Abstract
Amaranthus dubius is a vegetable consumed for its nutritional content in Kenya. In herbal medicine, A. dubius is utilized to relief fever, anemia and hemorrhage. Additionally, it is utilized to manage cognitive dysfunction and is considered to augment brain function, but there is no empirical evidence to support this claim. The contemporary study investigated cognitive enhancing potential of A. dubius in mice model of Alzheimer's disease (AD)-like dementia induced with ketamine. Cognitively damaged mice were treated with aqueous extract of A. dubius leaf upon which passive avoidance task (PAT) was used to assess the cognitive performance. At the end of passive avoidance test, brains of the mice were dissected to evaluate the possibility of the extract to inhibit hallmarks that propagate AD namely oxidative stress and acetylcholinesterase activity. Additionally, characterization of secondary metabolites was done using liquid chromatograph- mass spectrometry analysis. During PAT test, extract-treated mice showed significantly increased step-through latencies than AD mice, depicting ability of A. dubius to reverse ketamine-induced cognitive decline. Further, the extract remarkably lowered malondialdehyde levels to normal levels and effectively inhibited acetylcholinesterase enzyme. The study showed that A. dubius extract is endowed with phytoconstituents that possess anti-oxidant and anticholinesterase activities. Thus, this study confirmed promising therapeutic effects of 200, 300 and 400 mg/kg bw of A. dubius extract with potential to alleviate cognitive disarray observed in AD.
Introduction
Alzheimer disease is the most prevalent form of dementia contributing to 70% of the total disease burden. 1 It is featured by gradual memory deterioration, cognitive deficits and personality deviations as a result of neurodegeneration in the frontal cortex and hippocampus parts of the brain. 2 Brains of Alzheimer disease (AD) patients reveals nerve cell atrophy, neuronal death and astroglyosis. 1 Also, they contain widespread distribution of two biochemical hallmarks: intracellular neurofibrillary tangles (NFTs) and extracellular senile/amyloid plaques. 1 Mainly, amyloid plaques comprise of three insoluble proteolytic resistant variants namely Aβ38, Aβ40 and Aβ42 generated from breakdown of amyloid precursor protein by γ and β-secretase. 3 Neurofibrillary tangles comprise of aggregates of excessively herphosphorylated tau protein. 3
It is hypothesized that Aβ production recruits a series of downstream molecular events that orchestrate the clinical outcomes of AD. Formation of amyloid peptides induces oxidative stress, localized inflammation, excitoxicity and tau hyperphosphorylation. 4 Tau protein attaches to microtubules in neuronal cells to facilitate the transport system. 3 Overly phosphorylated tau protein misfolds into intraneuronal fibrils which results in microtubule disassembly with ultimate collapse of neuronal transport system. 3 Consequently, tau and Aβ act together to induce neurotoxicity with ultimate development of AD.5,6 Ultimately, gradual destruction of neuronal cells leads to deficiency and imbalance between numerous neurotransmitters chiefly acetylcholine eventually accomplishing the cognitive deficiencies observed in AD.3,4 Essentially, cholinergic hypothesis postulates that imbalance of acetylcholine in the cholinergic system markedly contributes to cognitive deficits observed in AD patients. 7
Besides, oxidative stress is implicated in pathogenesis of AD. 8 Notably, high metabolic rate in brain cells predispose them to oxidative stress than other body cells. 9 Particularly, major biomolecules (nucleic acids, lipids and proteins) are commonly oxidized in AD. 10 Neuronal cells contains high amounts of polyunsaturated fatty acids that are prone to assault by reactive oxygen radicals which lead to a series of self-sustaining lipid peroxidation reactions consequently, causing molecular damage in neurons. 11 Evidently, oxidative stress (OS) is major hallmark of AD, however, its origin remains widely debated. Research has proposed that β-amyloid (Aβ) deposition, mitochondrial dysfunction, 12 over phosphorylated tau 13 and inflammation are some of the fundamental mechanisms that stimulate oxidative damage in AD. 12 Previously, it has been demonstrated that blockade of OS enhances cognitive performance in Alzheimer's mice models. 14 Hence, inhibition of oxidative stress using antioxidant agents has been perceived as a reliable target for development of anti-Alzheimer's agents.13,15
Majority of the current AD therapeutics are cholinesterase inhibitors including galantamine, donepezil, rivastigmine and tacrine. 4 These compounds modulate the cholinergic system leading to increased levels of acetylcholine eventually alleviating symptoms of AD. Though unable to completely abolish the disease, the drugs have been largely successful in improving cognitive capabilities in AD patients. 16 Unfortunately, their clinical use is marred by severe adverse effects which limits their application. 16
Considering the growing contribution of AD to the growing global life-long disabilities and socioeconomic burden, 17 it is urgently fundamental to search for novel effective therapeutic agents with minimal side effects for management and treatment of AD. In fact, approximately 50 million people have AD and these cases are estimated to rise to 152 million people by 2050, globally. 18 Further, efforts to develop curative options for AD have been futile and to date no cure for AD exists. 16 Taken together, these factors necessitate continued efforts to explore for novel remedies to alleviate the increasing mortality and morbidity caused by AD.
