Abstract
Regulatory guidelines recommend systematic stability studies on a herbal product to establish its shelf life. In the present study, commercial extracts (Types I and II) and freshly prepared extract (Type III) of Centella asiatica were subjected to accelerated stability testing for 6 months. Control and stability samples were evaluated for organoleptics, pH, moisture, total phenolic content (TPC), asiatic acid, kaempherol, and high-performance thin layer chromatography fingerprints, and for antioxidant and acetylcholinesterase inhibitory activities. Markers and TPC and both the activities of each extract decreased in stability samples with respect to control. These losses were maximum in Type I extract and minimum in Type III extract. Higher stability of Type III extract than others might be attributed to the additional phytoconstituents and/or preservatives in it. Pearson correlation analysis of the results suggested that TPC, asiatic acid, and kaempferol can be taken as chemical markers to assess chemical and therapeutic shelf lives of herbal products containing Centella asiatica.
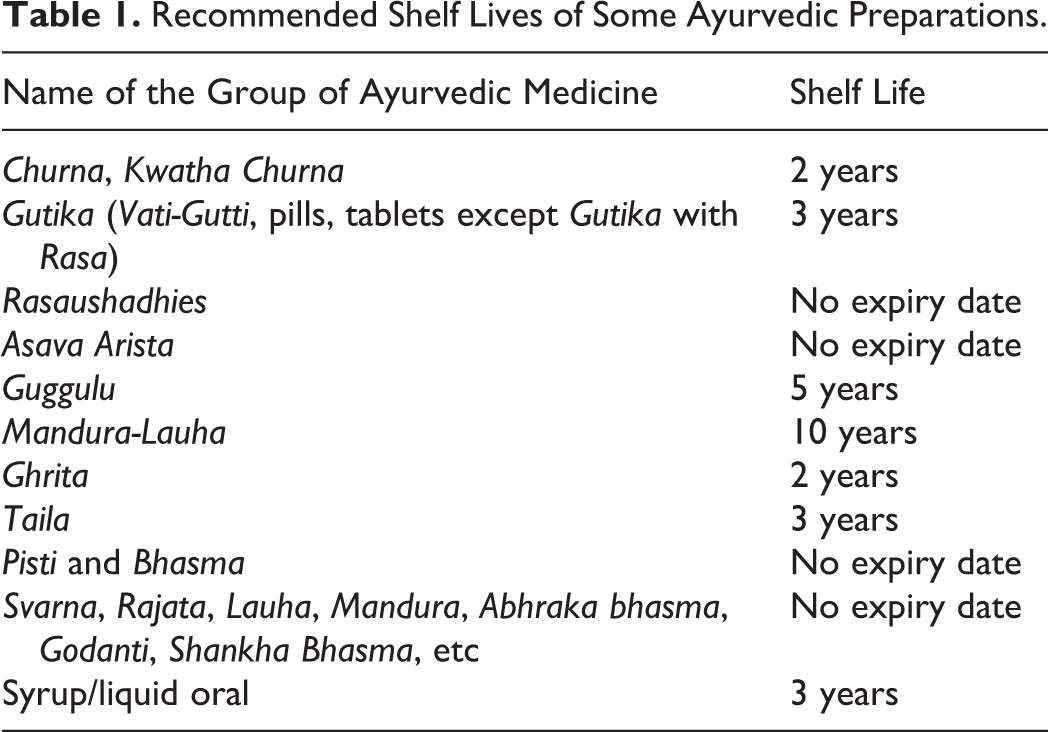
A large number of herbal products exerting varied therapeutic actions are available in the market worldwide. Despite the wide acceptability and availability of herbal drugs in medical health care systems in different countries, the attention toward their quality control is not at par with modern medicines. Assessments of chemical quality and therapeutic efficacies of herbal drugs/products are more challenging for establishing their shelf lives. Drugs regulatory agencies like the World Health Organization, European Medicines Agency, and the International Conference on Harmonization (ICH) have laid down various guidelines for establishing quality, safety, and efficacy of herbal products through stability studies. 1 –3 In India, herbal products bear shelf lives as recommended by the Department of AYUSH (Ayurveda, Yoga and Naturopathy, Unani, Siddha, and Homoeopathy), Ministry of Health and Family Welfare, Drug & Cosmetic Rules Amendment (Table 1). 4 However, there is no documented proof available that form the basis of these shelf lives mentioned in the gazette. It is undeniable that a herbal medicinal product is a very complex heterogeneous mixture, containing alkaloids, flavonoids, terpenoids, steroids, phenolics, and other constituents. Therefore, no 2 different herbal drugs/products can have the same chemical composition. From a scientific viewpoint, no 2 different herbal products in the same kind of formulation are necessarily expected to have the same shelf life owing to different kinds of phytoconstituents. Hence, the shelf life recommendation needs to be based on scientifically designed systematic stability studies of each herbal product in each formulation. Additionally, the biological activity of each stability sample is required to be evaluated to study the influence of storage conditions on product therapeutics. 5 Quality Council of India in its Annex A recommends that quality and stability of finished AYUSH products and, when necessary, of starting materials and intermediate products shall be evaluated. 6
Recommended Shelf Lives of Some Ayurvedic Preparations.
Centella asiatica, also known as Mandookaparni or Gotukola in Ayurveda, belonging to family Apiaceae, is a major constituent herb that is used among herbal products meant for acting on the central nervous system. This herb increases learning and memory by decreasing the levels of norepinephrine, dopamine, and 5-HT and their metabolites in brain. 7 The primary active constituents of C asiatica are triterpenoids that include asiaticosides, asiatic acid, madecassoside, and madecassic acid. Asiaticoside derivatives present in it prevent Alzheimer’s disease. 8 Ethanol extracts of its root, petioles, and leaves act as a potent antioxidant. 9 Different extracts of this herb have been formulated into commercial medicinal preparations. Madecassol, Centellase, Blastoestimulina, Gotu Kola capsules, Ayurvana, and Mayam are some of the commercial brands containing C asiatica extracts. 10
With this background, we aimed to apply the concept of comprehensive and systematic stability studies involving chemical, chromatographic, and pharmacological evaluations to assess the shelf life of a herbal drug. In this study, the candidate taken for study is C asiatica. So the present study is designed to (a) conduct systematic accelerated stability testing on freshly prepared and commercially available dried extracts of C asiatica at a temperature of 40°C and relative humidity of 75% for 6 months, as mentioned in ICH Q1A(R2) guidelines for stability testing, (b) evaluate stability samples withdrawn at periodic intervals for physical and chemical stability by chromatographic methods (high-performance liquid chromatography [HPLC] and high-performance thin layer chromatography [HPTLC]) with respect to control samples, and (c) evaluate control and stability samples for in vitro antioxidant and acetylcholinesterase (AChE) inhibitory activities to ensure therapeutic evaluation. The purpose is to ascertain the time period/shelf life during which the extracts of the selected herb will exert required therapeutic effects.
