Abstract
The purpose of this study was to show that some elements have a proven antioxidative effect and are essential for the proper development and functioning of the human body. The study also assessed the concentration of selected elements and total antioxidant status (TAS) in a group of male inhabitants of Białystok, Poland, who are professionally active in the metal industry. The study group comprised 163 men aged 55–64 years. The concentration of the analyzed elements was determined using flame (Zn and Cu) and electrothermal (Se, Cd, and Pb) atomic absorption spectrometry. Spectrophotometric test kits were used to assay the TAS and glutathione reductase (GR) activity in serum. The results suggested the mean concentration of Se in serum was 54.04 ± 12.10 μg/L, which was below the reference range. A significant negative correlation was observed between the concentration of Se in the serum and Pb and Cd concentrations in the whole blood of the studied men. The concentrations of the other elements, antioxidant potential, and GR activity were within the reference range. A statistically significant positive correlation was observed between Pb and Cd, indicating combined exposure and a considerable negative correlation between lead and selenium as well as between lead and TAS. Selenium and TAS are likely to modify the adverse effects of Pb in the bodies of the studied men. The results indicate that it is necessary to constantly monitor men who are occupationally exposed to heavy metals, maintain a healthy lifestyle, and introduce effective preventive measures at a local level.
Elements such as zinc (Zn) and copper (Cu) still arouse a great deal of controversy. While these elements have a proven antioxidative effect and are essential for the proper development and functioning of the human body, there are also reports of their increasing involvement in soil contamination, similar to heavy metals such as cadmium and lead. The largest threats to human health and the natural environment include coal and waste combustion, metal-bearing dust used for fertilizing sludge, and so forth (Zaborowska, Wyszkowska, & Kucharski, 2005). Zinc is a microelement that demonstrates antioxidative effects, and its function in the human body depends on the amount ingested via food. In the human body, zinc functions as a regulator—a structural, catalytic, and regulatory cofactor for over 300 enzymes involved in the synthesis and degradation of carbohydrates, lipids, and proteins (McCall, Huang, & Fierke, 2000). Small doses of zinc are essential for the proper functioning of the human body, yet high concentrations may have an adverse, toxic effect. Alternatively, zinc deficiency contributes to Alzheimer’s disease and infertility (Leitzmann et al., 2003; Pizent, Jurasović, & Telišman, 2003; Plum, Rink, & Haase, 2010; Samman & Roberts, 1987).
Copper is a component of numerous antioxidant enzymes that are involved in oxygen metabolism and is involved in forming cross-links in collagen and elastin, synthesizing skin and hair pigment, and is also necessary for the proper functioning of the nervous system. Excessive dietary copper intake results in various metabolic changes whose prolonged adverse effects mainly include liver changes, followed by renal failure, brain tissue damage, and coronary artery and heart muscle abnormalities. Copper deficiencies occur mainly in patients with parenteral nutrition who suffer from various metabolic disorders, malabsorption, and dietary intake shortages (Goodman, Mistry, Pasha, & Bosch, 2009). Clinical Cu deficiencies occur rarely in humans, yet significant deficiencies may lead to connective tissue abnormalities, anemia, central nervous system disorders, glucose and cholesterol metabolism disorders, and increased susceptibility to infections (National Institutes of Health, 2018; Puzanowska-Tarasiewicz, Kuźmicka, & Tarasiewicz, 2009).
Selenium (Se) is considered to be an essential element in the proper functioning and development of the human body (Zachara et al., 2001; Zwolak & Zaporowska, 2005), but Se has a toxic effect. Deficient or excessive Se causes numerous disorders, and the threshold between necessary and toxic amounts is very narrow (Rayman, 2000). In terms of selenium soil content, 77% of Poland, including Podlaskie Voivodeship, shows endemic diseases such as hyposelenosis (Haug, Graham, Christophersen, & Lyons, 2007; Wąsowicz, Gromadzińska, Rydzyński, & Tomczak, 2003; Zachara et al., 2001).
