Abstract
Insulin-like growth factor-1 (IGF-1) is known to be important for oligodendrocyte survival and myelination. In the current study, the authors examined the hypothesis that exogenous IGF-1 could reduce postischemic white matter injury. Bilateral brain injury was induced in near-term fetal sheep by 30 minutes of reversible carotid artery occlusion. Ninety minutes after ischemia, either vehicle (n = 8) or a single dose of 3 μg IGF-1 (n = 9) was infused intracerebroventricularly over 1 hour. White matter changes were assessed after 4 days recovery in the parasagittal intragyral white matter and underlying corona radiata. Proteolipid protein (PLP) mRNA staining was used to identify bioactive oligodendrocytes. Glial fibrillary acidic protein (GFAP) and isolectin B-4 immunoreactivity were used to label astrocytes and microglia, respectively. Myelin basic protein (MBP) density and the area of the intragyral white matter tracts were determined by image analysis. Insulin-like growth factor-1 treatment was associated with significantly reduced loss of oligodendrocytes in the intragyral white matter (P < 0.05), with improved MBP density (P < 0.05), reduced tissue swelling, and increased numbers of GFAP and isolectin B-4 positive cells compared with vehicle treatment. After ischemia there was a close association of PLP mRNA labeled cells with reactive astrocytes and macrophages/microglia. In conclusion, IGF-1 can prevent delayed, postischemic oligodendrocyte cell loss and associated demyelination.
Although injury of immature white matter is well known to be the dominant cause of neural handicap in very premature infants (Inder et al., 1999), the causes and treatment of white matter damage in the more mature infant and the adult have been relatively neglected (Petty and Wettstein, 2000). This was partially because white matter was believed to be less vulnerable to injury than gray matter (Marcoux et al., 1982). However, recent imaging data show that cerebral white matter injury also contributes to developmental disability after perinatal hypoxic–ischemic injury at term (Mercuri et al., 1999; Okumura et al., 1997). Experimentally, it is now increasingly recognized that differentiated oligodendrocytes and myelinated axons are also vulnerable to ischemic injury (Valeriani et al., 2000; Dewar et al., 1999; Jelinski et al., 1999; Nedelcu et al., 1999; Ikeda et al., 1998; Petito et al., 1998; Mandai et al., 1997; Pantoni et al., 1996). For example, in the 7-day-old rat, data from magnetic resonance imaging indicate that hypoxia–ischemia led to extensive secondary glial swelling and death, which followed an earlier phase of delayed neuronal death (Nedelcu et al., 1999). In contrast, after focal ischemia in the adult rat, oligodendrocyte loss developed earlier than neuronal injury (Pantoni et al., 1996). Similarly, the mildest lesion seen after asphyxia in the near-term fetal sheep was vacuolation and loss of myelin in white matter, rather than neuronal death (Ikeda et al., 1998).
The pathogenesis of demyelination after injury may be due to loss of mature oligodendrocytes (Mandai et al., 1997; Shuman et al., 1997) or may be secondary to other processes such as microglial activation or loss of trophic support after axonal degeneration (Shuman et al., 1997). Some evidence suggests that insulin-like growth factor-1 (IGF-1) may reduce primary and secondary postischemic white matter injury. IGF-1 promotes the proliferation and differentiation of oligodendroglia and up-regulates myelin production in vitro (Ye and D'Ercole, 1999; Wilczak and Keyser, 1997; Dercole et al., 1996; McMorris and Mckinnon, 1996; Shinar and McMorris, 1995). IGF-1 has broad, receptor-mediated antiapoptotic effects in vitro and in vivo (Parrizas et al., 1997; Galli et al., 1995; Yin et al., 1994) and specifically inhibits the apoptotic loss of oligodendrocytes associated with cytokine toxicity and metabolic insults (Mason et al., 2000; Ye and D'Ercole, 1999). Experimental demyelination is associated with distinctive patterns of induction of IGF-1 in astrocytes and the IGF-1 receptor in oligodendrocytes during subsequent regeneration (1 Hinks and Franklin, 1999; Komoly et al., 1992), suggesting that endogenous IGF-1 may play a key role in remyelination. Consistent with this hypothesis, IGF-1 also is intensely induced in reactive glia 3 to 5 days after hypoxic–ischemic injury, although the relationship with remyelination has not been examined (Lee and Bondy, 1993; Gluckman et al., 1992).
Currently, there is little information on the role of IGF-1 in oligodendrocyte survival or cerebral demyelination after hypoxic–ischemic injury in the developing brain. The authors previously have shown that postischemic administration of IGF-1 can protect cortical and striatal neurons from ischemic injury in the near-term fetal sheep (Guan et al., 2000; Johnston et al., 1996). The aim of the current study was to determine the effects of IGF-1 on cerebral white matter after ischemia in fetal sheep using proteolipid protein (PLP) mRNA expression to identify bioactive oligodendrocytes and immunohistochemistry to identify other reactive glia and quantify changes in myelin basic protein (MBP).
MATERIALS AND METHODS
Animals and surgery
All procedures were approved by the Animal Ethics Committee of the University of Auckland, New Zealand. Twenty-one Romney–Suffolk fetal sheep were instrumented at 117 to 124 days of gestation (term = 147 days) under general anesthesia (2% halothane in O2) using sterile techniques (Guan et al., 1997; Gunn et al., 2000). Ewes were given 5 mL Streptopen (Pitman-Moore, Wellington, New Zealand) intramuscularly for prophylaxis. Polyvinyl catheters were placed in both brachial arteries. Vertebral-occipital anastomoses were ligated bilaterally to restrict vertebral blood supply to the carotid arteries. A double-ballooned inflatable occluder cuff was placed around each carotid artery. Two pairs of electroencephalographic (EEG) electrodes (AS633–5SSF, Cooner Wire, Chatsworth, CA, U.S.A.) were placed on the dura over the parasagittal parietal cortex (5 mm and 15 mm anterior and 10 mm lateral to bregma), with a reference electrode sewn over the occiput (Gunn et al., 1997). To record cortical impedance, a third pair of electrodes (Cooner Wire AS 633–3SSF) was placed over the dura 5 mm lateral to the EEG electrodes. A 17-mm-long cannula was inserted into the left lateral cerebral ventricle at 4 mm anterior and 6 mm lateral to bregma (Guan et al., 2000). The fetus was then returned to the uterus and gentamicin (80 mg) (Roussel, Auckland, New Zealand) was administered into the amniotic sac before closure of the uterus. All catheters and electrodes were exteriorized through the maternal flank. A maternal femoral vein was catheterized.
Postsurgery sheep were housed together in separate metabolic cages with access to water and food ad libitum. They were kept in a temperature-controlled room (16°C, 50% humidity), in a 12-hour day/night cycle. A 3-day postoperative recovery period was allowed during which time antibiotics were administered daily to the ewe (600 mg crystapen (Biochemie, Vienna, Austria) intravenously for 4 days, and 80 mg gentamicin intravenously daily for the first 3 days). Fetal arterial blood was taken daily for blood gas analysis. Vascular catheters were maintained patent by continuous infusion of heparinized saline (40 U/mL at 0.2 mL/h). The lateral ventricle cannula was maintained patent by daily flushing with 200 μL artificial cerebrospinal fluid (aCSF) (Guan et al., 2000).
