Abstract
Background:
With the emergence of SARS-CoV-2, healthcare workers (HCW) have relied on reusable personal protective equipment (PPE), including respirators and face shields (FSs). The effectiveness of decontamination procedures outside experimental settings is unclear. We examined the prevalence of surface contamination on reusable PPE used by HCWs at a hospital incorporating daily centralized decontamination and post-use wiping by sampling for common pathogens.
Method:
Samples were collected from HCWs’ CleanSpace Halo respirator face masks (FMs) and FSs at the start of shift, immediately after use, and after cleaning with disinfecting wipes. Samples were analyzed for pathogens using the Applied Biosystems™ TaqPath™ COVID-19 Combo Kit and ThermoFisher TaqMan Array Card. Patient charts were reviewed for clinical correlation.
Findings:
Of the 89 samples, 51 from FMs and 38 from FSs, none tested positive for SARS-CoV-2, despite 58 being obtained from PPE used in the care of patients with COVID-19, many with recent aerosol-generating procedures. Four samples tested positive (4.5%) for Staphylococcus aureus, two each from FMs and FSs. FMs that tested positive were not worn concurrently with FSs that tested positive. The FM and FS samples testing positive were worn in the care of patients without diagnosed S. aureus infection. No FMs tested positive following wipe-based disinfection, but both positive FS samples were found after disinfection wiping.
Conclusion/Application to Practice:
Contamination of reusable PPE appears uncommon, especially with SARS-CoV-2, when regular decontamination programs are in place. The rare presence of S. aureus highlights the importance of doffing procedures and hand hygiene by HCW to prevent surface contamination.
Keywords
Background
Reusable respirator use increased during the COVID-19 pandemic due to shortages in disposable filtering facepiece respirators (FFRs; Hamby, 2020). Healthcare-based use of reusable respirators, such as elastomeric and powered air-purifying respirators (PAPRs), requires assurance that respirator surfaces do not harbor infectious agents or pose risk for fomite transmission to healthcare workers (HCWs) or patients. While COVID-19 infection arising from contact with infected surfaces is thought to be infrequent (Centers for Disease Prevention and Control [CDC], 2021), because contact transmission is possible, hospitals must ensure that reusable surfaces including respirators and face shield (FS) surfaces are adequately free of viral and other microbial contaminants.
Prior studies have demonstrated that viruses such as influenza and SARS-CoV-2 can be detected and may retain infectivity for several days on surfaces of personal protective equipments (PPEs) including respirators (Kasloff et al., 2021; Marques & Domingo, 2021; Meyerowitz et al., 2021). Experimental studies show that influenza-contaminated reusable elastomeric respirator surfaces are completely disinfected following submersion in water with neutral detergent and chemical disinfectant solution (Lawrence et al., 2017). Hospitals have incorporated standardized, centralized, end-of-shift cleaning and disinfection of reusable respirators according to these protocols (Chalikonda et al., 2020; Hines et al., 2021; Koh et al., 2020). In between centralized disinfection, some hospitals instruct HCWs to clean their reusable respirators with disinfectant wipes (Hines et al., 2020). While chemical disinfectants show efficacy against a variety of pathogens, including SARS-CoV-2, it is unclear whether real-world use of chemical disinfectant wipes during a work shift adequately eliminates microbial pathogens from surfaces that could serve as fomites (U.S. Environmental Protection Agency [EPA], 2020).
The CleanSpace ® HALO respirator is a reusable respirator that combines a tight-fitting clear, silicone face mask with a powered air mechanism, which was newly implemented for clinical use at several hospitals within a U.S. academic medical system in 2020. In a prior study, when CleanSpace HALO respirators were experimentally contaminated with influenza virus and simulated facial oil, cleaning of the face mask with detergent and disinfection with chemical disinfectant reliably eliminated the virus, but disinfection only with common hospital wipes did not (Hines et al., 2020). This experiment suggested that the practice of only requiring that a reusable respirator be wiped with a disinfectant wipe during healthcare use is inadequate with respect to the need for assurance of disinfection.
