Abstract
Ischemic–reperfusion (I/R) injury to cerebral white matter during the perinatal period leads to long-term cognitive and motor disabilities in children. Immature white matter oligodendrocytes are especially vulnerable to metabolic insults such as those caused by hypoxic, ischemic, and reperfusion injury. Consequences include an impaired capacity of oligodendrocytes to generate and maintain mature lipid-rich myelin needed for efficient neuronal conductivity. Further research is needed to increase an understanding of the early, possibly reversible myelin-associated pathologies that accompany I/R white matter injury. This experiment characterized I/R time-dependent alterations in cerebral white matter lipid profiles in an established fetal sheep model. Fetal sheep (127 days gestation) were subjected to 30 min of bilateral carotid artery occlusion followed by 4 h (
Introduction
Hypoxic–ischemic injury to the cerebral hemispheres in the perinatal period is the leading cause of long-term neurocognitive and motor deficits in children 1 . Ischemic injury causes tissue necrosis as the resulting critical reductions in blood flow compromise oxygen and nutrient delivery, threatening cellular energy metabolism and viability. Subsequent reperfusion could potentially salvage the injured tissue by flooding it with oxygenated blood and nutrients, but also could further damage the brain due to disruption of the blood–brain barrier and attendant calcium dyshomeostasis, oxidative stress 2 , and mitochondrial dysfunction. The resulting increase in mitochondrial permeability drives excess production of reactive oxygen species (ROS) 3 , damaging lipids and proteins, activating astrocyte and microglial inflammatory responses 4 , and transducing pro-apoptosis cascades in neurons, glia, and vascular elements 2 . Repeated and sustained hypoxic–ischemic insults cause central white matter injury ranging from myelin loss to coagulative necrosis 4 . In premature neonates, white matter ischemic–reperfusion (I/R) injury can progress to periventricular leukomalacia 1 , particularly when the immature mediators of cerebral blood flow autoregulation sustain significant damage 5 and can no longer control reperfusion. A better understanding of the early stages of I/R injury linked to permanent structural and functional white matter pathologies could aid in the development of targeted neuroprotective approaches for preterm infants 4 .
The major components of white matter include (1) axons, which transmit signals across different brain regions; (2) oligodendrocytes, which produce and maintain myelin to facilitate neurotransmission; (3) astrocytes, which provide a supportive matrix; (4) microglia, which participate in immune surveillance and injury responses; and (5) blood vessels, which are needed to maintain cellular and tissue viability, supply nutrients, remove waste, and together with astrocytes, furnish the blood–brain barrier.
Oligodendrocytes are especially vulnerable to hypoxic, ischemic, and reperfusion injury. Dysfunction or loss of oligodendrocytes results in the degeneration of myelin in the central nervous system (CNS) and impairment of nerve impulse conductivity. Also, the loss of integrity of myelin renders exposed axons susceptible to ischemic, metabolic, and reperfusion injury. These complications are exacerbated in the developing brain because the immature oligodendrocytes exhibit particular sensitivity to I/R due to their high metabolic demands. Many critical functions of immature oligodendroglia, including the capacity to proliferate, migrate, and sustain myelin synthesis, maturation, and homeostasis, are compromised by oxidative stress, excitotoxicity, inflammation, and mitochondrial dysfunction. Although oligodendrocyte precursor cells (OPCs) proliferate in response to hypoxic–ischemic injury and could potentially replace lost oligodendrocytes, severe hypoxic–ischemic insults can compromise the maturation of OPCs, limiting their capacity to generate mature myelin to ensure axonal conductivity 6 . Therefore, therapeutic strategies to enhance survival and maturation of oligodendrocytes that have sustained significant hypoxic–ischemic–reperfusion injury could potentially facilitate axonal remyelination and restore neurotransmission 7 in order to prevent or reduce neurocognitive and motor deficits.
Myelin in the central nervous system has a high dry mass of lipids (70%–85%) compared to proteins (15%–30%). Major myelin lipids include cholesterol, glycosphingolipids, sulfatides (STs), gangliosides, phospholipids (PLs), and sphingomyelin (SM). Abnormal metabolism and expression of PLs and STs occur in many CNS diseases 8 -10 . Membrane PLs regulate lipid rafts and receptor functions. Sulfatides, localized on the extracellular leaflets of myelin plasma membranes and synthesized by oligodendrocytes 11 , regulate neuronal plasticity, memory, myelin maintenance, protein trafficking, adhesion, glial–axonal signaling, insulin secretion, and oligodendrocyte survival 8 . Sulfatide degradation via galactosylceramidase and sulfatidase yields ceramide 11,12 , which promotes neuroinflammation, increases ROS and apoptosis, and impairs cellular signaling through survival and metabolic pathways 13 . Reductions in ST content disrupt myelin’s structure, function, and capacity to support neuronal conductivity 13 . Thus, imbalances in sphingolipid composition that reduce ST and increase ceramide probably act as important mediators of white matter (WM) degeneration.
Recent evidence indicates that alterations in white matter myelin lipid composition are found in many disease states including hypoxic–ischemic–reperfusion injury 14 . Despite considerable progress in understanding the effects of I/R on immature white matter oligodendroglia 6 , substantive information about the nature and degree of white matter lipid abnormalities or treatments that would be required to restore myelination in the developing brain is lacking. To address this question, we utilized matrix-assisted laser desorption/ionization (MALDI) mass spectrometry (MS) to characterize the alterations in cerebral white matter lipid expression that occurred within the very early time points following an I/R injury. The studies were performed in an established midgestation fetal sheep model 15 that mimics I/R injury in premature human infants.
For this study, the lipidomics data were acquired by the positive ion mode MALDI-MS. In a recent publication using the same model, we reported cerebral white matter lipidomics data acquired in the negative ion mode of MALDI-MS 16 . In contrast to the negative ion mode MALDI-MS which detects deprotonated lipid ions including phosphatidic acid (PA), phosphatidylethanolamine (PE), phosphatidylglycerol (PG), phosphatidylinositide, phosphatidylserine (PS), phosphatidylinositol phosphate, and ST 17 , the positive ion mode MALDI-MS favors the detection of protonated PLs, particularly phosphatidylcholine (PC), as well as ceramide, ganglioside, and SM 17 .