Experimental procedures
Fetuses were randomly assigned to either sham ischemia with no infusion (sham control group, n = 4), or ischemia groups who received intracerebroventricular infusions of 1 mL of either vehicle (aCSF, n = 8) or 3 μg rhIGF-I (n = 9), kindly provided by Dr. D. Hung (Chiron, Emeryville, CA, U.S.A.). Reversible cerebral ischemia was induced by inflation of both carotid cuffs with sterile normal saline for 30 minutes. Successful occlusion was confirmed by the onset of an isoelectric EEG signal within 30 seconds of inflation (Gunn et al., 1997). Before administration, the pH of the infusate was buffered to 7.33 to 7.39 with 1 mol/L NaHCO3, as previously described (Johnston et al., 1996). The dead space in the lateral ventricle catheter (0.7 mL) was primed with either rhIGF-1 or vehicle by infusion over 45 minutes. Ninety minutes after the reperfusion, IGF-1 or vehicle were infused over 1 hour. At the end of the experiment, 96 hours after ischemia, the ewe and fetus were killed by an intravenous overdose of pentobarbital. The fetus was rapidly removed through an abdominal incision, and the brain was perfusion fixed in situ with normal saline, followed by 10% phosphate-buffered formalin. Each brain was removed from the skull and fixed in the same fixative for a further 7 days before processing and embedding using a standard paraffin tissue preparation.
Immunohistochemistry
The following primary antibodies were used in the current study: rabbit antisera raised against MBP (Roche, Mannheim, Germany); isolectin B-4 (Sigma, St. Louis, MO, U.S.A.) to label reactive microglia; and glial fibrillary acidic protein (GFAP, Sigma) to label reactive astrocytes. Antibodies were diluted in 1% goat serum in phosphate-buffered saline (PBS) and 0.4% merthiolate.
Immunohistochemical staining was performed on coronal sections (6 μm), at the level of the parietal cortex, cut and mounted on chrome alum coated sides. Sections were deparaffinized in xylene, dehydrated in a series of ethanol, and incubated in PBS (0.1 mol/L). Sections were pretreated with 1% H2O2 in 50% methanol for 20 minutes, washed in PBS (3 × 10 minutes), and then incubated for 2 days at 4°C in the following primary antibodies at the dilutions indicated: MBP (1:200), isolectin B-4 (1:100), and GFAP (1:200). The primary antibodies were washed off with PBS (3 × 10 minutes) and then incubated with goat anti-rabbit biotinylated secondary antibody (1:200, Sigma) overnight at room temperature. Sections were washed, incubated in ExtrAvidin (Sigma, 1:200) for 3 hours, washed again in PBS-triton, and then reacted in 0.05% 3,3-diaminobenzidine tetrahydrochloride (DAB) and 0.01% H2O2 to produce a brown reaction product. Sections then were dehydrated in a series of alcohol to xylene and coverslipped with mounting medium. Control sections were processed in the same way except that the primary antibody was omitted from the incubating solution. Adjacent sections also were stained with thionin to examine apoptotic morphology.
Cloning of sheep proteolipid protein gene
Five micrograms total RNA extracted from near-term fetal sheep cortex was used to synthesize a single strand cDNA using SuperscriptII RNaseH− Reverse transcriptase (Gibco BRL, Gaithesburg, MD, U.S.A.). A 417 base pair fragment corresponding to nucleotide 79 to 495 of bovine partial PLP mRNA sequence (Genebank) was generated by polymerase chain reaction (PCR) with the following primers: upper 5′-ACCTATGCCCTGACCGTTG-3′, lower 5′-TGTGTGGTTAGAGCCTCGC-3′. The PCR conditions were as follows: 3 minutes at 94°C; 35 cycles of 30 seconds at 94°C, 30 seconds at 58°C, 45 seconds at 72°C; and 7 minutes at 72°C. The PCR product, which formed a single band at the expected size when resolved in agarose gel electrophoresis, was subcloned into plasmid pCR2.1 (Invitrogen, Carlsbad, CA, U.S.A.) by TA cloning and sequenced from both M13 reverse and forward directions. DNA sequencing identified the cloned fragment as sheep PLP. Compared with mRNA sequences of other species in the Genebank, the cloned sheep PLP sequence shared 100%, 95.7%, and 94.2% similarity with bovine, human, and rat PLP, respectively, at the nucleotide level. The sheep PLP cDNA fragment was released from the flanking EcoRI sites and resubcloned into the EcoRI site of pBluescriptIIKS (Stratagene, La Jolla, CA, U.S.A.), which was used as a template to make RNA probes.
In situ hybridization
Antisense or sense digoxigenin–labeled RNA probes were synthesized by in vitro transcription from HindIII or BamHI linearized template using DIG RNA Labeling Mix (Boehringer Mannheim) and T7 or T3 RNA polymerase (Gibco BRL), respectively, according to the manufacturer's instructions.
In situ hybridization was performed as described previously (Lai et al., 1996). Briefly, before hybridization, paraffin-embedded sections (6 μm) were dewaxed, rehydrated, and sequentially subjected to postfixation, proteinase K treatment and acetylation treatment. Dehydrated and air-dried sections were hybridized with probes in a humidified box at 58°C overnight. After hybridization, sections were treated with RNAse A and washed with 0.1 × standard saline citrate/dithiothreitol (SSC/DTT) at 65°C for 30 minutes. Sections then were washed 3 × 20 minutes with a buffer solution containing maleic acid (100 mmol/L), NaCl (150 mmol/L), and Tween 20 (0.1%, pH 7.5) (MABT buffer). Sections were incubated with block solution containing normal sheep serum (10%) and 2% blocking agent (Boeringer Mannheim), made in MABT buffer, at room temperature for 1 hour, and incubated with sheep anti-DIG-alkaline phosphotase Fab fragment (Boehringer Mannheim), diluted 1:300 in block solution, at 4°C overnight. Sections were washed 3 times with MABT buffer containing 2 mmol/L levamisole (Sigma) and freshly prepared staining buffer containing 100 mmol/L NaCl, 100 mmol/L Tris (pH9.5), 50 mmol/L MgCl2, 0.1% Tween 20, and 2 mmol/L levamisole. Signals were visualized with 225 μg/mL 4-nitro blue tetrazolium chloride (NBT, Promega, Madison, WI, U.S.A.) and 175 μg/mL 5-bromo-4-choro-3-indoyl-phosphate (BCIP, Promega) diluted in staining buffer at room temperature in a light-tight box for 3 hours. Slides were coverslipped for microscopy or used for immunohistochemical staining.