Experimental
Materials
Whole dried plant of C asiatica was purchased from Himalaya Herbs Store (Saharanpur, India). The plant was authenticated (Ref. No. NISCAIR/RHMD/Consult/2014/2469-48) from CSIR-NISCAIR (National Institute of Science Communication and Information Resources), New Delhi, India. Dried extract of C asiatica was procured from Natural Remedies (Bengaluru, India). TLC silica gel 60 F254 aluminum sheets (Batch HX360378) were purchased from Merck Millipore (Darmstadt, Germany). Asiatic acid, β-sitosterol, kaempferol, asiaticoside, diphenyl-β-picrazylhydrazyl (DPPH), tacrine, acetylthiocholine iodide, AChE (electric eel, type VI-S lyophilized powder, 480 U/mg solid, 530 U/mg protein), bovine serum albumin (BSA), and 5,5-dithiobis(2-nitrobenzoic acid) (DTNB) were purchased from Sigma-Aldrich (Bengaluru, India). Folin-Ciocalteau, phenol, methanol (HPLC grade), and acetonitrile (HPLC grade) were purchased from Merck Specialist Pvt Ltd (Mumbai, India). Gallic acid was purchased from Hi-Media Laboratories (Mumbai, India).
Instruments
Stability studies were performed in a stability chamber (Rolex Scientific Engineers, Ambala, India) capable of controlling temperature and relative humidity in the range of ±2°C and ±5%, respectively. The chamber was set at a temperature of 40°C and relative humidity of 75%. The HPLC system consisting of binary pumps (515), dual wavelength detector (2487), and Rheodyne manual injector (Waters, Milford, MA) was used for quantitative estimation of markers. The data were processed in Empower 2 software (Waters, Milford, MA). The chromatographic separations were achieved on Nucleodur C18 (250 mm × 4.6 mm; 5 μm) column. In addition, Inertsil C18 (250 mm × 4.6 mm; 5 μm) and XTerra C18 (250 mm × 4.6 mm; 5 μm) columns were used for validation studies. HPTLC analyses were performed on a HPTLC system equipped with Camag linomet V automatic spotting device (Camag, Anchrom). UV spectrophotometer (DU 640 B series, Beckman, USA) was used for spectrophotometric analysis. Moisture content in control and stability samples was determined by Karl-Fischer autotitrator (Model No. 841, Tetrando, Metrohm AG, Ionenstrasse, Switzerland).
Methods
Extracts
Dried extract procured commercially was marked as Type I. It was dry mixed with methyl paraben and propyl paraben in geometric progression, and the resultant extract was marked as Type II. Methyl paraben and propyl paraben were used as preservatives in a concentration of 1 mg/g and 0.1 mg/g, respectively. 11 The whole dried plant material was crushed in a grinder and extracted with absolute ethanol by soxhlet extraction method as reported by Thamarai Selvi et al. 12 This freshly prepared extract was mixed with methyl paraben and propyl paraben in the concentrations of 1 mg/ml and 0.1 mg/ml, respectively. 13 The solvent was recovered under reduced pressure to obtain dried fresh extract with preservatives, and marked as Type III extract.
Stability Studies
About 1 g of each extract was placed in each of 7 transparent glass vials separately, and each vial was tightly screwed. Two vials of each extract were stored at 4°C to serve as control. The others were placed in the stability chamber set at 40°C and 75% relative humidity. Vials were withdrawn at intervals of 1, 3, and 6 months, marked as stability samples AS1, AS3, and AS6, respectively, and stored at 4°C till physicochemical and biological studies were performed. All studies on each sample were performed in triplicate.
Physical Evaluation
Each of the control and stability samples was evaluated for its color, pH, and moisture content. For pH determination, a suspension of the sample in water (1 mg/mL) was sonicated for 15 minutes, and pH was measured using a pH meter. The moisture content of each sample was determined by Karl-Fischer titration.