The current study focused on evaluating cognitive enhancing potential of aqueous leaf extract of Amaranthus dubius as an alternative therapy for management of AD. A. dubius belong to the genus Amaranthus. 19 It's an annual green vegetable which particularly grows in Africa, besides other subtropical and tropical regions. It is one of the most widely eaten leafy vegetable green in Africa. It grows up to 200 cm tall with erect, green stem and petiole proximal ovate to elliptic leaves about 1.5-10 cm. 19 A. dubius is locally utilized for management cognitive associated disorders. 19 Besides, multiple Amarathus spp have been revealed to possess cognitive enhancing properties; Amaranthus tricolor 20 and Amaranthus paniculatus. 21
Phytochemistry of A. dubius has not been keenly investigated however qualitative screening shows that the plant the major classes of compounds in the plant include vitamins (B6, K, C, A, Folate and Riboflavin), phenols, flavonoids and alkaloids.19,22 Largely, Amaranthus spp have been shown to contain rutin, kaempferol, isoquercetin, hyperoside, quercetin, catechin, apigenin, naringenin, myricetin and catechin, 23 among others.
Some of these phytocompounds are associated with cognitive enhancing potencies. For instance, vitamin K is known to inhibit neural destruction in the brain, hence, its consumption is recommended for AD patients. 22 Further, various flavonoids and alkaloid have been demonstrated to inhibit oxidative stress, 24 amyloidogenesis 25 and inflammation, 26 halting key mechanism involved in AD pathogenesis.
Taking into account ethnopharmacological evidence and phytochemistry of A. dubius, it is conceivable that the plant may serve as an alternative remedy for management of cognitive disorders. However, there is paucity of information to confirm its efficacy on enhancing cognition. In the quest to tackle this gap, the current study assessed the effect of aqueous extract of A. dubius leaf on cognition. Anti-AChE and antioxidant activities of the extract were determined.
Materials and Methods
Plant Samples Collection and Preparation
Green leaves of Amaranthus dubius were gathered from Namakanyu, Muranga County, Kenya. The collected sample was taxonomically authenticated at the University Herbarium and assigned a voucher number DJK (001). The samples were dried under shade at 25 ± 2 °C after which they were milled to fine powdered, packed in a clean, labelled dry containers at room temperature (25 ± 2 °C) till extraction.
Aqueous Extraction
A total of 50 grams of powdered A. dubius were added to 500 ml of distilled water at 100 °C for 90 min. The extract was then cooled to 25 °C, decanted followed by filtration using Whatman Grade 1 filter paper. The filtrate was lyophilized to concentrate the crude extract that was later used for bioassays. The resulting extract was preserved at 4 °C. 27
In Vivo Cognitive Enhancement Studies
Study Animals
The animal models used in this study were male Swiss Albino mice weighing 20 ± 2 grams (4-5 weeks old). They were acquired from the University animal handling and breeding unit. The mice were transferred to the experimental laboratory where they were kept during the study period. They were reared in 30 cm × 20 cm × 13 cm size cages containing tender wood chips and fed on the standard rodent pellets and unlimited water. The mice were maintained in a natural night-day cycle, 25 C and 50% humidity throughout the study period. They were allowed seven days period to acclimatize to the study environment prior to commencement of the tests. The procedures for mice handling adhered to American Psychological Association's ethical guidelines for manipulating laboratory animals. 26 The study received ethical review and approval prior to experimentation; Approval Number: NACOSTI/P/22/17588.
Animal Grouping and Treatment
Animals were randomly assorted into 6 groups (n = 5) and labelled from roman I through VI. The mice were starved overnight prior to treatment. Group I (normal control) were given normal saline only, group II (negative control) received ketamine (15 mg kg−1 bw−1) only, group III (positive control) were administered donepezil (1 mg/kg bw) while group IV, V and VI received 200, 300 and 400 mg kg−1 bw−1 of the plant extract on the experimental day. Forty-five (45) minutes after receiving their respective treatment, group III through VI were intraperitoneally administered with 0.2 ml of ketamine (15 mg/kg bw) to induce cognitive damage. The extract doses used herein were selected based on pilot test. The choice of Ketamine dose was guided by previous studies. 28 Saline was the vehicle for all the treatments. 29 All treatments were administered orally except ketamine. Further, all treatments were administered daily for a period of three days. The experimental schedule was designed as outlined in Table 1.