Analysis
The number of isolectin B-4, GFAP, and PLP positive cells was counted in three areas in the intragyral white matter of the parasagittal cortex (Fig. 1, regions 1 to 3) and in one area in the corona radiata (Fig. 1, region 4) of both hemispheres by light microscopy (×20). Within the intragyral region, counts were made using full cross sections of the tract for a fixed length of 0.54 mm. In the corona radiata, a rectangular area of 0.54 × 0.36 mm (0.194 mm2) was used to count cell numbers. The counts in the three intragyral areas were averaged for comparison of the treatment groups by ANOVA (SPSS v10; SPSS). Where a significant effect of group was found, further post hoc comparisons were performed using the least significant difference test. The density of MBP from the same areas and their background was measured by image analysis (Sigmascan; SPSS). The difference between the MBP density and the background reading from adjacent grey matter was calculated and used for data analysis. The effects of IGF-1 treatment and region on the density of MBP were then assessed by two-way ANOVA, after transformation to normalize the data. The area of the parasagittal intragyral white matter tracts also was measured using the image analyzer. Because the vehicle group showed an increase in intragyral area compared with sham controls and the IGF-1 group, the authors reanalyzed the density of MBP within the intragyral region by ANOVA, after correction for relative white matter tract area. The relationship between MBP density and number of PLP-positive cells was examined by backward stepwise regression (SPSS). The colocalization of PLP-positive cells with GFAP and isolectin B-4, as well as cells with apoptotic morphology, was examined and photographed by light microscopy (Nikon E800; Nikon, Tokyo, Japan).
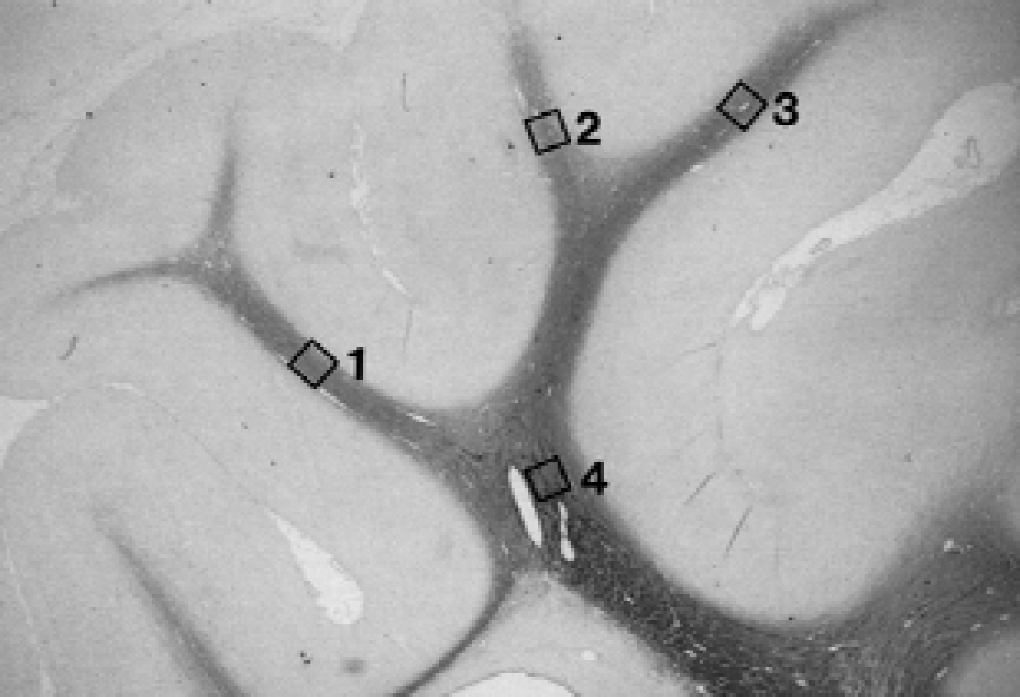
Photomicrograph indicating the regions used to count the number of cells expressing proteolipid protein, glial fibrillary acidic protein, and isolectin B-4 immunopositivity and to measure the average density of myelin basic protein in the intragyral white matter and the corona radiata. One area in the corona radiata (square 4) and three regions from the intragyral white matter (squares 1, 2, and 3) of both hemispheres were used for assessment. In the intragyral region, complete counts were made using the full cross-section of 0.54-mm-long portions of the tracts. In the corona radiata, the area used to assess these data was 0.194 mm2.
RESULTS
The authors have previously reported changes in striatal phenotypic neurons in the current study's cohort (Guan et al., 2000).
Proteolipid protein expression
In the sham control group, PLP mRNA-positive cells were present in greater density in the corona radiata compared with the intragyral regions (P < 0.001, Table 1, Fig. 2A). Morphologically, denser PLP mRNA staining was discontinuously distributed within the peripheral regions of the cytoplasm, typically showing a bipolar pattern of distribution (Fig. 2B, closed arrows). There was greatly reduced staining for PLP mRNA in the remaining cytoplasm and no staining in axons (Fig. 2B).
Effects of IGF-1 on PLP mRNA expression

The number of cells expressing proteolipid protein (PLP) mRNA was counted in the corona radiata and intragyral cerebral white matter tracts. The counts in the corona radiata are taken from an area of 0.2 mm2. The measurements in the intragyral region represent total cross-sectional counts from 3 areas of the tract, each 0.54-mm-long. Data are mean ± SD.
P < 0.01 sham control versus ischemia + vehicle.
P < 0.05 ischemia + vehicle versus ischemia + insulin-like growth factor-1 (IGF-1)-treated groups.