Chemical Evaluation
Total Phenolic Content
It was determined as milligram of gallic acid equivalent per gram (mg GAE/g) of dry extract by Folin-Ciocalteu’s method as reported by Singleton and Rossi 14 with some modifications. A series of standard solutions having concentrations of 50, 150, 250, 350, and 450 μg/mL of gallic acid in methanol was prepared. A 0.1 mL aliquot of each standard solution was mixed with 1900 μL of distilled water. The resulting solution was mixed with 1 mL of 1:10 diluted Folin-Ciocalteu’s reagent and incubated at room temperature for 5 minutes. Subsequently, 1 mL of saturated sodium carbonate solution (15% w/v) was added, mixed well, and the solution was left to stand for 30 minutes at room temperature. Absorbance of the resulting solution was read at 760 nm taking water as blank. A calibration curve was constructed taking concentration of gallic acid as abscissa and absorbance as ordinate. Calibration curves were constructed in triplicate using fresh sets of standard solutions of gallic acid each time. Each of the control and stability samples was rendered into solution in methanol (4 mg/mL) and was processed similar to the standard solution of gallic acid to determine total phenolic content by using the following equation: Total phenolic content = C × V × DF/M, where C is the concentration of gallic acid (mg/mL) measured from the calibration curve, V is the volume (mL) of the extract, DF is the dilution factor, and M is the weight (g) of the extract. 15
Quantitative HPLC Analysis
Asiatic acid and kaempferol were resolved on Nucleodur C18 (250 mm × 4.6 mm; 5 μm) column by isocratic elution with mobile phase consisting of acetonitrile and phosphate buffer (20 mM, pH 3.5; 50:50% v/v) at a flow rate of 1 mL/min. The injection volume was 20 μL, and the eluent was detected at 220 nm. Quantitative analysis was performed by using standard solutions of asiatic acid (0.05 mg/mL), kaempferol (5 μg/mL), mixture of propyl paraben and methyl paraben (0.01 and 0.1 mg/mL), control samples (2 mg/mL), and stability samples (2 mg/mL) in methanol. The HPLC method was validated by evaluating linearity, precision, accuracy, robustness, limit of detection (LOD), limit of quantification (LOQ), and specificity in accordance with ICH guidelines. 16
Qualitative HPTLC Analysis
Solutions of reference standards of markers (asiatic acid, 0.5 mg/mL; kaempferol, 0.5 mg/mL; β-sitosterol, 0.5 mg/mL; asiaticoside, 0.25 mg/mL; and mixture of methyl paraben and propyl paraben, 1 and 0.1 mg/mL), as well as of control and stability samples of Type I extract (20 mg/mL), Type II extract (20 mg/mL), and Type III extract (40 mg/mL) were chromatographed on precoated HPTLC plates to generate HPTLC fingerprints. All solutions were prepared in methanol. An 8 μL aliquot of each solution was loaded on HPTLC plate as a sharp band. The loaded plate was developed in mobile phase A (chloroform-toluene-ethyl acetate-formic acid, 5.5:2:1:0.2) for detection of asiatic acid, kaempherol, and β-sitosterol, whereas for detection of asiaticoside, the loaded plate was developed in mobile phase B (chloroform-glacial acetic acid-methanol-water, 5.5:3:0.5:0.4). The developed plates were dried in air. The spots due to markers and other constituents were detected by visualizing the developed dried plate in visible light and UV light (254 nm and 366 nm). After these visual recordings, additional bands were detected by spraying the plate with p-anisaldehyde reagent.
Biological Activity Evaluation
Free Radical Scavenging Activity
It was evaluated by DPPH method as reported by Locatelli et al 17 with some modifications. A solution of DPPH (100 μM, 1 mg/25 mL) was prepared in methanol. Control sample (1 mL), in different concentrations, of the extract was mixed well with an equal volume of DPPH solution and incubated in dark at room temperature for 180 minutes. Absorbance (A) of the resulting solution was read at 515 nm using methanol as blank. A plot between A versus the corresponding concentration for each control sample was constructed, and IC50 value (expressed as %w/v) of each extract was determined through probit regression using BLeSq software as reported by Locatelli et al. 17 Antiradical activity of control sample of each extract at concentration equal to its IC50 value was expressed as percent inhibition (I%) and calculated against negative control (where sample solution was replaced with same volume of methanol) using the following equation: I% = [(AN − AS)/AN] × 100, where AN is the absorbance of negative control and AS is the absorbance of stability sample. Subsequently, the antiradical activity (I%) of stability samples of each extract was evaluated at the concentration used for its control sample.
Acetylcholinesterase Inhibitory Activity Assay
AChE inhibitory activity of control and stability samples was evaluated by a spectrophotometric method as developed by Ellman et al 18 with some modifications. A mixture of 2500 μL of phosphate buffer (0.1 M, pH 8.0), 100 μL of control/stability sample (1 mg/mL), and 200 μL of AChE solution (0.3 U/mL) was incubated at 25°C for 15 minutes. The reaction was initiated by the addition of acetylthiocholine (2 mM, 100 μL) and DTNB (4 mM, 100 μL), and was quenched after 10 minutes by adding tacrine (1 mM, 100 μL). Absorbance was monitored after 15 minutes at 416 nm. For negative control, the control/stability sample was replaced with water. Percent inhibition (I%) of AChE was calculated as follows: I% = (AN − AS)/AN × 100.
Statistical Analysis
Three replicates of each sample were used for statistical analysis with values reported as mean ± SD. Pearson’s correlation analysis was done to correlate different biological activities as well as dependence of the each activity on total phenolic content and markers contents.
Results
Physical Stability Evaluation
Control samples of Type I and Type II extracts were light green in color, whereas Type III extract was sticky and dark green. There was no significant change in color and appearance of any accelerated stability sample. Variation in pH of different stability samples under accelerated stability conditions was also insignificant (Table 2), whereas moisture content in each extract was found to increase insignificantly (Table 2).
Moisture Content and pH of Control and Accelerated Stability Samples.

aValues are expressed as mean ± SD (n = 3).
Chemical Evaluation
Total Phenolic Content
The method was linear (y = 1.244x + 0.260; r 2 0.999) for gallic acid in the concentration range of 0.05 to 0.45 mg/mL. Based on this calibration curve, total phenolic content in control sample of Type I, Type II, and Type III extracts was found to be 219.90 ± 1.73, 221.26 ± 3.59, and 166.23 ± 1.49 mg GAE/g dry extract, respectively. It decreased gradually in stability samples over 6 months under accelerated stability conditions to 197.48 ± 2.62, 202.65 ± 1.85, 154.47 ± 2.11 mg GAE/g dry extract, respectively (Figure 1).

Total phenolic content in mg GAE/g of dry extract of different types of extracts of Centella asiatica under accelerated stability conditions.
Quantitative Analysis
HPLC Method
A few HPLC methods are available in the literature for simultaneous estimation of asiatic acid and kaempferol (Table 3). But there are certain limitations in these methods. The method reported by Devkota et al 19 employs different wavelengths for detection of these 2 analytes, which imply that either a dual wavelength detector or repeat analysis of the same sample is required for complete analysis. The method reported by Alqahtani et al 20 uses a single detection wavelength but it has not disclosed the LOD and LOQ. Moreover, both these methods require run times from 25 to 35 minutes. We have developed an economical and efficient method that can analyze the 2 markers simultaneously using a single wavelength within a run time of 15 minutes. The only point where the present method seems to lag behind that reported by Devkota et al 19 is higher LOD and LOQ. However, this decrease in sensitivity can be compensated by increasing the sample concentration.