Experimental Protocol.

Passive Avoidance Task (PAT)
Impact of aqueous extract of A. dubius on learning and memory was evaluated using PAT test. The test was conducted for six days using 25 cm×20 cm × 20 cm box partitioned into a lit compartment and a dark compartment 30 partitioned with a carton wall fitted with a sliding door (3 cm × 3 cm). The dark chamber was wired with an electro-circuit that could conduct a maximum of 0.5 mA. Initially, all the experimental animals were habituated to the passive avoidance box (PAB). Each mouse was transferred to the lit chamber for 20 s thereafter the sliding door was opened and let to explore PAB compartments. Once the mouse had completely occupied the dark partition of PAB, the door was locked and then, after 30 s, it was released from the dark compartment and taken back to its respective cage. Habituation trial was repeated after 30 min thrice 31 and the electro-circuit was off.
Habituation was followed by two-days training of all the animals for memory development using aggressive stimulus. Each mouse was allowed to roam in the lit chamber of PAB for twenty seconds. Afterward, the sliding door was opened and the mouse was let to explore the PAB for 5 min. After the animal entered the dark partition of PAB with both hind legs, foot electroshock of 0.5 mA was administered for three second. 32 The animal was held in the dark chamber for thirty seconds to relate the chamber with obnoxious stimulus, after which it was returned to its respective cage.
After 24 h following training, testing of cognitive memory was carried out for three days. After administration of various treatments, mice were separately transferred to the lit chamber and the sliding door was opened after 20 s. Step-through latencies of each animal wer monitored for 5 min, then returned to their respective cages. 32 The step-through latency was noted once the animal moved to the dark chamber with both hind legs.
During habituation, training and testing, both chambers were washed with 70% alcohol after every session to remove any olfactory signals. 33
Ex Vivo Assays
On the final day of PAT assay, mice were euthanized and their brains harvested. Each brain tissue was mixed with a 0.6 ml sodium phosphate solution (pH 7.4, 0.1 M) and homogenized. The resultant brain tissue homogenates were used for acetylcholinesterase and malondialdehyde activity assays.
Ex Vivo Acetylcholinesterase Activity Assay
Ellman method (1961)
34
was used to evaluate acetylcholinesterase inhibitory activity of the A. dubius. A volume of 0.5 milliliters of homogenized brain tissue was transferred to 1 milliliter of 0.1 M phosphate solution (pH 8.0) and centrifuged for ten minutes at 3000 rpm. A mixture containing 170 μl of 4% ellman's Reagent, 470 microliters of 0.1 M sodium phosphate (pH 8.0) and 66 microliters of the resultant supernatants was incubated at 37 °C for 5 min. Then, 280 microliters aliquots of 1 mM S-Acetylthiocholine iodide substrate were incorporated into the mixture. The resultant solution was incubated for 3 min at 37 °C following which the enzymatic activity was determined spectrophotometrically at 410 nm. A blank control comprised of all the contents in the assay was included. The optical density (OD) was recorded at the first and the fourth minute. Every sample was analyzed in triplicate. Enzyme activity was calculated as follows; ΔAs is the change in the absorbance of test sample from the first and the fourth minute. ΔAb is the change in the absorbance of blank control from the first and the fourth minute. ε is the molar extinction coefficient of AChE (13 600).
Time represents the time interval at which the absorbance was recorded
Tissue Conc. is the amount of the tissue used.
Anti-acetylcholinesterase activity of the extract was presented as percentage inhibition as inferred by the following formula;
Ex Vivo Malondialdehyde Activity Assay
Malondialdehyde (MDA), an oxidative stress marker, was measured using thiobarbituric acid assay.