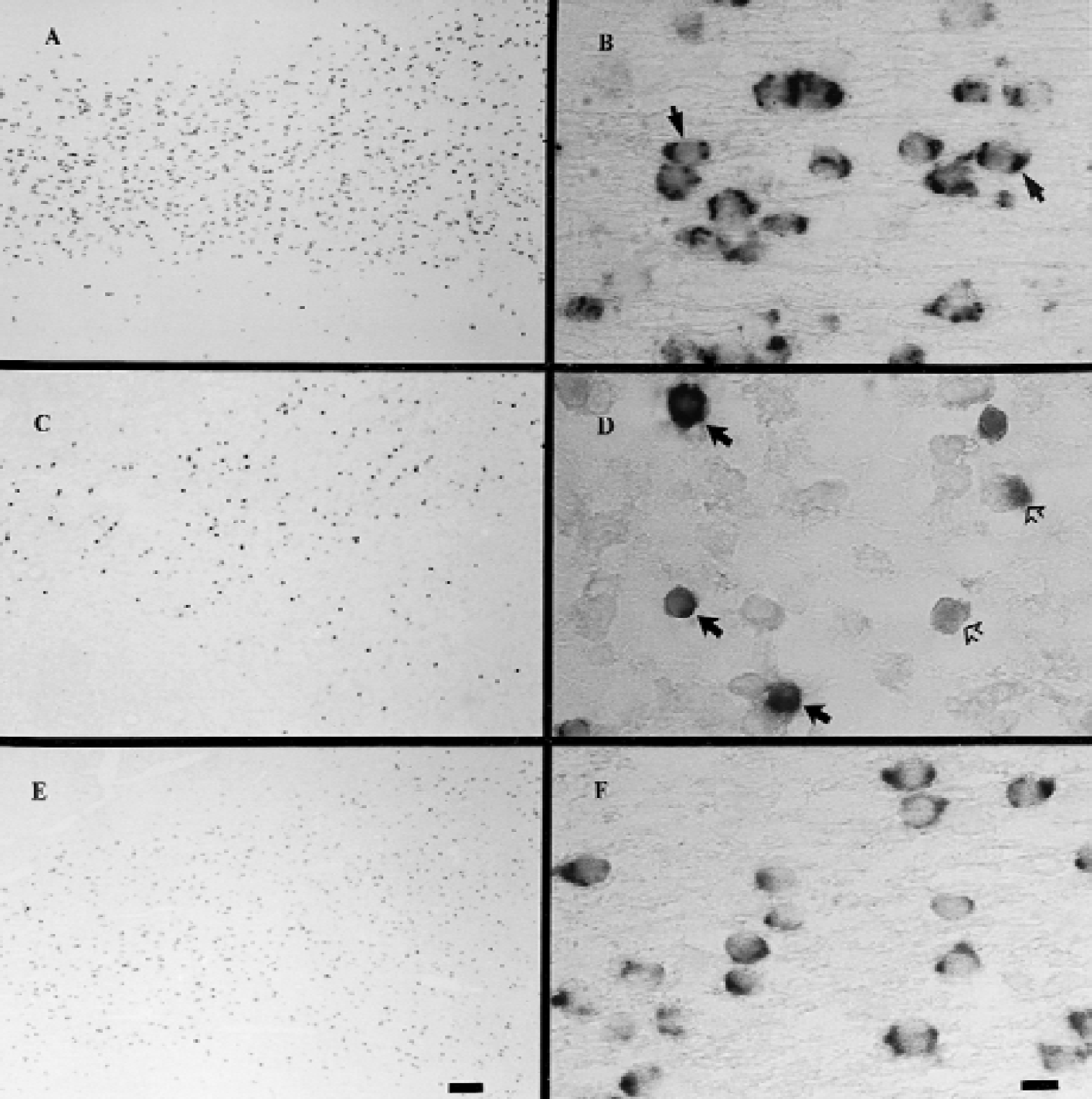
Photomicrograph showing the distribution
In the vehicle-treated ischemia group, a significant loss of PLP mRNA-positive cells was seen within the intragyral white matter tracts of the parasagittal cortex (P < 0.01, Table 1, Fig. 2C). Although there was a small reduction in the corona radiata, this was not significant (Table 1). Morphologically, many of the remaining PLP mRNA-positive cells showed an overall loss of PLP mRNA staining (Fig. 2D, open arrows), however, others showed continued dense staining, but with a loss of the normal peripheral distribution (Fig. 2D, closed arrow).
The reduction in the number of PLP mRNA-positive cells was accompanied by stromal edema and evidence of cell death in the white matter tracts, particularly the intragyral regions. Cells displaying apoptotic morphology, with either condensed nuclei or lobulated condensation of chromatin, were seen scattered within the white matter tracts (Fig. 3, closed arrows).
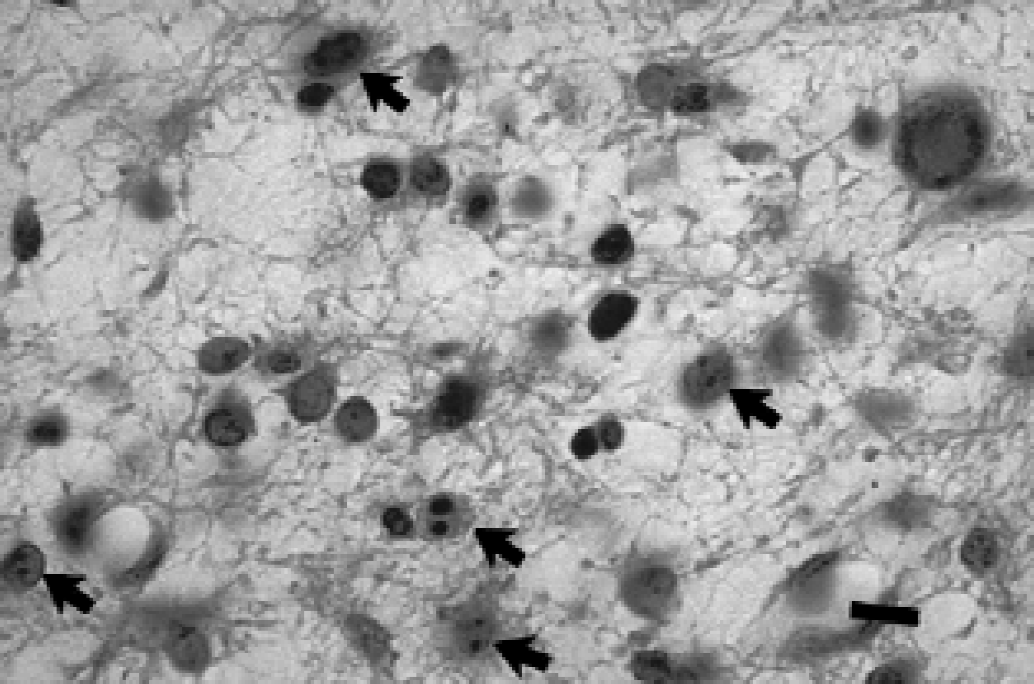
Photomicrograph showing white matter tracts in the vehicle-treated ischemia group, stained with thionin. Note the presence of condensation of chromatin or lobulated condensation fragmentation of the nuclei (stained blue), the morphologic changes associated with apoptosis (closed arrows). Scale bar = 10 μm.
Insulin-like growth factor-1 treatment was associated with a significant increase in the total numbers of PLP-positive oligodendrocytes in the intragyral white matter tracts, but not the corona radiata, compared with the vehicle-treated group (P < 0.05, Fig. 2E, Table 1). Furthermore, IGF-1 treatment was associated with a more normal cellular distribution of PLP mRNA in most of the oligodendrocytes (Fig. 2F).
Myelin basic protein immunoreactivity
In the sham control group, MBP staining was continuously distributed from the corpus callosum and external capsules into the corona radiata and intragyral white matter tracts (Fig. 4A). The average density was slightly greater in the corona radiata, gradually reducing distally in the intragyral white matter tracts (P < 0.01). The majority of MBP staining was associated with nerve fibers, which were seen to parallel each other in Fig. 4B, with no obvious staining in the cell bodies.