Comparative Account of Reported and Current HPLC Methods for Asiatic Acid and Kaempferol.
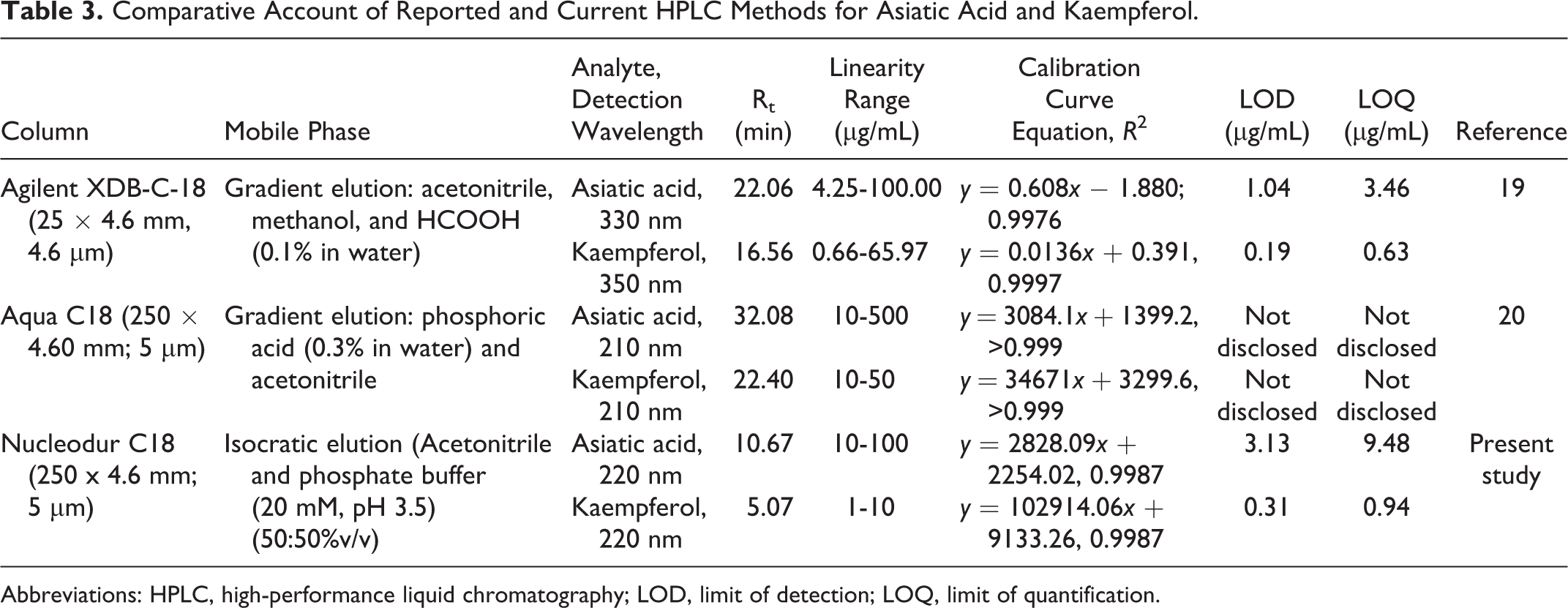
Abbreviations: HPLC, high-performance liquid chromatography; LOD, limit of detection; LOQ, limit of quantification.
HPLC Method Validation
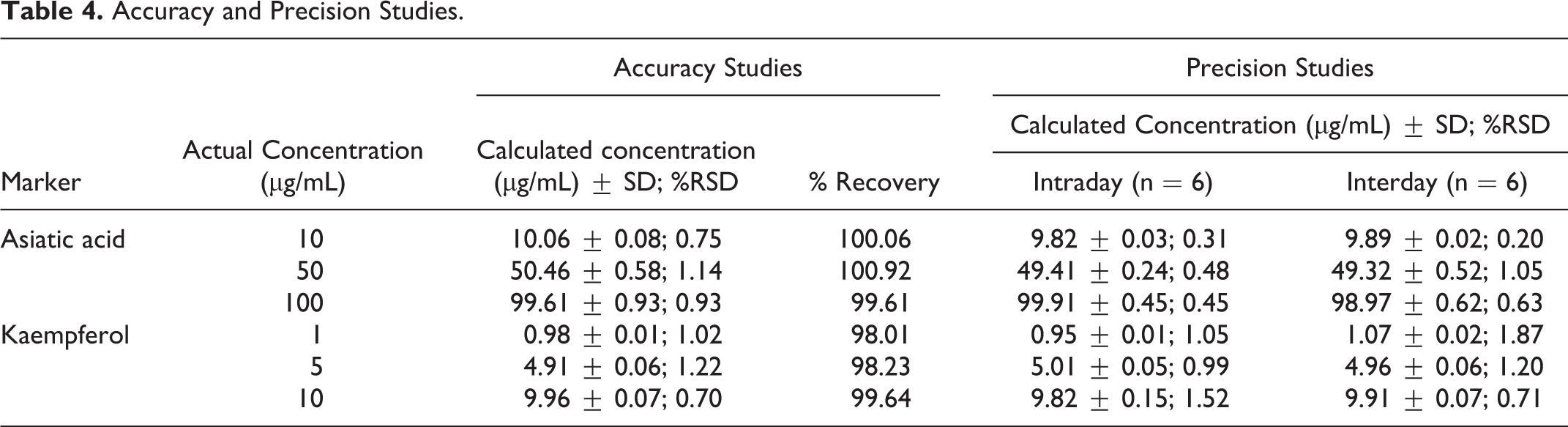
The method was linear for determination of asiatic acid and kaempherol in the concentration range of 10 to 100 μg/mL and 1 to 10 μg/mL, respectively. The mean (±SD) slope, intercept, and r
2
were, respectively, 2828.09 (±24.11), 2254.02 (±18.24), and 0.99866 (±0.0005) for asiatic acid, and 102914.60 (±14.01), 9133.26 (±29.91), and 0.99866 (±0.0005) for kaempferol. LOD and LOQ were 3.13 μg/mL and 9.48 μg/mL, respectively, for asiatic acid with percent relative standard deviation (%RSD) of 1.02 and 1.11, and 0.31 μg/mL and 0.94 μg/mL, respectively, with %RSD of 1.32 and 1.61 for kaempferol. Good recoveries of asiatic acid (99.61% to 100.92%) and kaempferol (97.4% to 99.65%) at each concentration level tested were achieved with %RSD less than 1.1 and 1.2, respectively (Table 4), which indicated the method to be sufficiently accurate for quantification of asiatic acid and kaempferol. The %RSD for interday and intraday precision was less than 1.8 (Table 4), which indicated that there was no significant variation in the concentration of marker compounds determined on any day. It established that the method was sufficiently precise for determining the marker concentrations in stability samples of dried extracts. In robustness studies, the % change in retention time (RT) as well as calculated concentration of asiatic acid and kaempferol was insignificant on deliberate small changes in different chromatographic conditions such as detection wavelength (λdet)
Accuracy and Precision Studies.