Materials and Methods
Experimental Model
We surgically prepared a fetal cerebral ischemia–reperfusion model at 118–122 days of gestation in mixed breed pregnant ewes
15,18
. Under 1% to 2% isoflurane anesthesia, the fetal carotid arteries were exposed, and then, the vertebral–occipital anastomosis and lingual arteries were ligated to restrict flow from the vertebral circulation and noncerebral vascular sources
19
. Two inflatable 4-mm vascular occluders were placed around each carotid artery to induce ischemia by inflating the occluders. Catheters were also placed in the brachial arteries and veins of the fetal sheep as previously described
19
. Reperfusion was achieved by deflating the occluders. Following recovery from surgery, on gestational days 125 to 130, 20 ewes/fetal sheep were assigned to one of the five groups to examine the effects of 30-min ischemia followed by 4 h (
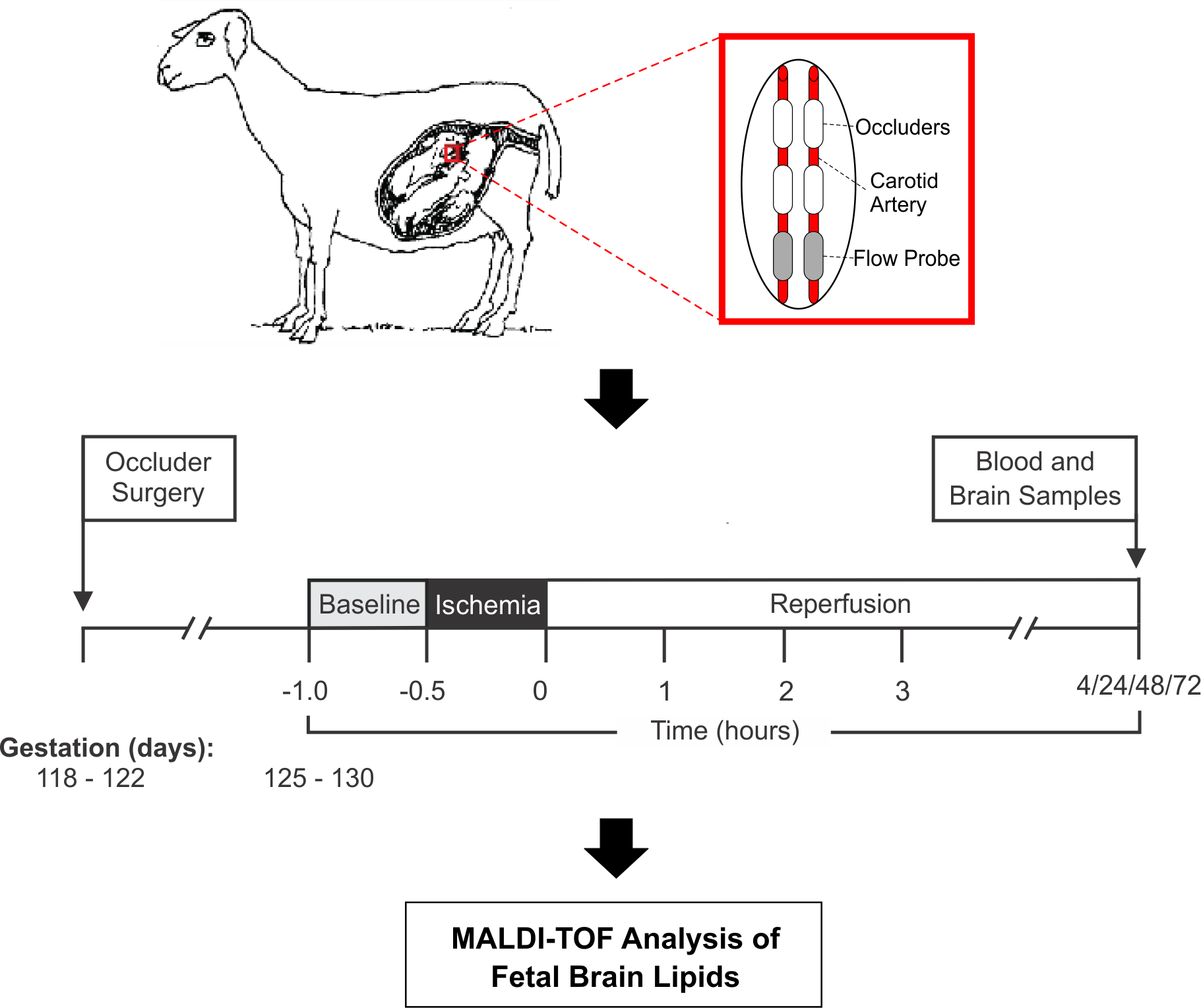
Fetal sheep model of cerebral ischemic–reperfusion. Midgestation fetal sheep were subjected to 30 min of brain ischemia (green vertical bars) followed by 4 h (
The ewes and fetuses were euthanized by intravenous pentobarbital (100–200 mg/kg). At the time of sacrifice, the fetal sheep were 80% to 85% of full gestation, and the brains of the fetal sheep at this time of gestation are approximately similar to near-term human infants 19 . Full-term gestation is 148 days. This research was approved by the Institutional Animal Care and Use Committees of the Alpert Medical School of Brown University and Women & Infants Hospital of Rhode Island, Providence, RI, USA (approval #1511000177). Postmortem fetal brains were removed promptly, and the samples of supraventricular and intragyral cerebral white matter were snap frozen in liquid nitrogen and stored at −80°C.
Lipid Extraction and MALDI
Standardized samples of fresh frozen fetal sheep white matter (50 ± 5 mg) were homogenized in 300 µL of sterile deionized water with steel beads and a TissueLyser (Qiagen N.V., Venlo, Netherlands) was used for 3 min at 20 Hz. Lipids in 150 µL of each homogenate were extracted with 1 mL of CHCl3:MeOH (2:1 v/v) according to the Folch method 20 . The dried pellets were solubilized in 200 µL high performance liquid chromatography (HPLC)-grade methanol and mixed at a 1:1 ratio with 2,5-dihydroxybenzoic acid (Sigma-Aldrich, St. Louis, MO, USA) as matrix 9 . 2,5-Dihydroxybenzoic acid was prepared at a concentration of 75 mg/mL with HPLC-grade methanol as the solvent. Prepared sample aliquots (1 µL each) were spotted in duplicate into a 384-well ground steel MALDI target plate (Bruker Daltonics, Bremen, Germany) along with mass calibration standards (Peptide Calibration Standard II, Bruker Daltonics, Bremen, Germany).
