Abstract
This study sought to investigate the anticholinesterase and antioxidative properties of quercetin and its glycosylated conjugate, rutin. The in vitro inhibition of acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) activities, inhibition of Fe2+-induced lipid peroxidation in rat’s brain homogenates, radicals scavenging, and Fe2+-chelating abilities of the flavonoids were investigated in vitro with concentrations of the samples ranging from 0.06 to 0.6 mM. Quercetin had significantly higher AChE and BChE inhibitory abilities than rutin. Quercetin also had stronger inhibition of Fe2+-induced lipid peroxidation in rat’s brain homogenates. Similarly, quercetin had higher radical scavenging abilities than rutin. Quercetin also had stronger Fe2+-chelating ability than rutin. The inhibition of cholinesterases and antioxidative properties are possible mechanisms by which the flavonoids can be used in the management of oxidative stress–induced neurodegeneration.
Several studies have linked the neuroprotective effects of phenolic compounds to their antioxidative 1,2 and anticholinesterase properties. 3,4 Antioxidants, such as flavonoids, can be effective in the management of neurodegenerative conditions as oxidative damage plays a crucial role in the development of such conditions. 5 The high use of oxygen by the central nervous system makes the tissues more susceptible to oxidative damage. 6,7 Furthermore, studies have shown that high amounts of lipid peroxidation products such as malondialdehyde (MDA) and hydroxynonenal are present in the brains of Alzheimer’s disease patients. 8,9 The inhibition of acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) enzyme activities is a modern therapeutic approach to the management of neurodegenerative conditions such as Alzheimer’s disease. 4 The inhibition of these enzymes leads to increased communication between nerve endings and increased activities in the cholinergic pathways in the brain, thereby alleviating the symptoms of Alzheimer’s disease. 10 BChEs are believed to play an important role in the onset of neurodegenerative diseases. 11 In the human brain, BChE appears to play a major role in the transformation of harmless amyloid plaques to pathogenic structures present in dementia and neurodegenerative diseases. BChE may also have an important role in breaking down AChE present in the extracellular portion of the brain. 12
Anticholinesterase drugs such as tacrine, caproctamine, and memantine, which are currently available for the treatment of Alzheimer’s disease, have side effects such as hepatotoxicity, gastrointestinal disturbance, dizziness, diarrhea, vomiting, and nausea, and this has led to the search for molecules from natural sources with fewer side effects. 13 Quercetin and rutin are abundant in nature and found in the edible portions of leafy vegetables, spices, herbs, legumes, tea, tubers, bulbs, and fruits. 14 However, like other flavonoids, quercetin usually occurs more in its glycosylated form in plants. 15 Albeit the anticholinesterase properties of flavonoids have been investigated, there is dearth of information on the effect of such glycosylation on the mechanisms by which flavonoids manage/prevent neurodegenerative conditions. This study therefore sought to compare the anticholinesterase properties and inhibition of lipid peroxidation in rat’s brain homogenates by quercetin and its glycoside (rutin).
Materials and Methods
Chemicals and Reagents
The chemicals quercetin, rutin, thiobarbituric acid (TBA), ABTS (2,2-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid), acetylthiocholine iodide, and butyrylthiocholine iodide were purchased from Sigma-Aldrich, Chemie GmH (Steinheim, Germany); acetic acid was procured from BDH Chemical Ltd (Poole, England). Except otherwise stated, all other chemicals and reagents were of analytical grade, while water was glass distilled.
Preparation of Flavonoid Stock Solutions
Stock solutions of the flavonoids (0.1 M) were prepared by dissolving in dimethyl sulfoxide (DMSO) and stored at 4°C for a maximum of 6 days. The stock solutions were then diluted with aqueous buffers depending on the individual assays. DMSO was used as negative control for all the assays.