35
Brain homogenates (0.1 ml) were transferred to a mixture comprising 1.5 millilitres of thiobarbituric acid (0.8%), 1.5 millilitres of 20% ethanoic acid (pH 3.5) and 0.2 millilitres of 8.1% Sodium lauryl sulphate. The resultant solution was heated for 60 min at 100 °C, cooled, then 5 millilitres of pyridine: n-butanol (1:15) and 1milliliter of distilled water were added. The contents were thoroughly vortexed then centrifuged at 2500 rpm for twenty minutes. Absorbance was then taken using a spectrophotometer at 532 nm. Concentrations of MDA were computed as outlined by Hodges et al (1999)
36
; As = absorbance of the analyte Ab = absorbance of blank control ε is the molar extinction coefficient of MDA (1.56 × 106 M− 1 cm− 1)
Phytochemical Profiling
Characterization of phytochemical contents of the aqueous extract of A. dubius was done following previously described protocols.37,38 The equipment used was Agilent MSD 6120-Triple Quadruple LC-MS/MS with an electrospray source (Palo Alto, CA). Separation was performed using Agilent Technologies 1200 infinite series reverse-phase eclipse plus C18 column (4.6 × 50 mm) (Agilents CA814112). Data was obtained in a full-scan negative ion mode at scan range between 100 to 1500 m/z.
Statistical Analysis
The study generated quantitative data on percentage yield of the identified compounds, passive avoidance tasks, acetylcholinesterase and malondialdehyde assays. Mass spectra for compound analyzed by LC-MS provided qualitative data. All the quantitative data were analyzed for descriptive summary and presented as mean ± standard error of the mean. Kolmogorov-Smirnov statistic revealed that the data was normally spread (P > .001). Homogeneity of variance was confirmed with Levene test (P > .001), hence validated for parametric tests. Statistical significance between means of the studied groups was tested with One-factor analysis of variance (ANOVA) followed by Fisher LSD at 99.9% confidence interval for pairwise comparison between the study groups. Statistical difference was inferred at p˂0.001. All data were analyzed in Minitab v19. The results were represented in figures and tablets.
Results
Effects of Aqueous Extract of Amaranthus dubius Leaf on Ketamine-Induced Cognitive Impairment in Mice
Generally, the extract showed remarkable in vivo cognitive-enhancing effects. During PAT test, all extract treated mice showed longer step-through latencies compared with the step-through latencies of negative control mice. On the first day of treatment, step-through latency of mice administered with A. dubius extract concentration of 300 mg/kg bw was significantly higher (P < .001) compared with step-through latencies of animals in all treatment groups except normal control mice where their step-through latencies were comparable (P > .001; Table 2). Thereby, indicating the ability of the extract to restore cognition to normal levels at this concentration.
Effects of Amaranthus dubius Aqueous Leaf Extract on Step Through Latency in Mice After Ketamine Induced Cognitive Impairment.
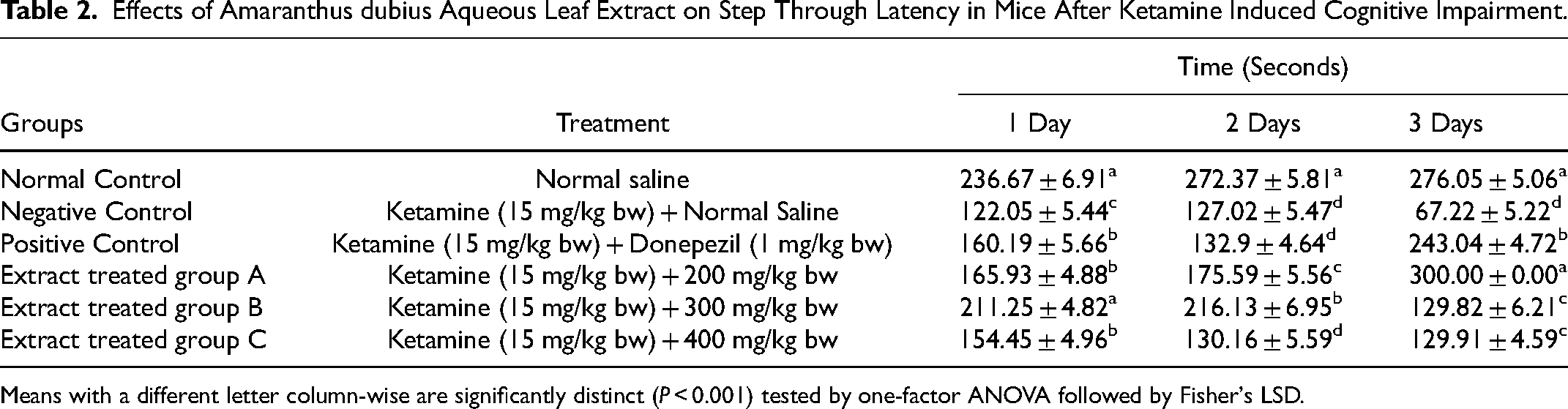
Means with a different letter column-wise are significantly distinct (P < 0.001) tested by one-factor ANOVA followed by Fisher's LSD.
