Abstract
Do different pharmaceutical product liability regimes in different countries induce propensity to patent? We exploit the variation in pharmaceutical liability and litigation rules across firms in the pharmaceutical industry and countries to explain the firm-level propensity to patent. Drawing on a large dataset from European Patent Office (EPO) covering over 9,950 pharmaceutical patents from 63 countries over the period 1991–2015, we compute the conditional probabilities of individual pharmaceutical firms to acquire a valid-based patent on the validation outcomes and examine whether different liability regimes encourage or deter firm-level propensity to patent. Our empirical strategy addresses firm-level idiosyncrasies, country-level unobserved effects, and common technology shocks that potentially invoke omitted variable bias in the effects of liability regimes on the propensity to patent. Our investigation reveals that liability regimes combined with damage caps, broad statutory excuses, and reversed burden of proof have a strong positive effect on the firm-level patent stock and a negative effect upon EPO patent validation rate. The evidence suggests that not all liability rules and related litigation procedures are created equal. Firms are systematically more likely to hold (firm-level patent stock) valid patents at the EPO when the liability and litigation rules are not complex and when the damage cap, broad statutory excuses, and reversed burden of proof are introduced.
Introduction
The essence of product liability is the appointment of the risks inherent in the modern mass production of goods. Particularly, the pharmaceutical product liability is a complex set of different legal regimes and regulations. In the last decades, scholarship devoted its attention toward the potential detrimental effects of different tort law regimes and product liability on the innovative activity (Blind, 2012; Carlin & Soskice, 2006; Crafts, 2006). Liability effects on the economic performance of the pharmaceutical industry have played a leading role in this debate (Green 1996). Over the last decades, the essence of liability law around the globe has shifted from negligence liability to the concept of strict product liability. This change has been applauded by many as a complete prevalence of consumer protection and consequently also of safer goods and services. Theory suggests that in such a world enhanced quality, safer products, and increase rate of innovation should materialize. However, scholars found that actually the reverse occurred (Herbig and Golden, 1994; Malott, 1988; McGuire, 1988). They show that product liability costs in the United States have prompted some manufacturers to abandon valuable new technologies, life-saving drugs, and innovative product designs (Herbig and Golden 1994; Malott, 1988; McGuire 1988).
Another stream of literature investigated the related issues of product liability and its detrimental effects on innovations. For example, Porter (1990) argues that in the “U.S. product liability is so extreme and uncertain that is actually retarding innovation.” Viscusi and Moore (1993) examined these competing effects of liability costs on product R&D intensity and new product introductions by manufacturing firms. They show that at “low to moderate levels of expected liability costs, there is a positive effect of liability costs on product innovation,” whereas at a “very high levels of liability cost, the effect is negative” (Viscusi & Moore, 1993). Pelkmans and Renda (2014) show that “more prescriptive EU regulation tends to hamper innovative activity, whereas the more flexible EU regulation induces better innovation” (Pelkmans & Renda, 2014). Mansfield (1986) shows that the innovation effects of the patent system are substantial and also find that the bulk of patentable inventions in the pharmaceutical industry are patented. Finally, in their recent work, Galasso and Luo (2020) examine the effect of product liability on innovation by exploiting a major quasi-exogenous increase in liability risk faced by U.S. suppliers of polymers. They show that “liability risk can percolate throughout a vertical chain and may have a significant chilling effect on downstream innovation” (Galasso & Luo, 2020).
However, the particular effects of exposure to different liability regimes on the propensity to patent and on the innovative activity measured by the stock of patents in the pharmaceutical industry are still out of sight. In other words, do stricter liability rules and other embodiments/materialization of liability costs also deter propensity to patent?
It has to be emphasized that not all patentable inventions are patented, since in some cases, firms rely instead on trade secrets or simply because patent protection, due to very difficult and costly copying, may not seem worthwhile. To achieve better understanding of the extent to which various firms make use of the patent system, Mansfield (1986) suggests the exploration of the propensity to patent.
This article examines the economic incentives of pharmaceutical companies stemming from product liability and other forms of litigation rules and considers effects on the firm-level number of patents and on the firm-level validation rates. It should be of interest to policymakers and policy analysts who seek to understand the economic effects of pharmaceutical product-liability regime on the propensity to patent in the pharmaceutical industry as well as to legal practitioners and policy advocates as they formulate their policy arguments. Namely, product-liability regime might be, under certain circumstances, a powerful boost to innovation, but can also, if too restrictive, disable innovation (Hay & Spier, 2005; Jarell & Peltzman, 1985; Leitzel, 2015; Posner, 2011; Schäfer & Ott, 2004; Shavell, 2007).
In this article, we exploit the variation in cross-country and cross-firm transaction costs to explain the propensity of firms to innovate. Drawing on a large dataset from European Patent Office (EPO) covering over 9,950 pharmaceutical patents from 63 countries over the period 1991–2015, we compute the conditional probabilities of individual firms to acquire a valid patent and construct firm-level propensities to innovate, and examine whether different liability and related litigation’s rules encourage firm-level stock of patents. Our empirical strategy specifically controls for firm-level idiosyncrasies, country-level unobserved effects and common technology shocks that potentially invoke omitted variable bias in the conditional effects of liability costs on the propensity to patent.
Our work contributes to the literature by considering the relationships between propensity to patent and litigation and liability laws generated by different legal systems. The paper defines propensity to patent as the probability to apply for and/or obtain a patent, given a patentable invention (Mansfield, 1986). The employed identification strategy is a standard difference-in-difference method exploiting several changes in the liability regime to estimate how many patent firms hold and how likely are they to push aggressively for patent protection. Our investigation suggests that current product liability regimes and related litigation laws across firms and countries do not account for the failure of pharmaceutical firms to acquire a valid patent. The results actually reveal that stricter litigation and liability rules across firms, between and within countries, have a significant strong negative effect on the stock of EPO patents, yet that such rules have a positive effect on the validation rate (validation success). Thus, product liability and related litigation laws might be perceived as a filter that screens opportunistic firms from filling dubious, rent-seeking EPO’s patent applications. However, firms are then systematically more likely to have (firm-level patent stock) valid patents at EPO when the damage cap, broad statutory excuses, and reversed burden of proof are introduced.
