Abstract
Oxytocin is associated with social behavior. In humans, several studies have assessed the relationship between social behavior and a single nucleotide polymorphism (SNP)—rs53576—within the gene encoding the oxytocin receptor (OXTR). Fewer studies have assessed the relationship between human sociability and the SNP rs3796863 found within the cluster of differentiation 38 (CD38) gene, whose product has been found to facilitate the secretion of oxytocin. This research examines whether these polymorphisms are associated with facial age discrimination. Young adults (N = 43) were genotyped at both the OXTR and CD38 loci. They discriminated aging in 1- to 4-, 5- to 8-, 9- to 12-, 13- to 17-, and 18- to 22-year-old male and female faces. Participants with the OXTR GG and the CD38 CC genotypes discriminated facial aging better than those possessing at least one A allele at either locus. These polymorphisms appear to be associated with social information processing involved in facial age discrimination.
There is a long history of research on the heritability of social behavior (e.g., Loehlin, 1992; Riemann, Angleitner, & Strelau, 1997). Advances in molecular biology have enabled a much more informed study of the impact of specific genes on social behavior. Limited research, however, has attempted to link specific genes to psychological processes involved in social cognition, and none of this research has examined the role of specific genes in facial age discrimination. The current research examines the relationship between single nucleotide polymorphisms (SNPs) within two genes associated with social behavior and cognition, that is, rs53576 located in the oxytocin receptor gene (hereafter OXTR) and rs3796863 located in the cluster of differentiation 38 gene (hereafter CD38), and the ability of young adults to discriminate facial aging in sets of faces ranging from 1 to 22 years of age.
Oxytocin, OXTR, and CD38
Oxytocin is a neurohypophysial hormone and nonapeptide neurotransmitter produced in the hypothalamus. Oxytocinergic physiology is associated with a range of social behavior, affiliation, and bonding (e.g.,Bakermans-Kranenburg & van Ijzendoorn, 2008; Feldman, 2012; Meyer-Lindenberg, Domes, Kirsch, & Heinrichs, 2011; Parker et al., 2014).
The OXTR gene encodes a G protein-coupled receptor to which oxytocin binds (Gimpl & Fahrenholz, 2001). Specifically, the OXTR SNP rs53576, located within intron 3 of OXTR, is associated with sensitivity to social stimuli and social behavior. Young adults with the OXTR GG genotype tend to be more responsive to infant cries (Riem, Pieper, Out, Bakermans-Kranenburg, & van Ijzendoorn, 2010), exhibit more maternal sensitivity to toddlers (Bakermans-Kranenburg & van Ijzendoorn, 2008), and show greater interest in infant faces (Marsh et al., 2012) than those with the AA or AG genotypes. They are also better able to infer emotional states from the eyes (Rodrigues, Saslow, Garcia, John, & Keltner, 2009) and are more sociable (Tost et al., 2010).
Whereas the OXTR gene encodes the receptor to which oxytocin binds, the enzyme encoded by the CD38 gene—ADP-ribosyl cyclase—facilitates the secretion of oxytocin via the mobilization of intracellular Ca2+ stores (Jin et al., 2007; Lerer et al., 2010). Little research has been conducted on the social cognitive correlates of CD38 expression. Mice without functional copies of the CD38 gene show poor maternal nurturing and social amnesia (Higashida et al., 2011; Jin et al., 2007). Among individuals exhibiting autism spectrum behavior, the C allele of the human CD38 SNP rs3796863 has been found to be overrepresented statistically (Munsue et al., 2010). In addition, this allele has been associated with reduced CD38 messenger RNA (mRNA) expression in cultured lymphoblastoid cells derived from individuals with Autistic Spectrum Disorder and their unaffected parents (Lerer et al., 2010). Young adults who possess the rs3796863 CC genotype also tend to respond slower to social stimuli and are less inclined to touch infants than those with the AC or AA genotypes (Feldman et al., 2012; Sauer, Montag, Wörner, Kirsch, & Reuter, 2012).