The samples were analyzed in the positive ion mode with the MALDI-tandem time-of-flight (TOF/TOF) Ultraflextreme mass spectrometer (Bruker Daltonics, Billerica, MA, USA) as previously described
9,21
. In brief, data were acquired by delivering 3,200 shots at different locations within the spotted sample using the Smartbeam II Nd:YAG laser. External mass calibration was performed using a 1:1 mixture of standard peptides (Peptide Calibration Standard II, Bruker Daltonics, Bremen, Germany) and α-cyano-4-hydroxycinnamic acid (Bruker Daltonics, Bremen, Germany). α-Cyano-4-hydroxycinnamic acid (25 mg/mL) was prepared with TA-50 as the solvent. A total of 1 µL of the standard was applied to the MALDI target plate. The mass calibration standards had masses ranging from 377 to 2,463 Da. Lipids with
Data Analysis
Matrix-assisted laser desorption/ionization data were processed using FlexAnalysis v3.4 (Bruker Daltonics, Billerica, MA, USA) and visualized with FlexImaging software v4.0 (Bruker Daltonics, Billerica, MA, USA). Results were analyzed using ClinProTools v3.0 (Bruker Daltonics, Billerica, MA, USA). Lipids were identified by their
Results
Characteristics of the Experimental Model
The duration of pregnancy in sheep is 148 days, but fetal growth occurs mainly during the last 60 days of gestation. The I/R models were generated at the midpoint of the third trimester of pregnancy. There were no statistically significant intergroup differences in mean fetal gestational age, body weight, or brain weight (supplemental Table 1A), or the terminal mean levels of arterial PO2, PCO2, base excess, blood pressure, hematocrit, and glucose before sacrifice (supplemental Table 1B) by one-way ANOVA. Thus, the groups were well matched for subsequent comparisons.
Lipid Ion Profiles
The Peak Statistic report identified 121 fetal sheep white matter lipids that had mass/charge (
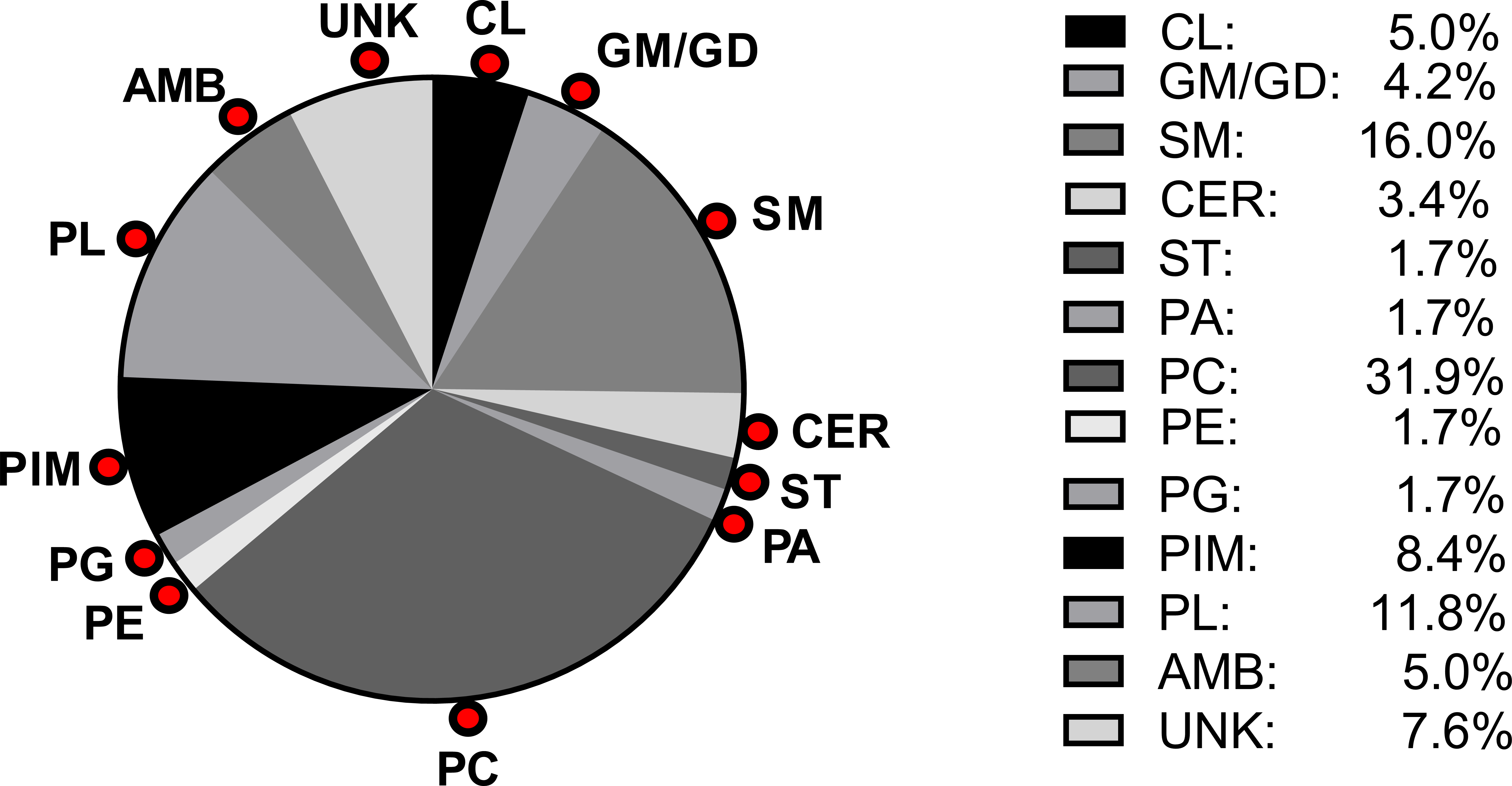
Pie chart showing percentages of each lipid subtype detected in control fetal sheep cerebral white matter by matrix-assisted laser desorption/ionization time-of-flight in the positive ionization mode. The lipids were identified as PA = phosphatidic acid; PC = phosphatidylcholine; PE = phosphatidylethanolamine; PG = phosphatidylglycerol; PIM = phosphatidylinositol mannoside; PL = phospholipid not further characterized; CER = lactosylceramides; GM/GD = ganglioside; SM = sphingomyelin; ST = sulfatide; AMB = ambiguous; or UNK = unknown. The numerical percentages of each lipid depicted in the pie chart are shown in the legend.
Ischemic–Reperfusion Effects on White Matter Lipid Profiles Demonstrated by χ2 Tests and Principal Component Analysis
To determine if I/R significantly altered white matter lipid profiles, χ2 tests were used to compare the percentages of samples within each group that had above-threshold levels of lipid ion expression (supplemental Fig. S1). Ninety-seven (79.5%) of 121 lipids detected in the positive ion mode were expressed in all samples, whereas 23 (18.9%) were expressed only transiently after I/R and 2 (1.6%) were detected de novo at all I/R time points but not in controls. χ2 tests revealed significant intergroup differences related to the proportions of samples that expressed PC(40:4)
Further comparisons of the I/R effects were made using principal component analysis (PCA) plots depicting the full spectra of white matter lipids expressed in each group. The PCA plots showed extensive overlap by two-way comparisons between control and each experimental group (Fig. 3), corresponding to the relatively few lipids that were expressed de novo or lost following I/R as illustrated in supplemental Fig. S1. These findings led to further studies to determine if I/R duration significantly altered the expression levels of specific lipids.