Particularly, obtained results suggest the following: (a) that existence—legal availability—of class action suits have no effect on the firm-level patent stock; (b) existence of broad statutory excuses (i.e., nonexistence of ex ante technological and scientific knowledge, or a federal pre-emption defense in United States) have a strong positive effect on the firm level stock of patents; (c) the evidence clearly suggest that procedural damage caps do not exert a systematic and statistically discernible effect on the firm-level patent stock; (d) burden of proof and causality (whether the claimant—injured party—needs to prove a causal link between defective drug and the damage) has a significant strong positive effect on the stock of patents; (e) existence of punitive damages has a strong negative effect on the firm-level stock of patents; (f) decision-making by juries has no effect on the stock of patents; and (g) special rules on pretrial discovery (which at least in theory should favor claimants—injured party) has a strong negative effect on the firm level stock of patents. In addition, results confirm the fundamental importance of low-cost litigation and liability costs in explaining the differential propensities to patent across firms and countries in the pharmaceutical industry. Moreover, this article in line with Galasso and Luo’s (2020) findings also shows that large firms and small firms indeed do react differently on the assessed types of laws.
However, several caveats should be stated. Namely, the aim of the article is not to impose a final word on the matter, but to undertake an exploratory analysis of the relationship between the pharmaceutical liability regime and firm-level propensity to patent. Moreover, one can never be sure that there still are omitted variables and indeed there are further issues that have to be addressed as for example issues of (a) sectoral heterogeneity, (b) contingency fees, (c) differential timing of changes in liability rules, (d) sample selection bias, and (e) pre-existing patenting trends. Furthermore, the employed coding of different rules and legal regimes is open to generalizations and conceptual limitations and may not reflect the entire complexity of assessed legal regimes. In addition, it has to be emphasized that valid patents and the propensity to patent, as Price (2020) convincingly shows, do not necessarily reflect the workable and safe innovation.
This article is organized as follows. In the “Conceptual and Legal Framework” section, we construct the conceptual framework for interpreting the effects of liability and related litigation and other administrative transaction costs on the stock of patents and on the validation rate (propensity to patent). This section also surveys the relevant legal concepts and liability regimes. Section “Data and Methods” discusses data and methodology while “Results—Product Liability, Related Litigation and Propensity to Patent” section discusses the results and presents our main findings and the robustness checks. “Conclusion” section concludes the article.
Conceptual and Legal Framework
Legal literature argues that “liability has a negative effect on firm’s willingness to develop new technologies” (Daemmrich, 2003; Lacetera & Orsenigo, 2001; Priest, 2011; Young, 1982) and often underlines the arguments for tort law reforms. Moreover, Galasso and Luo (2017) investigate the indirect and dynamic effects of tort law reforms upon the incentives to develop new technologies. They offer a theoretical framework that identifies channels through which tort reforms may affect innovation incentives and then empirically show that patenting in medical instruments is reduced roughly by 14% in the presence of caps on noneconomic damages (Galasso & Luo, 2017). Their model suggests that tort law reforms increase physician’s propensity to adopt riskier technologies by mitigating the “chilling effect” of high liabilities (Galasso & Luo, 2017). They also find that on average laws that “limit the liability exposure of health care providers are associated with a significant reduction in medical device patenting” (Galasso & Luo, 2017). Their result underscores the importance of considering dynamic effects in the economic analysis of torts and shows that tort laws have the strongest impact in medical fields in which the probability of facing malpractice claim is the largest (Galasso & Luo, 2017). Finally, in a recent paper, Galasso and Luo (2020) show that large incumbent firms are in their patenting activities significantly more responsive than small firms in terms of new product introductions. They also suggest that risk perception may be an important factor that drives innovation, shapes direction of technological progress and substantially affects the market structure (Galasso & Luo, 2019; 2020).
Product Liability and Legal Regimes
Pharmaceutical products are paradoxical, since they may save millions of human lives but can simultaneously, due to their powerful ingredients (and consequent side-effects), cause enormous damages and suffering. Due to this, potentially hazardous, characteristic of pharmaceuticals tort law is recognized as a very powerful mechanism that compensates victims harmed by defective drugs. Furthermore, such tort law may also “deter sale of defective pharmaceuticals and provide incentive for production and sale of safe pharmaceuticals” (Owen, 2005; Rabin, 2000; Schwartz, 1988).
Products liability law governs the private litigation of product accidents and defines the “legal responsibility of sellers, producers and other commercial transferors of products for harms resulting from product defects and misrepresentations about products safety or performance capacities” (Owen, 2005). In addition to this traditional substantive law, the European “Directive on the approximation of the laws, regulations and administrative provisions of the Member States concerning liability for defective products” (85/374/EEC, OJL 210) introduced also an important dimension of EU law (Fairgrieve & Vaque, 2005).
For example, in Germany, liability for drugs to some extent differed from the previously mentioned Directive (Lenze, 2005). Namely, the Drug Act of 1976 included “liability for development risks, rendered insurance compulsory and in 2002 incorporated rules on causation and special rules on disclosure of information” (Brock, 2002). In principle, the claimant has to prove a causal link between the defective drug and the damage. Yet, in 2002, German law-maker “reversed the burden of proof and actual causation was presumed if the drug in question was, in the actual circumstances capable of causing such damage” (Bollweg, 2004). However, such a reversed burden of proof contradicted the Article 13 of the “Directive on the approximation of the laws, regulations and administrative provisions of the Member States concerning liability for defective products” (85/374/EEC, OJL 210) and has been reversed to its previous, classical causation. Germany also features a “special compulsory insurance according to which a party placing a medical product on the German market must be insured against all liability” (Lenze, 2005). In consequence, the German insurance industry has created a “special pharma-insurance through which all major insurance companies pool the statutory strict liability risk” (Lenze, 2005). German system is also very restrictive in relation to pre-trial discoveries, as there is “no pre-trial discovery procedure and no consequential liability for non-disclosure of documents” (Lenze, 2005).
