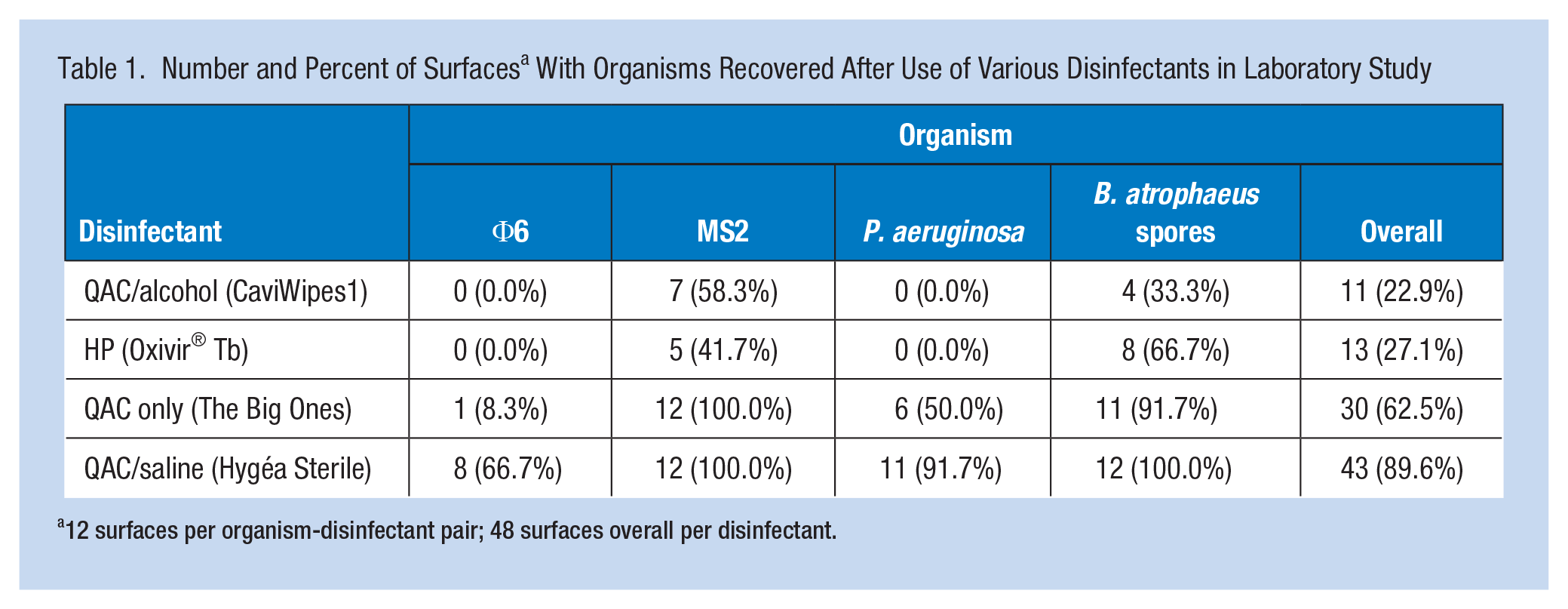
12 surfaces per organism-disinfectant pair; 48 surfaces overall per disinfectant.
All organisms demonstrated a median reduction of at least 4 log10 (Table 2). Log-rank tests found no significant differences across sites, so the remaining results use all sites combined. Reductions differed by disinfectant, X2(3)=71.7, p<.001. HP had the greatest reductions, but these were not significantly higher than QAC/alcohol, X2(1)=0.04, p=.8. QAC/alcohol had greater reductions than QAC only, X2(1)=12.4, p<.001, and QAC only had greater log10 reductions than QAC/saline, X2(1)=12.3, p<.001. Reductions also differed by organism, X2(3)=70.4, p<.001. P. aeruginosa reductions were greater than reductions of MS2, X2(1)=18.5, p<.001. P. aeruginosa was highly susceptible to HP and QAC/alcohol.