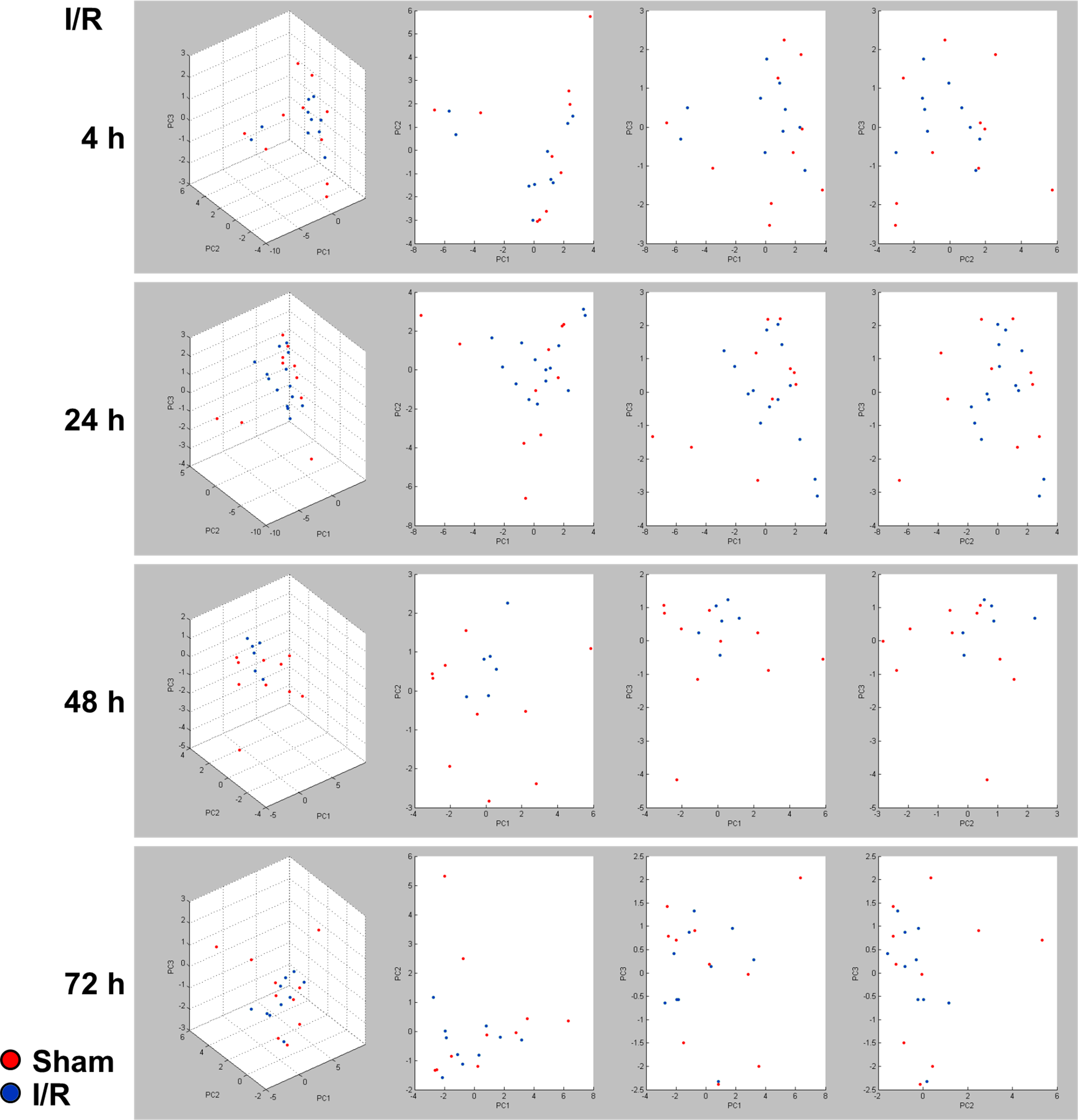
Principal component analysis of white matter lipid profiles. Matrix-assisted laser desorption/ionization time-of-flight (positive ion mode) lipid data (600–1,000 Da mass range) were compared between sham controls (red) and fetal sheep subjected to I/R (blue) with 30 min of ischemia and 4, 24, 48, or 72 h of reperfusion. Principal component analysis plots were generated with ClinProTools. Note the considerable overlap of the control and ischemic–reperfusion clusters at all time points.
Heatmap Analysis of Lipids Expressed in Relation to I/R Duration
The time-dependent effects of I/R on the relative levels of white matter lipids were depicted using a Java TreeView-generated heatmap with hierarchical clustering (Fig. 4). Lipids not detected in all the groups were excluded from the heatmap. Two broadly clustered effects of I/R were observed: The a1 cluster generally shows low levels of lipid expression in control white matter, while the a2 cluster depicts mainly high levels of lipid expression in control white matter. Intergroup differences were assessed by one-way ANOVA and the Tukey post hoc test. Significant differences are color coded in the figure.
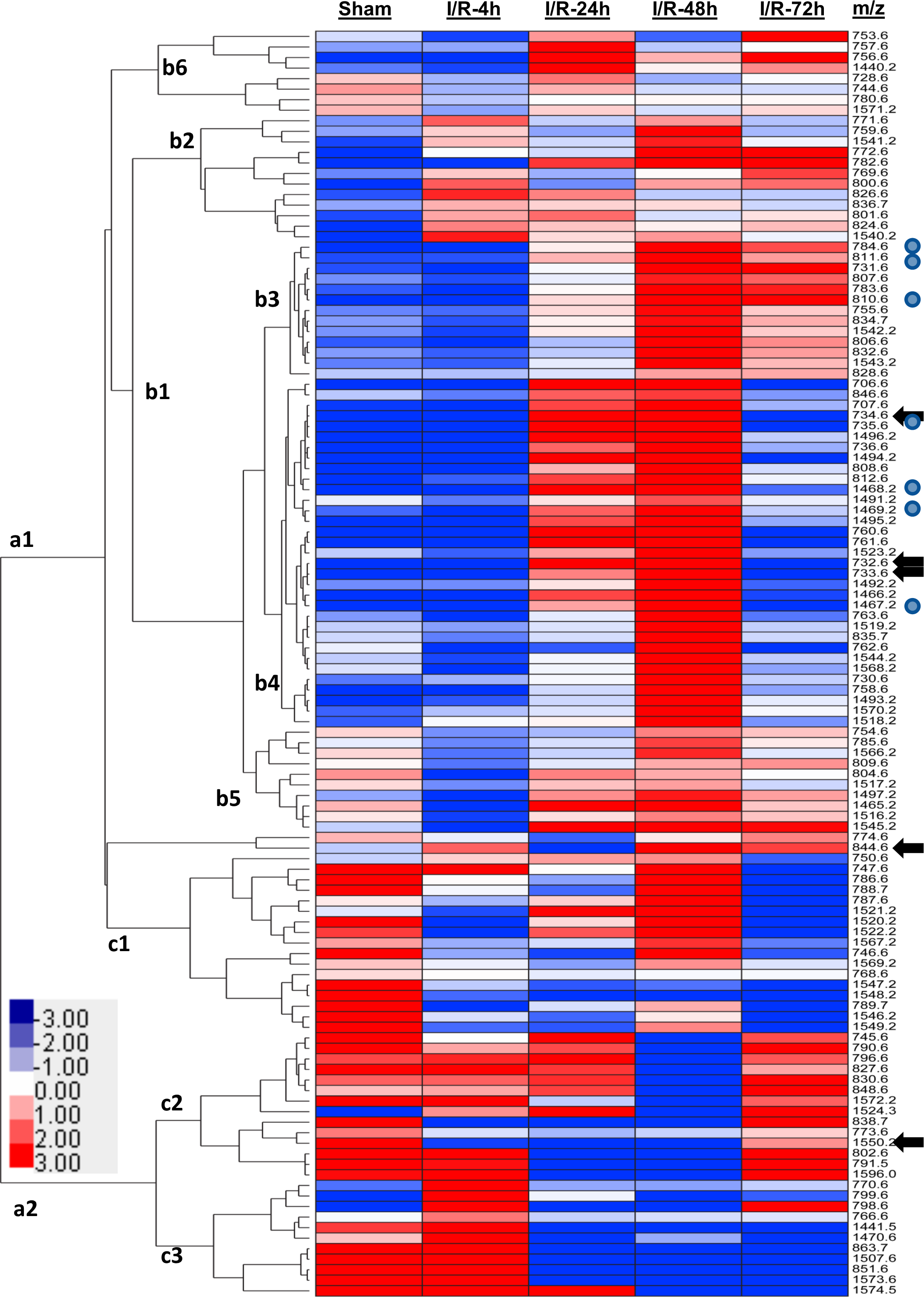
Heatmap illustrating ischemic–reperfusion’s duration effects on the relative expression levels of frontal white matter lipids. Fetal sheep were subjected to 30 min of ischemia followed by 4 h (I/R-4h), 24 h (I/R-24h), 48 h (I/R-48h), or 72 h (I/R-72 h) of reperfusion. Results were compared to the sham-operated controls. Ion intensities are displayed using a 7-color palette corresponding to
The a1 cluster had 6 subclusters: b1, the largest cluster, was subdivided into b2 through b5. Cluster b2 was associated with similarly increased lipid expression at all time points relative to control, whereas b3 and b4 were associated with the low-level lipid expression at I/R-4, similar to control, progressive upregulated lipid expression at I/R-24 and I/R-48, with peak responses occurring after 48 h of reperfusion, followed by modest (b3) or sharply (b4) reduced lipid expression at the 72-h time point (Fig. 4). In addition, samples within the b4 cluster exhibited virtually normalized levels of lipid ion expression at I/R-72. In subcluster b5, white matter lipid expression was midrange in controls, sharply reduced at I/R-4, and then elevated to similar or higher control levels at I/R-24, I/R-48, and I/R-72. The pattern in Cluster b6 was similar to b5, except that basal expression in controls varied from low to midrange.
The a2 cluster, which was mainly associated with high levels of lipid expression in control brains, was subdivided into c1, c2, and c3 clusters. In c1, lipid expression was broadly reduced at I/R-4 and I/R-24 and sharply increased toward control levels at I/R-48, while reducing to the lowest levels at I/R-72. For the c2 and c3 subclusters, high levels of lipid expression were present in control and I/R-4 groups, but lower in the I/R-24 and I/R-48 groups. In the c2 subcluster, lipid expression increased with the duration of I/R, eventually approaching control levels, whereas in the c3 subcluster, lipid expression remained suppressed across the time-span of I/R (Fig. 4). One-way ANOVA tests demonstrated significant intergroup differences for 734.6, 732.6, 733.6, 844.6, and 1,550.2, and trend effects for 1,467.2, 1,469.2, 1,468.2, 735.6, 810.6, 811.6, and 784.6, indicating pronounced variability in lipid expression in relation to I/R duration (Fig. 4).