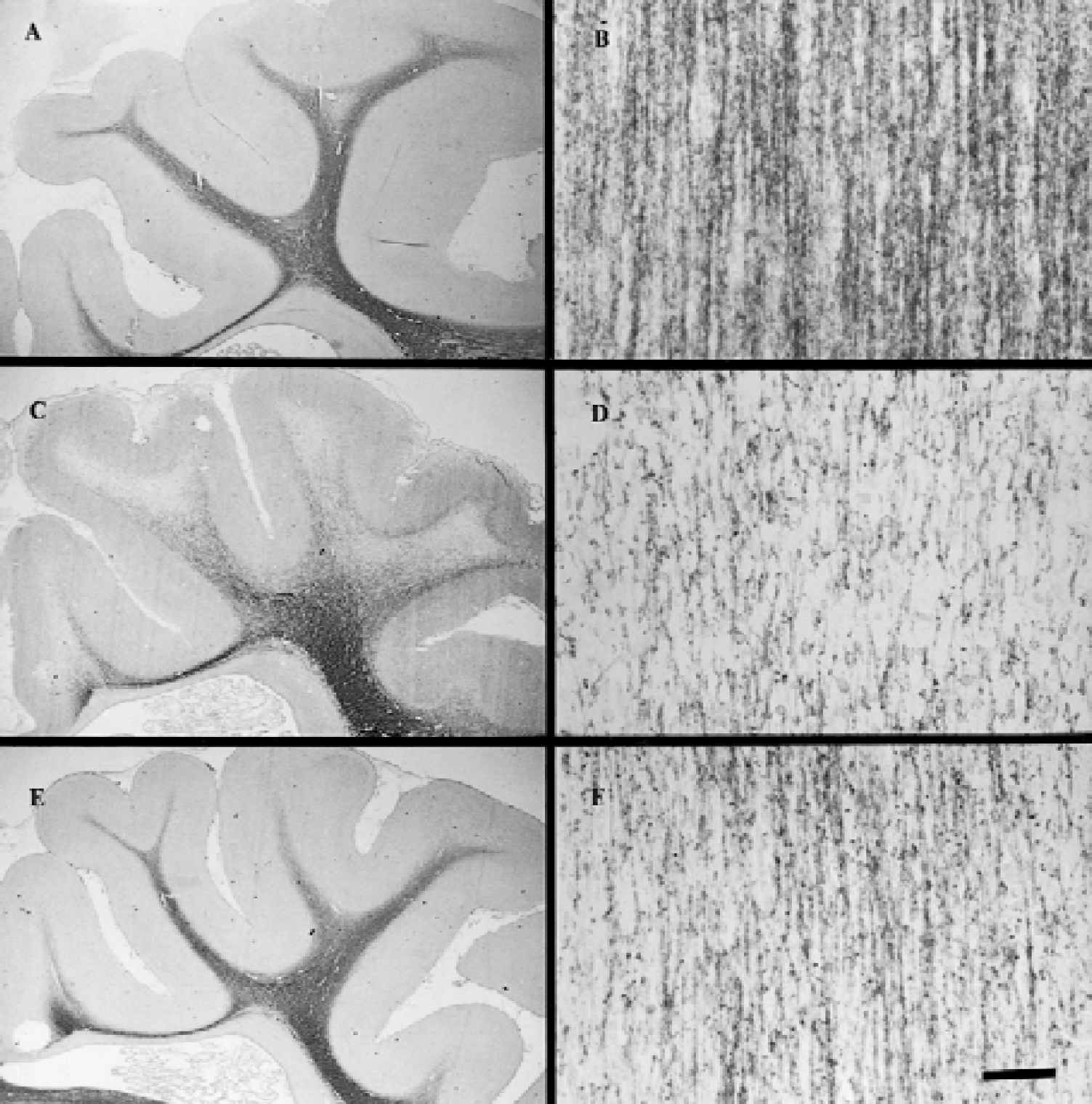
Photomicrographs showing the distribution
Ischemia resulted in a significant overall loss of MBP density (P < 0.05, Fig. 4C, Table 2). The loss of MBP density was most pronounced in the intragyral white matter tracts with only a trend toward a reduction in MBP staining in the corona radiata (Table 2). Morphologically, MBP staining with individual nerve fibers was weaker and less compact than in sham controls, and the normally parallel microstructure of the MBP fibers was disorganized and fragmented (Fig. 4D). Quantitatively, measurements of the area of the intragyral white matter tracts confirmed a significant increase in tissue swelling in the vehicle-treated groups (21.1 ± 5.3 mm2 in the vehicle group vs. 15.8 ± 2.3 in the sham control group, P < 0.05).
Effect of IGF-1 on MBP density
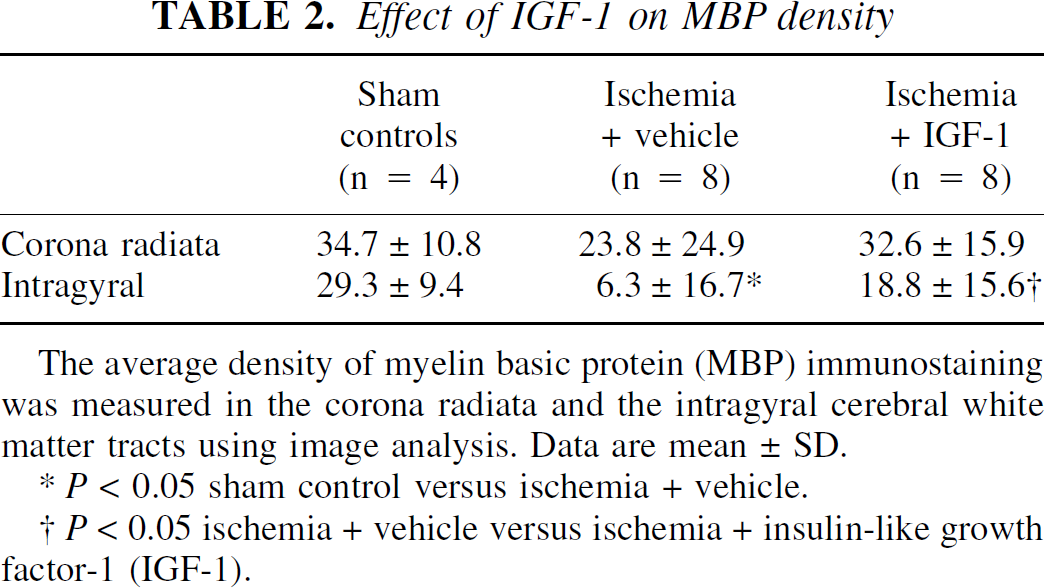
The average density of myelin basic protein (MBP) immunostaining was measured in the corona radiata and the intragyral cerebral white matter tracts using image analysis. Data are mean ± SD.
P < 0.05 sham control versus ischemia + vehicle.
P < 0.05 ischemia + vehicle versus ischemia + insulin-like growth factor-1 (IGF-1).