Facial Aging and Age Perception
Distinct changes occur in faces between infancy and early adulthood that give them a more mature appearance (Berry & Zebrowitz-McArthur, 1988). With age, the face elongates and becomes less rounded. There is a decrease in the size of the forehead and eyes relative to the face and an increase in the relative size and angularity of the chin. The placement of the eyes moves above the face’s midline, the nose becomes more pronounced and elongated, and skin becomes coarser and more varied in color.
Ecological theorists (e.g., McArthur & Baron, 1983; Zebrowitz & Montepare, 2006, 2008) argue that our response to these facial features has a biological basis and is rooted in our evolutionary past. How others perceive and respond to these features is associated with a number of social consequences. Youthful faces are perceived as cuter and evoke more physical attraction than older faces (Alley, 1988; Gross, 1997; Karraker & Stern, 1990; Stephan & Langlois, 1984). Immature faces are also perceived as more helpless and dependent than mature faces (Berry & Zebrowitz-McArthur, 1988; Friedman & Zebrowitz, 1992; Gross, 1997). From an evolutionary and ecological perspective (e.g., Eibl-Eibesfeldt, 1989; Mark, Shaw, & Pittenger, 1988; McArthur & Baron, 1983; Zebrowitz & Montepare, 2006, 2008), it is presumed that our response to faces is adaptive and stems from social selective pressures encountered in the course of human evolution. We ensure the survival of our young by having a genome that has been fine-tuned to bring about perceptual skills allowing us to recognize immature faces as young and in need of succor, nurturance, and protection (Schaschl et al., 2015; Vargas-Pinilla et al., 2015).
Evidence of a biological basis for age perception and associating mental and physical states with facial age comes from several sources. Perception of immaturity in faces occurs very early in life. By 4 months of age, children are sensitive to the facial features associated with aging (Kramer, Zebrowitz, San Giovanni, & Sherak, 1995; McCall & Kennedy, 1980; Montepare, Zebrowitz-McArthur, 1989). The perception of youthfulness in young faces and its association with dependence is also found across cultures (e.g., Zebrowitz, Montepare, & Lee, 1993). Furthermore, particular groups of individuals are deficient in their ability to perceive facial aging. Gross (2002) noted that, compared with neuro-typical children, those with autism had difficulty discriminating aging in nonhuman faces.
Overview and Hypotheses
The accurate perception of facial age is important to social interaction and, perhaps, even survival. Little research, however, has examined factors associated with sensitivity to facial aging. Given genetic influence on social behavior (Knafo & Plomin, 2006), it is reasonable to assume that the physiology associated with specific genes might influence the more explicitly defined social cognitive processes associated with facial age perception. To date, no one has examined a genetic basis for sensitivity to facial aging and facial age discrimination.
In the current study, young adults participated in a task of social cognition that required the detection of aging in sets of faces. In addition to completing the age discrimination task, participants provided buccal cell samples that were subsequently genotyped at the OXTR rs53576 and CD38 rs3796863 loci. Both OXTR and CD38 loci have been associated with oxytocinergic signaling and social functioning. Based on past research, it was hypothesized that participants with the GG genotype of OXTR or the AA/AC genotype of CD38 would perform better on the age discrimination task than participants possessing the OXTR GA/AA or CD38 CC genotypes. Given the ecological importance of detecting aging in very young faces and a presumed biological disposition for doing so, it was predicted that individuals possessing the GG genotype of OXTR or the AA/AC genotype of CD38 would more accurately identify aging in younger rather than older faces compared with participants with alternative genotypes.
Method
Participants
Prior to beginning this study and based on similar studies (Marsh et al., 2012; Sauer et al., 2012), we anticipated the need to recruit between 40 and 45 participants. Our sample consisted of nine male (two Asian-, two Hispanic-, five Caucasian Americans; M = 20.7 years, SD = 1.82) and 34 female (one Asian-, two African-, seven Hispanic-, 24 Caucasian Americans; M = 20.8 years, SD = 1.41) young adults. The distribution of participant genotypes at the OXTR and CD38 loci is shown in Table 1. Based on the convention of prior studies (e.g., Marsh et al., 2012; Sauer et al., 2012), data from participants with the OXTR GA/AA and CD38 CA/AA genotypes were combined for analyses.