In Vitro Experiments
Acetylcholinesterase and Butyrylcholinesterase Inhibition Assay
Inhibition of AChE was assessed by a modified colorimetric method. 16 The AChE activity was determined in a reaction mixture containing 200 μL of brain AChE solution (EC 3.1.1.7) in 0.1 M phosphate buffer, pH 8.0, 100 μL of a 3.3 mM solution of 5,5′-dithio-bis(2-nitrobenzoic) acid (DTNB) in 0.1 M phosphate buffered solution, pH 7.0, containing NaHCO3 (6 mM), test compound (0.1-0.4 mM), and 500 μL of phosphate buffer, pH 8.0. After incubation for 20 minutes at 25°C, acetylthiocholine iodide (100 μL of 0.05 mM water solution) was added as the substrate, and AChE activity was determined by ultraviolet spectrophotometry from the absorbance changes at 412 nm for 3.0 minutes at 25°C. Butyrylthiocholine iodide (100 μL) was used as a substrate to assay BChE enzyme (EC 3.1.1.8), while all the other reagents and conditions were the same. The AChE and BChE inhibitory activities were expressed as percentage inhibition.
Nitric Oxide Scavenging Activity
Nitric oxide scavenging assay was performed using the method reported by Susanta et al, 17 wherein 0.3 mL of sodium nitroprusside (5 mM) was added to 1 mL each of various concentrations (0-0.44 mM) of the flavonoids. The test tubes were then incubated at 25°C for 150 minutes. After 150 minutes, 0.5 mL of Griess reagent (equal volume of 1% sulfanilamide on 5% ortho-phosphoric acid and 0.01% naphthylethylenediamine in distilled water, used after 12 hours of preparation) were added. The absorbance was measured at 546 nm.
Hydroxyl Radical Scavenging Activity
The method of Halliwell et al, 18 was used to know the ability of the flavonoids to prevent Fe2+/H2O2 induced decomposition of deoxyribose. The flavonoids (0.15-0.6 mM) were added to a reaction mixture containing 120 μL of 20 mM deoxyribose, 400 μL of 0.1 M phosphate buffer, 40 μL of 500 μM FeSO4, and the volume were made up to 800 μL with distilled water. The reaction mixture was incubated at 37°C for 30 minutes and the reaction was then stopped by the addition of 0.5 mL of 28% trichloroacetic acid. This was followed by addition of 0.4 mL of 0.6% thiobarbituric acid solution. The tubes were subsequently incubated in boiling water for 20 minutes. The absorbance was measured at 532 nm in a spectrophotometer.
Fe2+ Chelation Assay
The Fe2+ chelating ability of both flavonoids were determined using a modified method of Minotti and Aust. 19 Freshly prepared 500 μM FeSO4 (150 μL) was added to a reaction mixture containing 168 μL 0.1 M Tris–HCl (pH 7.4), 218 μL saline, and the flavonoids (0-0.32 mM). The reaction mixture was incubated for 5 minutes before the addition of 13 μL 0.25% 1,10-phenanthroline (w/v). The absorbance was subsequently measured at 510 nm in a spectrophotometer. The Fe (II) chelating ability was subsequently calculated.
Lipid Peroxidation Assay Using Rats’ Brain Homogenates
Experimental animals
Twenty male Wistar strain rats weighing between 190 and 250 g were purchased from the Central Animal House, Department of Biochemistry, University of Ilorin, South West, Nigeria. They were housed in stainless steel cages under controlled conditions of a 12 hour light/dark cycle, 50% humidity, and 28°C temperature. The rats were allowed access to food and water ad libitum. The handling and use of the experimental animals was as approved by the Animal Ethics Committee of the School of Sciences, Federal University of Technology, Akure, Nigeria, with protocol reference SOS/14/02, and was in accordance with the NIH guide for the use and handling of experimental animals.
