Abstract
Objective:
To understand the real-world clinical practice patterns and variation in Alzheimer’s disease (AD) diagnostic and screening tool utilization by primary care physicians (PCPs), including tools used for assessing dementia/AD severity and subsequent treatment patterns.
Methods:
This retrospective observational study used de-identified primary care data from electronic medical records (EMR) data provided by the researchers from Queen’s University, Ontario, Canada from August 2011 to August 2021. Individuals ≥50 years old with dementia or AD were identified using AD and dementia-related diagnostic codes, medications, and keywords searched using natural language processing (NLP) and Artificial Intelligence (AI) algorithms from EMR chart notes. Diagnostic and screening tools included scales, neuroimaging, and laboratory tests. Medications examined were cholinesterase inhibitors, memantine, antidepressants, and antipsychotics.
Results:
The study cohort included 417 individuals with all-cause dementia (mean [standard deviation: SD] age: 78.86 [0.19] years), and 71 individuals with AD (mean [SD] age: 76.13 [1.07]). The most-used scale was the Montreal Cognitive Assessment (MoCA; dementia: 53.2%, AD: 84.5%). The mean [SD] frequency of MoCA administration doubled in the year following AD index date compared to the year prior (0.29 [0.82] to 0.67 [1.19] times per patient-year). Severity scores, often unspecified, suggested various stages of cognitive impairment. Among the medications examined, cholinesterase inhibitors were prescribed in 27.8% (n = 116) and 57.8% (n = 41) of people with dementia and AD, respectively. Antidepressants were the most frequently prescribed medication examined (dementia: 49.6%; AD: 71.8%).
Conclusion:
PCPs play an important role in the early detection and management of dementia/AD. As new biomarkers and therapies emerge for early AD, there is a need for connected health system data to guide PCPs through the early diagnostic process.
Introduction
Dementia is a clinical syndrome marked by changes in memory, language, problem-solving, and other cognitive skills, resulting in functional impairment. 1 Between 2011 and 2031, the number of Canadians living with dementia is predicted to double, 2 possibly reaching almost 1 million by 2030. 1 Alzheimer’s disease (AD), the most common and most studied cause of dementia, is a chronic, progressive neurodegenerative disease marked by the accumulation of β-amyloid plaques and hyperphosphorylated tau tangles, leading to synaptic dysfunction and neuronal loss.3 -5 AD progresses on a cognitive continuum with 2 marked clinical stages of prodromal AD or mild cognitive impairment (MCI) due to AD, followed by AD dementia. 6 Biomarker profiles are now defining AD, 7 and disease-modifying therapies (DMTs) for early AD (i.e., MCI due to AD, or mild AD dementia) are emerging to slow cognitive and functional decline.8,9
Detecting AD early is difficult, and the window for detecting, assessing eligibility, and prescribing prior to functional impairment is relatively small. Primary care physicians (PCPs) (general practitioner physicians who deliver services across the entire spectrum of care), as the first point of healthcare contact, play key roles in early detection. The 5th Canadian Consensus Conference on the Diagnosis and Treatment of Dementia (CCCDTD) emphasized the importance of PCPs being attentive for early symptoms.10,11 These guidelines also recommended using validated scales to assess cognition given signs of a cognitive disorder or other risk markers,11,12 but there is no gold standard; scale choice depends on clinical judgment or preference.