Median Log Reduction Values by Organism/Disinfectant in Laboratory Study
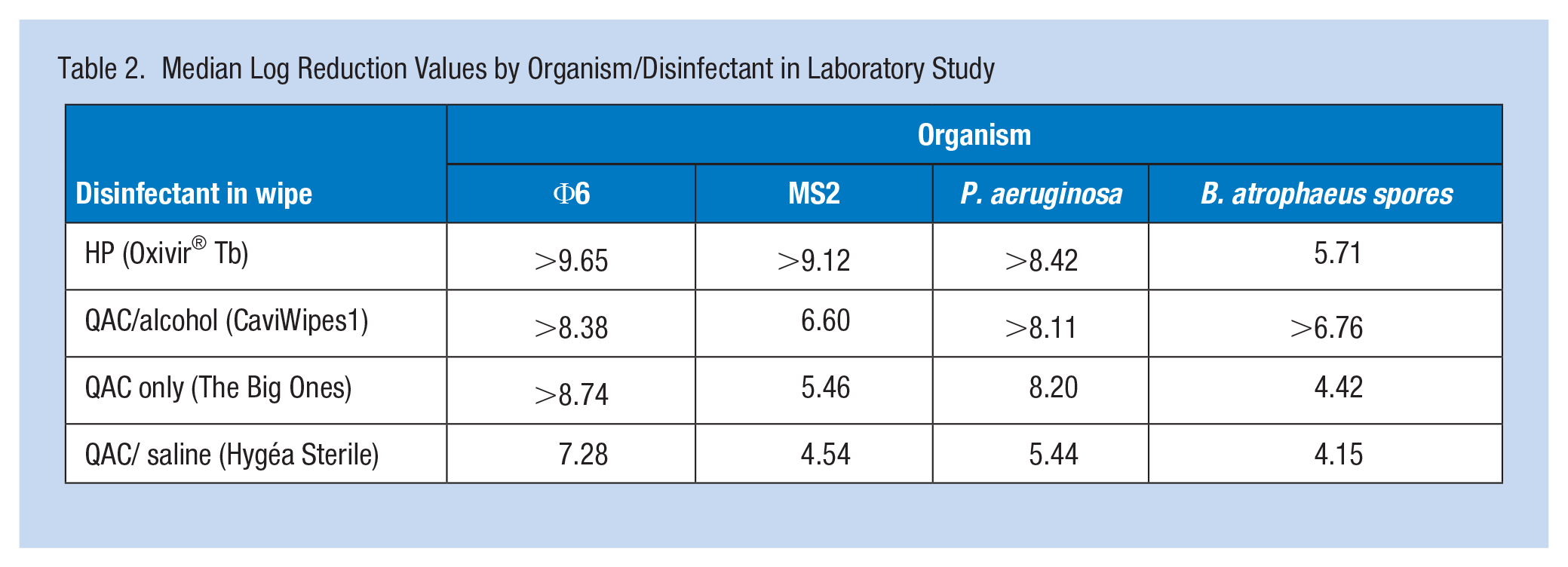
Φ6 was highly susceptible to all disinfectants (overall median log10 reduction >8.62) (Table 1). Reductions of Φ6 were greater than for P. aeruginosa, X2(1)=4.7, p=.03. Susceptibility varied for MS2. Median reduction for MS2 was highest using HP and lowest using QAC+saline. HP showed higher reduction than QAC only, median >9.12, X2(1)=8.5, p=.003. However, reduction of MS2 was not significantly different between QAC+alcohol and HP, X2(1)=1.5, p=.2, or QAC only and HP, X2(1)=1.8, p=.2. The lowest reductions were observed for B. atrophaeus, but these were not significantly lower than MS2, X2(1)=2.2, p=.1, B. atrophaeus was least susceptible to all disinfectants, with viable spores recovered on 72.92% of surfaces. Median reduction ranged from 4.15 (QAC+saline) to >6.76 (QAC/alcohol). The reduction for spores was higher following disinfection with QAC+alcohol (median>6.76) compared to HP (median=5.71); however, the difference was not statistically significant, X2(1)=1.5, p=.2. HP resulted in a higher reduction for B. atrophaeus compared with QAC only, median=4.42, X2(1) =13.7, p<.001.