Preparation of brain homogenates
The rats were decapitated under mild diethyl ether anesthesia and their brains were rapidly isolated and placed on ice and weighed. These tissues were subsequently homogenized in cold saline (1/10 w/v) with about 10 up-and-down strokes at approximately 1200 rev/min in a Teflon glass homogenizer. The homogenate was centrifuged for 10 minutes at 3000 g to yield a pellet that was discarded, and a low-speed supernatant (S1) was kept for lipid peroxidation assay. 20
Lipid peroxidation and thiobarbibutric acid reactions
The lipid peroxidation assay was carried out using the modified method of Ohkawa et al. 21 Briefly, 100 μL S1 fraction was mixed with a reaction mixture containing 30 μL of 0.1 M pH 7.4 Tris–HCl buffer, test compound solution (0.11-0.44 mM), and 30 μL of 250 μM freshly prepared FeSO4. The volume was made up to 300 μL with water before incubation at 37°C for 1 hour. The color reaction was developed by adding 300 μL 8.1% SDS (sodium dodecyl sulfate) to the reaction mixture containing S1, which was subsequently followed by the addition of 600 μL of acetic acid/HCl (pH 3.4) mixture and 600 μL 0.8% thiobarbituric acid. This mixture was incubated at 100°C for 1 hour. Thiobarbituric acid reactive species produced were measured at 532 nm and the absorbance was compared with that of standard curve using MDA.
Data Analysis
The result of replicate experiments (n = 6) were pooled and expressed as mean ± standard deviation (SD). The means were analyzed using one-way analysis of variance (ANOVA), and Duncan test was used for the post hoc treatment. Significance was accepted at P < .05.
Results
