Abstract
Large and small safety accidents caused by experiments frequently occur in laboratories, and preventive measures are very important to prevent human, physical, and research and development (R&D) losses. It is very important to maximize R&D performance and to produce excellent R&D results through the efficient and optimal operation of laboratories, which can be done through R&D laboratory safety management activities. Research managers worldwide are obliged to engage in research activities with equal emphasis on good R&D performance and R&D laboratory safety management activities. In this article, the strategic R&D laboratory safety management status in South Korea was studied. Toward this end, an analytic hierarchy process analysis was conducted, and the degrees of importance and priority levels of the established factors affecting laboratory safety management, and their subfactors were analyzed based on a 5-point scale. Finally, a strategic management system for R&D laboratory safety was proposed.
Introduction
With the continued advances in science and technology, new infrastructure and technologies are being developed, and research is continually being carried out for the implementation of such technologies (Alaimo et al., 2010). In laboratories, various experiments are performed as part of studies that seek to discover important related phenomena and principles, to synthesize new materials, and to implement new technologies (Benedict, 2002). Laboratory research or experimentation uses various research equipment and various harmful substances (Lippman & Moon, 2004). The process of handling materials can cause safety and health problems like fires, explosions, poisoning, and environmental pollution due to the chemical, biological, mechanical, electrical, and radiological hazards involved, and new materials and experiment facilities may pose unknown hazards (Hurley et al., 2005). The workers in laboratories with insufficient safety management systems or that lack safety devices may be exposed to danger while conducting various experiments (Berbano, 2012; Gilling-Smith et al., 2005; Lupotto & Hognon, 2011). Despite these risk factors, laboratory researchers may not be aware of the importance of safety management activities (Giacomelli et al., 2005; Jin et al., 2012). As such, in South Korea, the government enacted a law on the creation of a safe environment for laboratories to provide legal grounds for managing accidents in laboratories. Such law has been in effect since April 1, 2006. In accordance with this act, the South Korean government develops and distributes laboratory safety management models; provides various support modalities, including laboratory safety diagnosis; and conducts activities for laboratory safety management, including establishing safety management organizations and enacting safety management regulations. Nevertheless, safety accidents caused by a lack of safety awareness continue to occur, and the amount of national losses and the negative effects of the damage are on the rise. These laboratory events and accidents result in lower safety management performance compared with the efforts made for laboratory safety management and the direct and indirect costs incurred by such. The fact that despite the high level of interest in laboratory safety management, the related policy and investment costs are still low can be seen as overlooking because there is no visible social achievement.
The term “laboratory” is defined as a research facility with equipment, materials, and so on installed by a university or a research institute for science-and-technology-related research and development (R&D) activities. Each laboratory has research workers, who are faculty members, researchers, university students, or research assistants engaging in science-and-technology-related R&D activities in universities and research institutes. Laboratory operations include safety management, which refers to maintaining safe conditions in the laboratory, free from risks or worker injuries. One of the activities that form part of laboratory safety management is safety inspection, an activity by a safety expert involving the investigation of the risks inherent in a laboratory. Another is precision safety diagnosis, which is conducted by qualified individuals based on the standards set by the government, for the purpose of discovering potential risks and establishing safety improvement measures to prevent accidents in the laboratory.