Stability Sample Analysis
Kaempherol and asiatic acid were eluted at 5.07 and 10.67 minutes, respectively, whereas methyl paraben and propyl paraben (preservatives) were eluted at 4.41 and 7.83 minutes, respectively (Figure 2). Presence of both the markers and preservatives in control samples was ascertained through matching of retention times and spiking studies. Comparison of HPLC-UV chromatograms of control samples of the 3 extracts revealed that the commercially procured extract and freshly prepared extract have similar constituents (Figure 3). The content of asiatic acid and kaempherol in each accelerated stability sample of each extract decreased with respect to the respective control (Table 5). More than 20% of asiatic acid was degraded in Type I and Type II extract after 6 months of storage. Degradation of asiatic acid was less in the stability samples of Type III extract than in those of Type I and Type II extracts. Degradation of kaempherol in the stability samples of Type I and Type II extracts was more than 23%, whereas the same in the stability samples of Type III extract was less than 17% after 6 months. HPLC-UV chromatograms of the 3 extracts stored under the accelerated stability conditions along with standard asiatic acid and kaempherol are shown in Figure 3.

HPLC-UV chromatograms of preservatives, methyl paraben (mp), and propyl paraben (pp) mixture (A), asiatic acid standard solution (B), kaempferol standard solution (C), and of control samples of Type I (D), Type II (E), and Type III (F) extracts of Centella asiatica. Peaks of asiatic acid and kaempferol marked as 1 and 2, respectively.
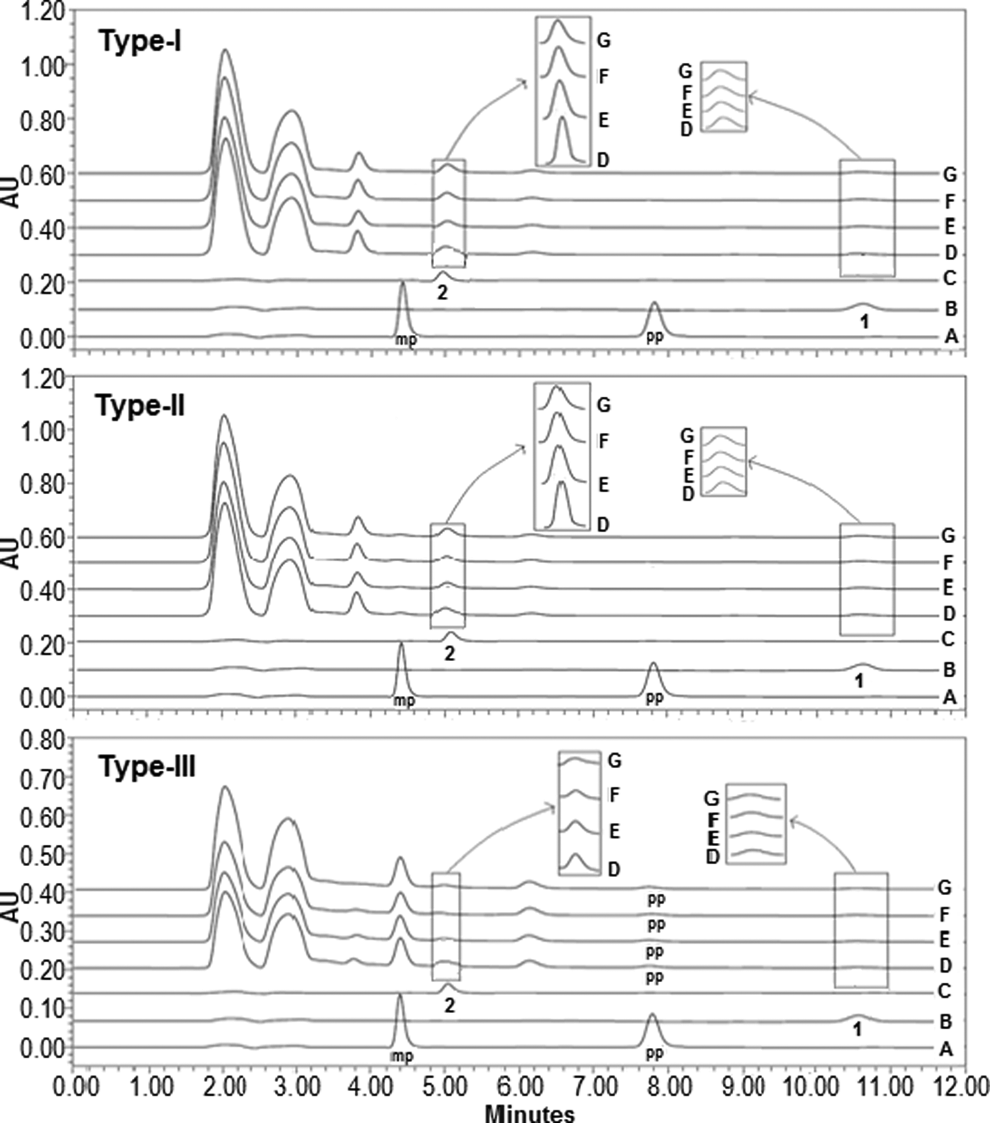
HPLC-UV chromatograms of preservatives, methyl paraben (mp), and propyl paraben (pp) mixture (A), asiatic acid standard solution (B), kaempferol standard solution (C), and of control sample (D) and stability samples of different extracts of Centella asiatica after 1 month (E), after 3 months (F), and after 6 months (G) under accelerated conditions. Peaks of asiatic acid and kaempferol marked as 1 and 2, respectively.