Volunteer Study
Fifty-four subjects (22 nurses, 3 advanced practice practitioners [APP], 2 nurse technicians, and 28 nursing students) completed a study visit (17 two-wipe, 19 five-wipe, and 18 naive). Nurses, APPs, and techs had median 6 years’ experience (M=13) in their current role. Home units included COVID and non-COVID. They had been wearing respirators an average of 4.5 years (SD=7.7, median=1.5). Pre-pandemic, 70% had worn N95 FFRs; during the pandemic, most wore N95 FFRs. Both pre and during, only one wore an EHMR.
An average of two wipes was used in the naive cohort. In the two- and five-wipes, some used more than the specified number, mostly on splash guards; there was variation in the duration each wipe was used (Figure 1). There was no statistically significant difference in total disinfection duration for the three cohorts, F(2,51) = 2.01, p=.144,; average for the naive cohort was 1 min 44 s (95% confidence interval [CI] = 1 min 17 s–2 min 11 s), two-wipe 2 min 17 s (95% CI = 1 min 52 s–2 min 47 s), and five-wipe 2 min 10 s (95% CI = 1 min 46 s–2 min 38 s). Controlling for cohort, subjects spent significantly more time wiping the parts of the respirator surface outside of the four inoculated sites (>40 s) than they did the four inoculated sites themselves (<40 s; p < .0001 for all pairwise comparisons). Significantly more time was spent on the strap than the filter covers (p < .0001 for both pairwise comparisons). There were no significant differences between cohorts or cohort-by-site interactions in disinfection duration.
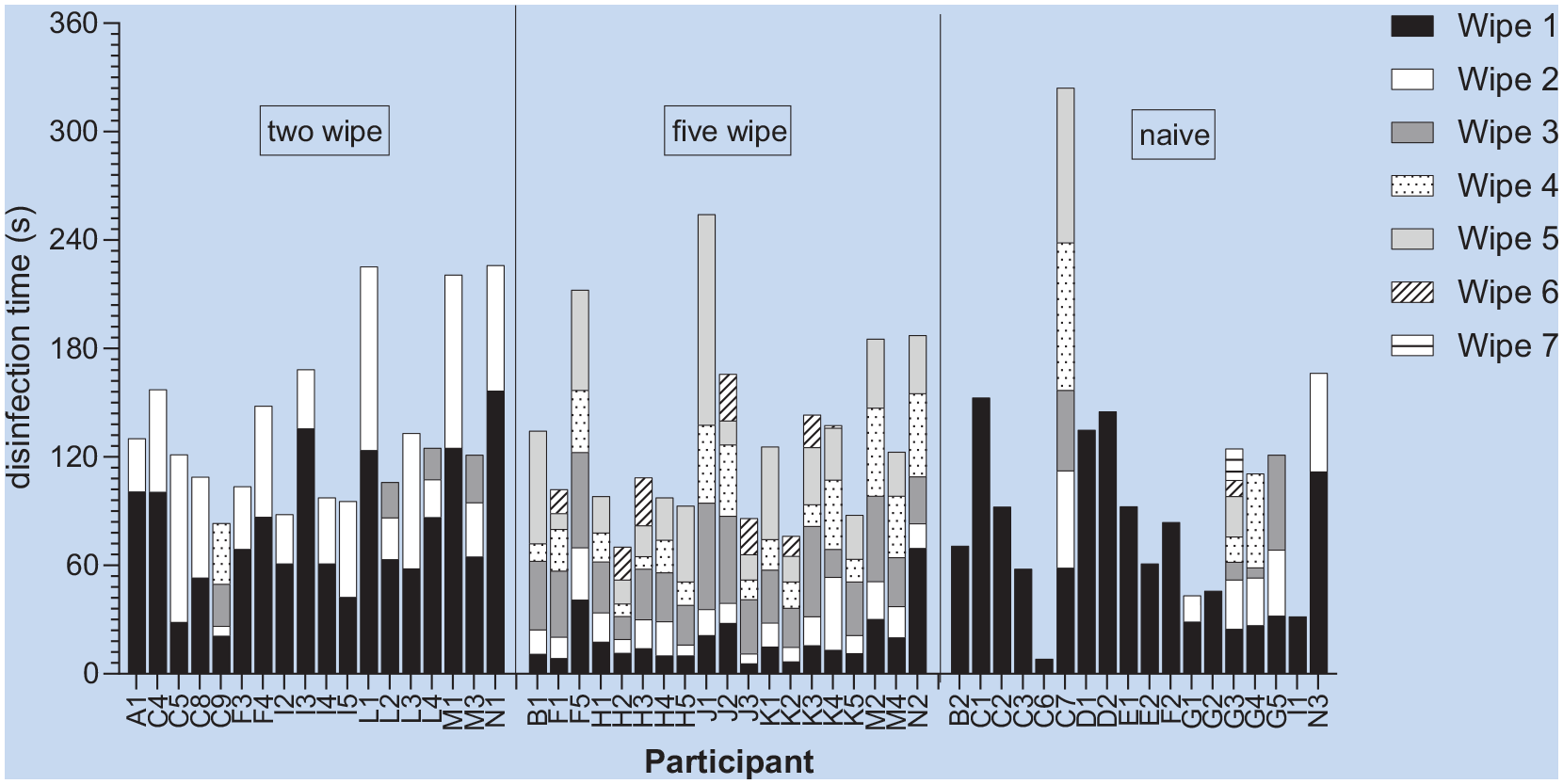
Number of Wipes Used and Duration of Use for Each Wipe Within Total Disinfection Time for Each Individual Subject (Categories: Two Wipes= Two Wipes and Instructions, Five Wipes=Five Wipes and Instructions, Naïve=No Instructions or Specified Number of Wipes).
Log10 reductions varied within and between subjects (Figure 2). Overall, reductions were greater for R. terrigena than MS2, although there is substantial variability within the cohort and site for both. The number and proportion of observations at the detection limit, along with median log10 reductions of R. terrigena and MS2 stratified by site and cohort, are shown in Table 3. Reductions of MS2 were overall lower than those of R. terrigena.
Log10 Reduction of R. terrigena and MS2 for All Subjects (N=54) Stratified By Site and Cohort
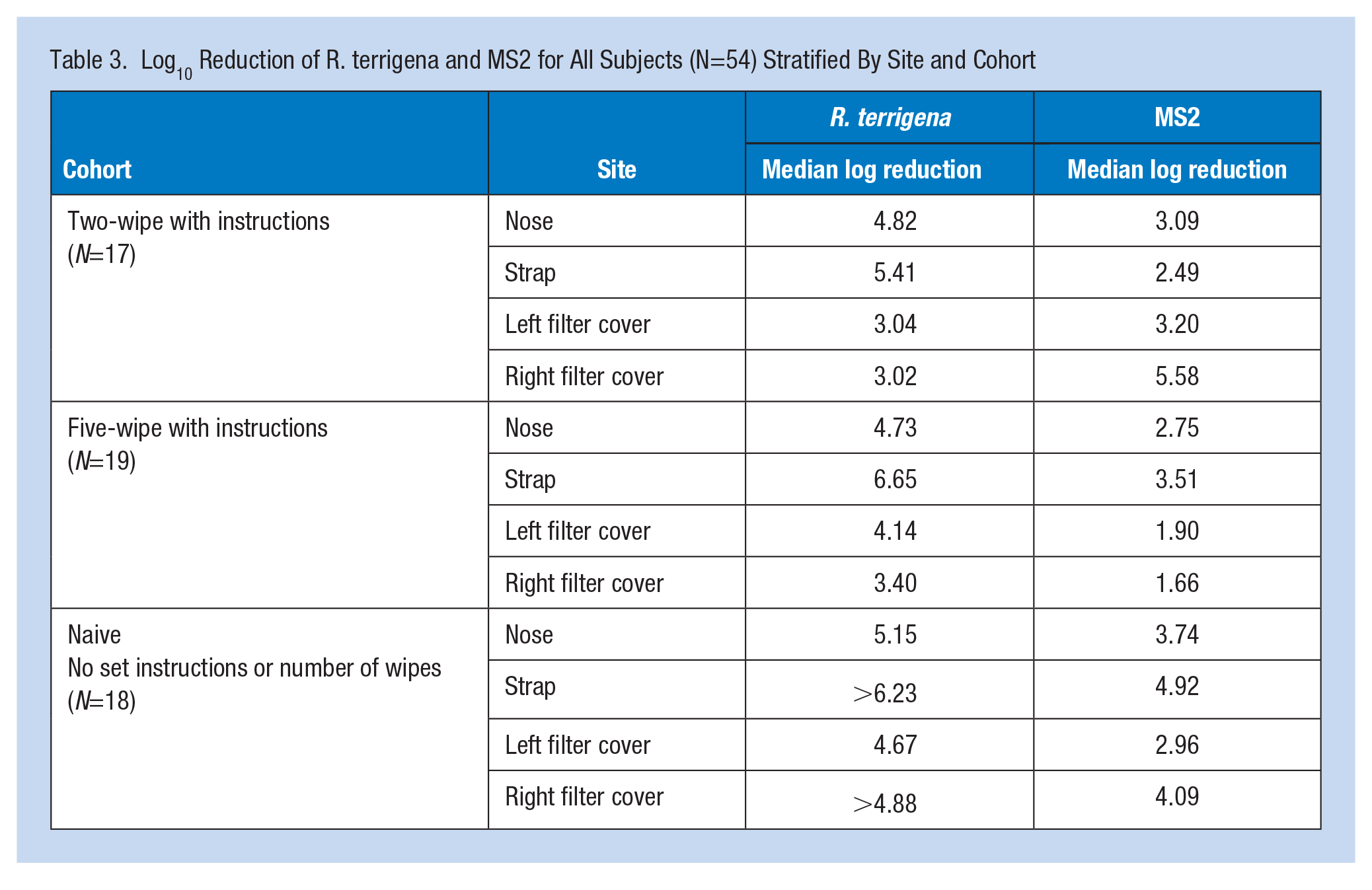
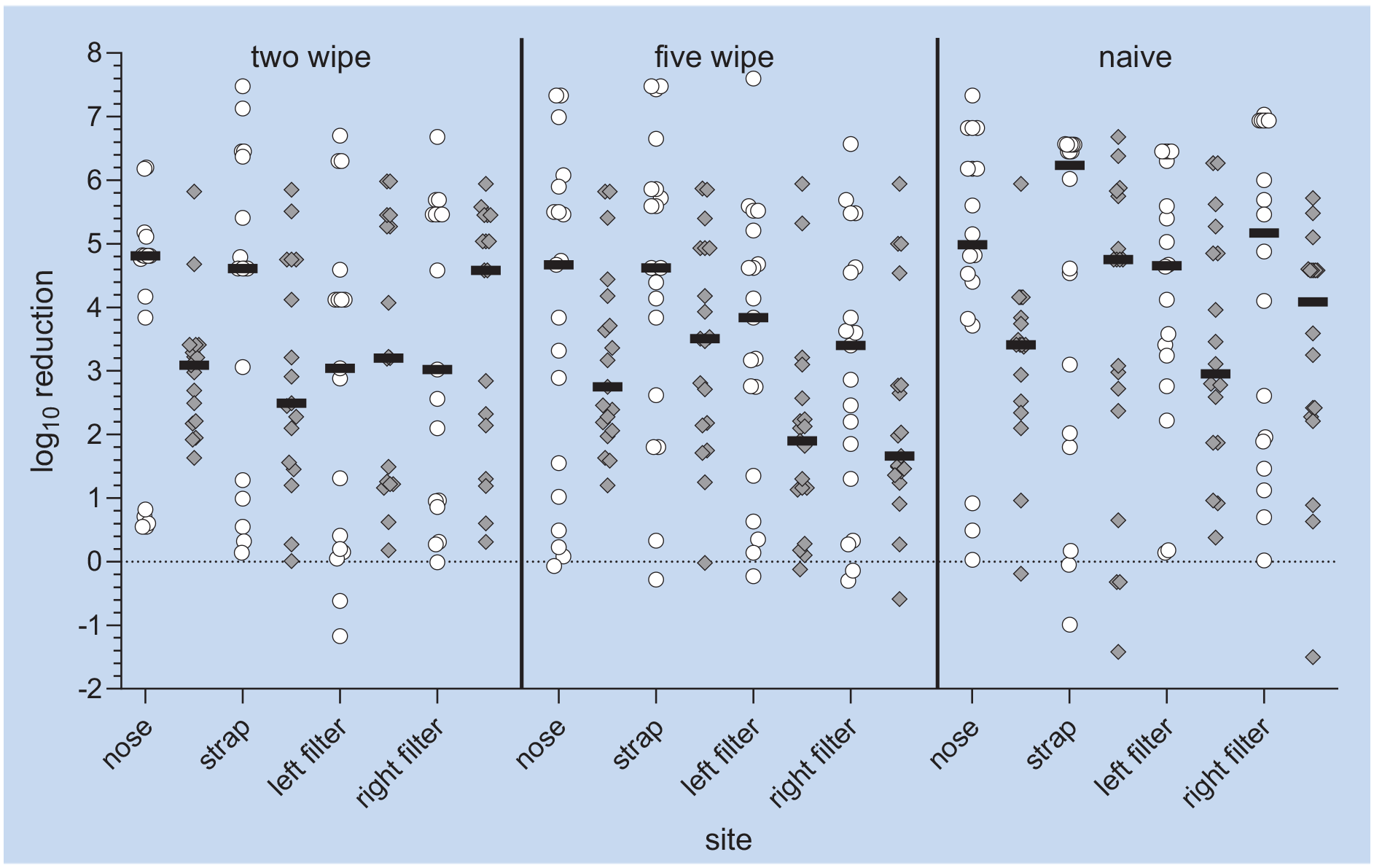