Data Bar Displays of the Relative Effects of I/R on Lipid Ion Abundance
The ANOVA tests detected relatively few significant (
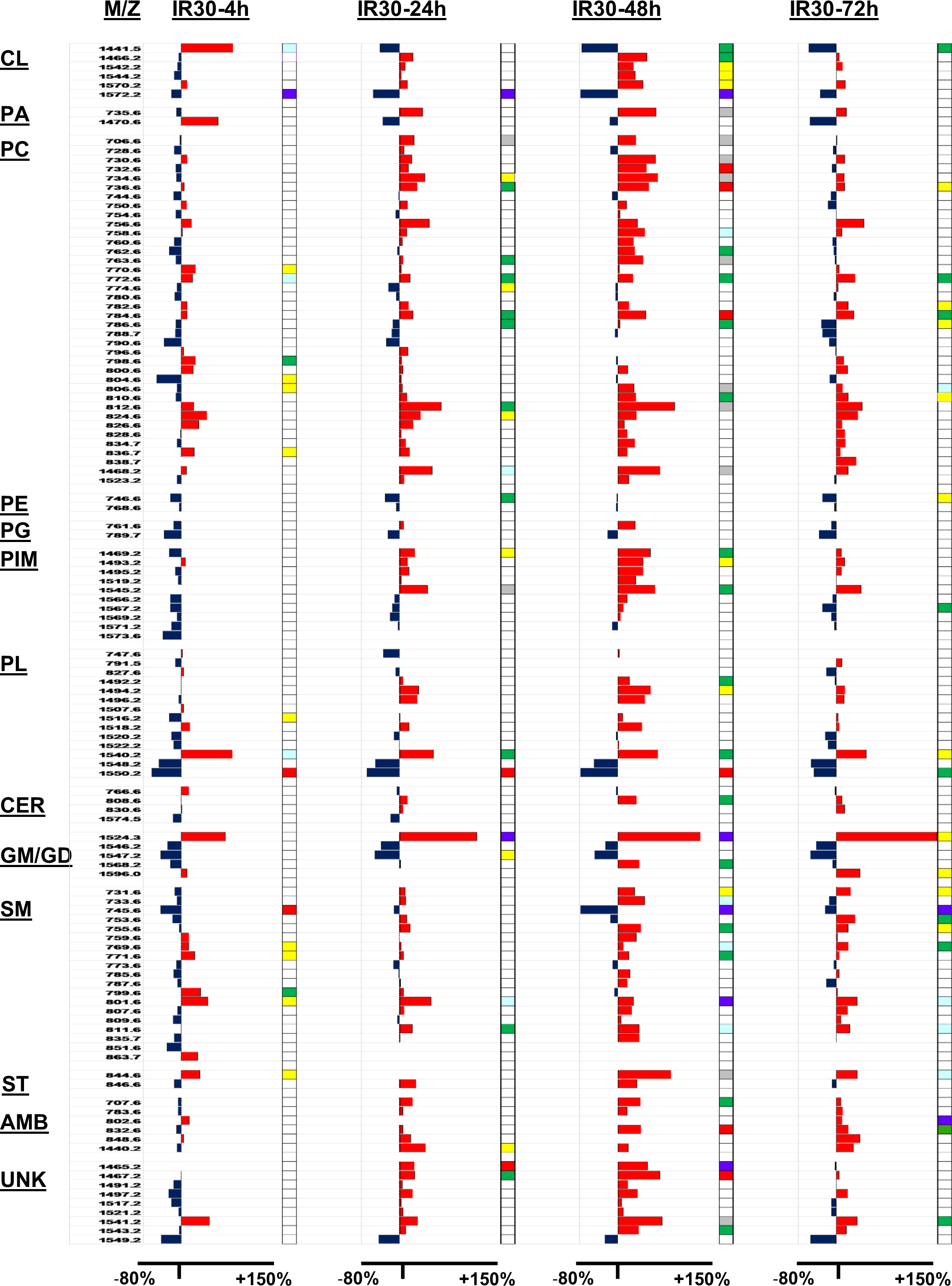
Data bar plot illustrates paired analyses of ischemic–reperfusion’s effects on relative mean lipid ion abundance after 30 min of ischemia followed by 4 h (IR30-4h), 24 h (IR30-24h), 48 h (IR30-48h), or 72 h (IR30-72h) of reperfusion. Blue bars to the left indicate I/R-associated reductions in lipid expression relative to control and red bars to the right reflect increases in lipid expression. The lipids were first grouped by their subtypes and then sorted by
Overall Effects of I/R on Lipid Expression (Fig. 5)
Generalizable effects of I/R on white matter lipid expression were as follows: (1) the smallest differences from control typically occurred at I/R-4, whereas the largest differences mainly occurred at I/R-48; (2) the directional responses at I/R-4 were frequently opposite to those that were observed at I/R-24, I/R-48, and I/R-72; (3) with increasing duration of reperfusion, 57% of the lipids increased in expression, whereas 8% declined, and 35% were modulated by less than 5%, that is, their mean levels were virtually unchanged relative to control; and (4) the responses at I/R-72 were muted primarily in comparison with I/R-48, but in some instances, I/R-24 as well.
Effects of I/R on the Expression of Specific Lipid Subtypes
Cardiolipin
Cardiolipins are mitochondria-specific PLs that maintain integrity and functionality of membranes
26
. Among 6 CLs that were detected in the white matter, CL (
Phosphatidic Acid
Phosphatidic acids are structural components of cell membranes and have functional roles in intracellular signaling during growth and proliferation
27
. Two PAs detected had opposite time course responses with I/R. Phosphatidic acid (
Phosphatidylcholine
Phosphatidylcholines comprise the majority of eukaryotic cell membrane lipids and have important roles in signal transduction. Disruption of PC homeostasis can lead to cellular apoptosis 28 . Ischemic–reperfusion increased the expression of 25 PCs (66%), modestly reduced the expression of 4 PCs (10%), and minimally altered the expression of 9 PCs (24%). Although modestly upregulated PC expression was detectable at I/R-4 in some instances, progressive and often statistically significant increases mainly occurred from I/R-24 to I/R-48. By I/R-72, the levels had declined from their peak responses but still moderately or significantly higher than control. Although several PCs were reduced at various I/R time points relative to control, for the most part, statistically significant or trend effects were not observed.
Phosphatidylethanolamine
Phosphatidylethanolamines are structural components of the inner leaflet of plasma membranes
29
. Only 2 PEs were detected in the positive ion mode of MALDI. Phosphatidylethanolamine (
Phosphatidylglycerol
Phosphatidylglycerols have important roles in maintaining cell membrane homeostasis 30 . Only 2 PGs were detected. The effects of I/R varied but there were no significant or trend effects detected relative to control.
Phosphatidylinositol monomannosides
This lipid subtype has no known function in mammalian cells
31
. Thus far, PIMs have been characterized only in relation to prokaryotic organisms in which they provide lipid anchors of lipoglycans, lipomannan, and lipoarabinomannan
32
. Nonetheless, PIMs and LPIMs were detected in brains of both control and I/R-treated fetal sheep. PIM/LPIM