In this study, we aimed to measure surface contamination on FSs and CleanSpace HALO respirator face masks (FMs) by SARS-CoV-2 and other pathogens during routine clinical use. We also evaluated differences in microbial surface contamination on FMs and FSs before and after wipe-based disinfection. We hypothesized that there would be low detectable levels of microbial contamination on the surfaces of FSs and FMs in routine clinical use and that real-world use of disinfectant wipes would significantly reduce levels of surface contamination.
Method
Setting
The study was conducted in the intensive care unit (ICU) of a 272-bed community hospital that is part of a larger academic medical system in August 2021, during the delta variant surge of the COVID-19 pandemic. This 24-bed ICU housed patients with and without COVID-19 and included several airborne isolation rooms with negative-pressure ventilation. The hospital had not previously utilized CleanSpace HALO or reusable elastomeric respirators prior to the COVID-19 pandemic. With the launch of this new respirator, CleanSpace trainers provided live virtual education to small groups of designated respirator “super users.” Education included background about how the HALO respirator worked, its level of protection provided, and how to use, clean, and disinfect the respirator. Each super user practiced donning and doffing and adjusting the respirator for the best fit in front of a web camera, receiving feedback from the CleanSpace trainers. Then, super users fit-tested and trained HALO end users in these same tasks. End users received instructions on wiping the respirator after each doffing and instructions on where to drop off and collect FMs. At the conclusion of training, new users demonstrated respirator donning, doffing, and decontaminating according to a proficiency checklist. This hospital had instituted a program for HCWs to obtain HALO respirator FMs that had undergone centralized decontamination prior to the start of each shift. This decontamination was performed by dedicated staff and included cleaning the FMs by submersion in neutral detergent and water, followed by submersion in a chemical disinfectant and rinse, as per a procedure previously described by Bessesen et al. (2015). During the work shift, after exiting each patient room, HCWs disinfected their reusable PPE, including HALO FMs and power units and FSs, with EPA-registered hospital disinfecting wipes containing 55.5% isopropyl alcohol (IPA), 0.25% quaternary ammonium compound (QAC), and 0.25% benzyl ammonium chloride compounds. This product claimed germicidal activity after 2 min of contact time against 32 microbial pathogens, including SARS-CoV-2, Staphylococcus aureus and Methicillin-resistant S. aureus (MRSA) (EPA, 2020; “Super Sani-Cloth® Germicidal Disposable Wipe—PDI Healthcare,” n.d.). HCWs were expected to don their respirators and FSs and perform hand hygiene before entering patient rooms. They were also instructed to wear disposable procedure masks over the exhalation valve of the HALO FM as a form of source control from potential asymptomatic shedding of SARS-CoV-2 in exhaled breath (Chang et al., 2020). Upon exiting patient rooms, HCWs performed hand hygiene, removed their FSs and respirators, then donned gloves and disinfected their FSs and respirators (both FMs and power units) with wipes. FSs and HALO FMs remained with the HCW during the work shift. HALO power units were returned to charging stations in the nursing unit after use.
Respirator
HCWs used CleanSpace HALO, a PAPR with HEPA filtration. A silicone mask sits over the nose and mouth and is secured by a harness made of silicone and polycarbonate. A power unit composed of polycarbonate and containing an encased filter sits on the back of the neck. An adjustable neck support made of Acrylonitrile Butadiene Styrene plastic fits into the power unit. The entire respirator can be repeatedly decontaminated in accordance with manufacturer instructions and reused (CleanSpace Technology Pty Ltd., 2018).