Median log10 Reduction of R. terrigena and MS2 by (a) Site on Elastomeric Half Mask Respirator and (b) Cohort (Cohorts: Two Wipes= Two Wipes and Instructions, Five Wipes=Five Wipes and Instructions, Naïve=No Instructions or Specified Number of Wipes. White Circles=R. terrigena; Gray Diamonds=MS2; Black Lines=Median)
No significant differences for cohorts were found in either MS2 model. Although more time was spent disinfecting the strap than other surfaces, the time a participant spent disinfecting a given site was not a significant predictor of log10 reduction for either organism. No significant interactions were found between site and cohort when selecting the four models.
Discussion
This study explored the feasibility of using commercially available hospital disinfectant wipes for reducing bacteria and viruses on EHMRs under controlled laboratory conditions and by volunteer HCPs. Previous studies have evaluated SOPs for wipe disinfection (Bessesen et al., 2015), the effects of wipe disinfection on filter performance (Vo et al., 2024), and disinfection practices HCPs engaged in during the pandemic (Hines et al., 2020, 2023; Thurman et al., 2023). There is limited literature evaluating disinfection efficacy using respirators inoculated with organisms, although one study found 4.5 log10 reduction using manual cleaning with detergent and bleach immersion (Lawrence et al., 2017). All disinfectants produced greater than 3.3 log10 reductions, and the type of organism influenced efficacy. 0.5% HP and QAC/alcohol most frequently removed all detectable organisms and produced the largest reductions, and QAC/saline had the least activity against all organisms. After disinfection with HP, which has good virucidal, bactericidal, and sporicidal properties (Lin et al., 2020; Omidbakhsh & Sattar, 2006; Rutala & Weber, 2019), Φ6 and P. aeruginosa were undetectable on all sampled areas, with a greater than 8 log10 reduction overall for both. Evidence from studies by other investigators shows that 0.5% HP with 1-min contact time produced a 4 log10 reduction in enveloped viruses and a 6 log10 reduction in bacteria including P. aeruginosa on nonporous inanimate surfaces (Lin et al., 2020; Omidbakhsh & Sattar, 2006). Other investigators also found that HP activity against non-enveloped viruses was similar to activity against MS2 in our study (Omidbakhsh & Sattar, 2006). In our study, HP was not as effective against B. atrophaeus spores as against R. terrigena or MS2; this is consistent with evidence from the literature using HP on rubber, titanium and steel surfaces, suggesting that a higher concentration and a longer contact time may be required for spores (Sagripanti & Bonifacino, 1996).