Generally, in the United States, product liability encompasses strict liability for manufacturing defects and liability for warning and design defects manufacturers of pharmaceutical (Baldwin et al., 2009; Goldberg & Zipursky, 2010). Moreover, in the United States, pharmaceutical products are heavily regulated by the Food and Drug Administration Agency, which “regulates the design of prescription drugs, appropriate warnings and related user’s instructions” (Owen, 2005). In addition, the area of pharmaceutical product liability is a “constant shuffle between supporters of consumer protection and of dynamic commercial innovativeness” (Hodges, 2005).
This article, due to large variety of legal rules, focuses solely on the following, the most significant legal rules: (a) strict liability (whether liability is a strict one or whether the assessed country has any other liability regime—for example, contributory negligence); (b) class actions (whether class actions are possible); (c) special information duty (existence of special information disclosure duties—for example, the German disclosure duty); (d) existence of broad statutory excuses (i.e., nonexistence of ex ante technological and scientific knowledge, or a federal pre-emption defense in United States; (e) damage cap; (f) causality—burden of proof (whether the claimant needs to prove a causal link between defective drug and the damage); (g) existence of punitive damages; (h) decision-making by juries; (i) existence of contingency fees; (j) looser pays all costs rule; and (k) special rules on pretrial discovery (which at least in theory should favor claimants).
Furthermore, one should also note the so-called “Federal preemption” in the United States which is an affirmative defense on which the defendant has the burden of proof. It arises, and a product liability of a firm is foreclosed, when the “claim somehow conflicts with a federal product safety statue or regulation specifying design, marketing or manufacturing standards” (Ausness, 2004; Owen, 2005; Scordato, 2001; Smith and Grage, 2000).
Data and Methods
Patent Applications
Our data on the legal status of patent applications are from European Patent Office’s (EPO) PATSTAT database, which contains bibliographic and legal status firm-level patent data from leading industrialized and developing countries for the period 1995–2015. Sixteen different forms of legal statutes are broadly classified and systematized into four broad categories. The first category entails the patent applications sent to EPO. This category is used to code firm-level observations based on whether the patent application has been submitted to EPO. The second category comprises the pooled firm-level observations for which the patent application has been approved and official validated. This category comprises the firms for which a valid patent has been approved in a given year. The third category comprises the firms whose patent application has been rejected by EPO on various grounds, which exceed the scope of this article. Fourth, the remaining forms of legal status were coded into miscellaneous category, which amounts to a minor fraction of the whole set of applications and which are omitted from the empirical analysis.
In Table 1, we present the key descriptive statistics on patent applications and the associated legal status for the core sample comprising 63 countries. For the sake of consistent comparison, small countries are omitted from the table. The key descriptive parameters are based on the patent application data from 8,875 firms from EU and non-EU countries. The total number of patent applications in the period 1995–2015 amounts to 24,362. The number of cells equals 318,400 taking into account the number of observations, the number of policy treatment variables, and the number of covariates. This also implies that the use of multi-way clustering is preferred and this is the strategy that we undertake in our empirical investigation.
Patent Applications and Legal Status by Country, 1995–2015.

About 55% of the applications have been validated and granted a valid patent. In absolute terms, most applications came from Germany followed by the United States, Italy and Switzerland while most patent applications were validated for German firms followed by the U.S., Swiss, Italian, and French firms. Excluding small and micro states with population less than 100,000, in per applicant firm terms, the highest number of valid patents was approved to the firms originating in South Korea, Switzerland, Japan, Luxembourg, the United States, and Belgium. The least successful countries in terms of the number of valid patents are Bulgaria, Chile, Russia, Croatia, Latvia, and Iceland. The median number of valid patents per applicant firm corresponds to 1.00. Using similar exclusion criteria, in per capita terms, most valid patents were acquired by the firms originating in Luxembourg, Switzerland, Austria, Slovenia, Denmark, Belgium, and Sweden whereas as the least successful firms in per capita-based patent validation outcomes are from Russia, South Africa, Argentina, Mexico, India, Brazil, and China. Excluding outliers, the median per capita number of valid patents corresponds to 3.24, which lies in the observed range between Czech Republic and South Korea.
Considering the validation outcomes statistics, firms with the highest validation rate are most likely to originate from countries with a low per capita valid patent count such as Estonia, China, Malaysia, Brazil, and South Korea. Excluding small and microstates, the lowest rate of validation is observed in Greece, Malta, Slovakia, Croatia, Latvia, Iceland, and Albania. The mean patent validation rate in our sample is 0.54, which corresponds to the same level as the one observed for Austria and United Kingdom. Twenty-nine countries, or roughly 46% of country-level sample, have validation rates below the median while the validation rate in the remaining 54% of the sample is located above the median. The mean rejection rate on patent applications is 40%. In percentage terms, firms with the rejection on patent applications are most likely to originate in Iceland, Latvia, Croatia, and Slovakia while the lowest rejection rates are found among firms from Malaysia, China, Estonia, and Brazil. Considering the EU subsample, the highest rejection rates are found among firms from Latvia, Croatia, Slovakia, Bulgaria, and Hungary while the lowest ones are observed in Estonia, Norway, Slovenia, Sweden, and Poland. Twenty-five countries have the rejection rate below the sample mean, which comprises roughly 39% of the country-level sample. The descriptive statistics clearly suggests a wide dispersion in patent validation outcomes across firms and countries, which indicates the source of heterogeneity in patenting activity which deserves further attention. A widespread variation in patent validation outcomes across firms and countries is further emphasized by the country-level means on the rate of approved patents (validation rate), and on the rejection rate.