The dose-dependent inhibitions of AChE and BChE activities by quercetin and rutin are presented in Figures 1 and 2, respectively, and the IC50 values in Table 1. Quercetin had significantly (P < .05) higher AChE (IC50 = 0.181 mM) and BChE (IC50 = 0.203 mM) inhibitory abilities than rutin (AChE [IC50 = 0.219 mM]; BChE [IC50 = 0.288 mM]). Similarly, Figure 3 reveals that quercetin (IC50 = 0.322 mM) had significantly (P < 0.05) stronger inhibition of Fe2+-induced MDA production in rat’s brain homogenates than rutin (IC50 = 0.381 mM). Furthermore, Figures 4 to 6 show that quercetin also had significantly (P < 0.05) higher radical (DPPH [IC50 = 0.799 mM]; NO [IC50 = 0.374 mM]; OH [IC50 = 0.284 mM]) scavenging abilities than rutin (DPPH [IC50 = 0.938 mM]; NO [IC50 = 0.420 mM]; OH [IC50 = 0.344 mM]). The Fe2+ chelating abilities of the flavonoids (Figure 7) revealed that quercetin (IC50 = 0.164 mM) had higher chelating ability than rutin (IC50 = 0.184 mM).

Inhibition of acetylcholinesterase activity by quercetin and rutin.

Inhibition of butyrylcholinesterase activity by quercetin and rutin.
IC50 Values for Inhibition of Acetylcholinesterase and Butyrylcholinesterase Activities and Fe2+-Induced MDA Production in Rats Brain Homogenates, DPPH•, NO•, OH• Scavenging, and Fe2+ Chelating Abilities of Quercetin and Rutin (mM)*.
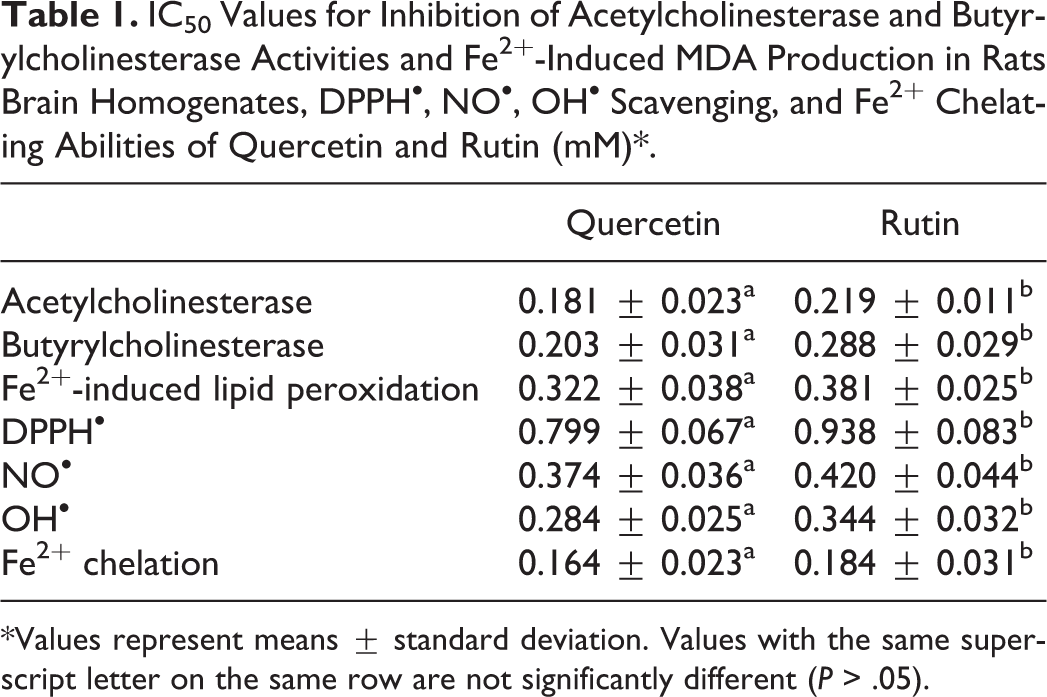
*Values represent means ± standard deviation. Values with the same superscript letter on the same row are not significantly different (P > .05).

Inhibition of Fe2+-induced lipid peroxidation by quercetin and rutin.

DPPH• scavenging ability of quercetin and rutin.

NO• scavenging activity of quercetin and rutin.
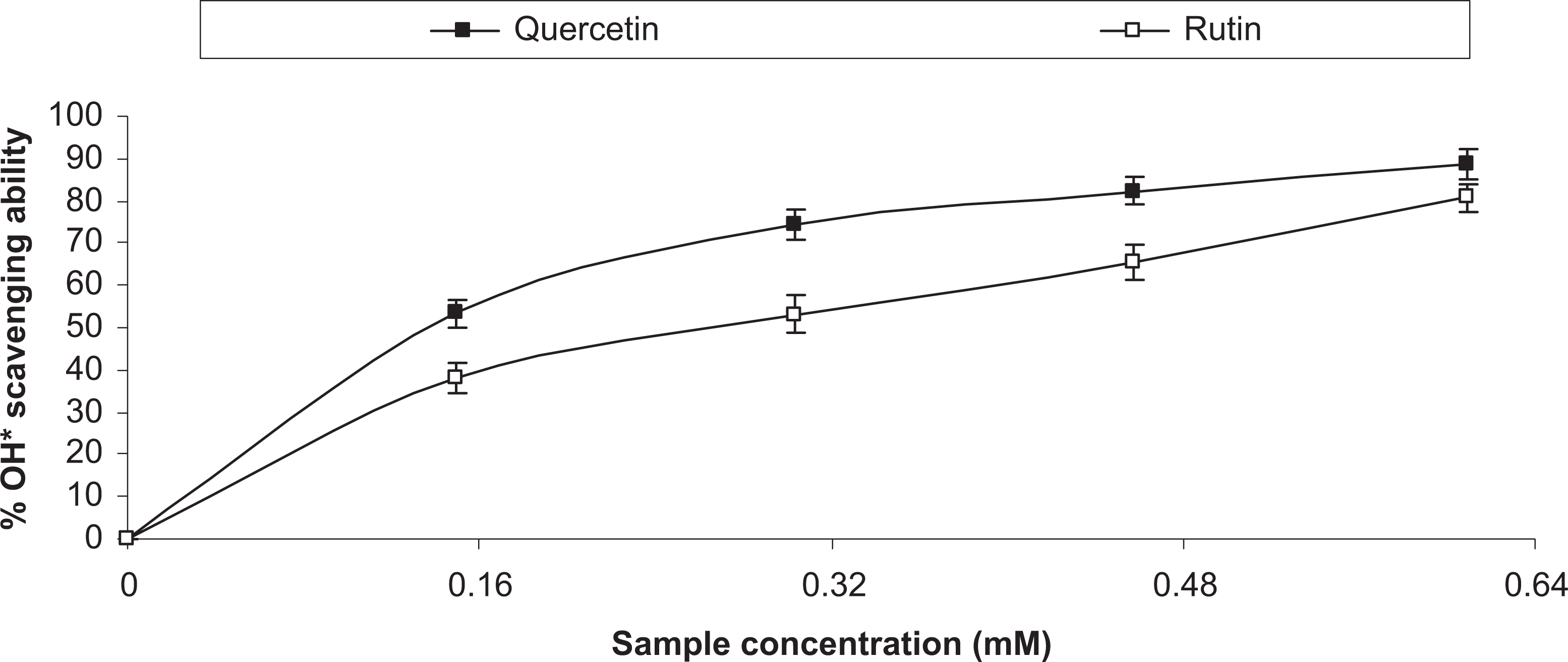
OH• scavenging activity of quercetin and rutin.