Asiatic Acid and Kaempferol Contents in Control and Accelerated Stability Samples.

aValues are expressed as mean ± SD (n = 3).
Qualitative Analysis by HPTLC Fingerprinting
A few HPTLC methods are reported in the literature for simultaneous detection of asiatic acid, madecassic acid, asiaticoside, and madecassoside (Table 6). 21 –23 But in the present study, we have selected asiatic acid and asiaticoside as central nervous system active markers, kaempferol as an antioxidant marker, and β-sitosterol as analytical marker. These compounds were not eluted/resolved by the chromatographic conditions reported in the literature. Hence, we attempted to develop a fresh HPTLC method that could resolve the 4 selected markers as well as that could resolve maximum possible number of other unknown constituents in the extracts.
Comparative Account of Reported and Current HPTLC Methods for Centella asiatica.
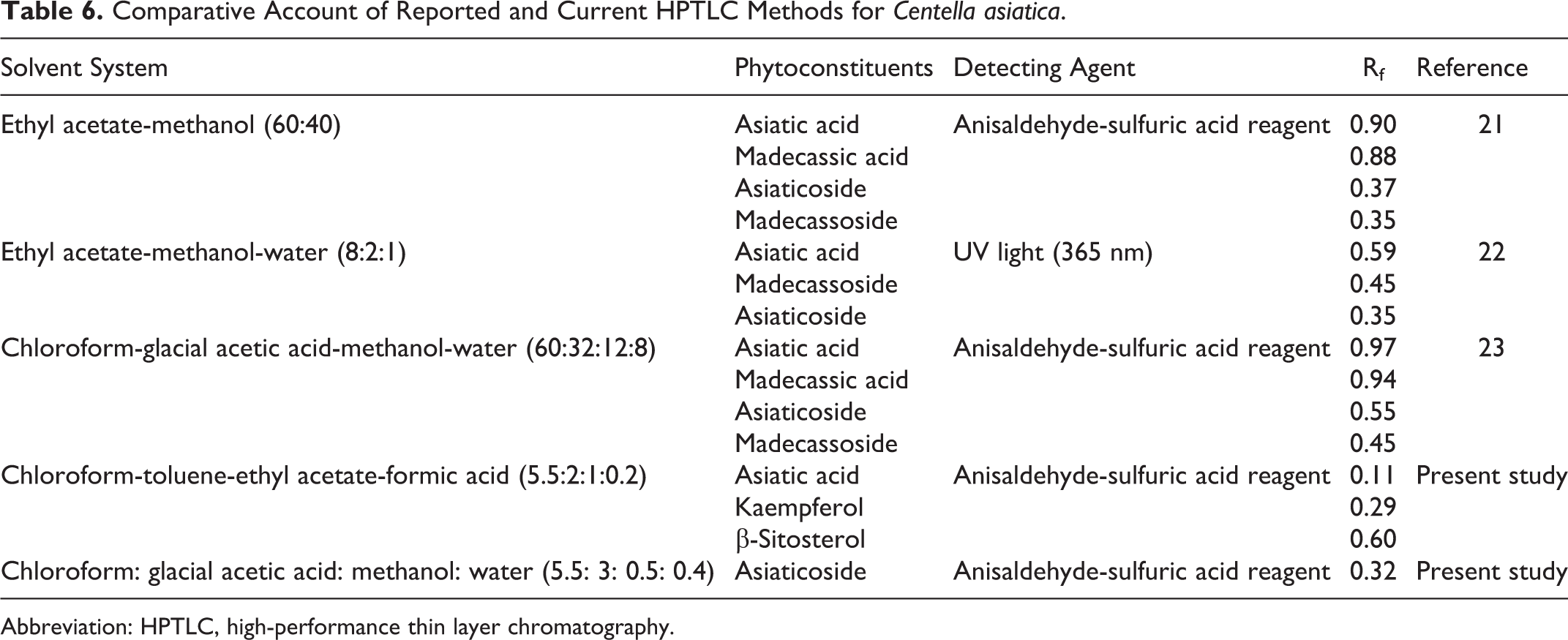
Abbreviation: HPTLC, high-performance thin layer chromatography.
Asiatic acid, β-sitosterol, and kaempferol were resolved with mobile phase A, and detected as blue, violet, and orange colored bands, respectively, after treatment of the developed plate with p-anisaldehyde (Figure 4). Kaempherol was also detected in visible light as well as UV light. Each of these 3 markers was present in the control sample of each extract (Figure 4). Comparison of fingerprints of the 3 extracts developed in mobile phase A and visualized in 365 nm revealed that fingerprint of Type III extract was congested than that of Type I and Type II extracts. It suggested that more number of constituents were present in freshly prepared extract of C asiatica. As an intense fluorescence is exhibited by compounds containing aromatic and rigid planar structures, 24 the additional bands in Type III extract may be due to additional flavonoid constituents in C asiatica. Many of such flavonoid constituents are reported by Chong and Aziz 10 and Vasantharuba et al, 25 and these are detected in the wavelength range of 325 to 370 nm. 26 These findings also indicated that many flavonoids, reported to be present in C asiatica, may be absent in commercially prepared extract vis-a-vis the freshly prepared one (Figures 4 and 6), and it may extrapolate to altered pharmacological effectiveness of the extract. In mobile phase B, asiaticoside was detected as a blue band after p-anisaldehyde treatment (Figure 6). It was detected in control samples of all 3 types of extracts. More number of bands were observed in mobile phase B during fingerprinting of commercially procured extract than in the freshly prepared one, when visualized in 254 nm. In other visualization modes, there was no difference between fingerprints of the 2 types of extracts (Figure 6). Comparison of fingerprints of stability samples with control sample of each extract (Figures 5 and 7) revealed that all bands noted in the control sample were also present in all stability samples. The actual colored figures of the HPTLC chromatograms can be accessed in the Supplementary Figures 4S to 7S (available online at http://chp.sagepub.com/supplemental) for the perfect view of the HPTLC data.