Literature Review
A Total Quality Management plan was developed and implemented on a chemical engineering laboratory at the University of California, Berkeley. Feedback from the researchers in the lab indicated that the safety measures introduced in this report successfully improved laboratory safety (Zakzeski, 2009). Personal protective equipment (PPE) compliance was highly associated with researchers’ perception of the level of risk in their lab; PPE compliance generally declined with lower perceived risk. In contrast to industry and government labs, PPE compliance of researchers in academia was significantly positively influenced when their safety behavior was monitored (Schröder et al., 2016). Laboratory safety must be an integral part of today’s chemistry—a science based upon laboratory inquiry (Hill, 2007). Laboratory safety teams (LSTs) are collaborative groups that seek to improve the safety culture within a department or multiple departments. They usually consist of a partnership between departmental faculty, staff, and students and have the goal of providing concrete opportunities for all of these stakeholders to continuously improve safety practices within their department (Miller & Tyler, 2019). Recognition of chemical hazards, an essential component of laboratory safety, depends upon the availability of clear and accurate information about specific chemical hazards on labels and other sources, such as Material Safety Data Sheets (MSDSs; Robert, 2009). Laboratory safety must always be a top priority in today’s research laboratories (Cadwallader & Pawelko, 2019). A flipped-classroom model highlighting case studies of the laboratory safety course creates a concrete experience that actively engages course participants (Hill et al., 2019). According to the most recent data from the federal Chemical Safety and Hazard Investigation Board, between 2001 and 2011, more than 120 university laboratory accidents have caused injuries and millions of dollars in damages (Zhu et al., 2018). Lab safety is an important and critical component to the success of any medical laboratory. Professional biosafety practices are affected by biological, chemical, radiological, fire, and electrical hazards (Eden & Hanson, 2019). The updated approach to educate on the core of laboratory safety was well-received by the researchers. The new program tackles many of the challenges associated with auditing and managing safety at a large research university (Tumidajski, 2016). Although some regulatory frameworks for the occupational health and safety of nanotechnology workers have been developed, worker safety and health issues in these laboratory environments have received less attention than many other areas of nanotechnology regulation (Ahn et al., 2016). Globally Harmonized System of Classification and Labeling of Chemicals (GHS) that seeks to protect human health and the environment by standardizing labeling and hazard information for all chemicals sold or traded in worldwide commerce (Hill, 2010). The laboratory safety training can achieve the desired goal of increasing the safety knowledge and improving the safety attitudes of students (Cheung & Spickett, 2007). The risk perception, when framed with regard to the risk of not performing the safety procedures, is related to safety behavior, as is supervisor commitment to safety (Taylor & Snyder, 2017). The Occupational Safety and Health Administration implemented its Laboratory Standard in 1990—It is a performance standard designed to protect employees working in laboratories (Hill, 2016). Assessment is the means of determining the effectiveness of a laboratory’s quality management system. Standards, as well as other normative documents that provide guidelines, form the basis for assessment. They may be developed at international, national, or local level. Organizations that establish norms or standards, and that provide for accreditation or certification of laboratories, play a vital role in the assessment process (World Health Organization, 2020). Laboratory Safety Standards are in use in clinical and chemical labs, testing labs, and R&D labs in both industrial and educational facilities. Lab safety standards pertain to protective clothing and equipment such as lasers, as well as procedures and lab designs that promote safety (American National Standards Institute, 2020). ISO 15189 can be used by medical laboratories in developing their quality management systems and assessing their own competence. It can also be used for confirming or recognizing the competence of medical laboratories by laboratory customers, regulating authorities, and accreditation bodies (Clinical and Laboratory Standards Institute, 2020).
Safety Management for R&D Laboratories
Laboratory safety is a state in which there is no danger or fear of the occurrence of an accident in a laboratory due to measures that have been taken to prevent harm to the researchers (Southworth & Heffernan, 2012). A safe laboratory does not happen naturally; various factors must be considered and various efforts must be made to ensure it (Karapantsios et al., 2008). It necessitates laboratory safety management, which can be defined as preventing and minimizing damages in a laboratory resulting from injury, disease, or property damage from hazards through the creation of a physical environment that can reduce the risk to and loss of life and property due to laboratory accidents (Mortland & Mortland, 2003). The goal of laboratory safety management is to ensure that no accidents occur in the laboratory so as to protect the lives and well-being of the researchers as well as the laboratory assets, and ultimately to improve the productivity and minimize losses (Winch et al., 2012).
R&D Laboratory Accidents in South Korea
According to the accident analysis report of the Ministry of Science and ICT (MSIT), the number of R&D laboratory accidents in South Korea gradually increased from 166 in 2014 to 203 in 2015 and 204 in 2016. Of the 204 cases in 2016, 164 were university laboratory accidents, 18 were research institute laboratory accidents, and two were company laboratory accidents. The analysis results showed that the increase in the number of laboratory accidents was due to the lack of safety management policies by laboratories, such as continuous field guidance and inspection, and the insufficient law enforcement capabilities. It is difficult, however, to obtain accurate and timely accident information regarding laboratory accidents as the accident damage information and causes of accidents in laboratories are often recorded in an unclear way, which makes it hard to prevent the recurrence of such accidents. Also, according to a survey conducted by MSIT in South Korea, the number of separate regulations, guidelines, and procedures regarding the recording, maintenance, and management of laboratory safety accident data in the country is still low. The laboratory accident situation in the country, such as lack of safety awareness and lack of investment in safety facilities, can be seen in Table 1.