The synergistic relation between selenium and iodine is the cause of increased abnormalities in thyroid gland functions, especially when these two microelements are deficient. Moreover, selenium has affinity with hematopoietic elements, that is, Zn, Cu, Fe, and Co (Czeczot, 2001; Fairweather-Tait et al., 2011; Kornhauser et al., 2008; Tinggi, 2008).
Cadmium (Cd) and lead (Pb) are toxic elements, which when ingested in food and drinking water, accumulate in blood and bones as well as organs such as the liver, kidneys, and brain (Krzywy, Krzywy, Pastuszak-Gabinowska, & Brodkiewicz, 2010). These metals are capable of displacing essential microelements from metalloenzymes. Their interactions with Zn, Cu, and Se, all of which are elements that are vital to the human body, result in serious morphological and functional changes in some organs. Excretion or elimination of cadmium by living organisms is difficult and is it is only extracted in small amounts in urine and/or bile. The biological half-life of Cd in the human body is 16–30 years (Krzywy, Krzywy, Peregud-Pogorzelski, Łuksza, & Brodkiewicz, 2011), and the half-life of Pb in the body ranges from 30 days to 27 years (Krzywy et al., 2010, 2011).
The aim of the study was to assess the concentration of selected elements and total antioxidant status (TAS) in a group of male inhabitants of Białystok who are professionally active in the metal industry.
Methods
Data Collection
The study group consisted of 163 men employed in a plant in Białystok in the year 2008 who formerly worked in the metal industry. The men’s age ranged from 55 to 64 years (average age: 59.6 ± 2.5 years).
The test material was venal blood collected in accordance with procedures for trace element assays. The blood for Se, Zn, Cu, TAS, and GR assays was collected in Vacutest vacuum test tubes with a separating gel and coagulation accelerator to produce serum; the blood for Pb and Cd assays was collected in Vacutest vacuum test tubes with EDTA-K2 coagulant.
All of the tests conducted in this study were approved by the Bioethics Commission of (no. 124-03565P); the patients agreed to participate in the study (written and verbal) and were informed that they could drop out at any stage.
The concentrations of Zn and Cu were assayed using flame atomic absorption spectroscopy (air-acetylene) in a Hitachi Z-5000 spectrophotometer. The concentration was determined based on the calibration curve set by the spectrophotometer for each element. Four calibrations solutions were used to assay Cu in the following concentrations: 0, 1, 2, and 3 mg Cu/L. The calibration curve for Zn was established based on 0, 0.5, 1.0, and 1.5 mg Zn/L concentration standard solutions.
The concentration of Se, Cd, and Pb was assayed using electrothermal atomic absorption spectrometry with argon in a Hitachi Z-5000 apparatus. The concentration was determined based on the calibration curve set by the spectrophotometer for each element, using calibration solutions in the following concentrations: 40 μg/L for Pb, 60 μg/L for Se, and 4.0 μg/L for Cd.
The obtained values for the calibration solutions of all the elements as well as the tested samples are the mean of the three tests that were performed. The analysis of selected elements, TAS, and GR activity in the blood of the patients was assayed at the Department of Biochemical Diagnostics at the Medical University of Białystok.
Test kits were used to determine the TAS in blood serum in a ultraviolet-visible (UV-VIS) spectrophotometer (Total Antioxidant Status [TAS] by Randox Laboratories Ltd.). The accuracy of the assay was ensured using a Randox Total Antioxidant Control assay kit by Randox Laboratories Ltd. (https://www.randox.com/ [assay kits]; Miller, Rice-Evans, Davies, Gopinathan, & Milner, 1993).
Ready-made test kits were employed to assay the activity of GR in blood serum in a UV-VIS spectrophotometer (Glutathione Reductase [GR] by Randox Laboratories Ltd.). Accuracy and repeatability were ensured using Randox Glutathione Reductase Control Sera GR 2608 by Randox Laboratories Ltd. (https://www.randox.com/ [assay kits]), a control serum for GR.