Pharmaceutical Industry and Firms
The data on the firm-level stock of patents and validation rates for the pharmaceutical industry are from hand-picked dataset derived and gathered from the entire EPO dataset. The data on 11 different types of liability rules are matched with each pharmaceutical firm in our sample in considering the effects of such costs on firm-level innovation (stock of patents) and patenting activity. We have examined every legal regime of analyzed countries in relation to the liability rules and classified them accordingly (see Appendix for further details). Moreover, in our study, we focus solely on the legal regimes of 56 countries including the United States, the United Kingdom, India, China, South Korea, Japan, Brazil, and so on (see Appendix for further details). The liability rules and related litigation rule indexes ranges from 0 to 1 where zero value indicates nonexistence of such legal instrument and where value 1 indicates the existence (and de facto enforcement) of such legal institution (and consequently high or low litigation and liability costs for pharmaceutical industry).
Liability rules and related litigation rules
Strict liability
The strict liability variable is defined as liability of pharmaceutical company without fault. Fault is generally considered to be a “synonym for intentional or negligent conduct, and this then implies that strict liability is liability without intentional or negligent conduct” (Koch and Koziol, 2002; van Dam, 2007). However, one should note that strict liability for defective products holds some elements in “the requirement of defect that resemble the elements of negligent conduct” (Koch and Koziol, 2002). Consequently, we have classified each observed legal regime according to whether a particular legal regime has for pharmaceutical products a system of strict liability or any other alternative liability regime (e.g., contributory negligence).
Class action
The class action variable is denoted as a class action lawsuit where one of the parties is a group of people (consumers) who are represented collectively by a member of that group. The class action although originated in the United States (Posner, 2011), also Canada and several European countries have, in recent years, introduced class action. For example, on November 1, 2005, Germany enacted the “Act on Model Case Proceedings in Disputes under Capital Markets Law” allowing sample proceedings to be “brought before the courts in litigation arising from mass capital markets transactions” (Halfmeier & Fees, 2014). It does not apply to any other civil law proceeding and it is not like class actions in the United States—it only “applies to parties who have already filed suit and does not allow a claim to be brought in the name of an unknown group of claimants” (Steinberger, 2016). To capture the potential effect of class actions upon the firm-level stock of patent rates, we have classified each assessed legal system according to whether a particular legal regime has a possibility of class actions in place or whether it forbids such class actions.
Special information duty
The special information disclosure duty variable depicts the existence or nonexistence of special information disclosure duties in different legal regimes. Namely, German law for example contains a specific disclosure duty and in pharmaceutical product liability cases (Pfeiffer-Gersche et al., 2013), a claim for information can be made under section 84a of the German Federal Drug Act (BGBl. I S. 3394). The right refers to effects, side-effects and interactions known to the pharmaceutical entrepreneur as well as suspected cases of side-effects and interactions brought to his attention and all further knowledge that could be of significance in assessing the justifiability of harmful effects. Thus according to such duty, a claimant can, from the pharmaceutical firm, demand the disclosure of information, particularly in relation to adverse events.
Broad statutory defenses
This “broad statutory defenses” variable depicts the existence or nonexistence of broad statutory defenses and excuses available to pharmaceutical industry in different legal regimes (i.e., non-existence of ex ante technological and scientific knowledge, or a federal pre-emption defense in the United States; Ginsburg & Huntington, 2013). Namely, such broad statutory defenses to a product liability claim are in line with the Product Liability Directive (Council Directive 85/374/EEC of 25 July 1985). Under such broad statutory excuses, a pharmaceutical’s liability is excluded if the producer can prove any of the following: “(a) the producer did not put the product into circulation, (b) in the circumstances it is probable that the defect which caused the damage did not exist at the time when the producer put the product into circulation, (c) the product was not manufactured by the producer for sale or any other form of distribution for economic purposes, or manufactured or distributed by him in the course of his business, (d) the defect is due to compliance with mandatory regulation at the time the product was put into circulation, and (e) the state of scientific and technical knowledge at the time when the producer put the product into circulation was not such as to enable the existence of the defect to be discovered—development risk defense (Ginsburg & Huntington, 2013).” Furthermore, a producer of a component part is not liable if it proves that the defect is attributable to the design of a product in which the component part has been fitted, or to the instructions given by the producer of the product (Ginsburg & Huntington, 2013). Moreover, under the general tort law, further defenses are available (Posner, 2011). As the liability is fault based the “producer can claim not to have acted negligently” (Shavell, 2007). A claim will be dismissed if the action is “time-barred and the defendant invokes the statute of limitation” (Ginsburg & Huntington, 2013).
In addition, we classified as such broad statutory defense also the so-called federal pre-emption defense in the United States (Ginsburg & Huntington, 2013; Davis, 2004; Owen, 2004). The federal pre-emption is generally referred to as the “power of the federal government under the Supremacy Clause of the United States Constitution to displace a state law in favour of a federal law or regulation, when the state law conflicts with the federal law or regulation” (Burnham, 2006; Caleb & Roosevelt, 2019).
Damage caps
The damage cap variable reflects the existence or non-existence of damage caps in different procedural laws and whether under such laws, there is or there is no limit on the type of damages recoverable. Namely, assessed statutes sometimes do establish strict liability and set a maximum limit on the producer’s liability. For example, the German Product Liability Act sets “a limit of EUR85 million for damages to persons caused by the same defect” (Product Liability Act of 15 December 1989). If the defect has “caused damage to property other than the product itself, this limit does not apply” (Zekoll, 1989). Moreover, under the Federal Drug Act, liability is capped for “lump sum payments to EUR600,000 and for annual pension payments to EUR36,000 per claimant” (Magnus, 2016).