Status of Laboratory Accidents in South Korea (Unit: Number).

University laboratory accidents accounted for 86% of the total number of laboratory accidents in South Korea from 2012 to 2016, followed by company laboratory accidents (5.3%) and research institute laboratory accidents (8.7%). The reason for the high number of university laboratory accidents is that while the university and graduate students engaging in R&D activities may have high knowledge, their safety awareness tends to be lower than that of the researchers in the company or research institute laboratories, whose activities are directly and predominantly related to research. Universities deal with relatively fewer risk factors, process development activities using new facilities and equipment, and R&D activities with potential hazards, so it is difficult to predict the scope and size of risks by stereotyping them. The laboratory accident types by institutional nature based on the 2016 survey on safety management conducted by MSIT are shown in Table 2.
Types of Laboratory Accidents in South Korea by Institutional Nature (Unit: %).

The operation of R&D laboratories in South Korea is very dangerous as most of the devices are driven at high temperatures and pressure, and as there are insufficient safety management systems in place and a lack of safety education programs. Due to the insufficient budget support for laboratory safety management and the insufficient construction of accumulated data management systems, there is a high possibility of large-scale laboratory accidents due to the design and structural conditions of laboratories, which generally reflect a lack of safety awareness. Lack of safety awareness and various other factors can cause laboratory accidents, and the ensuing material damage cost is almost the same as the laboratory’s maintenance cost. The financial losses on account of laboratory accidents by institutional nature based on the 2016 survey on laboratory safety management conducted by MSIT are shown in Table 3.
Financial Losses on Account of Laboratory Accidents (Unit: Won).

Many safety experts in South Korea point to the lack of budget support for R&D laboratory safety environment infrastructure projects, but the laboratory cost is relatively overlooked. The 2015 laboratory safety maintenance costs based on the 2016 survey on laboratory safety management conducted by MSIT are shown in Table 4.
2015 Laboratory Safety Maintenance Costs (Unit: Won).

R&D Laboratory Safety Management in South Korea
In South Korea, a law on the creation of a safe laboratory environment was promulgated on March 31, 2005, due to the need to systemically ensure safety management for laboratories and research workers. The Laboratory Safety Management Act aims to efficiently manage the research resources and help revitalize R&D activities in South Korea by ensuring the safety of the laboratories in universities and research institutes and ensuring proper compensation for the damages caused by laboratory accidents. The institutions that are subject to the Laboratory Safety Management Act (especially Article 10, 18, and 25) and its enforcement decree and rules are shown in Table 5.
Institutions Subject to the Laboratory Safety Management Act.

The Laboratory Safety Management Act applies to universities and research institutes that have been established for R&D activities. The act does not apply in its entirety if less than 10 research workers are engaged in research activities in a laboratory set up by a university or research institute. The act guarantees the safety of students and researchers through the conduct of safety inspection, precision safety diagnosis, and insurance subscription by the research director to efficiently manage the research resources, such as the research laboratory managers and research workers. In South Korea, as the Law on the Creation of Safety Environments for Laboratories was promulgated in 2008, the Laboratory Safety Management Act stipulated necessary matters concerning the contents, methods, and procedures of the safety inspection and precision safety diagnosis of laboratories by the research manager. The guidelines for such safety inspection and precision safety diagnosis are shown in Table 6.
Guidelines for the Safety Inspection and Precision Safety Diagnosis of Laboratories in South Korea.