Underlying etiologies for dementia are less often explored in primary care than in specialty care. DMTs require AD biomarker confirmation, and most recent diagnostic tests for biomarkers are still exploratory. 13 Recent advances in AD treatment have shifted toward disease-modifying therapies targeting amyloid-β pathology. Anti-amyloid monoclonal antibodies such as lecanemab and donanemab have shown efficacy in slowing cognitive decline in early AD by reducing brain amyloid burden. Symptomatic treatments, including cholinesterase inhibitors and memantine, remain standard for managing cognitive symptoms, while ongoing research explores tau-targeting agents and neuroinflammation modulators.14 -17 Detecting AD for early therapeutics necessitates coordination of care and access to treatments and swift referrals, to improve patients’ quality of life. Moving forward, PCPs will likely take on new roles4,18 together with specialized services.19,20
In general, PCPs deliver first-contact health care services and coordinate patients’ health care services to support continuity of care and ease of movement across the health care system when more specialized services are needed from specialists or in hospitals. As AD detection becomes more time-sensitive, few Canadian studies17,20,21 have examined the current role of PCPs to guide clinical practice in the evolving dementia landscape.
With DMTs emerging, PCPs are expected to play a bigger role in early detection and referral, yet many might be unprepared for this evolving responsibility. We aimed to understand real-world practice patterns surrounding the detection and management of dementia and AD in primary care by studying anonymized electronic medical record (EMR) data from a single academic family practice in Eastern Ontario. Our study objectives were to identify a real-world cohort of individuals with dementia and AD, describe diagnostic tools used in clinical practice, and disease severity scores.
Methods
Study Design and Data
A retrospective observational study of people with dementia and AD was conducted using de-identified EMR data from Queen’s University Family Medicine Restricted Data Environment (QFAMR) between August 1, 2011 and July 31, 2021 (Figure 1). The Queen’s Family Medicine Health Team (QFMT) is an academic Family Medicine Health Team comprising 25 PCPs serving 18,000 patients within the Eastern Ontario Network (EON). EON is a practice-based research network comprising 16 health teams with over 150 PCPs and 200,000 patients.

Study schematic.
Natural Language Processing (NLP) was applied on the unstructured chart note data to identify patients with dementia or AD using dementia and AD related keyword search algorithms. Additionally, algorithms were developed to examine diagnostic and screening tools (e.g., Montreal Cognitive Assessment, neuroimaging) and disease severity scores used by physicians to assess patients’ cognitive, behavioral, and functional capabilities.
NLP algorithms were developed through multiple phases to extract diagnosis dates, diagnosis codes, and patient demographic information. For disease severity, our algorithm was designed to identify the scale scores and to extract the scores if they were in alignment with the valid scale range for comparison with other published work in the literature. To validate our algorithms, manual chart inspection was performed among a random selection of patient charts to compare and report the performance metrics.
The data in the current study were analyzed in a secure research environment which provides robust controls and safeguards to enable the secure transfer of sensitive data into a highly secure environment where it can be stored and analyzed by approved research team members. 22 Only approved researchers from the academic family practice clinic accessed the data within the controlled environment.
The study was approved by the Queen’s University Health Sciences and Affiliated Teaching Hospitals Research Ethics Board with ethical approval registration number 2021QSRC004. Reporting adhered to the STROBE statement. 23 QFAMR database was chosen after a rigorous feasibility assessment of available data sources in Canada.
Study Population
Inclusion Criteria for the Dementia Cohort
Patients ≥50 years of age at index date, with dementia indication based on dementia case definition and at least 1-year pre- and post-index data for identification of clinical characteristics and assessment of screening and diagnostic tools:
Dementia case definition is (Supplement 1):
a. The presence of 1 dementia-related International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) code (including sub-codes) in the Billing, Encounter Diagnosis or Health Condition Tables; OR
b. The presence of 1 dementia-related medication prescription; OR
c. Dementia diagnosis or use of dementia or Alzheimer’s diagnostic and screening tools as indicated in chart notes using NLP based on a key word search within the patient charts, comprising of AD and diagnostic and screening tools, such as the MoCA, MMSE, cerebral fluid analysis, and imaging techniques
Inclusion Criteria for the AD Cohort
Patients ≥50 years of age at index date with AD diagnosis and confirmation after index date, as indicated by either having a minimum of 2 AD diagnostic codes (331.0) in 2 visits at least 1 year apart or AD keywords must have been reported in the patient’s chart a minimum of 5 times within 3 years; and with at least 1 year of pre- and post-index data for identification of clinical characteristics and assessment of screening and diagnostic tools, severity, and treatment.