Data Analysis
The assumed research scheme included a medical interview with each patient in a professional medical center, followed by an interview with a dietitian based on a nutritional and lifestyle questionnaire. The day after the interview, blood was taken from each patient for testing in a certified laboratory. The planned study of this group of men was carried out from mid-January to mid-April; the nutritional interview included once-a-day observation of the men’s lifestyle.
The patients filled out surveys on their consumption frequency of the following food groups: white bread; wholemeal bread; pastries (sweet rolls, cakes, chocolate); flour-based meals (noodles, pastas, pancakes); grains, rice, and cereals; milk and dairy drinks; curd cheeses; aged and processed rennet cheeses; meat; poultry; sausages; quality smoked meats; cured meat products; pork belly, lard, and pork fat; meat preserves; fish preserves; fresh fish; eggs; butter; margarine; oils; potatoes; vegetables; fruit; fruit juices and beverages; jam; honey and sugar in beverages; products made from pulses; beer; wine; vodka; tea; and coffee.
During the study, a 24-hour dietary intake survey was conducted, which was preceded by doctor consultations, although the available Polish nutrient program did not include assessment of all of the same analyzed elements in the patients’ diets, that is, TAS, GR, Pb, and Cd.
The survey questionnaires included questions on lifestyle, such as smoking tobacco, alcohol consumption, and physical activity. The statistical analysis did not demonstrate any significant correlation between the concentration of evaluated elements or antioxidant potential and the lifestyle of the studied group of men.
In accordance with the principals of statistical analysis, the obtained results were processed in Statistica v.13.1 (by StatSoft) software suite, using a Student’s t test and multiple stepwise forward regressions to compare the effects of dietary habits on the concentration of the assayed elements, GR activity, and TAS. Pearson correlation coefficients (r) between particular properties in the analyzed groups were determined. The borderline statistical significance level was p < .05.
Results
Table 1 presents the average concentrations of the analyzed elements and the antioxidant potential in the blood of the men working in the metal industry.
The Average Concentrations of the Analyzed Elements and the Antioxidant Potential in the Blood of the Men Working in the Metal Industry (n = 163).

Note. X = mean; SD = standard deviation; Me = median; Min = minimum; Max = maximum.
Neumeister & Böhm, 2015. bPearson linear correlation coefficient.
Table 2 presents the reference ranges of the Zn, Cu, Se, Pb, Cd, TAS, and GR activity, and the number of subjects classified as above, below, or within the reference range.
The Reference Ranges of the Zn, Cu, Se, Pb, Cd, TAS, and GR Activity, and the Number of Subjects Classified as Above, Below, or Within the Reference Range (n = 163).

Note. GR = glutathione reductase; TAS = total antioxidant status.
Table 3 presents multiple regression analysis of the effects of food consumption frequency on the concentrations in serum and blood of selected elements and antioxidant potential in the studied group.
Multiple Regression Analysis of the Effects of Food Consumption Frequency on the Concentrations in Serum and Blood of Selected Elements and Antioxidant Potential in the Studied Group.

Note. GR = glutathione reductase; TAS = total antioxidant status; β factor = standard error; p = significance level; R2 = multiple regression.
Table 4 presents correlation between some elements from dietary intake and concentration of Zn, Cu, Se, TAS, and GR activity in serum and concentration of Pb and Cd in whole blood in the studied group.
Correlation Between Some Elements From Dietary Intake and Concentration of Zn, Cu, Se, TAS, GR Activity in Serum, and of Pb and Cd in Blood in the Studied Group (n = 163).