Research Design
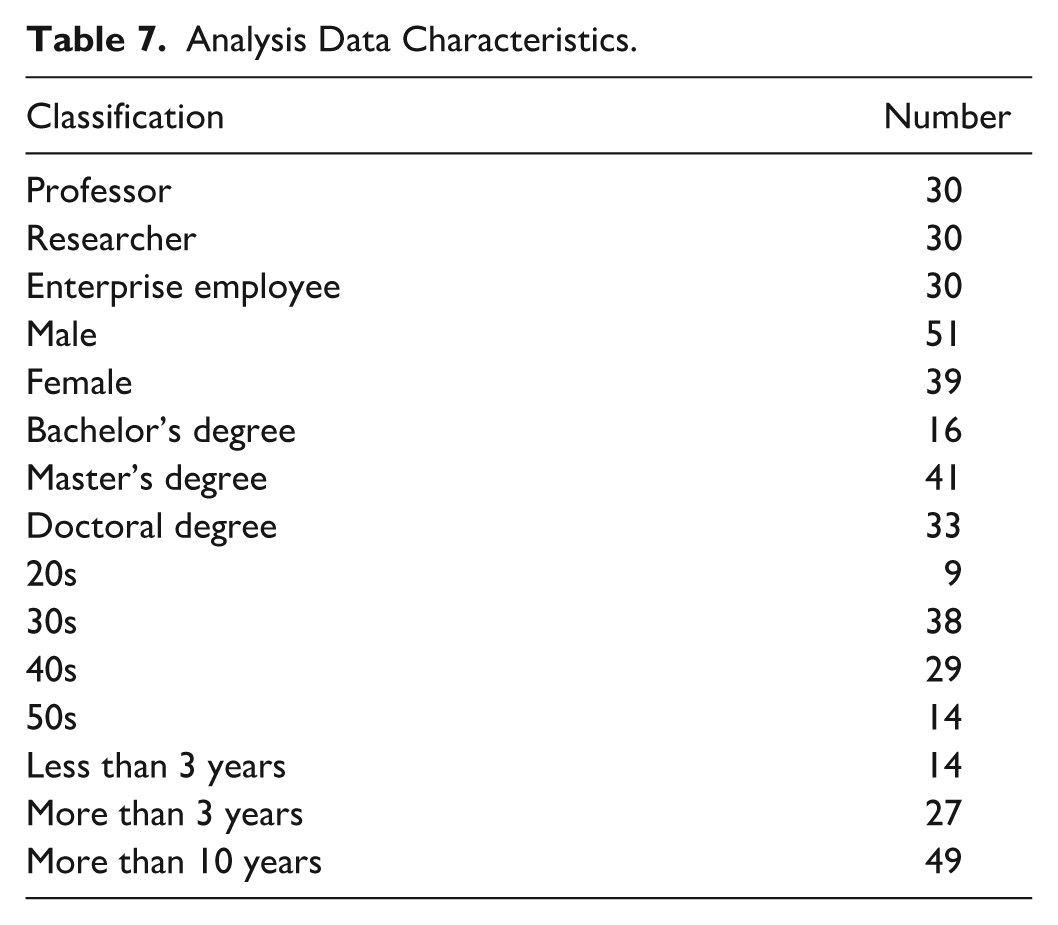
In this article, we used the 2018 data (30 professors, 30 researchers, and 30 company employees) of the MSIT (Information and Communications Technologies) of South Korea. Analytic hierarchy process (AHP) analysis was conducted, and the importance and priority levels of the established factors affecting laboratory safety management, and their subfactors, as perceived, based on a 5-point scale were analyzed. AHP analysis is a decision-making method that systematically evaluates the relative importance of various alternatives (Saaty & Vargas, 1991). In this study, we adopted the AHP analysis because it can make reasonable decision-making and systematic evaluation. The analysis data characteristics is shown in Table 7. The analysis data characteristics included professors, researchers, and company employees, in the same proportions. Most of the analysis data characteristics had master’s degrees, were in their 30s, and had worked in the laboratory for more than a decade.
Analysis Data Characteristics.
In this study, the legal system, program, and organization management factors were set as the factors influencing laboratory safety management. In the case of the legal system factor, mandatory regulation and sanction tightening were established as subfactors; for the program factor, safety education and training and accident prevention activities; and for the organization management factor, organization structure improvement and safety awareness strengthening. The AHP hierarchy for safety management is shown in Figure 1.