Further, step-through latencies of mice that received A. dubius extract dosages of 200 and 400 mg/kilogram bodyweight were statistically different (P < .001) to step-through latency of normal control mice. On the other hand, findings revealed that the step-through latencies of mice given 200 and 400 mg kg−1 bw−1 of A. dubius extract were statistically alike to the step-through latency of mice that received the conventional drug (donepezil) (P > .001; Table 2). Thus, indicating that the extract at these doses had similar cognitive-enhancing effects to conventional drug (donepezil). In addition, cognitive-enhancing activities of aqueous extract of A. dubius leaf on the first day of treatment was demonstrated by the significantly higher step-through latencies in all experimental groups of animals compared with step-through latencies of the negative control mice (P < .001; Table 2).
During the second day of treatment, mice given A. dubius extract at the dosage of 300 mg/kilogram body weight demonstrated significantly higher step-through latency compared with step-through latencies of mice given aqueous extract of A. dubius at the dosages of 200 and 400 mg kg−1 bw−1 (P < .001; Table 2). Furthermore, mice given A. dubius extract doses of 300 and 200 mg/kg bw showed significantly higher step-through latencies compared with step-through latency of animals that were given the standard drug (P < .001; Table 2), indicating better extract efficacy in restoring cognition compared with the conventional drug (donepezil).
Moreover, step-through latency of animals given A. dubius extract concentration of 400 mg/kg bw was statistically similar to step-through latency of mice that received reference drug (P > .001; Table 2), which was indicative of similar cognitive-enhancing efficacies. Findings of this study further showed that step-through latencies in all experimental animals were significantly below the step-through latency of normal control mice (P < .001; Table 2).
On the third day of treatment, animals treated with aqueous extract of A. dubius leaf at 200 mg kg−1 bw−1 dosage showed significantly higher step-through latency compared with animals in all treatment groups (P < .001; Table 2) except mice in normal control group, which had statistically similar step-through latency (P > .001; Table 2). In addition, mice that received A. dubius extract concentration of 200 mg/kilogram bodyweight demonstrated significantly higher step-through latency compared with mice given the standard drug (donepezil) (P < .001; Table 2), an indication that A. dubius extract concentration of 200 mg/kilogram bodyweight had better cognition enhancement effects than the conventional drug. Furthermore, mice that received extract of A. dubius at doses of 300 and 400 mg/kg bw had significantly higher step-through latencies compared with amnesiac mice.
Ex Vivo Effects of Aqueous Extract of Amaranthus dubius Leaf on Malondialdehyde Levels in Mice After Ketamine-Mediated Cognitive Damage
Aqueous leaf extract of A. dubius was deciphered to enhance cognition by its potency to reduce amount of MDA in brains of cognitively damaged mice. The brains of normal control mice and extract treated mice had statistically similar MDA levels (P > .001; Figure 1). Further, MDA concentration in brains of mice administered with the studied doses of A. dubius extract were statistically similar (P > .001; Figure 1), indicating that the studied doses equally reduced MDA levels in mice.

Effects of Amaranthus dubius aqueous leaf extract on levels of MDA in the brain of mice following ketamine induced cognitive impairment. Bars with a different letter are significantly distinct (one-factor ANOVA followed by Fisher's LSD; P < .001).
Notably, brains of A. dubius-treated mice, at all doses, showed significantly lower MDA levels than brains of mice that received the reference drug (P < .001; Figure 1). Cognitive-promoting capacity of A. dubius extract were further demonstrated by significantly low levels of MDA in brains of all animals treated with the plant extract compared with MDA amounts in brains of ketamine-treated mice (P < .001; Figure 1).
Ex Vivo Anti-acetlycholinesterase Activities of Aqueous Leaf Extract of Amaranthus dubius in Cognitive-Impaired Mice
The present study established that A. dubius had potent inhibitory activities on acetylcholinesterase enzyme activity in cognitively-impaired mice. This was indicated by percentage inhibition of acetylcholinesterase activity. A. dubius extract dose of 200 mg kg−1 bw−1 had significantly higher anticholinesterase activity compared with anticholinesterase activities in mice administered with the reference drug and the other extract doses (P < .001; Figure 2). The extract concentration of 400 mg kg−1 bw−1 and reference drug had comparable efficacies (P < .001; Figure 2). Additionally, the extract dose of 300 mg kg−1 bw−1 showed the lowest efficacy against acetylcholinesterase enzyme (P > .001; Figure 2).

Anti-AChE effects of A. dubius aqueous leaf extract in the brains mice after ketamine induced cognitive impairment. Bars with a different letter are significantly distinct (P < .001) tested by one-factor ANOVA followed by Fisher's LSD; P < .001.