Fe2+-chelating ability of quercetin and rutin.
Discussion
The inhibition of AChE and BChE enzyme activities has been broadly established as a first-line treatment/management for the symptoms of neurodegenerative conditions, such as Alzheimer’s disease. 22 The results of our research revealed that both flavonoid compounds inhibited AChE activity in a concentration-dependent manner, with quercetin exhibiting a slightly higher inhibitory ability when compared with rutin as revealed by their IC50 values. Our result is in agreement with that of other researchers who showed the AChE inhibitory ability of quercetin 4 and a number of other polyphenols from various plant sources. 23
The results of our research revealed that both flavonoid compounds inhibited BChE activity in a concentration-dependent manner, with quercetin exhibiting a higher inhibitory ability when compared with rutin as revealed by their IC50 values. Research work has linked the ability of the flavonoids to inhibit BChE potently to the presence or absence of –OH groups on the side phenyl ring of the compounds. 24 The hydroxyl groups of flavonoids form hydrogen bonds with important amino acid residues at the active site of the enzyme. 25
It has been observed that brains of Alzheimer’s disease patients show several signs of oxidative stress such as lipid peroxidation, protein oxidation, and damage to mitochondrial and nuclear DNA. 26 The gradual accumulation of Cu2+ and Fe2+ in the plaques of the brain has been linked to oxidative stress, which is important to Alzheimer’s disease pathology. 27 Also, metal ion levels in Alzheimer’s disease individuals have been observed to be much higher than those in healthy people. 28 Quercetin and rutin exhibited several antioxidant properties as typified by their radicals’ (DPPH, OH, and NO) scavenging ability, Fe2+ chelation, and inhibition of Fe2+-induced lipid peroxidation. In all of these antioxidant assays, quercetin showed a greater ability than rutin as exemplified by the IC50 values. The antioxidative properties of the flavonoids could enhance the ability of the 2 molecules to serve as therapeutic molecules in Alzheimer’s disease management. Some researchers have suggested that due to the multifactorial nature of Alzheimer’s disease, it is important to get molecules that not only modulate the activity of the cholinesterase enzymes but also exhibit other biological activities, such as antioxidant activities as well. It is believed that these would lead to a more effective management of the disease. 28 Furthermore, it should be noted that the bioactivity of the flavonoids reported in this in vitro study might not translate to direct in vivo effect due to the low bioavailability of flavonoids. 29
Conclusion
This study revealed that quercetin had a stronger inhibition of key enzymes linked to neurodegenerative conditions (AChE and BChE) and antioxidant properties when compared with its glycoside (rutin). However, more work still needs to be done on the structure–activity relationships to establish, authoritatively, the functional groups responsible for these abilities.
Footnotes
Authors Contributions
AOA participated in the design of the research, conducted the experiment, and drafted the article. GO participated in the design of the experiment, supervised the experiment, and provided mentorship support. FB participated in the design of the experiment and conducted the experiment. POA participated in the design of the research and drafted the article. All authors read and approved the final version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The handling and use of the experimental animals was as approved by the Animal Ethics Committee of the School of Sciences, Federal University of Technology, Akure, Nigeria, with protocol reference SOS/14/02, and was in accordance with the NIH guide for the use and handling of experimental animals.