Distribution of Participant Genotypes: OXTR rs53576 X CD38 rs3796863.
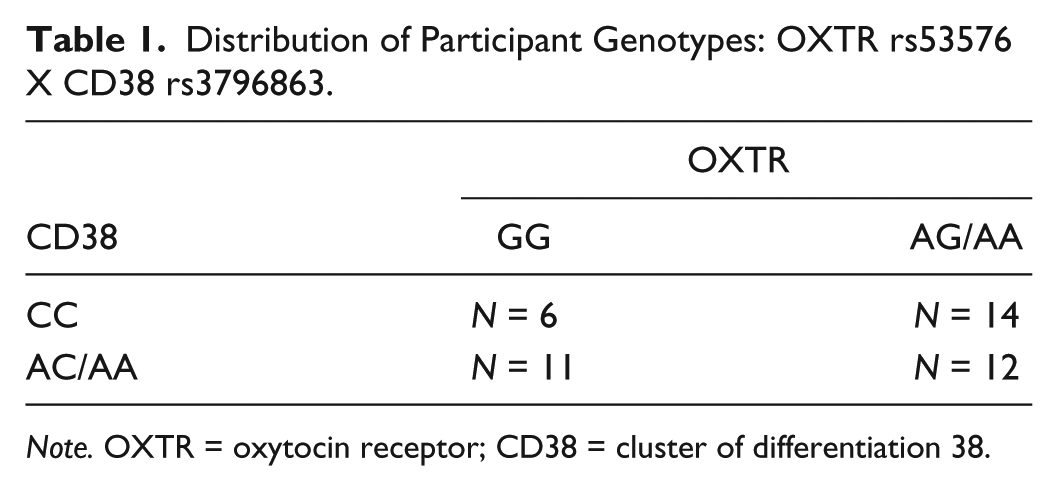
Note. OXTR = oxytocin receptor; CD38 = cluster of differentiation 38.
Neither OXTR nor CD38 allele frequencies deviated from Hardy-Weinberg equilibrium—chi-square goodness of fit: χ2(1)OXTR = 0.69, p = .41; χ2(1)CD38 = 0.03, p = .87. With respect to the two genotyped loci, alleles segregated independently within the sampled population—chi-square test of independence: χ2(1) = 0.72, p = .40. Participants were treated in accordance with ethical standards established by the American Psychological Association and received $10.00 for their participation.
Facial Age Perception Task
Participants were asked to respond to 10 facial age sets. Half of the face sets consisted of male and half of female faces. For both male and female faces, one set consisted of faces that were aged from 1 to 4, 5 to 8, 9 to 12, 13 to 17, and 18 to 22 years.
To construct each set, full face, color photographs were obtained of the same four individuals as they appeared at a younger (e.g., 5 years old) and older (e.g., 8 years old) age. Faces had no adornment, a pleasant expression, and closed mouths. Photographs of the four younger and four older faces were morphed into composite prototypical images of the younger and older face. These younger and older prototypical faces were then morphed across 28 steps to create the 10 sets of 29 faces that, within each set, gradually changed from the youngest to the oldest face. Photographs were framed in oval masks to eliminate hair and clothing (Figure 1). This procedure for studying facial aging was adapted from previous methodology used to assess discrimination of social properties of the face (e.g., ethnicity, gender; for example, Corneille, Huart, Becquart, & Brédart, 2004; Ng, Ciaramitaro, Anstis, Boynton, & Fine, 2006). 1

An example of a facial array with a 1-year-old male face morphed (beginning in the upper left-hand corner and proceeding to the lower right hand corner) over 28 steps into a 4-year-old male face.
Participants were assessed individually. Using a computer program developed in PsyScope (Cohen, MacWhinney, Flatt, & Provost, 1993), participants were shown, one set at a time, 10 sets of 29 faces. Stimuli were presented on a computer console. At the start of each trial, faces were randomly placed into an array of 29, 4.5 × 5 cm windows. Participants were directed to examine the faces in the array and to mouse-click on the face that appeared the youngest. After clicking on a face, the face was removed from the array and the participant was instructed to, again, select the youngest appearing face from the remaining 28. Participants continued in this fashion until all 29 faces in the set had been selected. The computer program recorded the order in which faces were selected and the amount of time that it took participants to make their selections. Upon completing one set, another set of faces was presented on screen. Male and female face sets alternated between trials. The age of the face sets varied randomly across presentations. Each participant responded to each of the 10 face sets.