Quantitative Phytochemical Profiles of the Aqueous Extract of A. dubius Leaf
The LC-MS analysis showed presence of seventeen phytocompounds in the aqueous leaf extract of A. dubius (Table 3). Majority of the identified compounds were flavonoids and phenols. Phenolic metabolites included vanillic acid, gallic acid, ellagic acid 4-hydroxycinnamic and ferulic acids. Flavonoids comprised of catechin, luteolin, naringenin, apigenin and quercetin. Other secondary metabolites identified were fatty acids (octadecanoic, arachidic and oleic acid), phytosterol (campesterol and beta-Sitosterol) and sterols (Stigmasterol) (Table 3). Figure 3 indicates the LC-MS spectra for the identified compounds.
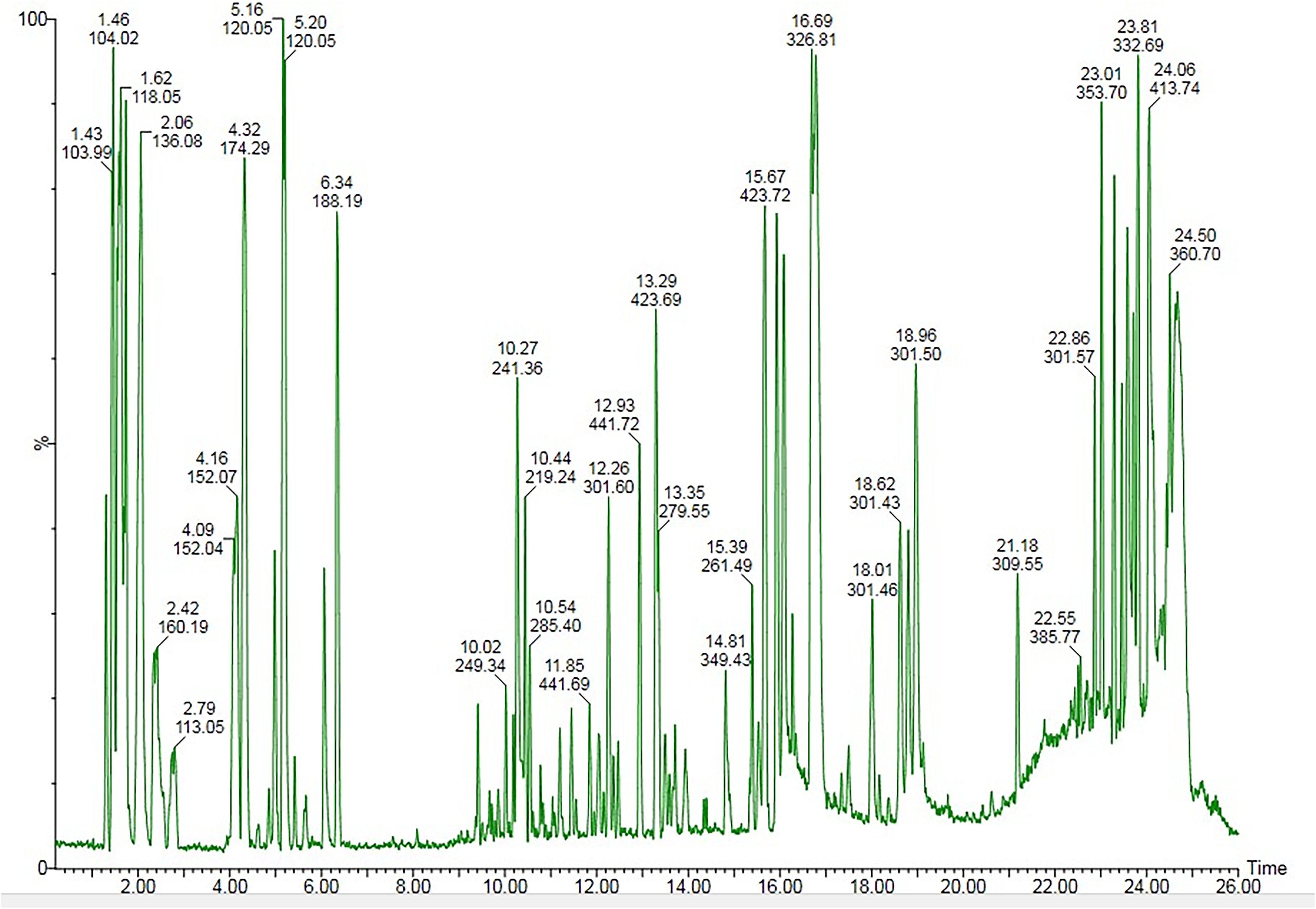
LC-MS chromatogram for aqueous extract of A. dubius leaf with retention time.