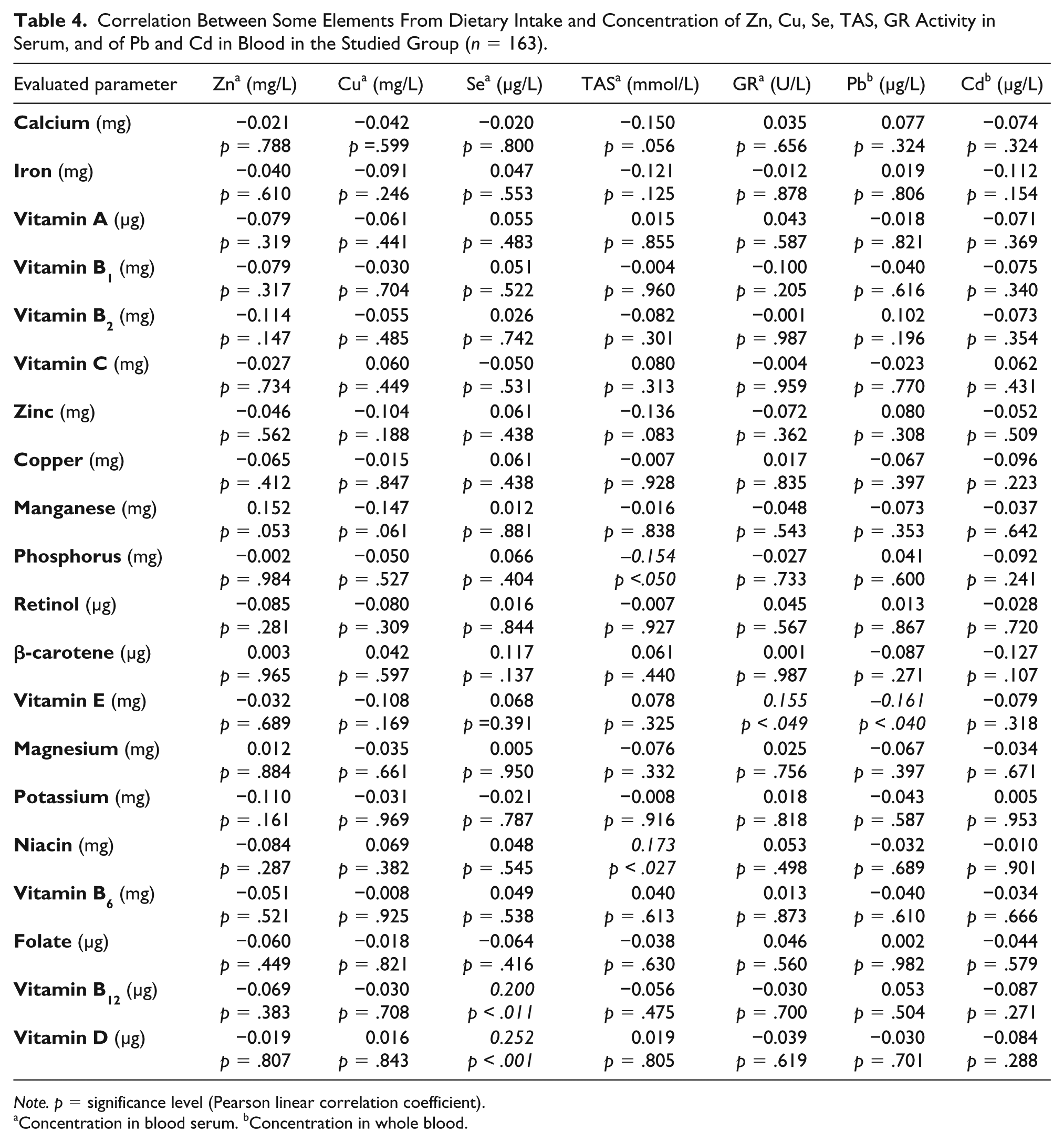
Note. p = significance level (Pearson linear correlation coefficient).
Concentration in blood serum. bConcentration in whole blood.
Discussion
It is assumed that the normal concentration of Zn in human blood serum ranges from 0.6 to 1.2 mg/L (Neumeister & Böhm, 2015). In this study 46 samples exceeded the established reference level. A similar result was observed in the blood serum of 33 men in Zabrze (Kasperczyk, Ostałowska, Grucka-Mamczar, & Birkner, 2008). A younger group of 49 men in China demonstrated a slightly lower average concentration of Zn in blood serum (Wang, Du, & Zheng, 2008). A study performed on 120 fertile and 100 infertile men in Lebanon indicated a relationship between infertility and environmental and occupational exposure to heavy metals (Inhorn et al., 2008).