Genotyping
Immediately preceding or following the administration of the facial age discrimination task, participants rinsed their mouths thoroughly with 40 mL of bottled water. They then vigorously swished 10 mL of a .9% (m/v) saline solution about the mouth for 30 s before depositing it into a conical tube that was placed immediately on ice.
DNA was isolated from 200 µL of resuspended buccal cells obtained following an initial pelleting of 1 mL of the 10 mL saline rinse obtained from participants. DNA isolation was conducted using a QIAamp DNA Blood Mini kit (Qiagen, Valencia, CA) within 2 hr of sample collection for all participants. Genotypic calls were generated using TaqMan SNP Genotyping Assays (Applied Biosystems, Foster City, CA) specific to each locus, either OXTR (C__3290335) or CD38 (C__1216944_10). Genotypic assignments for each participant were verified in duplicate at both loci. Reactions were conducted using a Type-it Fast Probe PCR Kit (Qiagen) in a Bio-Rad C1000 thermal cycler equipped with a CFX96 optical reaction module (Bio-Rad, Hercules, CA). Individual PCR reactions contained 1x Type-it Fast SNP Probe PCR Master Mix, 1x TaqMan SNP Genotyping Assay, approximately 20 ng of participant genomic DNA, and .5x Q-Solution. Each reaction was conducted in a total volume of 25 µL. Thermal cycling conditions consisted of an initial step of 5 min at 95°C followed by 40 cycles of 15 s at 95°C, then 30 s at 60°C. Fluorometric readings for each reaction were acquired after every cycle. Genotypes were assigned using the CFX Manager software (Bio-Rad).
Quantification of Salivary 17β-Estradiol
Several previous studies examining individual differences within the OXTR and CD38 genes have attempted to control for inter-subject variability in female sex hormone levels (Marsh et al., 2012; Sauer et al., 2012). Because some research (e.g., Marečkova et al., 2014; Miyahara, Morita, Yamaguchi, Nonaka, & Maeda, 2000) has found that estradiol concentration in females may influence sensitivity to faces and it was anticipated that many participants in our sample would be female, salivary estradiol concentration (pg/mL) was assessed for each participant and used as a covariate in our analyses. Participants provided approximately 2 mL of saliva by way of passive drool immediately prior or subsequent to their completion of the facial age discrimination task. Saliva collection and sample storage were in accordance with methods detailed by Salimetrics. Samples were assayed at the Arizona State University Institute for Interdisciplinary Salivary Bioscience Research using an immunoassay (Salimetrics, State College, PA) specific to 17β-estradiol (inter-assay %CV = 0.9, intra-assay %CV = 2.4).
Results
Age Discrimination Accuracy
Age discrimination scores were Spearman rank order correlation coefficients reflecting the degree of correspondence between the actual ranking (youngest to oldest) and the participant’s rankings for each facial age set. Initial analyses of these data failed to find significant effects or interactions involving participant gender, so participant gender was excluded from further analyses.
A 2 × 2 × 2 × 5 ANOVA with two between-subjects factors (OXTR genotype: GG or AG/AA; and, CD38 genotype: CC or AC/AA) and two within-subject factors (face gender: male or female; and, face age: 1-4, 5-8, 9-12, 13-17, 18-22 years of age) was conducted on age discrimination scores. Including estradiol concentration as a covariate had no appreciable effect on outcomes. The results reported are for analyses without estradiol as a covariate. The F-statistics for the analyses with estradiol as a covariate are indicated in brackets. The least significant difference (LSD) test was used in post hoc analyses of mean differences.