In the IGF-1–treated group, the overall density of MBP was improved compared with the vehicle-treated group in the intragyral white matter (Fig. 4E, Table 2, P < 0.05). There was reduced tissue swelling in the intragyral regions after IGF-1 treatment (intragyral white matter area 16.0 ± 4.3 mm2, P < 0.05 vs. vehicle treatment, Mann–Whitney U test). The increase in intragyral MBP density was significant after correction of the density measurements in the vehicle group for white matter tract area (18.8 ± 15.6 vs. 8.3 ± 21.9 (6.3 ± 16.7, uncorrected), P < 0.05). The density of MBP and the number of PLP-positive cells were closely correlated across all 3 groups in both the intragyral cortex (r = 0.73, P < 0.001) and the corona radiata (r = 0.59, P < 0.007), as shown in Fig. 5. Treatment group did not significantly influence this relationship (P = 0.71). On visual assessment, compared with the vehicle-treated group, fibers with MBP staining were denser, more compact, and less fragmented (Fig. 4F).
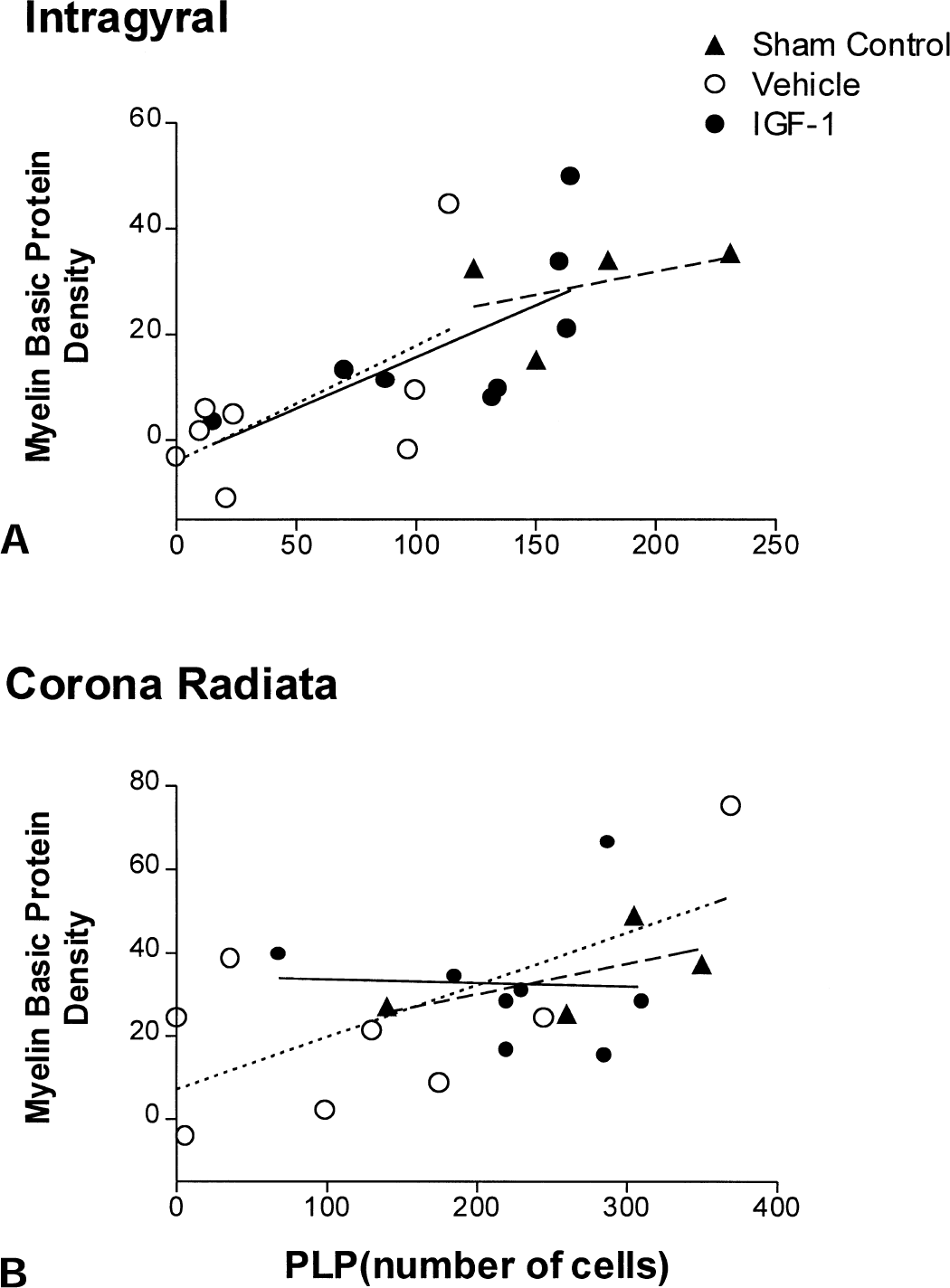
Relationship between density of myelin basic protein and number of proteolipid protein (PLP)-positive cells in the intragyral white matter
Reactive glia
No reactive glia were seen in the sham controls. After ischemia, there was marked induction of isolectin B-4-and GFAP-positive cells within white matter. Morphologically, isolectin B-4–positive cells showed strong staining in the cytoplasm, with a small number of processes. The number of isolectin B-4 cells was significantly increased after IGF-1 treatment in the intragyral matter tracts and in the corona radiata compared with the vehicle-treated group (Table 3).
Effect of IGF-1 on reactive glia

The number of glial fibrillary acidic protein (GFAP) or isolectin B-4 immunopositive cells counted in the corona radiata and the intragyral cerebral white matter tracts. Data are mean ± SD. IGF-1, insulin-like growth factor.
P < 0.005,
P < 0.001 ischemia + vehicle versus ischemia + IGF-1.
The GFAP-positive cells showed multiple cell processes, with GFAP staining mainly in the processes, with weak or no staining in the cell bodies. IGF-1 treatment also significantly increased the number of GFAP immunopositive cells in the intragyral tracts and the corona radiata compared with the vehicle-treated group (Table 3, P < 0.05).
Association of PLP mRNA positive cells with reactive glia
Proteolipid protein mRNA digoxigenin labelling followed by GFAP or isolectin B-4 immunohistochemical counterstaining demonstrated that some PLP mRNA-positive cells were closely associated with either GFAP (Fig. 6) or with isolectin B-4–positive cells (Fig. 7). Associated GFAP and PLP cells were seen distributed throughout the corona radiata and the intragyral tracts. The PLP mRNA and GFAP staining often was difficult to separate, but typically could be seen to represent two separate cells, with GFAP-positive cells wrapping around the PLP mRNA containing cell bodies (Fig. 6B and 6C, arrows). Within the most severely damaged tissues, the PLP/GFAP associations were more isolated and had fewer and shorter GFAP positive processes (Fig. 6C, arrows).