Phospholipids
A total of 14 PLs could not be further categorized. The PL responses to I/R varied over the time course, but 6 exhibited progressive patterned alterations in expression with the duration of reperfusion such that 4 lipids increased from I/R-4 to I/R-48 then declined somewhat by I/R-72 and 2 remained consistently reduced relative to control over the full-time course of study. In contrast, I/R had minimal or no consistent effect on the expression of 8 PLs.
Ceramide
Galactosylceramide (GalCer) is the most abundant glycolipid in myelin
33
, and glucosylceramide (GlcCer) is a precursor for generating lactosylceramides
34
. Four Cer species were detected. Overall responses to I/R were minimal except for GlcCer (40:1;
Gangliosides
Gangliosides are generally located on the outer surface of plasma membranes and regulate cell-to-cell interactions
35
. Ischemic–reperfusion had variable effects on the expression levels of 5 white matter gangliosides. Two were moderately reduced from I/R-4 to I/R-72. One was prominently increased at I/R-4, I/R-24, and I/R-72, but unchanged at I/R-48 relative to control. Two others had moderately reduced expression over the full I/R time course. Two were significantly or trend-wise increased at either I/R-48 or I/R-72, and the remaining one, GD1a (
Sphingomyelin
Sphingomyelins help to compartmentalize signaling events and promote neuronal differentiation, synapse formation, synaptic transmission, and glial–neuronal interactions in the CNS 36 . Among 19 SMs detected, I/R increased the expression of 14 (73.7%) at one or more time points, and for the most part, peak-level stimulation occurred at I/R-48 followed by I/R-72. Modest inhibitory responses occurred mainly at I/R-4.
Sulfatide
Sulfatides assist in stabilizing plasma membranes 37 . Two STs were detected. Both were mainly increased by I/R and had peak responses at I/R-48.
Ambiguous Lipids
A total of 6 lipids were designated as AMB because their
Unknown/Unidentified Lipids
A total of 9 lipids were designated as UNK because they could not be identified from the LIPID MAPS database or published literature. As a group, most were either modestly inhibited or unchanged at I/R-4, but after longer periods of reperfusion, their expression levels increased and tended to peak at I/R-48. In contrast, the expression levels of 1 UNK lipid (
Discussion
The primary goal of this research was to characterize the early biochemical abnormalities associated with perinatal cerebral I/R injury because attendant white matter damage can lead to long-term cognitive and motor disabilities. Since immature oligodendrocytes are especially vulnerable to metabolic insults such as those caused by hypoxic, ischemic, and reperfusion injury, and their main functions include synthesis and maintenance of lipid-rich myelin which is needed for efficient neuronal conductivity, we hypothesized that the associated impairments in oligodendroglial function would be marked by substantial alterations in white matter myelin lipid composition. Improved understanding of oligodendroglial and myelin-associated responses to injury, particularly in the early stages, could lead to new strategies for reducing, reversing, or limiting their long-term adverse effects. Herein, we used MALDI-MS to assess shifts in myelin lipid profiles that occur after 30 min of ischemia followed by up to 72 h of reperfusion in an established midgestation fetal sheep model 15,18,19 .