Phytochemistry profile of Aqueous Leaf Extract of A. dubius.

Discussion
In the present study, multiple assays were carried out to validate cognitive enhancing properties of A. dubius. Phytochemical screening was conducted to reveal phytoconstituents of A. dubius extract that could mediate the cognitive effects.
During the study, all mice except normal control were treated with ketamine to induce cognitive dysfunction. Ketamine is non-competitively blocks N-methyl-D-aspartate receptor through which it selectively induces analgesic effects.39,40 Long-term use of ketamine causes severe cognitive harm including learning and memory loss. 41 Cognitive damage by ketamine has partly been associated with its effects on major neurochemicals such as acetylcholine, resulting in decreased cholinergic firing. 42 Further, ketamine causes severe oxidative damage to the brain. 43 It is also established that ketamine induces severe hippocampal-associated memory deficits. 44 It has been established that transient tau hyperphosphorylation is induced following clinical ketamine administration. 45 Collectively, these mechanisms result in cognitive decline in both humans and rodents. 41 Findings of the present study correlates with previous reports demonstrating efficacy of ketamine to induce cognitive impairment in rodent models.44,46
Effect of A. dubius in enhancing cognition was evaluated using passive avoid task (PAT). Previously, PAT has been found to be an adequate preclinical correlate applied to determine outcomes induced by pharmacological substances on learning capabilities and memory efficiency in rodents. 47 PAT is carried out using a passive avoidance box (PAB) containing a dark and lit chamber. Hence, it was adopted for the present study. Rodents are known to naturally prefer dark environments. 48 However, the dark chamber was fitted with aversive stimuli (electric shock). 30 During the training phase, mice developed memory on unfavorable stimuli in the dark chamber and resisted to move to the dark compartment recording high step through latency. However, ketamine treatment induced loss of memory acquired during training and the negative control mice predominantly revealed short step-through latency into the electrocuting dark chamber. Extract of A. dubius was seen to reverse this trend by increasing step through latency in ketamine treated mice demonstrating potential to improve cognitive functionality.
During the passive avoidance test, it was noted that the extracts enhanced cognition in a non-dose dependent trend. This could be attributed to irregular drug absorption rates associated with oral root of drug administration used on this study. It has been documented that oral route of drug administration could cause individual variability in drug absorption due to differing gastrointestinal pH, volume and emptying, 49 among others
This study evaluated the likely underlying avenues through which aqueous A. dubius leaf extract ameliorated ketamine induced cognitive dysfunction. It was postulated that the extract could have enhanced cognition by decreasing AChE activity, a major hallmark implicated in AD pathogenesis. 50 Indeed, majority of conventional therapeutics used to manage AD inhibit AChE activity which achieves activation of cholinergic system and positively improving AD symptoms. 51 Hence, inhibitors of AChE are considered as promising candidates for development of effective therapeutics to alleviate cognitive symptoms manifested in AD. 52 In the present study, brains of mice treated with ketamine exhibited elevated AChE activity compared with the normal control mice. Remarkably, aqueous leaf extract of A. dubius significantly decreased AChE efficacy compared with the negative control mice, demonstrating anti-AChE properties of the extract. Therefore, the ability of the studied extract to relieve cognitive damage mediated by ketamine could be accredited to its ability to attenuate the rate of acetylcholinesterase enzyme activity. The observations of the present study are in tandem with previous reports that have noted many therapeutic plants exert cognitive improvement through inhibition of acetylcholinesterase activity.53,54
Similarly, it was noted that anticholinesterase activity of A. dubius was non-dose dependent. This could be attributed to varying absorption rates of the orally administered extracts. 49
Another likely mechanism leading to restoration of cognitive function by A. dubius extract could involve restoration of oxidative homeostasis. The brain being highly metabolically active, consuming approximately 20% of body oxygen, generates huge loads of oxidants.43,55 Ultimately, this exposes the brain to severe oxidative stress, triggering cognitive instabilities by damage of neuronal cells and their associated biomolecules (DNA, RNA, lipids and proteins). 56 It is, thus, a major contributor to pathophysiology of neurodegenerative disorders, including AD. 57 Therefore, antioxidant substances are a promising target for reducing rate of AD development and progression. 58 In this study, ketamine treated mice exhibited increased oxidative stress in their brains. On the other hand, A. dubius extract reduced oxidative stress in cognitively damaged mice. This was deduced by low MDA amounts in extract treated mice brains compared with negative control mice. Malondialdehyde is an established biomarker of lipid peroxidation which occurs under oxidative assault. 59 Thus, reduced levels of MDA in ketamine treated mice after extract administration demonstrated the potential of the extract to alleviate oxidative assault in the brain of AD-like mice models, proving an effective antioxidant agent. Similar mechanism of numerous antioxidant agents has been reported elsewhere.27,60
Furthermore, phytochemical characterization of A. dubius extract was performed to identify secondary metabolites that would be correlated with its cognitive enhancing effect. The extract contained numerous flavonoids, phenols, fatty acids and phytosterols, whose neuropharmacological efficacies are extensively documented.61,62 These phytocompounds may have alleviated cognitive deficits noted in the AD-like mice models resulting in the positive outcomes.