Sample Collection and Processing
Samples were obtained from respirator FMs and FSs by swabbing surfaces using a standardized protocol with a BDTM Universal Viral Transport System and sterile swab applicator. This system is designed to transport specimens at room temperature. FMs were sampled by swabbing the exterior twice horizontally across the entire FM and twice vertically along the front of the FM. FSs were sampled by swabbing the exterior twice horizontally and twice vertically. Samples were collected at the start of shift (including from FMs that were just obtained from the decontamination station), immediately after use in patient rooms but before doffing, and after doffing and cleaning with disinfecting wipes. Patient charts were reviewed for the presence of documented infections, time from diagnosis of infection, use of airborne isolation precautions, and recent aerosol-generating procedures (AGP) within 6 hr. Samples were transported to the laboratory in insulated containers, processed within 24 hr, and stored at −80°C until analysis.
Laboratory Analysis
Processed samples were analyzed for bacterial and viral pathogens, including SARS-CoV-2, with quantitative reverse transcription polymerase chain reaction. Nucleic acids were first extracted with the MagMax Pathogen RNA/DNA Kit (Applied Biosystems) on KingFisher Duo Prime (ThermoFisher Scientific), following manufacturer protocols specific to sample type, the same day that PCR was to be run. Samples were assayed for SARS-CoV-2 using the Applied Biosystems™ TaqPath COVID-19 Combo Kit. Samples were also assayed using the TaqMan Array Card (TAC, ThermoFisher Scientific) and TaqMan Fast Virus One Step Master Mix (Applied Biosystems). The TAC is a low-density microfluidic card that can detect 21 individual respiratory pathogens including influenza A, influenza B, rhinovirus, adenovirus, Streptococcus pneumoniae, S. aureus, and Hemophilus influenzae. The level of detection (LoD) for the TaqPathTM is 250 copies per sample and TAC LoD ranges between 6,250 and 625,000 copies per sample depending on the target organism.
Sample results were evaluated in relation to factors that potentially could contribute to the presence of surface contamination. The study was approved by the University of Maryland-Baltimore Institutional Review Board (IRB). The IRB granted a waiver of consent for the patient chart reviews and a waiver of written informed consent from the participating HCWs, who provided verbal consent after reviewing an approved information sheet.
Results
A total of 89 samples were obtained, 51 from FMs (57.3%) and 38 from FSs (42.7%). Nine samples were from PPE that had not yet been used that day for patient care, including 7 FMs (one of which had not undergone central decontamination) and 2 FSs. None of the 89 samples tested positive for SARS-CoV-2. Four (4.5%) samples tested positive, all of which were positive for S. aureus. Both positive FM samples came from the same FM—the first obtained at the start of shift prior to first use and the second after use but prior to cleaning with a disinfectant wipe. None of the samples that tested positive for S. aureus were used in the care of patients that had an S. aureus infection. Table 1 shows characteristics of various factors thought to potentially impact PPE surface contamination, including patient characteristics (infection with SARS-CoV-2 or other organism, intubation) and use in a negative-pressure patient room.