Matrix-assisted laser desorption/ionization time-of-flight enables high throughput lipidomics analysis of white matter myelin abnormalities linked to various diseased states including injury and degeneration. The expanded use of this investigational tool could facilitate highly sensitive diagnostic applications. Despite the rapid growth in structural analysis, the regulation of biosynthesis, turnover, and function of specific lipid ions has not been well documented. Moreover, since specific classes of lipids may share similar functions, their expression levels may be coordinated 16 . One goal of lipidomics profile research is to identify clustered responses that characterize disease states.
White matter lipids in the CNS include cholesterol, glycosphingolipids, STs, gangliosides, and PLs consisting of glycerophospholipids (PA, PC, PE, PG, phosphatidylinositol (PI), PS, and plasmalogens), and SM. Sphingomyelin is composed of ceramide plus a phosphocholine or phosphoethanolamine polar head group. Several CNS diseases have already been linked to altered metabolism or expression of PLs and STs 8 . but the long-term functional consequences are poorly understood. This study employed the positive ionization mode MALDI-MS which is primarily suited for detecting protonated PLs, particularly PC, as well as SM, gangliosides, and ceramides.
The previous studies in the midgestation fetal sheep model demonstrated marked reductions in white matter myelin content within 48 h of I/R and little or no structural recovery over 72 h of reperfusion 15 . Instead, reactive inflammatory and astrocytic responses and oligodendrocyte loss accompanied the white matter damage, particularly at I/R-48 and I/R-72. Since the MALDI-MS studies were performed with lipids extracted from white matter, oligodendrocyte membranes and myelin were likely the main sources. Principal component analysis plots demonstrated that I/R caused relatively few changes in the composition of lipids that were expressed. Instead, the major conclusion drawn from the heatmap and data bar plots was that cerebral I/R caused striking and rapid shifts in the expression levels of PLs and sphingolipids over the 72-h time course, with peak responses occurring mainly at I/R-48. Furthermore, for the most part, I/R increased the expression of CL, PC, phosphatidylinositol monomannoside, SM, and ST, which mainly contribute to the functional support of neuronal membranes, synapses, metabolism, and signal transduction.
The heatmap with hierarchical clustering demonstrated 2 broad patterns of dynamic shifting in the levels of lipids expressed over the time course of study. One broad group was characterized by abundant lipid expression in controls and reduced levels with I/R, while the other showed low levels of lipid expression in controls and striking upregulation in response to I/R, particularly at 24 h and later time points. The very early (4 h) generally inhibitory effects of I/R may have been due to acute oligodendrocyte injury and abrupt cessation of biosynthetic activity. In contrast, the subsequent striking and progressive increases or declines in lipid expression with increasing duration of reperfusion could have reflected (1) changes in oligodendrocyte metabolism leading to substantial alterations in cell membrane and myelin lipid composition; (2) increased inflammatory cell infiltration in response to injury; or (3) activation of astrocytes and vascular elements. The trends toward normalized lipid expression levels at I/R-72 are consistent with the concept that the earlier responses (I/R-24 and I/R-48) were likely reactive and transient, whereas the later responses beginning at I/R-72 were adaptive and possibly reflective of a return to metabolic homeostasis.
The data bar plots revealed that more than half of the CL, PC, PIM, SM, ST, AMB, and UNK/unidentified lipids and the GD1a [M1-Neu5Ac-CO2+Na]+ganglioside were increased by I/R, particularly at I/R-48. In contrast, the expression levels of 4 ceramides and 4 of 5 gangliosides were modestly altered and often inhibited by I/R. Conclusions cannot be drawn concerning PE, PG, or PA since only 2 of each were detected; one was increased and the other decreased by I/R. However, data captured by the negative ionization mode MALDI-MS more effectively revealed the time course effects of I/R on PA, PE, PI, and PG 16 , whereas the positive ionization mode MALDI-MS was more suitable for detecting PC and CL.
Since CL is uniquely localized in the inner mitochondrial membrane, comprises 20% of its lipid mass, and has a critical role in optimizing the function of mitochondrial enzymes 38 -41 , its upregulated expression following I/R could represent a compensatory or reparative response needed to restore bioenergetics and sustain the viability of oligodendroglia. Furthermore, increased CL is neuroprotective via increased mitophagy which mediates the elimination of damaged mitochondria in the setting of traumatic brain injury induced ROS production and neuronal apoptosis 42 .
Phosphatidylcholines are major structural components of biological membranes and they are important sources of diacylglycerol and fatty acid-generated second messengers 43 . The previous reports showed that PC and PE expressions declined following global cerebral ischemia and do not subsequently increase with reperfusion 44 . Our discordant finding of increased PC expression after I/R may have been due to inherent differences between the present fetal model and the adult rat models utilized in the previous publications. In another study using adult rat brains, I/R increased PC[M+Na] and depleted PC[M+K] in injured brain regions 45 , whereas in fetal sheep white matter, we detected an elevated expression of [M+H]+, [M+Na]+ and [M+K]+, particularly at I/R-24 and I/R-48. Conceivably, the I/R-associated striking increases in PC sodium, potassium, and hydrogen adduct ions observed in fetal sheep white matter may have been mediated by significant disruption of the blood–brain barrier and attendant massive cation influx with edema 45 .
Sphingolipids are derived from cell membranes and function as signaling molecules that mediate a broad range of functions including proliferation, survival, apoptosis, migration, and adhesion 33,36,46 . The upregulated expression of SM in fetal sheep white matter following I/R agrees with findings in the previous reports that showed sphingolipid accumulation during reperfusion and restoration of blood flow to ischemic brain tissue 44,47 . These responses may indicate that with increasing duration of reperfusion, compensatory processes become activated to help re-establish synaptic connectivity, glial–nerve interactions, neuronal repair and differentiation, and intracellular signaling 36 . On the other hand, studies in adult rat models showed that sphingolipid expression was significantly altered by cerebral ischemic injury and during postischemic repair 48 such that ischemic stroke led to progressive and sustained reductions in SM and increases in ceramide 49 . Mechanistically, SM hydrolysis via sphingomyelinase increased ceramide generation. Both the loss of SM and the accumulation of ceramide exacerbate brain injury. In our fetal sheep model, I/R prominently increased the expression of SMs. Those responses may have been protective or restorative as they were associated with either reduced or minimal change in the expression of all but one of the five ceramide species were detected.