Causality and burden of proof
The procedural rules on causality and reversed burden of proof depict the fact whether in assessed legal regimes either the claimant or the pharmaceutical firm needs to prove a causal link between defective drug and the sustained damage. For example, under the German Product Liability Act and in tort law, the claimant (consumer) and not the producer must “prove beyond reasonable doubt that the product is defective and has caused damage” (Magnus, 2016). Prima facie evidence can suffice if “the damage is a typical result of the product defect, unless the producer can prove a possible alternative cause for the damage” (Magnus, 2016). However, the producer might also have the “burden of proof for any defenses it claims” (Magnus, 2016). Namely, generally, liability under tort law is “fault-based and linked to a negligent breach of a duty of care” (Zekoll, 1989). The producer must than typically prove that it complied with its duty of care and a “possible defense is that the producer’s production processes, including quality control, complied with the state of the art at the relevant time” (Magnus, 2016).
Punitive damages
The punitive damage variable reflects the existence or nonexistence (or de fact nonenforcement) of punitive damages remedy in addressed legal regimes. Punitive damages are generally defined as “damages assessed to punish the defendant for outrageous conduct and/or to reform or deter the defendant and others from engaging in conduct similar to that which formed the basis of the lawsuit” (Posner, 2011). Although the purpose of punitive damages is not to compensate the plaintiff, the “plaintiff will receive all or some of the punitive damages award” (Shavell, 2007). For example, van Dam (2007) suggests that in the United States tort law has become infamous for its punitive damages (or the so called exemplary damages), which are extraordinary high awards of millions of dollars awarded by juries who want to punish the tortfeasor for his wrongful conduct.
Thus, we classified the existence and enforcement of such punitive damages in each of the assessed states. Moreover, we classified the existence of such punitive damages also where the elements of punitive damages could be recognized in different cases of infringements of a person’s rights.
Juries
The “juries” variable relates to the procedural decision-making (dispute-resolution mechanism) by juries or merely by expert judges. We define a jury as a “panel of peers from the surrounding community called upon to decide a legal matter” (Posner, 2011). When the laws of a state authorize or require the jury to ascertain or determine certain facts, to make certain findings or to fix the amount of damages, the value of property or the value of the use, hire or rent of property and a jury trial is waived or the court or judge trying the case is authorized to find and determine the facts as well as the law, then the word “jury” or “juries” may include the court or judge trying the case. (Heffer, 2005)
Special rules on pretrial discovery
The special rules on pretrial discovery index depict whether such special rules exist in the assessed legal regimes (Spears, 1980). One should note that as Adams (1995) suggests “at least in theory such special rules on pre-trial discovery should favor claimants (consumers).” Namely, in civil cases in the United States, “evidence is primarily gathered by parties using the formal investigatory pre-trial discovery” process (Adams, 1995). The current U.S. Federal Rules were originally adopted in 1938 and provide extremely broad discovery provisions, requiring parties to disclose “any non-privileged matter that is relevant to any party’s claim or defense (Federal Rules of Civil Procedure, rule 26(b) (I) “Discovery Scope and Limits”). A party who strongly believes it has been wronged can “file a claim even if it has hardly any evidence, and use the evidence gathered during pre-trial discovery to build up their case” (Adams, 1995). In the United States, disclosure is compulsory and has an extremely broad scope (De Lummen, 2013). Two key characteristics of pretrial discovery are that “(a) it takes place, for the most part, extra-judicially with minimal court intervention and (b) a party can actually force the other to provide them evidence, whether or not it is of genuine interest or relevance to the case” (Adams, 1995). However, one should note that “in general such a pre-trial disclosure or discovery rule does not exist in civil law countries” (Adams, 1995).
Existence of contingency fees
The existence of contingency fee variable denotes whether assessed legal regimes have in place the system of contingency fees. Contingency fee is a “fee for services (as of a lawyer) paid upon successful completion of the services and usually calculated as a percentage of the gain realized for the client” (MacKinnon, 2008). In a typical tort case in the United States, the “plaintiff’s attorney receives his compensation in form of a contingent fee” (MacKinnon, 2008). Under this payment scheme the attorney gets a “share of the judgment if his client wins and nothing if his client loses” (Posner, 2011). A common practice is to use a sliding scale: “the attorney gets one-third if the case is settled without trial, 40% if the plaintiff wins at trial, and 50% if a judgment for the plaintiff is affirmed on appeal” (MacKinnon, 2008). Contingent legal fees are widely used in the United States. In a well-known empirical study, Kritzer (1990) observes that “individual litigants tend to use contingent fees. In Europe contingent legal fees were strictly forbidden.” Moreover, it should be noted that this “existence of contingency fees” variable is employed as covariates in the model specification.
Looser pays all costs rule
The loser pays all costs variable relates to the existence of different renumeration systems in different procedures. Particularly, the existence or nonexistence of the so-called “loser-pays-all-costs” (Posner, 2011) rule in the assed legal regimes. In the field of law and economics, the so-called English is “a rule controlling assessment of lawyer’s fees arising out of litigation” (Posner, 2011). The looser-pays-all-costs rule provides that “the party who loses in court pays the other party’s legal costs” (Shavell, 2007). This rule contrasts with the American rule, under which “each party is generally responsible to pay its own attorneys’ fees (the same rule also prevails in civil law jurisdictions), unless a statue or contract provides for that assessment” (Posner, 2011). The rationale for the “looser-plays-all-costs rule is that a litigant is entitled to legal representation and, if successful, should not be left out of pocket by reason of his or her own legal fees” (Posner, 2011). It should be borne in mind that, in virtually all English civil litigation, damages are merely compensatory (Giliker, 2014). Moreover, it should be noted that this “loose-pays-all-cost” variable is employed as covariates in the model specification.