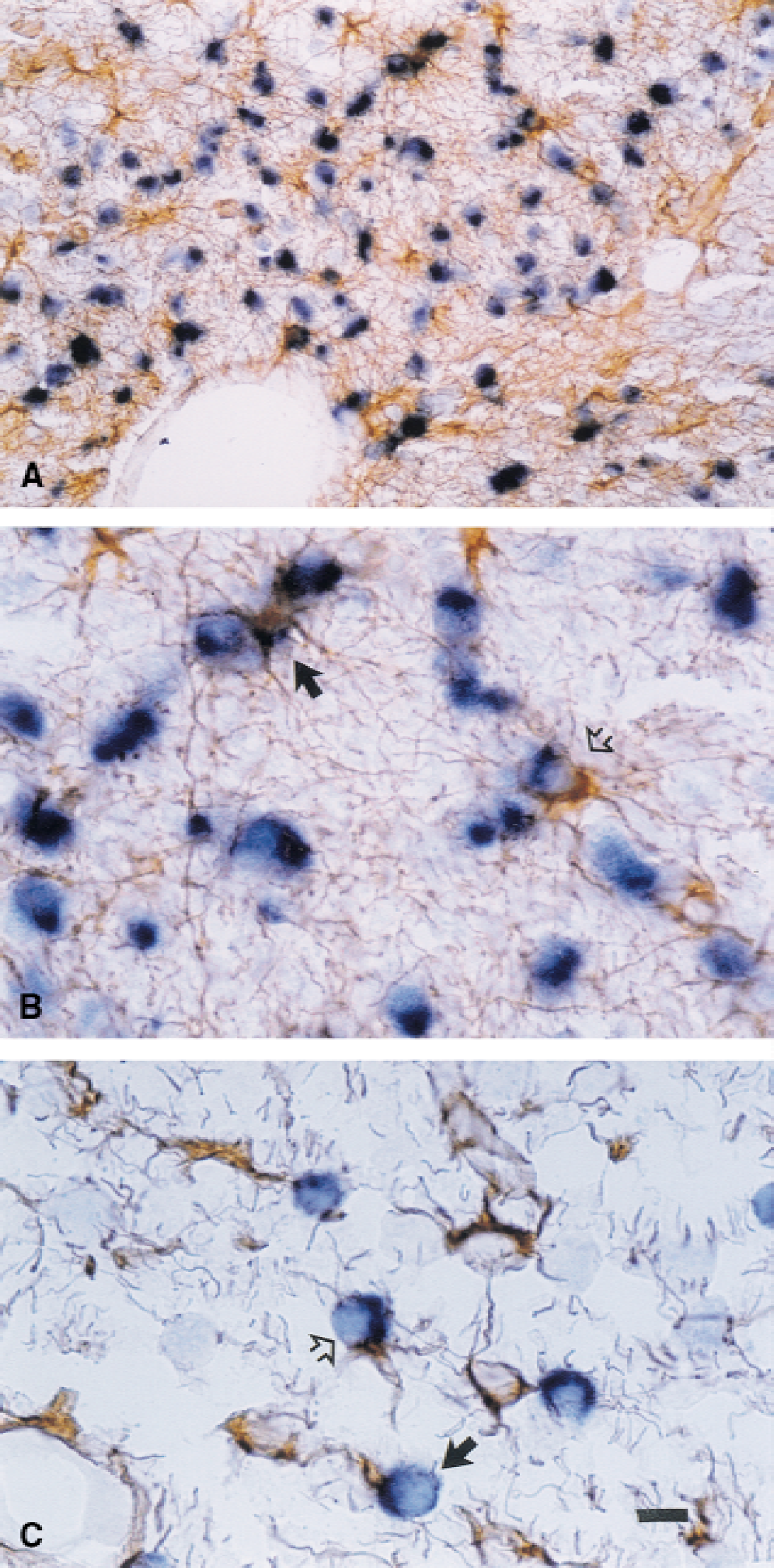
Photomicrograph showing the relationship between digoxigenin labelling for proteolipid protein (PLP) mRNA (blue) and glial fibrillary acidic protein (GFAP) immunopositive staining (brown, arrows).
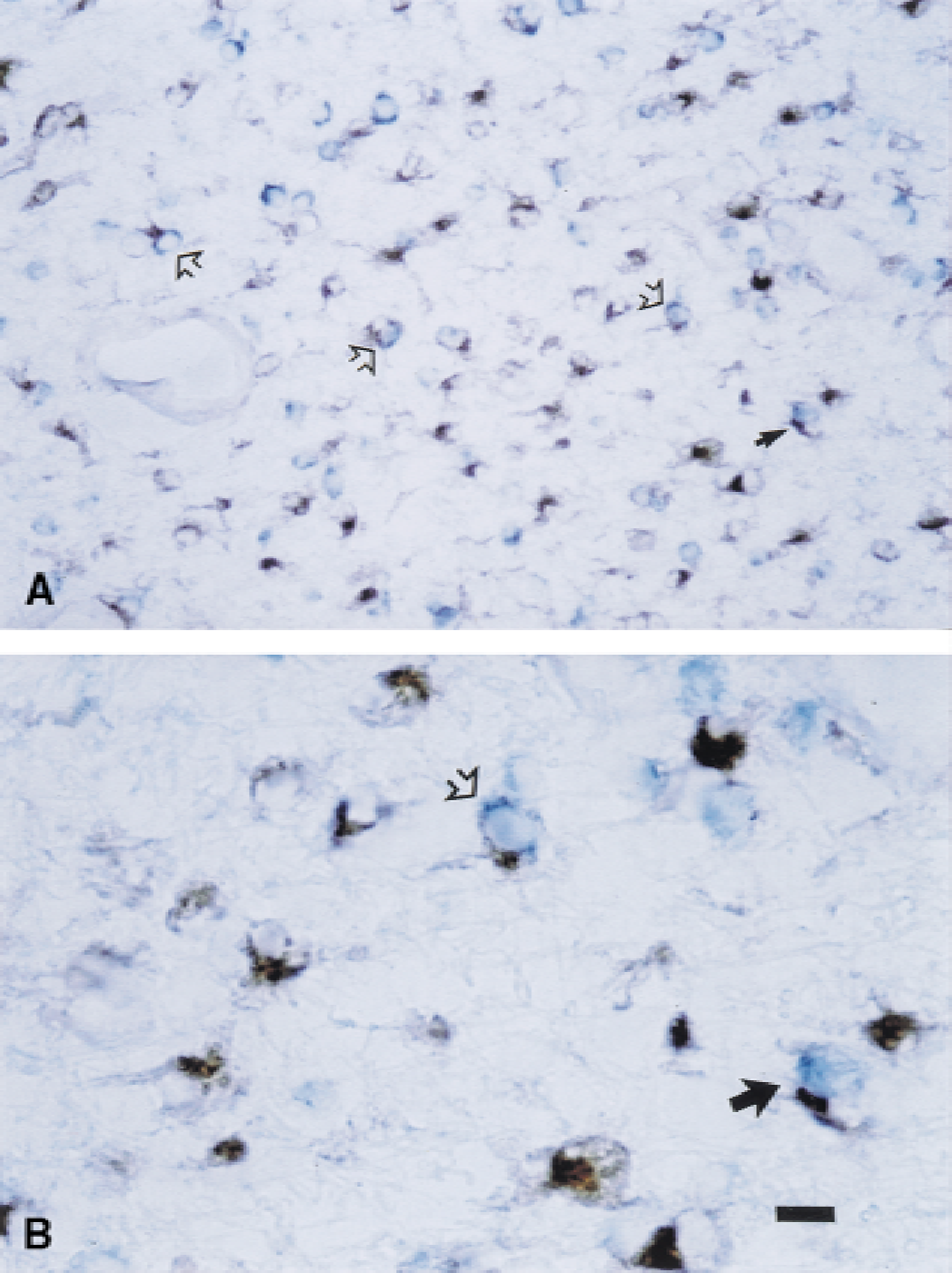
Photomicrograph of the relationship between digoxigenin labelling for proteolipid protein (PLP) mRNA (blue) and isolectin B-4 immunostaining (brown). These low power (
The association of PLP and isolectin B-4–stained cells was less common than for PLP and GFAP cells, and this type of association was seen primarily within areas showing background tissue infarction (Fig. 7, arrows). In contrast with GFAP staining, isolectin B-4 positively labeled in the cytoplasm. Again, the two labels often were difficult to resolve. As shown in Fig. 7A and 7B (arrows), however, in most cases it was possible to distinguish them as PLP mRNA-labeled cells that were closely adjacent to isolectin B-4–positive cells.