In the volunteer study, although overall reductions in MS2 were lower than R. terrigena, between-participant variation was considerable for both. Organism recovery was associated with the respirator site. R. terrigena was less likely to be recovered from the strap than from the filter covers and nose. Among sites with detectable MS2 after disinfection, reductions were greatest on the nose. One study with influenza virus found no difference in organism recovery between respirator sites (Lawrence et al., 2017). However, in that study, the respirator was disassembled before the components were disinfected, which may be more appropriate for end-of-shift than between-patient. The evaluation of efficacy had limitations; all wipes contained disinfectant, so it was not possible to separate the effects of mechanical wiping alone on microbial removal or determine if wiping transferred microbes between sites on the surface. The first wipe used might have performed more of a cleaning function, facilitating mechanical removal while subsequent wipes disinfected the cleaned surface. Although participants were not told where contamination was located on the respirator surface, they may have been able to see some small residual dried spots, possibly drawing attention to some sites when disinfecting.
In clinical use, the ideal outcome is to have a surface with no remaining viable microbes after disinfection, which differs from the ideal experimental design. In these experiments, having remaining viable microbes on the respirator surface after disinfection makes it possible to quantify the total log10 reduction. Measuring total log10 reduction reflects the fact that the measurable reduction of any microbe by a disinfectant is always relative to the starting quantity of the microbe pre-disinfection. Measuring log reduction, rather than the absolute number of microbes remaining on the surface after disinfection, allows us to calculate the predicted number of surviving microbes on a respirator from any starting concentration. For example, if the disinfectants tested can achieve a 3 log10 reduction of a particular microbe when used in the volunteer study, it is possible that a respirator contaminated with up to 1,000 microbes could have no surviving microbes after disinfection. The higher the log reduction value, the more likely it is that a contaminated respirator could be free of viable microbes after disinfection. In this study, even with individual variation, on average 3-4 log10 reductions of bacteria and viruses were achieved. Based on reductions, HP alone and QAC/alcohol in combination have the greatest efficacy. Efficacy was higher for vegetative bacteria than viruses and was lowest for spores.
Future studies of the role of mechanical removal, higher disinfectant concentrations, and other training and instruction modalities may be needed to optimize the reduction of all classes of organisms on EHMRs. Duration of disinfection differed by site but not by cohort, and subjects spent more time disinfecting straps and noses than covers. While instructions said which sites to wipe, subjects were not told how long to wipe each site. Instructions and the number of wipes used did not significantly affect reduction. It may be possible that multiple strategies and formats could be used to provide training and instructions; however, these would need evaluation to determine if they affect individual-level variation or microbial reduction overall. Adoption of EHMRs requires addressing logistics (Bowdle et al., 2021; Chalikonda et al., 2020; Pompeii et al., 2020), HCP attitudes (Hines et al., 2017), life of components (Baffoe-Bonnie et al., 2022; Patolia et al., 2021), and choice of disinfection method (Bessesen et al., 2015; Subhash et al., 2014). For reusable respirators, the possible presence of disinfectant residual on the surface needs to be evaluated, as it can have both health effects and effects on the acceptability of respirators, such as if it leaves an unacceptable odor. Hospitals may have options for uniform, replicable centralized processing procedures (Grossman et al., 2020) or devices (Lindsley et al., 2015); however, for initial cleaning and disinfection by HCP between patient encounters, disinfectant wipes have been a suggested option. This study explored the potential utility of disinfectant wipes to be used by the respirator wearer without extensive training or following strict cleaning protocols. The findings indicate that these wipes may be a feasible approach; however, further exploration into this approach is needed to verify its utility.
Footnotes
Acknowledgements
The authors recognize Edward Fisher, Mihili Edirisooriya, and Ashley Whitson from the National Personal Protective Technology Laboratory for their thoughtful reviews of this document.
Attributions
N95 is a certification mark of the U.S. Department of Health and Human Services (HHS) registered in the United States and several international jurisdictions.
Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention. Mention of any company or product does not constitute endorsement by the National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Centers for Disease Control and Prevention, National Institute of Occupational Safety and Health, cooperative agreement 75D30118C02645.