Analyses revealed significant main effects for OXTR genotype, F(1, 39) = 5.37 [6.02], MSE = .22, p = .03,
Participants with the OXTR GG genotype demonstrated significantly better facial age discrimination (M = .73, SD = .16) than participants with the AA or AG genotype (M = .62, SD = .16). Participants with the CD38 CC genotype performed better (M = .74, SD = .14) on the face discrimination task than participants with either the AC or AA genotypes (M = .61, SD = .17). Male faces were discriminated better (M = .79, SD = .14) than female faces (M = .57, SD = .24). For female faces, the 1- to 4-year-old faces were discriminated better than the 5- to 8-year-old faces which were discriminated better than the older faces. The age of male faces did not significantly influence facial age discrimination (Figure 2).

Means and standard deviations (error bars) for Spearman’s rho as a function of face gender and face age.
Response Time
In addition to the accuracy with which participants identified aging in stimulus sets, the amount of time participants’ spent discriminating aging in each of the facial age sets was recorded by the PsyScope program with millisecond resolution. There was a significant relationship between participants’ overall age discrimination accuracy (the sum of the 10 age discrimination scores) and the overall amount of time participants took to make judgments about facial age, r(41) = .41, p = .006. Participants who performed better on the age discrimination task took more time discriminating aging in facial age sets.
As with the accuracy analysis, response time data expressed in minutes to set completion were subjected to 2 × 2 × 2 × 5 ANOVA with two between-group factors (OXTR genotype: GG or AG/AA; and CD38 genotype: CC or AC/AA) and two within-subject factors (face gender: male or female; and face age: 1-4, 5-8, 9-12, 13-17, 18-22 years). Again, F-statistics with estradiol as a covariate are included in brackets and LSD tests were used in post hoc analyses of mean differences. A significant main effect was obtained for face gender, F(1, 39) = 7.36 [7.2], MSE = .36, p = .01,
Marginal interactions were found between OXTR and CD38, F(1, 39) = 3.52 [3.2], MSE = 11.83, p = .06,
Mean Time in Minutes (SD) to Complete Face Age Sets as a Function of CD38 (CC and AC/AA) and OXTR (GG and AG/AA) Genotypes.
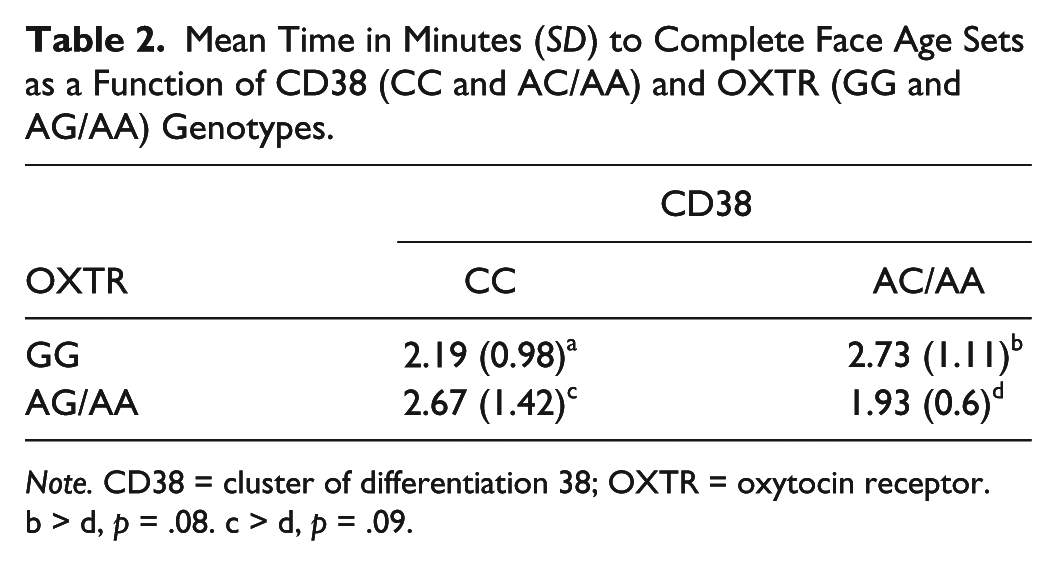
Note. CD38 = cluster of differentiation 38; OXTR = oxytocin receptor.
b > d, p = .08. c > d, p = .09.