Empirical Methods
The aim of empirical strategy is to examine the contribution of pharmaceutical liability regimes to patenting activity and firm-level innovation consistently. The basic Poisson fixed-effects relationship between the number of patents applied for by a firm at EPO and liability regime that takes places is:

where
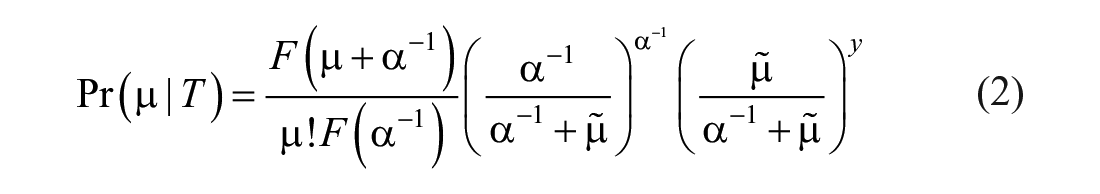
where

Specifically, our interest lies not in the estimated coefficients from Equation 3 but in the marginal effects of the change in liability regime on the number of validated patents. The marginal effects clearly indicate not only whether transaction costs might induce firm-level innovation but also how much a hypothetical change in liability costs would affect the patenting activity. Taking the first-order derivative of Equation 3 through liability, cost covariate leads to:

which denotes the partial marginal effect of the change in liability from the initial level
We establish the probabilistic model of patent validation to determine how much a change in the liability regimes shapes the probability that the patent application is approved or rejected. Our outcome of interest is a simply binary variable indicating whether the patent application of ith firm from jth country at time t has been approved or rejected which self-suggests the following outcome distribution scheme:

Using the potential outcome distribution from Equation 5, we examine the effects of liability costs on the patent validation decision using a simple unconstrained logit model with fixed effects to control for unobserved heterogeneity:

where
Results—Product Liability, Related Litigation, and Propensity to Patent
Baseline Results
In Table 2, the marginal effects of the change in potential liability costs on the firm-level innovation (stock of patents) are presented using the number of valid EPO patents as the outcome variable. Each model specification is estimated using the negative binomial and Poisson model to check for the potential discrepancy arising from the variance overdispersion relative to the mean valid patent count. Columns (1) and (2) report the coefficients on the special information disclosure duties for all and for the civil law countries (existence of special information disclosure duties—for example, the German disclosure duty). The evidence clearly suggests that procedural information disclosure duties do not exert a systematic and statistically discernible effect on the firm-level patent stock (level of innovation). The estimated responses of the valid patents to the change in procedural information disclosure duties are not statistically significantly different from zero even at artificially high significance levels even after addressing the overdispersion in the valid patent count.
Effects of Pharmaceutical Liability Regimes on Firm-Level Patent Stock.

Note. The table presents the marginal effects of pharmaceutical liability regimes on the firm-level patent stocks. Standard errors are adjusted for arbitrary heteroskedastic distribution of random error variance and serially correlated stochastic disturbances using Huber–White sandwich estimator. The marginal effects are obtained using the delta method expansion of the random variable function around the mean estimate with one-step Taylor series approximation with Oehlert (1992) valued functions of random vectors. Asterisks denote statistically significant marginal effects at 15% (a), 10% (*), 5% (**), and 1% (***), respectively.
In Columns (3) and (4), the effects of the existence of broad procedural statutory excuses (i.e., nonexistence of ex ante technological and scientific knowledge, or a federal pre-emption defense in the United States) are reported. The evidence suggests a highly positive effect on the firm-level of patent stock (firm-level innovation) across all jurisdictions. The point estimate from Column (4) implies that a hypothetical decrease of such costs by 1% would increase by the firm-level valid number of patents by 0.6% holding everything else fixed. Columns (5) and (6) report the effects of the liability costs generated by the damage caps on the firm-level of patent stock (firm-level innovation). The evidence clearly suggests that procedural damage caps do not exert a systematic and statistically discernible effect on the firm-level patent stock (level of innovation).
In Columns (7) and (8), the marginal effects of reversed burden of proof (causality—burden of proof depicts the notion of whether the claimant needs to prove a causal link between defective drug and the damage) are displayed. Controlling for the potential over-dispersion in patent variance across firms, the evidence suggests that existence of such a reversed burden of proof tends to encourage firm-level patent stock (firm-level innovation measured by the stock of patents). In a hypothetical scenario, expanding the reversed burden of proof by 1% would increase the firm-level valid patent count by 0.7% at 5% significance level, respectively. The established effect does not depend on the model selection and is sensitivity to the over-dispersion. In Columns (9) and (10), the marginal effects of the procedural requirement that court cases should be heard by jury are displayed. The evidence clearly suggests that procedural jury requirement does not exert a systematic and statistically discernible effect on the firm-level patent stock (level of innovation). The estimated responses of the valid patents to the change in procedural requirement of having juries are not statistically significantly different from zero even at artificially high significance levels even after addressing the overdispersion in the valid patent count.
The effects of special procedural rules on pretrial discovery (which at least in theory should favor claimants) are displayed in Columns (11) and (12). The existence of such special procedural rules on pretrial discovery that favor claimants has a significant negative effect on the firm-level patent stock (firm-level innovation) and may be associated with significant decrease in the number of valid patents. Having the possibility of class action suits is not associated with a marked decrease in firm-level innovation given the evidence from Columns (13) and (14).
In Columns (15) and (16), the effects of punitive damages associated with firm-level patent stock are examined. The evidence clearly suggests that the existence of such punitive damages has a significant negative effect on the firm-level patent stock (firm-level innovation) and may be associated with significant decrease in the number of valid patents.