HPTLC chromatograms of standard solutions of asiatic acid (1), β-sitosterol (2), kaempferol (3), methyl paraben and propyl paraben mixture (4), and control samples of Type I (5), Type II (6), and Type III (7) extracts. The mobile phase was chloroform-toluene-ethyl acetate-formic acid (5.5:2:1:0.2), and plates were visualized in visible light, UV light (254 and 365 nm), and in visible light after treatment with p-anisaldehyde.

HPTLC chromatograms of standard solutions of asiatic acid (1), β-sitosterol (2), kaempferol (3), methyl paraben and propyl paraben mixture (4), and of control samples (5) and stability samples of extracts of Centella asiatica after 1 month (6), after 3 months (7), and after 6 months (8) under accelerated conditions. The mobile phase was chloroform-toluene-ethyl acetate-formic acid (5.5:2:1:0.2), and plates were visualized in visible light, UV light (254 and 365 nm), and in visible light after treatment with p-anisaldehyde.

HPTLC chromatograms of standard solutions of asiaticoside (1), control samples of Type I (2), Type II (3), and Type III (4) extracts. The mobile phase was chloroform-glacial acetic acid-methanol-water (5.5:3:0.5:0.4), and plates were visualized in visible light, UV light (254 and 365 nm), and in visible light after treatment with p-anisaldehyde.

HPTLC chromatograms of standard solutions of asiaticoside (1) and of control sample (2) and stability samples of extract of Centella asiatica after 1 month (3), after 3 months (4), and after 6 months (5) under accelerated conditions. The mobile phase was chloroform-glacial acetic acid-methanol-water (5.5:3:0.5:0.4), and plates were visualized in visible light, UV light (254 and 365 nm), and in visible light after treatment with p-anisaldehyde.
Biological Activity Evaluation
Antioxidant Activity
An incubation time of 180 minutes for evaluating free radical scavenging activity was optimized through kinetic studies on control samples of each extract. The antioxidant activity expressed as IC50 value of control samples of Type I, Type II, and Type III was found to be 0.031 ± 0.01, 0.026 ± 0.03, and 1.621 ± 0.01 mg/mL. Based on these IC50 values, a concentration of 0.03 mg/mL for Type I and Type II extracts and 1.6 mg/mL for Type III extract was selected for determining antiradical activity (I%) of control and stability samples of the extracts (Table 7). The antiradical activity of each stability sample of each type of extract was decreased after 6 months (Figure 8). However, the decrease was less in stability samples of Type II than in Type I extract and minimum (only 3%) in Type III extract.
Antioxidant Activity of Control and Accelerated Stability Samples of the Extracts.

Abbreviation: DPPH, diphenyl-β-picrazylhydrazyl.
aValues are expressed as mean ± SD (n = 3).
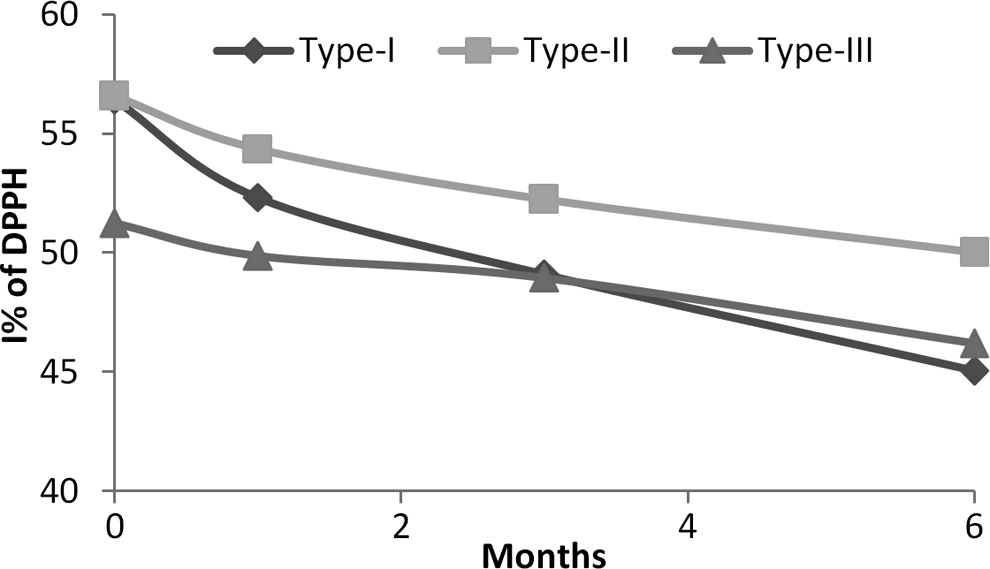
Percent inhibition of DPPH radical activity by different extracts at different time intervals under accelerated stability conditions.
AChE Inhibitory Activity Assay
Percent inhibition (I%) of AChE activity by control and stability samples of each type of extract (Table 8) revealed that AChE was inhibited almost equally by Type I and Type II extracts. However, this inhibition was significantly less by Type III extract, which revealed that Type III extract was a poor AChE inhibitor than the Type I and Type II extracts. Furthermore, the activity of each stability sample was decreased with time (Figure 9), and this decrease was observed to be minimum in Type III extract.
AChE Inhibitory Activity of Control and Accelerated Stability Samples of the Extracts.