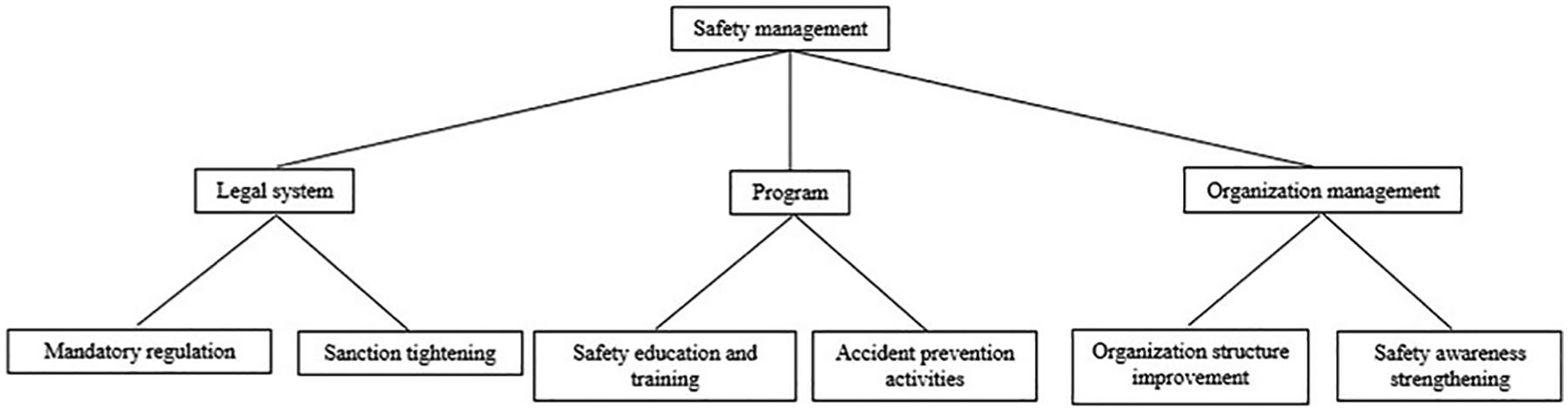
Analytic hierarchy process hierarchy for safety management.
Analysis and Results
The importance and priority levels of the legal system, program, and organization management factors affecting laboratory safety management as perceived are shown in Table 8. Perceived as the most important factor, which should be prioritized, was the legal system (0.391), followed by program (0.346) and organization management (0.310).
Importance and Priority Levels of the Factors Affecting Laboratory Safety Management as Perceived.

The importance and priority levels of the subfactors under the factors affecting laboratory safety management (legal system, program, and organization management) as perceived are shown in Table 9. For the legal system factor, mandatory regulation (0.447) was perceived to be more important than sanction tightening (0.391) and was thus considered the higher priority. In the case of the program factor, safety education and training (0.569) was perceived to be more important than accident prevention activities (0.380) and was thus considered the higher priority. Finally, for the organization management factor, safety awareness strengthening (0.618) was perceived to be more important than organization structure improvement (0.380) and was thus considered the higher priority.
Importance and Priority Levels by Factor Affecting Laboratory Safety Management as Perceived.

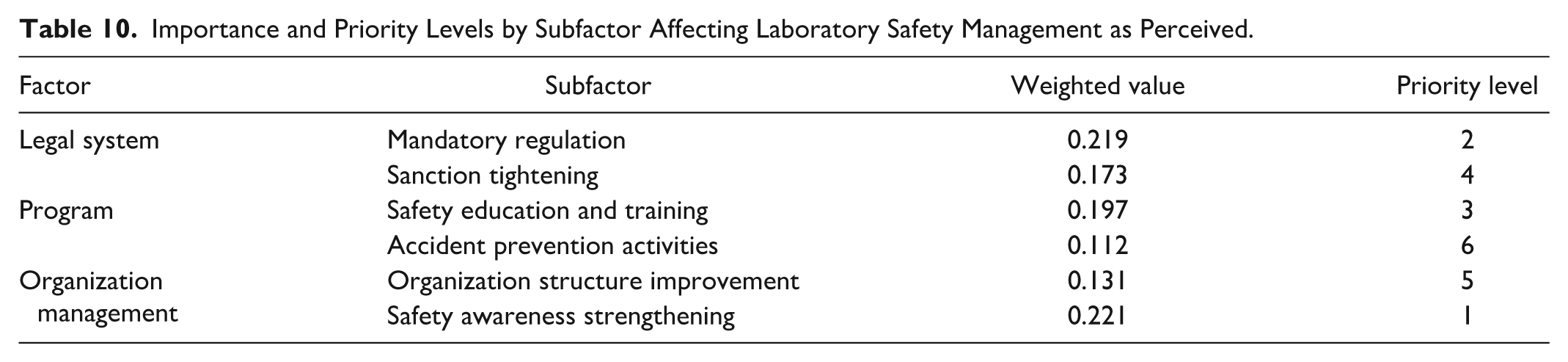
The total weighted values and priority levels of the subfactors under the factors affecting laboratory safety management are shown in Table 10. The analysis results showed that safety awareness strengthening (0.221) was perceived as the most important subfactor affecting laboratory safety management. Mandatory regulation (0.219) landed second place, but with only a slight difference from the first place. Next came safety education and training (0.197), sanction tightening (0.173), organization structure improvement (0.131), and accident prevention activities (0.112).