Zinc demonstrates a detoxifying effect against Cd and Pb because (similar to Cu and Fe) it competes with these elements for bonding sites. This antagonism is also observed between Zn and Cu as well as between Zn and calcium/magnesium (Ca/Mg; Puzanowska-Tarasiewicz et al., 2009). The main source of dietary Zn is wheat products, meat, and processed meat. Milk and dairy products contain dietary Zn, while vegetables and fruit contain less of this element (Rutkowska & Wojtasik, 2002; Szponar, Sekuła, & Rychlik, 2003). This study indicated that among all of the analyzed nutritional factors, the concentration of Zn in the blood serum of the studied group was also positively increased by an additional 14% by poultry, products from pulses, and eggs; it was negatively affected by cooking oil (Table 3). Zinc is better absorbed from products of animal origin than from plant-based products. The type of amount of protein in the diet also impacts Zn absorption, which is higher in diets rich in highly nutritional animal protein (Plum et al., 2010; Samman & Roberts, 1987). The presence of cysteine and histidine in animal protein increases the absorption of this microelement, while tryptophan reduces absorption. Selected amino acids, dietary citric acid, refined products, and simple sugars (such as glucose) improve Zn absorption. Improper stomach pH, the presence of bicarbonate ions, and selected heavy metals (Cd, Hg) significantly reduce Zn absorption (Zaborowska et al., 2005).
The reference range of Cu in blood is 0.8–1.2 mg/L (Neumeister & Böhm, 2015). Respectively, 19 and 18 subjects exceeded or were under the reference level. Wang et al. (2008) analyzed Chinese males, 49 of whom demonstrated lower blood Cu concentration (Wang et al., 2008). On the other hand, in their study of males in Lebanon, Inhorn et al. observed an average Cu concentration that was below the recommended reference level (Inhorn et al., 2008).
In this study, a statistically significant effect on Cu blood serum levels (11%) was observed in cases of frequent consumption of cured meats and butter (positive) and milk (negative; Table 3). Some elements have a negative effect on Cu absorption (i.e., Fe, Zn, Ca, and P); absorption is also inhibited by phytates, fiber, and sulfur compounds that occur in dietary products (Kozłowska, Mikołajczyk, Boroń, Kasperczyk, & Pawlas, 2015; Rutkowska & Wojtasik, 2002; Wang et al., 2008). The average dietary absorption of Cu is approximately 35%–50% and its absorption increases when there is a shortage of Cu in the body.
Selenium deficiency is harmful and disrupts the functioning of multiple systems in humans (Gać & Pawlas, 2011; Rayman, 2000). Se shortage occurs when its concentration drops below 85 μg/L (Hogberg & Alexander, 2007; Kozłowska et al., 2015), and the daily limit value is 450 μg/day. Over 900 μg/day toxic of Se is considered toxic and leads to selenosis (Kozłowska et al., 2015; Raisbeck, 2000). The concentration of Se in the healthy European population ranges from 38.71 to 432 μg/L (Kozłowska et al., 2015). The highest occupational exposure limit is 0.1 mg/m3, while the blood content limit is in the range 70–140 μg/L (Neumeister & Böhm, 2015). In this study, the average concentration of Se in the group of men working in the metal industry was below the reference range. A higher average was observed by Kozłowska et al. (2015), who studied a group of males working in Zn and Pb mills in Śląskie Voivodeship, Poland. The lower concentration of this element in this study may be the effect of low Se content in soil (Haug et al., 2007) and, consequently, low levels of Se in the diet of the population of Podlaskie. Additionally, this study indicated a negative correlation between Pb levels and Se concentration, similar to the findings of Kozłowska et al. (2015). It was identified that Se’s antioxidant action protects the body from heavy metals such as Cd, Pb, Hg, and Cd, thus reducing their toxic effects on the body (Hogberg & Alexander, 2007; Kozłowska et al., 2015). Similarly to Cd, Se is absorbed by erythrocytes and is therefore released and bound with proteins and carried away in the blood. Consequently, Se has a high affinity to Cd, with which it has a 1:1 binding ratio. Selenium forms a complex with Pb particles bound with erythrocytes, thus reducing the level of free Pb ions in blood serum (Kasperczyk, Prokopowicz, Dobrakowski, Pawlas, & Kasperczyk, 2012; Kozłowska et al., 2015). Moreover, Se levels that are too low in various populations lead to higher susceptibility to free oxygen species and, consequently, to the development of numerous disorders. Elevated concentration of heavy metals leads to decreased Se absorption (Gać & Pawlas, 2011; Kozłowska et al., 2015).