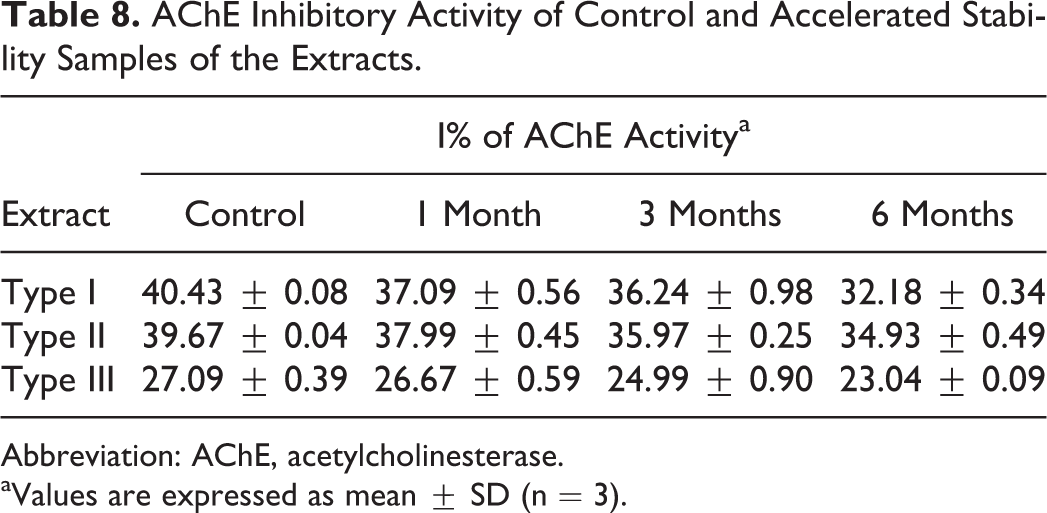
Abbreviation: AChE, acetylcholinesterase.
aValues are expressed as mean ± SD (n = 3).

Percent inhibition of AChE by different extracts under accelerated stability conditions.
Correlation Among Total Phenolic Content, Antioxidant Activity, Markers Content, and AChE Inhibitory Activity
Correlation between different biological activities as well as dependence of the each activity on total phenolic activity and marker contents was determined by Pearson correlation coefficient calculator, which is a test used to measure the strength of a linear association between 2 variables. A correlation coefficient (r) of 1 indicates a strong positive correlation between the 2 tested variables, whereas a correlation coefficient of −1 reflects a strong negative correlation. In the present study, the antioxidant activity was strongly correlated positively with total phenolic content with r values of 0.998, 0.989, and 0.987 for Type I, II, and III extracts, respectively. A strong positive correlation was also noted between AChE inhibitory activity and total phenolic content with r values of 0.989, 0.964, and 0.971 for the 3 extracts. These findings were consonant with the results reported by Ahmed et al, 27 who have reported a significant positive correlation between total phenolic content and anti-AChE activity of methanol extracts of Nardostachys jatamansi and Acorus calamus. There was also a strong positive correlation between AChE inhibitor activity and antioxidant activity (r values of 0.983, 0.992, and 0.977 for Type I, II, and III extracts, respectively). These results were in agreement with studies reported by Jo et al 28 wherein a positive correlation is shown between AChE inhibitory and antioxidant activities of seed and pericarp of Camellia sinensis L. AChE inhibitor activity was also correlated positively with content of asiatic acid (r values of 0.990, 0.964, and 0.956) as well as kaempferol (r values of 0.970, 0.99, and 0.976) for Type I, II, and III extracts, respectively. These results are in agreement with the studies conducted by Nasir et al, 29 who revealed that asiatic acid has effective AChE inhibitory properties, as well as by Priya, 30 who have reported that kaempferol isolated from methanol extract of Morus alba L. leaves show significant AChE inhibitory activity.
Discussion
Total phenolic content, content of markers, and antioxidant and AChE inhibitory activities of each extract decreased gradually in accelerated stability samples with respect to control. These changes were maximum in Type I extract followed by Type II and Type III extracts. It might be attributed to the altered chemical composition of Type III extract because of it being from a different source, which in turn may be accounted for batch to batch variation in chemical composition and therapeutic efficacy of natural products. Comparison of stability data of Type I and Type II extracts through HPLC analyses and biological evaluation indicated that preservatives have marginally protected the markers. Comparison of HPTLC fingerprints of the stability samples with control sample of each extract suggested that there was no gross change in chemical composition of the extracts on storage under accelerated stability conditions. Additionally, more number of bands was observed in stability samples of Type III extract under 365 nm in mobile phase A and under 254 nm in mobile phase B. Furthermore, antioxidant constituents are protected to maximum extent in Type III extract. The higher stability of Type III extract than Type I and II extracts was attributed to the presence of additional phytoconstituents as revealed by HPTLC fingerprints and/or preservatives. Pearson correlation analysis revealed a linear positive correlation between the different chemical and biological parameters. These findings suggested that total phenolic content, asiatic acid, and kaempferol in C asiatica can be taken as chemical markers to assess chemical as well as therapeutic shelf lives of herbal products containing C asiatica as per the World Health Organization guideline.
Footnotes
Acknowledgements
The authors are grateful to the National Institute of Science Communication and Information Resources (New Delhi, India) for authentication of the plant and to Natural Remedies (Bengaluru, India) for providing dried extract of Centella asiatica as generous gift sample.
Author Contributions
Gulshan Bansal conceptualized the study, oversaw data collection, and made the manuscript word perfect. Nancy Suthar designed the study, oversaw data collection, and commented on the manuscript. Ishtdeep Kaur executed the study, performed experimental work, and drafted the manuscript. Jasmeen Kaur did an important job of re-representation of the data and improving resolution of the figures in the manuscript. Yogita Bansal oversaw the data of antioxidant activity assay and commented on the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are thankful to DBT-Punjabi University Patiala Interdisciplinary Programme in Life Sciences for Advance Research and Education for providing partial financial assistance for carrying out the study (File No. 102/IFD/SAN/4650/2011-2012). Thanks are also due to the All India Council for Technical Education (New Delhi, India) for disbursing a fellowship to one of the authors (Ishtdeep Kaur).
Ethical Approval
The study did not involve use of any animals. All biological testing was done through in vitro methods. So no approval from the animal ethical committee was required.