Characteristics of Samples Obtained From Personal Protective Equipment
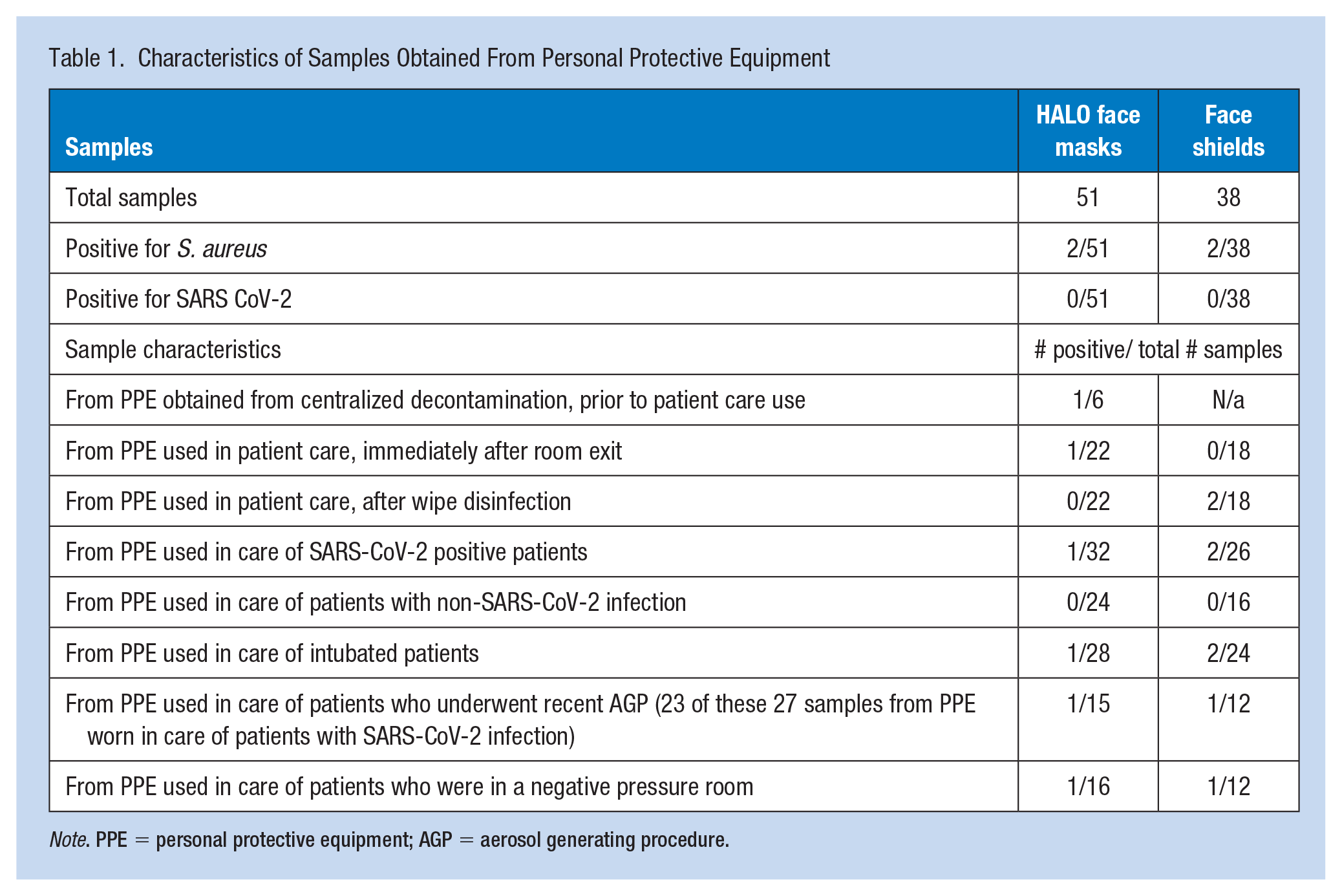
Note. PPE = personal protective equipment; AGP = aerosol generating procedure.
Among samples collected from PPE used during care of patients infected with SARS-CoV-2, these patients had become symptomatic an average of 12.3 days prior and had positive PCR testing 9.1 days prior to sampling. Among the 44 samples obtained from FMs used in patient care prior to sampling, HCWs had worn either a disposable procedure mask or FS over their FM in 36 cases (81.8%) (not shown).
Table 2 shows the potential impacts of the duration of PPE use and time since wipe disinfection on surface contamination detected by wipe samples. No consistent patterns were observed in either length of time of PPE use or in time since wipe disinfection associated with positive or negative samples.