Importance and Priority Levels by Subfactor Affecting Laboratory Safety Management as Perceived.
The first-level analysis (factors affecting laboratory safety management) showed that professor, researcher, and enterprise employee considered the legal system the top priority because they believed that the establishment of laboratory safety management regulations by each institution as well as the definition of the responsibilities and duties of the research managers, research safety officers, and employees involved in research activities will affect laboratory safety management. In other words, professor, researcher, and enterprise employee believed that reinforcement is necessary in terms of legal systems. The second-level analysis (subfactors affecting laboratory safety management), on the contrary, indicated that professor, researcher, and enterprise employee saw mandatory regulation (0.447) as more important than sanction tightening (0.391) between the two legal system subfactors, and believed that the mandatory provision of safety management regulations should be prioritized, and that sanctions for non-compliance with such regulations should be put in place only if the regulations are not followed. In other words, the organization’s duties and responsibilities should be prioritized. For the program factor, professor, researcher, and enterprise employee perceived safety education and training (0.569) as more important than accident prevention activities (0.310). For instance, they believed that the installation of safety labels and of sufficient safety protection equipment and facilities requires strengthening laboratory safety education and training, such as the treatment of harmful reagents. With regard to the organization management factor, professor, researcher, and enterprise employee perceived safety awareness strengthening (0.618) as more important than organization structure improvement (0.380), which is consistent with such subfactors’ weighted values and priority levels in the second-level analysis. The second-level analysis (weighted values and priority levels of the subfactors affecting laboratory safety management) showed that professor, researcher, and enterprise employee perceived safety awareness strengthening (0.221) as the most important subfactor, followed closely by mandatory regulation (0.219).
Safety Management and Improvement Policy in the Laboratory
The establishment and operation of safety management regulations are very important for laboratory safety management. The safety management policy should include matters concerning the safety management system and tasks, the designation and authority of the safety management personnel by laboratory, the implementation of routine safety checks, the installation of safety signs, the implementation of emergency countermeasures, and the conduct of safety inspection and precision safety diagnosis in the event of an accident. The laboratory’s safety manager should keep all the relevant records on the inspection log after conducting routine inspections before beginning each test, and should establish a security system so that if an abnormality occurs, actual safety inspection can be carried out. In addition, safety education must be provided to research workers. To prevent safety accidents, laboratories handling hazardous materials should post safety signs and experiment safety rules in appropriate spaces. The laboratory equipment, jockeys, materials, and so on should always be organized. Most dangerous machines function as power transmission devices and are located close to the operator, so adequate safeguards are essential as there is a high risk of an accident taking place. The use methods and safe operation rules of all machine equipment should be posted in appropriate spaces, and all research workers should be aware of these before starting an experiment. The person in charge of safety management should conduct regular legal safety inspection of dangerous equipment and should see to it that all equipment are performing normally and optimally. Safety should be maintained when operating dangerous equipment, considering the safety precautions from the manufacturer. If high-pressure gas is used, all the facilities and equipment using it should be maintained to prevent gas leakage, and it should be ensured that the pressure gauges of various devices as well as all leakage alarms and gas shut-off devices are always operating normally. In the gas storage room, the name of each gas should be indicated, and danger signs and piping should be installed per gas and should be painted using a different color for each gas. Gas containers in the laboratory should not be left unattended, and those that have expired should not be used, because the corrosion of the valve can cause gas leakage, and because it is difficult to secure the corrosion stability of the container itself. If mobile high-pressure gas is used, it should be secured to a wall or an anchorage with a chain or belt to prevent it from conducting. Dispose of all containers that have no longer been in use for a long time, or install protective caps on them to keep them safe. Electrical power can lead to major accidents in the laboratory due to electric shock, so exercise caution in this regard. To prevent electric shock, use only equipment with three-prong plugs, and make sure that all multi-plug adapters have circuit breakers or fuses. Do not overload the electrical wires in the laboratory or use multiple power sources in one outlet, and equip all laboratory circuit breakers with a box. Also, do grounding work when installing an electric box to keep the ground resistance low. Waterproof socket-outlets and distribution boxes should be installed in humid places. In laboratories, switchboards should be made easily accessible by leaving no load nearby, and name stickers should be attached to the switchboard for efficient management. When introducing new equipment, check the load power capacity to prevent overload. All wires should be treated with conduits or molding to prevent the researchers from tipping over or damaging the sheath when walking. Appropriate fire extinguishers should be installed to enable initial suppression in the event of a fire in the laboratory. Laboratories handling expensive equipment can protect the equipment by installing gas meter fire extinguishers. One or more fire extinguishers should be installed in a location with a 33 m2 or more floor area in the laboratory, and all locations with a 20 m2 or more floor area should be equipped with a fire detector. In the event of a fire, shelter and emergency guidance lights play an important role in minimizing human damage, so make sure that they are always sufficiently charged and operating. The emergency exit should always be open, and it must be ensured that it can be opened by pushing it from the inside. All flammable substances in the laboratory should be stored in a safe place as they may cause a fire explosion when ignited. Fire safety should be fully considered and ensured in the design of the building, and fire damage should be minimized by using flame-resistant materials for the building interior. A MSDS should be properly accomplished or prepared in accordance with the law, and should be displayed in the place indicated in the applicable law, so that the researchers will know how to use hazardous materials correctly and will be able to use them accordingly. The applicable materials herein are physical, health, and environmental hazards.