DISCUSSION
The current study demonstrates for the first time that IGF-1 can reduce postischemic demyelination in the intracerebral white matter tracts. Ischemia led to a “watershed” distribution of white matter injury, greatest in the parasagittal gyii, consistent with the previously reported distribution of neuronal loss in this model (Gunn et al., 1997; Johnston et al., 1996). This type of injury is common after perinatal asphyxia at term (Mercuri et al., 1999; Okumura et al., 1997). IGF-1 administration 90 minutes after reperfusion from the ischemic insult was associated with greater oligodendrocyte survival, improved myelin density, and reduced tissue swelling in the parasagittal intragyral white matter tracts, with increased numbers of reactive astrocytes and microglia in both the intragyral white matter and coronal radiata.
Proteolipid protein mRNA expression is a biologic marker for myelination at the transcript level and its expression in the CNS is classically reported only in differentiated oligodendrocytes, although trace amounts may be found earlier in development (Delbigio et al., 1997). Potentially, a loss of PLP mRNA could be the result of either loss of oligodendrocytes or reduced myelin production by surviving but inactive oligodendrocytes. However, the number of cells expressing PLP in the vehicle-and IGF-1–treated groups was closely correlated with the density of myelin basic protein, particularly in the intragyral white matter tracts (Fig. 5A and 5B). This close relationship suggests that postischemic demyelination was primary, that is, because of loss of oligodendrocytes. Cerebral ischemia was associated with marked tissue swelling because of cellular lysis, as illustrated by Fig. 2, with an increase in the area of the intragyral tracts compared with sham controls (Table 2). The reduction in injury with IGF-1 treatment also significantly reduced tissue swelling. This difference in tissue swelling of approximately 30% will have accounted for part of the apparent reduction in myelin density in the vehicle group; however, the improvement in estimated myelin content of the intragyral area was still significant after adjustment for this systematic change.
These data suggest that IGF-1 is able to interrupt critical processes occurring early in the recovery phase that lead to white matter injury. In the current study, the mode of cell death within the intracerebral white matter tracts appeared to be mixed, however, cells showing classic apoptotic changes were detected (Fig. 3), which is consistent with previous reports (Petito et al., 1998; Mandai et al., 1997). The capacity of IGF-1 to interrupt programed cell death or apoptosis is well described in both neurons (D'Costa et al., 1998; Russell et al., 1998) and myelinating glia in vitro (Delaney et al., 1999; Ye and D'Ercole, 1999). This antiapoptotic activity is receptor mediated and involves the phosphatidylinositol 3′-kinase and mitogen-activated protein kinase pathways (Parrizas et al., 1997).
Ischemia was associated with marked morphologic changes, including very dense PLP expression in some surviving oligodendrocytes and loss of its normal cytoplasmic distribution. IGF-1 treatment was associated with restoration of the normal distribution of PLP. The increase in PLP density after ischemia may reflect a compensatory increase in PLP production in these surviving cells (Fig. 2D). This is consistent with previous studies showing that surviving oligodendrocytes in the borders of traumatic lesions display up-regulation of mRNA for MBP or PLP, as early as two days after injury in some cases (Frei et al., 2000; Bartholdi and Schwab, 1998). The striking loss of the bipolar distribution after ischemia (Fig. 2) raises the possibility that ischemia may have impaired the intracellular distribution of PLP mRNA, possibly because of cytoarchitectural damage or axonal damage (Petty and Wettstein, 2000).
In addition to increasing oligodendrocyte survival, IGF-1 treatment was associated with an increased glial reaction. Potentially the increase could be related either to enhanced proliferation (Vergelli et al., 1995; Chernausek, 1993) or to increased survival of these cell types after ischemia. Improved glial survival may play a role in preventing demyelination or even improving remyelination after ischemia. Reactive astrocytes and microglia have been considered to have opposing actions. Microglia release neurotoxic factors, whereas astrocytes release factors that promote neuronal survival and can counteract microglial toxicity (Norenberg, 1994; Giulian et al., 1993; Gluckman et al., 1992). However, in coculture, microglia have been shown to promote astrocyte proliferation (Giulian et al., 1994), and under some circumstances, microglial conditioned media also increases neuronal survival in vitro (Watanabe et al., 2000; Toku et al., 1998). Finally, induction of IGF-1 mRNA after brain injury has been identified in microglia as well as in reactive astrocytes, suggesting that both cell types may contribute to white matter recovery after ischemia (Beilharz et al., 1998; Gluckman et al., 1992).
Furthermore, in the current study the authors also found evidence that oligodendrocytes in the ischemic regions were very closely associated with both reactive astrocytes and microglia. These data are consistent with the previous report of a close association between dividing oligodendrocytes and reactive astrocytes and macrophages/microglia after severe focal ischemic injury in the adult rats, in the periphery of cortical infarcts (Mandai et al., 1997). In the current study, associated GFAP and PLP cells were seen throughout the injured area. This difference likely reflects the global rather than focal nature of ischemia in the current model. However, as in the study of Mandai and colleagues (1997), microglia were primarily seen associated with PLP mRNA in the infarcted areas. This association has been suggested to reflect a key role for the reactive glia in supporting oligodendrocyte recovery and migration into the damaged areas of the brain (Mandai et al., 1997).
Conclusion
White matter damage is an important contributor to disability after hypoxic-ischemic injury. It has been proposed that increased IGF-1 expression by reactive glia after traumatic and hypoxic-ischemic injury may play a significant autocrine/paracrine role in recovery from injury. The present study demonstrates that exogenous IGF-1 can reduce post-ischemic loss of oligodendrocytes and demyelination, probably by inhibiting post-ischemic programed cell death. These data, combined with the increased glial response after IGF-1 administration, and the close association between PLP mRNA expressing cells and reactive astrocytes and microglia, support the hypothesis that reactive glial responses may have a role in limiting white matter injury.