Timing of PPE Use and Disinfection Among Positive and Negative Samples
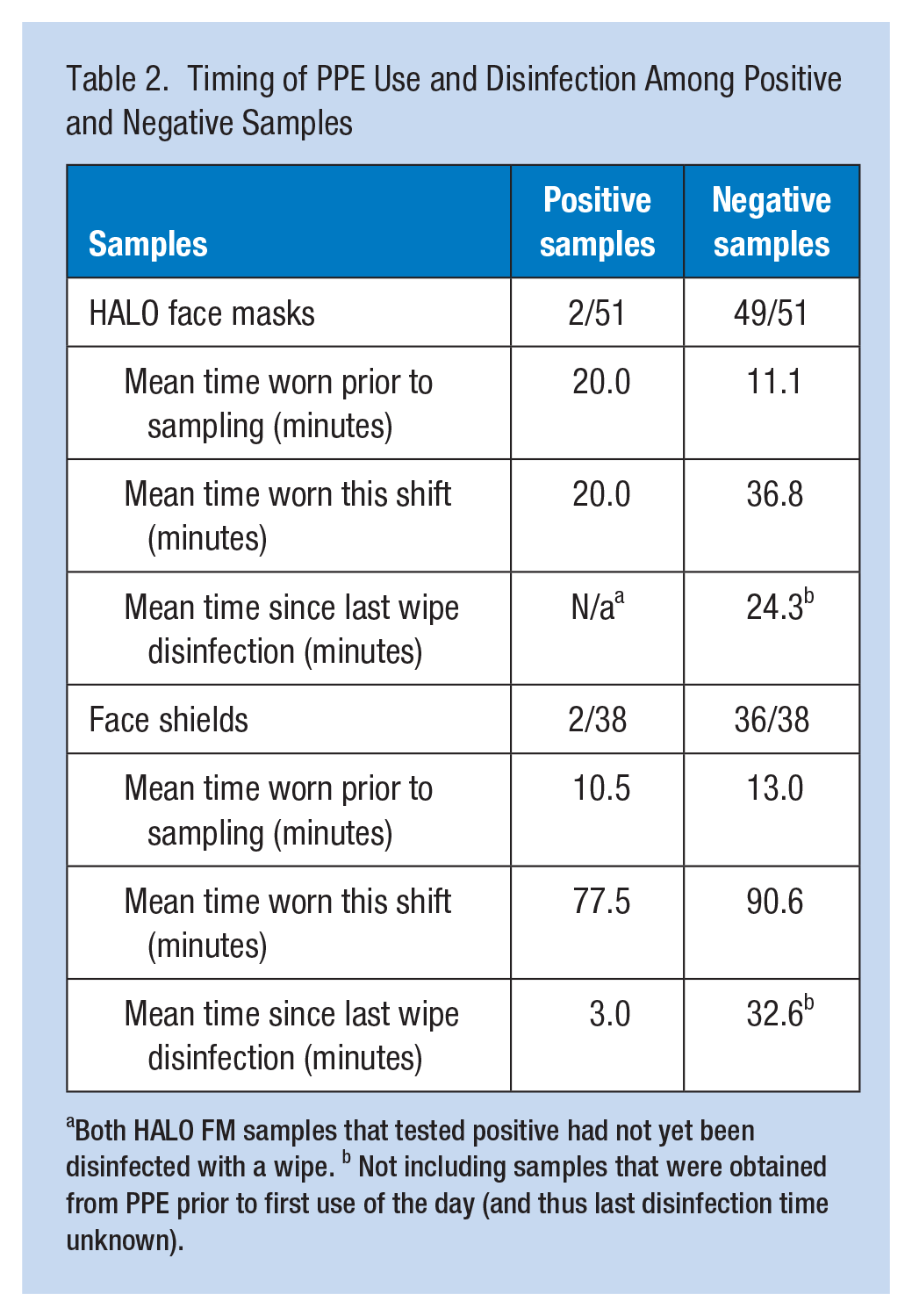
Both HALO FM samples that tested positive had not yet been disinfected with a wipe. b Not including samples that were obtained from PPE prior to first use of the day (and thus last disinfection time unknown).