Analysis of the diets of this group of men reported that 13% of the serum Se concentration is determined by frequent consumption of meat and sugar, which significantly decreases the level of this microelement (Table 3). Most likely these food products are contaminated with heavy metals, that is, Pb, as seen by the negative correlation Pb versus Se (Table 1). By creating hardly soluble selenides, which are excluded from biochemical processes and are immediately expelled from the body, selenium compounds are responsible for the removal of toxic substances from the body, including heavy metals (Cd, Pb). Selenium facilitates the retention of Pb ions in the extracellular matrix of the liver and kidneys, where it binds with methionine and inhibits the absorption of Se in the body (Czeczot, 2001). Also, it is significant that the soil in Podlaskie is poor in Se, thus causing hyposelenosis in this area (Wąsowicz et al., 2003).
The reference range of TAS is 1.30–1.77 mmol/L. Significantly lower results in terms of TAS were observed in males working as roadside petrol dispensers (Adamu et al., 2018) as well as dislipidemic and hypertensive Nigerian males (1.02 ± 0.15 mmol/L; Ogunro et al., 2009). TAS values had a negative correlation with Pb concentration in the blood of the studied males. TAS in the diets of the studied males was positively affected by frequent consumption of eggs and honey, both of which contain antioxidant vitamins A, E, and D and active substances (enzymes and bacteriostatic agents), while increased milk consumption significantly reduced the antioxidant status (Tables 3 and 4).
The reference range for GR in blood is 33–73 U/L. This had a positive correlation with Cu concentration in blood serum. Mecdad et al. (Mecdad, Ahmed, ElHalwagy, & Afify, 2011) observed significantly lower GR values in their assessment of a group of 95 males working in agriculture who were exposed to pesticides and fungicides. Analysis of the data presented in Table 3 leads to the conclusion that the consumption of selected types of food had a 9% impact on GR dietary activity. A positive, statistically significant impact was demonstrated in frequent consumption of eggs and meat as these products are rich in Cu and B vitamins; a negative effect was observed in the case of milk consumption (Tables 3 and 4).
Currently, the biological limit value of Pb varies between 270 and 300 µg/L (Jakubowski, 2014; Neumeister & Böhm, 2015), and the blood Pb level limit is 700 µg/L. People who are occupationally exposed to lead should be under medical surveillance when their blood lead level is greater than 400 µg/L [Council directive 98/24/EC]. In this study, the average concentration of Pb in the group of men working in the metal industry was below the biological limit. A higher average was observed by Kozłowska et al. (2015), who studied a group of male employees of Zn and Pb mills in southern Poland (Sląskie Voivodeship). It must be noted that the two results did not vary significantly despite the difference in geographical location, where the region Podlaskie is considered to be the area in Poland with the least pollution. On the other hand, the studies conducted by Poręba et al. (2010) on 171 workers who were occupationally exposed to acute effects of heavy metals in mills in Legnica and Głogów indicated a 10 times higher concentration of Pb. Research conducted by Glenn et al. (2006) on 575 car battery plant employees in South Korea indicated an even higher value that affects systolic and diastolic blood pressure (Glenn et al., 2006). Similarly, Schwartz et al. (2005) conducted a study on 803 workers who were occupationally exposed to lead in South Korea; they demonstrated a significant relationship between manual dexterity and peripheral vibration threshold test scores as well as neurobehavioral test scores (Schwartz et al., 2005). This study indicated a positive correlation between Pb levels and Cd content, similar to the findings of Kozłowska et al. (2015).