Ceramides function as important second messengers in apoptosis and cellular signaling in response to stress, including ischemia/reperfusion 50 . Ceramides are generated via SM hydrolysis, recycling of complex sphingolipids, or de novo biosynthesis 51 . The previous studies reported that cerebral ceramide levels increase following I/R 50 or fetal asphyxia 52,53 . Ceramide accumulation due to SM hydrolysis is associated with declines in SM, which can occur with I/R injury 50 . In contrast after mild I/R, ceramide accumulates via de novo biosynthesis rather than SM degradation. Ischemic–reperfusion induced ceramide accumulation within mitochondria contributes to mitochondrial dysfunction 54 -56 . In the present study, in light of the prominently increased expression of SM, the selective increase in GlcCer (40:1) may have resulted from de novo synthesis in injured oligodendroglial cells. Alternatively, it could have been driven by the influx of inflammatory cells associated with responses to I/R injury 57 .
Gangliosides are synthesized from ceramide by sequential glycosyltransferase reactions used to add sugar moieties 58 . Since gangliosides have an important role in helping to maintain plasma membrane integrity, their transient upregulation with I/R may reflect a compensatory response to injury. However, of 5 gangliosides (sialylated glycosphingolipids) detected, only GD1a [M1-Neu5Ac-CO2+Na]+ was strikingly elevated across most I/R time points. Although GD1a is normally expressed in gray matter, particularly the cerebral cortex, increased levels in the white matter have been observed in neurodegenerative diseases. The elevated levels of GD1a following I/R may have contributed to the white matter pathology by functioning as a nerve cell ligand for myelin-associated glycoprotein which inhibits nerve regeneration 46 , or as a driver of neuroinflammation via activation of brain microglia 59 .
Sulfatide is a major lipid component of CNS myelin and has broad functions related to developmental signaling, growth regulation, protein trafficking, neuronal plasticity, cell adhesion, and membrane structure 60 . However, in disease, ST may promote inflammation via aberrant activation of microglia and astrocytes and paradoxically may play a role in demyelination 61 . Therefore, the upregulated expression of ST following I/R could reflect a pro-inflammatory state corresponding with the progressive injury and degeneration of cerebral white matter observed in the fetal sheep model 16 .
Conclusions
In conclusion, these studies identified major rapid reperfusion duration-dependent shifts in lipid composition within fetal sheep cerebral white matter following transient ischemia. Ischemic–reperfusion in fetal sheep causes pathophysiological changes reminiscent of those observed in white matter of preterm human infants 6,62 -64 . The timing of injury in preterm infants cannot be ascertained easily in the clinical setting, and the overall impact of the injury cannot be determined until structural pathology, that is, cystic or diffuse white matter injury is detected by neuroimaging weeks or months after the initial insult 65 . The rapid shifts in lipid composition observed in fetal sheep brains indicate that I/R-related white matter injury in neonates begins long before it can be detected using the current ultrasound or magnetic resonance imaging, and highlights the need to develop more sensitive noninvasive biomarker assays.
The strength of this work was that a novel approach was applied to demonstrate early and progressives shifts in myelin lipid expression following I/R in an established fetal sheep model that shares properties with human preterm infants. The use of MALDI-MS revealed significant alterations in myelin lipid composition corresponding to the effects of ischemia–reperfusion injury. The fact that significant differences were detected indicates that the study was adequately powered, despite relatively small group sizes. Nonetheless, future efforts should be directed toward repeating this study with larger sample sizes and longer durations of post-I/R recovery to assess long-term effects on WM myelin lipidomic development.
Supplemental Material
Figure S1 - Supplemental material for Critical Shifts in Cerebral White Matter Lipid Profiles After Ischemic–Reperfusion Brain Injury in Fetal Sheep as Demonstrated by the Positive Ion Mode MALDI-Mass Spectrometry
Supplemental material, Figure S1, for Critical Shifts in Cerebral White Matter Lipid Profiles After Ischemic–Reperfusion Brain Injury in Fetal Sheep as Demonstrated by the Positive Ion Mode MALDI-Mass Spectrometry by Suzanne M. de la Monte, Gina M. Gallucci, Amy Lin, Ming Tong, Xiaodi Chen and Barbara S. Stonestreet in Cell Medicine
Supplemental Material
Table S1 - Supplemental material for Critical Shifts in Cerebral White Matter Lipid Profiles After Ischemic–Reperfusion Brain Injury in Fetal Sheep as Demonstrated by the Positive Ion Mode MALDI-Mass Spectrometry
Supplemental material, Table S1, for Critical Shifts in Cerebral White Matter Lipid Profiles After Ischemic–Reperfusion Brain Injury in Fetal Sheep as Demonstrated by the Positive Ion Mode MALDI-Mass Spectrometry by Suzanne M. de la Monte, Gina M. Gallucci, Amy Lin, Ming Tong, Xiaodi Chen and Barbara S. Stonestreet in Cell Medicine
Supplemental Material
Table S2 - Supplemental material for Critical Shifts in Cerebral White Matter Lipid Profiles After Ischemic–Reperfusion Brain Injury in Fetal Sheep as Demonstrated by the Positive Ion Mode MALDI-Mass Spectrometry
Supplemental material, Table S2, for Critical Shifts in Cerebral White Matter Lipid Profiles After Ischemic–Reperfusion Brain Injury in Fetal Sheep as Demonstrated by the Positive Ion Mode MALDI-Mass Spectrometry by Suzanne M. de la Monte, Gina M. Gallucci, Amy Lin, Ming Tong, Xiaodi Chen and Barbara S. Stonestreet in Cell Medicine
Footnotes
Author Contributions
Suzanne M. de la Monte conceived of the collaborative research, directed and supervised the collection of primary data including sample analysis, analyzed the data, and wrote the manuscript. Gina M. Gallucci processed the brain samples for MALDI, performed bioinformatics analysis of the results, helped prepare the tables and graphs, and contributed to the preparation of the manuscript. Amy Lin performed bioinformatics analysis of the results, helped prepare the tables and graphs, and contributed to the preparation of the manuscript. Ming Tong organized and guided the brain sample processing for MALDI, and reviewed the manuscript. Xiaodi Chen generated the experimental model, provided critical results about the experimental model, and reviewed the manuscript. Barbara S. Stonestreet designed the in vivo experimental model and critically reviewed the manuscript.
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
All procedures in this study were approved and conducted in accordance with the Institutional Animal Care and Use Committees of the Alpert Medical School of Brown University and Women & Infants Hospital of Rhode Island, Providence, RI, USA (Approval #1511000177).
Statement of Informed consent
There are no human subjects in this article and informed consent and human ethical approval is not applicable for this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This research was supported by Grants AA11431, AA024092, NS096525, and HD057100 from the National Institutes of Health.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