Discussion
In this study, we found that surface contamination on FSs and CleanSpace HALO FMs was uncommon and when present was due to S. aureus, an organism commonly found on the skin (Taylor & Unakal, 2022) We measured microbial surface contamination by SARS-CoV-2 and other pathogens on in routine clinical use during the COVID-19 pandemic. We also assessed for differences in microbial presence pre- and post-disinfection with common hospital wipes.
These results were found in a setting where HCWs decontaminated their respirators and FSs with disinfectant wipes following each patient room exit and submitted their FMs for central cleaning and disinfection at the end of each shift. Our results suggest that this strategy is associated with low risk for HCW contact with SARS-CoV-2 on CleanSpace HALO FM surfaces. We found S. aureus on two FM samples. Both positive samples came from the same FM, the first obtained immediately prior to and the other immediately following patient care but before disinfection wiping. It is unclear if contamination occurred from contact with the HCW’s skin, a surface where the FM may have rested before donning or contact with skin or a surface following centralized decontamination. The sample obtained immediately post-wipe from this same FM showed no contamination, as expected. Both FS samples testing positive for S. aureus, however, were obtained immediately following post-use disinfection wiping. Samples from both FSs that had been taken immediately post-use, but before disinfection, showed no contamination. The FMs from these HCW did not test positive for S. aureus. This suggests that the HCW wiping the FS contaminated the FS with their own skin, rested the FS on a contaminated surface during drying, or that the disinfection wipe did not completely remove surface contaminants.
Studies of surface contamination by SARS-CoV-2 have shown variable patterns. Our finding of no surface contamination by SARS-CoV-2 is similar to findings from some authors. These studies have found low rates of contamination, ranging from 0% to 8% of specimens, where positive specimens were found on HCW gowns, stethoscopes, phones, a shoe, and an endotracheal intubation tube (Nakamura et al., 2020; Ong, Tan, Chia et al., 2020; Ong, Tan, Sutjipto et al., 2020; Peyrony et al., 2020). Other studies, however, have reported frequent SARS-CoV-2 surface contamination, ranging from 15% of HCW PPE to 78% of HCW cell phones and 57% of patient rooms with any positive surface samples (Chia et al., 2020; Jung et al., 2020; Pasquarella et al., 2020; Santarpia et al., 2020). Given the variability in the prevalence of surface contamination, multiple factors likely contribute, including the amount and duration of viral shedding from the patient, porosity and antimicrobial properties of surfaces, and the presence of engineering controls in the room.
Our findings of limited S. aureus contamination are like studies showing bacterial surface contamination of items used repeatedly in healthcare. Many studies have cited high frequencies of HCW cell phone bacterial contamination, commonly by S. aureus (Kalra et al., 2021; Malhotra et al., 2020; Panigrahi et al., 2020). Furthermore, some data suggest that even disinfected stethoscopes maintain significant rates of bacterial contamination (Kalra et al., 2021). Thus, our findings are consistent with other studies showing that S. aureus surface contamination is not uncommon.
Our study has several limitations. We did not assess viral viability with culture; however, our detection of no viral ribonucleic acid by PCR, a more sensitive indicator, suggests that this does not affect the interpretation of our results. Second, we sampled surfaces following short periods of use. While this differs from the extended use periods that occurred commonly at the beginning of the pandemic, short duration use likely reflects common patterns for healthcare respirator use (Kobayashi et al., 2020). Third, we did not measure air samples, which would have aided our understanding of risk of room contamination from SARS-CoV-2. The surface samples in this study were obtained from PPE worn in the care of patients who were several days into their illnesses, intubated, and mainly in airborne isolation rooms with negative pressure room ventilation. Studies have suggested that surface contamination from COVID-19 patients is more likely to occur early in the disease course, before day 7 (Chia et al., 2020; Santarpia et al., 2020). Thus, while our results may not reflect the highest risk periods for HCW exposure, it likely reflects the exposure scenarios during care of prolonged acute COVID-19 patient illness. In addition, while many HCWs wore procedure masks over the exhalation valve of the respirators, which likely limited FM surface contamination, the lack of SARS-CoV-2 on FSs, which also cover the FMs, suggests that this does not significantly alter the interpretation of our results.