Conclusions and Research Limitations
With regard to the importance levels, weighted values, and priority level results of the first-level analysis in this study as perceived, the priorities were legal system → program → organization management, in descending order, with the laboratory’s safety management infrastructure being the top priority. The weighted-value and priority level analysis showed that safety awareness strengthening under the organization management factor was perceived as the top priority, and that professor, researcher, and enterprise employee believed that the safety awareness of those involved in research activities needs to be improved. The results of the weighted-value and priority level analysis also showed that mandatory regulation under the legal system factor was perceived as the second priority, and that professor, researcher, and enterprise employee believed that it is necessary to formulate and organize safety management regulations for laboratories, and that through active promotion and training, the laboratory’s directors, research managers, and research workers should be fully informed of safety measures. Regarding the reinforcement of safety awareness, professor, researcher, and enterprise employee believed that it is necessary to stipulate the responsibilities, duties, and compliance by rank, and that a system for the safety management of the laboratories within an institution is required. Sanction tightening, which ranks fourth in terms of priority level, was perceived as an essential system for laboratory safety management. Sanction tightening education and training was found to be the third most important subfactor affecting laboratory safety management for professor, researcher, and enterprise employee, and they believed that laboratory safety education needs to be developed and provided by the government institutes. A laboratory safety rating system and a laboratory emergency training program are also believed to be required for laboratory safety management improvement. Under the program factor, accident prevention activities was ranked sixth in importance, and professor, researcher, and enterprise employee believed that the distribution of hazardous materials, safety protection equipment, and facilities needs to be supported and improved. Professor, researcher, and enterprise employee also believed that improving the organization structure requires the training of the safety environment managers and the fostering of experts. In addition, it was believed that the safety management committee should be well maintained by each institution.
The following improvement measures for laboratory safety management are thus suggested. First, more systematic laboratory safety management can be carried out if the regulations will be revised to allow each institute to appoint additional research workers in charge of research activities when the upper limit of research workers has been reached. Second, if the head of an institute is allowed to establish a drug center or build related facilities, the damage will be minimized in the event of an accident. This requires legislation, management, supervision, and support from the government institutes. Third, regular education and training of research workers is required. Development of education programs for research managers, safety management personnel in research laboratories, and research workers is required. Fourth, if the Laboratory Safety Management Act stipulates the responsibilities and duties of the director, laboratory safety and environment manager, laboratory manager, laboratory safety management officer, and research worker, the laboratory safety management regulations of all institutions can also be clarified, and laboratory safety management can be stabilized. Fifth, the laboratory safety management can also be stabilized by enacting a law on the conduct of precision safety diagnosis by research institutes, and by addressing the recommendations made during regular laboratory inspections with the aim of optimizing the safety facilities.
This study aimed to objectively determine the laboratory safety management status in South Korea based on the laboratory safety management status data of each institution, but there were limitations in data collection. That is, the institutions holding the relevant data were reluctant to release such data, claiming that these were confidential and raising concerns about the deterioration of their image as an institution if they released these. This made it difficult to analyze the causes of laboratory safety accidents in South Korea, and to conduct analysis to prevent the recurrence of such accidents. This situation must be overcome so that the aims of laboratory safety management research can be fully attained.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