The dietary levels of calcium (Ca) and phosphorus (P) affect reduced Pb absorption from the gastrointestinal tract. Ca and P dietary deficiencies increase Pb absorption by up to 63%. Conversely, the antioxidant effects of Zn reduced aromatic L-amino acid decarboxylase deficiency (ALAD) inhibition by Pb. Also, Fe deficiencies have observed effects on Pb absorption (Mecdad et al., 2011). Table 3 reported that frequent consumption of fish and canned fish preserves increased lead concentration in the blood in the studied group; this might be the consequence of the presence of this metal in the water bodies where the fish were bred.
The current biological limit value of blood cadmium is 5 μg/L according to the American Conference of Governmental Industrial Hygienists (ACGIH, 2010; https://www.acgih.org/, 2010. ACGIH) and 2.7 μg/L in blood according to Neumaister (Neumeister & Böhm, 2015). In this study on a group of males working in the metal industry, the concentration of Cd in the blood was below the biological limit. However, in the study conducted by Kozłowska et al. (2015) on males exposed to Cd, the concentration of the element was almost five times higher, which was in line with the results obtained by Poręba et al. (2010). A negative correlation was observed between blood Cd levels and Se concentration in serum, as has also been confirmed by other researchers studying occupational and environmental exposure in populations in the industrial region of Górny Śląsk, Poland (Dynerowicz-Bal et al., 2005; Jakubowski, 2012, 2014; Kozłowska et al., 2015).
The main dietary sources of Cd include the following: cereals, root vegetables, offal from adult animals, some shellfish, meat, and fish, and other seafood. In this study, it was observed that frequent consumption of pastries and products from pulses had significant effects on blood cadmium levels in males with occupational exposure (Table 3). Also, keeping food in enameled containers may lead to increased Cd levels, especially in the case of acidic liquids (Jakubowski, 2012).
Study Limitations
To confirm the results of the research, repeated observation of the same group of men should be carried out. In further research, a control group and groups with various disorders should be distinguished; this was not done in the current study due to a different research plan and financial limitations. The period in which the study was conducted presented that almost the entire study group of men had a deficiency in serum Se, which was the result of reduced consumption of, for example, Brazil nuts (limited or expensive imports) and eggs (poor availability on the market). The studied group of men were first educated in healthy lifestyle and nutrition 20 years ago by the same doctor who participated in the current study (Charkiewicz et al., 2018). This study reported that the group still has the same level of knowledge as before. The fact that these men do physical work and mostly only have lower education should also be taken into account.
Conclusion
Occupational exposure to Pb and Cd may reduce blood selenium levels in the studied group of males. A statistically significant positive correlation was observed between Pb and Cd, indicating mixed exposure and a considerable negative correlation between Pb and Se as well as between Pb and TAS. Selenium and TAS were likely to affect the adverse effects of Pb in the bodies of the studied men. Low Se concentrations may contribute to the development of lifestyle diseases, that is, ischemic heart disease, diabetes, stroke, and selected forms of cancer. The results indicate that it is necessary to constantly monitor men who are occupationally exposed to heavy metals in order to preserve a healthy lifestyle and introduce effective preventive measures on a local level.
The results obtained in this work provide new information on the impact of the frequency of consumption of particular types of food products, their antioxidant status, and concentration of heavy metals in blood on the nutritional status of the Polish working men. They can be used in practice to improve the nutritional status of men working in very difficult conditions to prevent the occurrence of broadly understood lifestyle diseases.
Footnotes
Author Contributions
BM has received consultation and/or lecture honoraria from Abbott, Roche, Cormay and Biameditek.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