Although the testing positivity was low for both FMs and FSs, these findings highlight several areas for focus with healthcare reuse of PPE. First, HCWs must completely wipe the surfaces of their PPE. Second, doffing areas should be clean and free of microbial contaminants and support a standardized post-doffing practice. Third, disinfectants should have adequate properties against common hospital microbial pathogens. Although the wipes used in this setting were IPA and QAC-based disinfectants with claims against S. aureus, some literature suggests that hydrogen peroxide-based disinfection may be more effective against S. aureus, including MRSA, compared with routine cleaning methods in hospital settings (Dancer, 2014; French et al., 2004). Finally, given that FSs may be worn at times when respirators are not required, FSs may face more hand-to-surface and surface-to-surface contact. These devices require particular attention to decontamination.
Our findings contribute to understanding of healthcare reusable PPE use in several ways. First, centralized respirator decontamination adds a layer of standardized cleaning and disinfection that likely significantly decreases the opportunities for sustained microbial contamination. Like other reusable equipment such as blood pressure cuffs, reuse of PPE should include attention to general housekeeping measures to limit contamination from commonly encountered microbes, especially those found on the skin. The use of appropriate disinfectant strategies against these microbes must be a routine component of policies and procedures addressing reusable PPE.
In summary, we found no surface contamination with SARS-CoV-2 and infrequent contamination with S. aureus on reusable FSs and CleanSpace HALO respirator FMs disinfected after each patient room exit during the COVID-19 pandemic. Reuse of PPE allows hospitals to maintain sustainable resources for HCW safety. Routine, evidence-based disinfection strategies can provide assurance that HCWs have PPE that effectively provides protection and does not pose risks for fomite transmission of microbial contaminants.
Implications for Occupational Health Practice
HCWs have increasingly relied on reusable PPE since the onset of the COVID-19 pandemic. This equipment, while necessary to protect workers, could potentially act as a fomite for transmission of SARS-CoV-2 or other pathogens. The efficacy of decontamination and cleaning practices outside of experimental studies was not known. This study demonstrates that post-shift centralized decontamination and routine post-doffing use of disinfecting wipes are associated with a low risk of contamination of reusable PPE with SARS-CoV-2 and other respiratory pathogens. These results are reassuring for HCWs involved in the care of patients with common respiratory pathogens.
Applying Research to Occupational Health Practice
This study finds a low rate of contamination of reusable personal protective equipment (PPE) with common respiratory pathogens, including SARS-CoV-2, at a site using centralized daily PPE decontamination and post-doffing use of hospital disinfecting wipes to decontaminate respirator face masks and face shields. Hospital respiratory protection and infection prevention leaders should ensure appropriate processes are in place for the decontamination of PPE. Healthcare workers must be diligent in practicing hand hygiene and regularly disinfect PPE to minimize the risk of surface contamination.
Footnotes
Acknowledgements
The authors would like to thank Jason Heavner, MD, Jordan Assadi, DO, Sandra Thomas, RRT, and Carol Ann Sperry, RN, MS for their assistance in completing the study.
Authors’ Note
Data collection instruments may be obtained by contacting the authors.
Conflict of Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A family member of SH works as an instructional trainer for CleanSpace Technology and was not involved in any of the current research endeavors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant to the University of Maryland School of Medicine, contract CCT 3809-19 (Hines, PI) by CleanSpace Technology Pty Ltd.
Human Subjects Review
The study was approved by the University of Maryland-Baltimore Institutional Review Board (IRB) (HP-00096953). The IRB granted a waiver of consent for the patient chart reviews and a waiver of written informed consent from the participating HCWs, who provided verbal consent after reviewing an approved information sheet.