Means and standard deviations (error bars) showing time in minutes to complete facial age sets as a function of face age and CD38 (CC and AC/AA) genotype.
Discussion
As anticipated, SNPs within the OXTR and CD38 genes were significantly associated with young adults’ performance on the facial age discrimination task. These relationships were found for both accuracy and response rate. The response rate and accuracy measures identified the same alleles (i.e., OXTR A, CD38 A) as conferring relative impairment on the facial age discrimination task. Although this assignment of allele risk is internally consistent, it is at odds with most literature regarding the CD38 locus (Feldman et al., 2012; Lerer et al., 2010; Sauer et al., 2012).
Participants found some facial age sets to be more difficult to discriminate than others. Notably, they were less accurate when discriminating aging in older female faces. Presumably due to increased task difficulty, participants took more time making judgments about female faces and 9- to 12-year-old faces (i.e., faces that, with 18- to 22-year-old faces, were least accurately discriminated) relative to 1- to 4- and 5- to 8-year-old faces. The increased challenge of discriminating female faces, and older ones in particular, may reflect the earlier maturation of female relative to male faces (e.g., Ferrario, Sforza, Poggio, & Schmitz, 1998; Mellion, Behrents, & Johnston, 2013). Older female faces may have appeared more similar with respect to age-differentiating features. In addition, mathematically averaged faces are perceived as more attractive than individual images used to generate them (Rhodes, Sumich, & Byatt, 1999; Valentine, Darling, & Donnelly, 2004). Because attractive female faces tend to retain youthful features (Cunningham, 1986), it is possible that the female face stimuli reflected an atypically low rate of aging that decreased discrimination accuracy.
Young children’s faces are perceived as helpless and dependent (Berry & Zebrowitz-McArthur, 1988; Gross, 1997). Assuming that sensitivity to youthful features is, in part, based in our biology and has adaptive social consequences, it was anticipated that individuals having genotypes favoring sociability would perform better when discriminating younger rather than older faces.
Although both OXTR GG and CD38 CC genotypes were significantly associated with better discrimination accuracy, contrary to what was anticipated, there was no Genotype × Facial Age interaction. This suggests that individuals with the OXTR GG and CD38 CC genotypes possess more generalizable (rather than age-specific) information-processing skills (e.g., scan rates, perceptual disembedding, memory) or specific social information-processing skills (e.g., increased sensitivity to facial features and/or configurations) (Richler, Cheung, & Gauthier, 2011) that sensitize them to facial aging across a wide range of ages rather than only to younger faces and those typically in greatest need of care.
An interaction between OXTR and CD38 on the rate of responding was observed in two trending interactions. Participants possessing both OXTR GA/AA and CD38 CA/AA genotypes (i.e., at least one risk allele at both loci) tended to complete the facial age discrimination task more rapidly than those possessing either genotype in isolation. In addition, a CD38 × Facial Age interaction was detected, in which CD38 CC participants spent more time discriminating 9- to 12-year-old faces (the more difficult facial age discrimination) compared with those with the CA/AA genotype at this locus, whose rate of responding did not significantly differ across any of the facial age groups.
These findings are in accordance with those noted by Sauer et al. (2012), who observed CD38 CA/AA participants responding more rapidly than those of CC genotype on an extended version of the Hariri Emotional Face Task (Hariri, Tessitore, Mattay, Fera, & Weinberger, 2002). As this paradigm involves a simple one target, two probe match to sample, it is understandable that the authors were unable to find significant effects for CD38 genotype on response accuracy, an outcome they themselves attributed to a strong ceiling effect. Notably, no ceiling effect was found in the present study, for which distinct main effects of the CD38 genotype upon facial recognition accuracy were observed.
In contrast to the Sauer et al. (2012) finding, the present research suggests a beneficial role for the CD38 CC genotype, potentially by way of its impact upon participants’ rate of responding. Although Sauer et al. suggest that longer response latency to social stimuli is indicative of relative impairment on a face recognition task, the present study found no such relationship. In fact, a significant, moderate correlation was noted between rate of responding and response accuracy. Those who took more time to make facial age judgments discriminated facial aging better.