In Table 3, the marginal effects of the change in pharmaceutical liability regimes on the patent validation rate are presented using the number of validated EPO patents as the outcome variable. Again, each model specification is estimated using the negative binomial and Poisson model to check for the potential discrepancy arising from the variance overdispersion relative to the mean valid patent count. Columns (1) and (2) report the coefficients on the special information disclosure duties for all and for the civil law countries (existence of special information disclosure duties—for example, the German disclosure duty). The evidence clearly suggests that procedural information disclosure duties do not exert a systematic and statistically discernible effect on the firm-level patent validation rate. The estimated responses of the patent validation rate to the change in procedural information disclosure duties are not statistically significantly different from zero even at artificially high significance levels even after addressing the overdispersion in the valid patent count.
Effects of Pharmaceutical Liability Regimes on Patent Validation Rate.

Note. The table presents the marginal effects of pharmaceutical liability regimes on the firm-level patent validation rate. Standard errors are adjusted for arbitrary heteroskedastic distribution of random error variance and serially correlated stochastic disturbances using Cameron et al. (2011) non-nested multiway clustering scheme for finite-sample empirical distribution function. The marginal effects are obtained using the delta method expansion of the random variable function around the mean estimate with one-step Taylor series approximation with Oehlert (1992) and Rice (1994) valued functions of random vectors. Asterisks denote statistically significant marginal effects at 15% (a), 10% (*), 5% (**), and 1% (***), respectively.
In Columns (3) and (4), the effects of the existence of broad procedural statutory excuses (i.e., nonexistence of ex ante technological and scientific knowledge, or a federal pre-emption defense in the United States) are reported. The evidence clearly suggests that broad procedural statutory excuses do not exert a systematic and statistically discernible effect on the firm-level patent validation rate. The estimated responses of the patent validation rate to the change in broad statutory excuses are not statistically significantly different from zero even at artificially high significance levels even after addressing the overdispersion in the valid patent count. Columns (5) and (6) report the effects of the liability costs generated by the damage caps on the firm-level patent validation rate. The evidence clearly suggests that procedural damage caps do not exert a systematic and statistically discernible effect on the firm-level patent validation rate.
In Columns (7) and (8), the marginal effects of reversed burden of proof (causality—burden of proof depicts the notion of whether the claimant needs to prove a causal link between defective drug and the damage) are displayed. Controlling for the potential over-dispersion in patent variance across firms, the evidence suggests that the existence of such a reversed burden of proof tends to have no significant effect on the firm-level validation rate. In Columns (9) and (10), the marginal effects of the procedural requirement that court cases should be heard by jury are displayed. The evidence clearly suggests that procedural jury requirement does not exert any systematic and statistically discernible effect on the firm-level validation rate in the civil law countries. However, it has a slightly positive effect in common law countries.
The effects of special procedural rules on pre-trial discovery (which at least in theory should favor claimants) are displayed in Columns (11) and (12). The existence of such special procedural rules on pretrial discovery that favor claimants has a slightly positive effect on the firm-level validation rate in the civil law jurisdictions and may be associated with increase in procedural efficiency of firms filling in the patent applications. Having the possibility of class action suits is actually associated with a slight increase in firm-level validation rate in the civil law jurisdictions given the evidence from Columns (13) and (14).
In Columns (15) and (16), the effects of punitive damages associated with firm-level patent validation rate are examined. The evidence clearly suggests that the existence of such punitive damages has in civil law jurisdictions a significant positive effect on the firm-level patent validation rate and may be associated with significant increase in the number of validated patents (effectiveness of patent applications). This could be associated with the screening effect of such legal instrument on the patenting activity (preparations of a file, procedural effectiveness of firms, novelty, etc.) of pharmaceutical firms.
Effect by Firm Size
One of the most obvious caveats against the estimated impacts of pharmaceutical liability regimes on firm-level innovation concerns the presence of potential effect heterogeneities. One of the key sources of such heterogeneities is firm size. In particular, both the capacity and ability of firms to innovate and acquire a valid patent may respond differently to the policy-related liability changes across firm sizes. In this respect, large firms may respond differently to the liability policy thresholds than small- and medium-sized firms. The neglect of such caveats may pose a constraint on the understanding of both channels and policy implications behind the estimated impacts.
To address these concerns, we reestimate both patent stock and validation rate specifications for samples of large and nonlarge firms separately. Estimating the effects of pharmaceutical liability regimes on these two samples may inform us whether large, and small- and medium-sized firms react differently to these types of liability regimes. We reestimate the saturated specifications for firm-level patent stock and validation model across the samples of large and nonlarge firms (we consider the threshold of 250 employees and above as the criteria to define a large firm) with the full set of legal policy covariates, country-level control variables and time-fixed effects, and cluster the standard errors at the firm level. Our focus is the comparison of the interaction effects between the pharmaceutical industry indicator variable and the liability regime policy variable to parse out the potential heterogeneity behind the impact of liability regimes on firm-level innovation.
Figure 1 reports the marginal interaction effects of pharmaceutical liability regimes on patent stock for large and nonlarge firms. The marginal effects of interaction terms are obtained employing the delta method expansion of the random variable function around the mean estimate with one-step Taylor series approximation with Oehlert (1992) and Rice (1994) random vector-valued functions. The evidence indicates both marked similarities and dissimilarities in the impact of liability regimes on the firm-level patent stock. More specifically, we find that large firms do not react different to the presence of punitive damages given the insignificant difference in the coefficient between both size thresholds. By contrast, the presence of jury may disproportionately reduce the expected patent stock of large firms while having no discernible impact on small firms. Another source of impact heterogeneity emanates from information disclosure requirements. Our finding highlights a considerably negative impact of such requirement on firm-level patent stock among large firms opposed to the significant, positive, and substantial impact among small- and medium-sized firms. On balance, this may reveal a potential intricacy where information disclosure requirements foster firm-level innovation among small- and medium-sized firms while having potentially negative effects on the pursuit of innovation among large firms. In contrast, we find no perceptible differences in the impact of class action suits given that both large and small- and medium-sized firms are about equally affected by class-action suits. In stark contrast to information disclosure requirements, patent stock of large firms is positively affected by damage caps opposed to zero average effect of caps on small- and medium-size firms. For pretrial discovery, similarly beneficial impact is found for large firms opposed to almost zero impact for small- and medium-sized firms. Overall, the evidence emphasizes substantial heterogeneity in the impact of pharmaceutical liability regime that arises from the firm size.