For instance, gallic acid has been found to inhibit cognitive decline via antiradical activity, 63 reduction of amyloid βeta 1–42 protein aggregates 64 and inhibition of neuroinflammation 65 in mice brain tissues. Vanillic acid has been revealed to restore cognitive integrity by inhibiting oxidative damage, 66 anti-inflammatory and anti-AChE activities. 67 Ellagic acid has been demonstrated to have potent antioxidant, anti-AChE and antiinflammatory effects. 68
Hydroxycinnamic acid as well as its derivatives, ferulic and p-coumaric acid, have demonstrated anti-lipid peroxidation 69 and anti-AChE activities in vitro. 70 Furthermore, hydroxycinnamic acid has been reported to inhibit Aβ40 fibrillation and reduce Aβ40 activated neurotoxicity. 71
Quercetin has anti-acetylcholinesterase, 72 anti-lipid peroxidation activities, 73 reduces tauopathy and β-amyloidosis. 74 Catechins, blocks formation and aggregation of Aβ fibril, lipid peroxidation and oxidation of protein. 75
Luteolin ameliorates cognitive dysfunction by alleviating oxidative stress and neuroinflammation. 76 Naringenin is known to reverse cognitive deficiency in rats by inhibiting acetylcholinesterase activity, tau-excessive phosphorylation, Aβ generation, neuroinflammation and enhancing endogenous antioxidant defensive mechanism. 77 Apigenin remedies cognitive dysfunction through epigenetic modulation and anti-inflammatory activity. 78
Further, β-sitosterol, campesterol and stigmasterol have been found to inhibit Aβ deposition, oxidative assault and acetylcholinesterase activities, 79 neuroinflammation, 80 all of which offer neural protection enhancing cognitive integrity. More so, octadecanoic and oleic acids have been found to inhibit acetylcholinesterase activity 81 and oxidative stress. 82 Evidently, the secondary metabolites present in A. dubius exerted diverse neuropharmacological mechanisms. Thus, it is conceivable that the therapeutic cognitive enhancing outcomes shown by A. dubius were achieved through the diverse mechanisms mediated by diverse secondary metabolites in the extract.
It worth to underlined that AD is multifactorial disorder caused by diverse etiological pathways. 83 Consequently, futuristic therapeutic approaches are focusing on development of therapeutic cocktails and drugs that simultaneous target different disease mechanisms in AD. 83 Aqueous A. dubius leaf extract demonstrated such multifunctional neuroprotective efficacy by simultaneously inhibiting both oxidative stress and AChE activity. Furthermore, numerous neuroprotective mechanisms of the secondary metabolites contained in the extract were highlighted in present study. This way, it's discernible that the promising cognitive enhancing effects of A. dubius were as results of synergistic activity of the diverse pharmacologically active phytochemicals present in extract.
Conclusions
In a nutshell, this study provides empirical evidence for therapeutic efficacy of aqueous A. dubius leaf extract to reverse cognitive dysfunction through inhibition of oxidative stress and acetylcholinesterase activity. Besides, anti-acetylcholinesterase and attenuation of oxidative damage, the study concludes that cognitive enhancing efficacy of A. dubius may have occurred through diverse mechanisms associated with its phytochemical principals. The study revealed therapeutic value of A. dubius for alleviating AD-like memory impairments. Remarkably, these findings present A. dubius as an ideal candidate that could provide several leads to develop potential novel agents for management of AD.
Footnotes
Acknowledgements
The authors would like to acknowledge Mr Daniel Gitonga of Kenyatta University for his technical support.
Data Availability
To access the data for this study, contact the corresponding author.
Authors Contributions
All the authors contributed equally to the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
National Commission for Science, Technology and Innovation (NACOSTI) provided approval to conduct the study (Approval number: NACOSTI/P/22/17588)