Although it remains to be substantiated by further research, it is possible that those possessing risk alleles, as assigned within the context of this study, failed to recognize and compensate for the relative ambiguity of facial age in certain stimulus sets. This interpretation is particularly compelling when considering trends evident in participants’ age discrimination accuracy when these data are collapsed across stimulus gender and both genetic polymorphisms. Notably, a statistically significant decrease in facial age discrimination accuracy is present in responses to the 9- to 12-year-old condition relative to the 1- to 4-year-old and 5- to 8-year-old conditions. It is thus possible that individuals with the CD38 CC genotype were uniquely in a position to perceive and respond to this presumed change in difficulty (i.e., by slowing down) in a way that facilitated a consistent, more accurate pattern of age discrimination across all age levels.
The present study was not without limitations. We note our small sample size relative to sample sizes found in contemporary genetic association studies. Nonetheless, we believe this study highlights an interesting question and possible explanations regarding the relationship between genetic foundations and social cognition.
We are also unable to fully account for the apparent advantages afforded by the CD38 CC genotype within the context of our research task. Although social cognition is central to facial age perception, related research assessing the CD38 locus in relation to social functioning has consistently identified the C allele and particularly the CC genotype as conferring risk (e.g., Feldman et al., 2012; Lerer et al., 2010; Munsue et al., 2010).
Such “allele flips” (i.e., study-to-study variations in indicating which allele is associated with risk) are not uncommon across different association studies of oxytocinergic genes (Michalska et al., 2014; Parker et al., 2014) and are thought to arise due to genetic heterogeneity between the sampled populations (e.g., diverging haplotypes, differing allele frequencies) as well as discordant environmental factors between populations (Clarke & Cardon, 2010). It is unclear to what extent our sample may have differed from previous studies along these parameters and we cannot exclude their potential impact upon our assignment of allele risk.
It may then be premature to assign risk to the CD38 CC variant. Feldman et al. (2012), for example, noted that parents with the CD38 CC genotype had lower levels of plasma oxytocin and touched their infants less than parents with alternate genotypes. However, the relationship between plasma and cerebrospinal fluid oxytocin concentrations is unclear (Winslow, Noble, Lyons, Sterk, & Insel, 2003). As plasma oxytocin is excluded from the central nervous system by the blood-brain barrier, its relationship to CD38 variation, although demonstrated, may have inconsistent bearing upon human social behavior and cognition.
The findings demonstrated by Lerer et al. (2010) in autistic children and their parents, however, may suggest a somewhat counter intuitive explanation of CD38’s role in facial age perception. Individuals with autism exhibit strength in specific aspects of visual information processing, being adept at identifying fine detail in complex figures and separating figure from ground (de Jonge, Kemner, & van Engeland, 2006; Happé & Frith, 2006). As our paradigm assessed participants’ ability to make subtle discriminations within the context of a single stimulus set, it may have disproportionately favored individuals (i.e., those of CD38 CC genotype) possessing greater aptitude for these feature-focused, information-processing skills.
Conclusion
This research has suggested a relationship between two SNPs, OXTR rs53576 and CD38 rs3796863, associated with oxytocinergic signaling and facial age discrimination. For OXTR, allele assignments identified in this study are consistent with those made in previous studies linking this polymorphism to social behavior. For CD38, a gene less studied in relation to social cognition, the risk allele identified in this study is different from that identified previously. We have argued that, given the lack of research on the CD38 gene and its social behavioral/cognitive correlates, it may be premature to conclusively state which allele of the CD38 SNP examined here is protective of facial age discrimination ability. Nonetheless, it would appear that facial age discrimination is one of a growing number of human social behaviors and cognitive processes appreciated as being impacted by oxytocinergic physiology and genes.
Footnotes
Acknowledgements
We are grateful for the support of the staff at the University of Redlands Stauffer Center for Science and Mathematics, University of California at Riverside Institute for Integrative Genome Biology, and the Institute for Interdisciplinary Salivary Bioscience Research at Arizona State University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