Effects of pharmaceutical liability regimes on patent stock by firm size.
Figure 2 presents the effects of pharmaceutical liability regimes on the patent validation rate by firm size. Note that the marginal effects are obtained using a simple delta transformation method for the probit estimator. The evidence indicates marked similarities and differences in the impact magnitude that arises from firm size. In particular, we find that the presence of the jury has relatively strong and negative effects on the patent validation rate of larger firms in contrast to zero expected effect on small firms. Concerning broad excuses and class action suits, we find no discernible differences in the magnitude of the effect between large and nonlarge firms. Testing the differences between both coefficients on marginal interaction effects using Chow test confirms no structural differences in the impact magnitude with high p-value. The effect of information disclosure requirements and punitive damages on the validation rate is both positive and comparable between both samples.

Effects of pharmaceutical liability regimes on patent validation rate by firm size.
By contrast, pretrial discoveries may have reasonably large and positive effects on the validation rate large firms in comparison with a weak negative effect on small- and medium-sized firms which appears to be statistically indifferent from zero. Leveraging the impact heterogeneities against the baseline, we find that firm size plays an important role in mediating the effect of pharmaceutical liability regimes on firm-level propensity to patent and validation rate. While large firms may be reasonably strongly affected by pretrial discovery, damage caps and broad excuses, the presence of information disclosure requirements has disproportionately beneficial for small- and medium-sized firms. This implies that considering the firm size, some dimensions of pharmaceutical liability laws may be especially pertained to large firms while others may importantly shape the innovation capacity of small- and medium-sized firms.
Conclusion
From the law and economy perspective, the relationship between liability, risk, uncertainty, and investments might be a key to understanding economic growth and prosperity. Our investigation suggests that current product liability regimes and related litigation laws across firms and countries do not account for the failure of pharmaceutical firms to acquire a valid patent. The results actually reveal that stricter litigation and liability laws across firms, between and within countries, have a positive effect on the firm-level validation rate. Thus, product liability and related litigation laws might be perceived as a filter that screens hazardous innovation in the pharmaceutical field and provides incentive for an efficient, productive, and safe innovations and simultaneously also deter opportunistic litigants. However, obtained results also suggest that moderate liability costs (induced by strict and prudent EPO’s validation procedures) as such costs tend to address moral hazard, opportunistic behavior, and other sources of asymmetric information and deter potential copy-cat imitator firms from entry (from obtaining validated patent). The results confirm the fundamental importance of reasonable liability–litigation–cost regulatory policy and institutional environment in explaining the differential propensities to patent across firms and countries in pharmaceutical industry.
Moreover, this article also shows that large firms and small firms indeed do react differently on the assessed types of laws. Namely, investigation suggests that considering the firm size, some dimensions of pharmaceutical liability laws may be especially pertained to large firms while others may importantly shape the innovation capacity of small- and medium-sized firms. However, this article does not aim to present a conclusive argument that will end the policymaker’s debates on the proper tort law regimes, potential trade-offs, and related different welfare effects on liability and innovation. Rather, it aims to contribute to the existing pool of, evidence-based, research on liability risk and innovation.
Footnotes
Appendix
Pharmaceutical Liability Legal Regime’s Characteristics: 0 (No) in 1 (Yes).

| Country | Strict liability | Class actions | Special information disclosure duty | Broad excuses—no ex ante technological and scientific knowledge | Damage cap | Causality burden of proof on injured person | Punitive damages | Jury | Pretrial discovery | Protection | Contingency fee | Looser pays all costs |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| United States (1972) | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 0 |
| United Kingdom (1987) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 |
| India (1986) | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 1 |
| Ecuador (2008) | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 |
| China (2009) | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 1 |
| South Korea (2000) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Japan (1995) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| Israel 2006 (1980) | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 1 |
| Australia (1995) | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 1 |
| Taiwan (1994) | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 |
| Turkey (2014) | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Canada (2002) | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Brazil (2012) | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 1 |
| Argentina (1998) | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 1 | 0 |
| Mexico (1992) | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Chile (2014) | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Russia (1992) | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 |
| Ukraine (2011) | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Kazakhstan (2007) | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 |
| Indonesia (1999) | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 |
| Philippines (1992) | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 1 | 0 |
| New Zealand (1986) | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| South Africa (2006) | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Albania (1994) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Austria (1993) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Belgium (1991) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| Bulgaria (2007) | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Croatia (2013) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Czech Republic (2012) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Cyprus (1999) | 1 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 1 |
| Denmark (1989) | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Estonia (2004) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Finland (1993) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| France (1998) | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Germany (1990) | 1 | 0 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Greece (1991) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Hungary (1993) | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Ireland (1991) | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 1 |
| Italy (1988) | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Lithuania (2000) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Latvia (2000) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Luxemburg (1988) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Malta (2000) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 1 |
| Netherlands (1990) | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Norway (1993) | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Poland (2004) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Portugal (1989) | 1 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Romania (2007) | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 |
| Spain (2007) | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 1 |
| Slovenia (1998) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Slovakia (2004) | 1 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 |
| Switzerland (1994) | 1 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
| Sweden (1992) | 1 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 0 | 1 |
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received no financial support for the research and/or authorship of this article from Javna Agencija za raziskovalno dejavnost Republike Slovenije – ARRS (project: Challenges of inclusive sustainable development in the predominant paradigm of economic and business sciences; project number: P5-0128).