Exclusion Criteria for the Dementia and AD Cohort
Patients <50 years of age at index date, who do not meet the dementia identification criteria or who do not meet AD-specific identification criteria and with <1 year of pre- and post-index data.
Individuals were identified with dementia if they had 1 dementia-related ICD-9-CM diagnostic code or medication prescription (Supplement 2 21 ). Individuals were identified with AD if they had 1 AD diagnostic code (331.0) recorded on 2 separate visits ≥365 days apart. Dementia and AD case definitions were supplemented by keyword searches of unstructured data using NLP (Supplement 2). A dementia diagnosis or use of a diagnostic or screening tool in the unstructured data was required for inclusion in the dementia cohort. For the AD cohort, AD keywords had to be reported in the chart at least 5 times within 3 years. The index date was defined as the earliest date of meeting eligibility criteria for dementia or AD. For both cohorts, individuals must have been ≥50 years old at index with ≥1 year of pre- and post-index data. Among those with AD, a 2-year washout period identified newly diagnosed (incident) cases.
A sample size calculator informed the validation sample size,24,25 Based on 50% prevalence with expected sensitivity of 80%, specificity of 80%, a 95% confidence interval, and 10% precision. Quality measures included macro-precision, macro-F-score, macro-recall, and accuracy. The total number of patients’ records in the study is 488 (417 individuals with dementia and 71 with AD).
Study Variables and Outcomes
Demographic and clinical variables included age (years) at index, sex, geographic region, and comorbidities of interest based on ICD-9-CM diagnostic codes (Supplement 3). Diagnostic and screening tools identified using NLP included scales of cognition, behavior, and function (Supplement 2; Supplement 4), neuroimaging, and laboratory tests. Dates of administration and outcome of the diagnostic and screening tools were obtained using NLP. Medications included cholinesterase inhibitors (ChEIs: donepezil, galantamine, rivastigmine), memantine, antidepressants, and antipsychotics (Supplement 5).
Statistical Methods
NLP techniques involved a stepwise process of separating words, transforming words, and keyword matching. In-house algorithms verified sentence structure and the dementia/AD keywords. Manual chart validation was conducted using a randomly selected sample of individuals with dementia or AD to determine the accuracy of data extraction and NLP techniques. 25 Manual validation is the process where human evaluators assess and verify the accuracy, quality, or correctness of an NLP model’s outputs. It is often used to ensure that the model is performing well and generating meaningful results.
Baseline characteristics at index were summarized descriptively for the dementia and AD cohorts; comorbidities were assessed in the 1-year pre-index and follow-up periods. Diagnostic and screening tools were summarized as the number and proportion of individuals administered each tool in the 1-year pre-index period. Usage rates for each repeat-administration tool were within 1-year pre-index and follow-up periods and reported per patient-year. Disease severity scores for the AD cohort were extracted at index or within the 1-year follow-up period. Summary measures included mean/median, range, and time from index to first severity assessment (months). The number and proportion of individuals receiving each medication of interest, dosing range, cumulative medication duration, and time to first prescription (months) over the follow-up period were summarized. Those without a subsequent visit for 2 years following their last visit were censored.
All statistical analyses were performed using Python® version 3.79, Sklearn version 0.23.2, pandas version 1.1.3, and NumPy version 1.19.1.
Results
The cohort comprised 417 individuals with dementia and 71 with AD (Figure 2). Everyone with AD was newly diagnosed.

Study cohort inclusion flowchart.
Manual validation of the dementia and AD case definitions included 125 people with dementia and 104 people with AD. The dementia validation sample included individuals who met the dementia cohort criteria as well as those who did not. The AD sample represented all NLP-identified people prior to applying exclusion criteria. For the dementia cohort, the macro-precision, macro-F-score, macro-recall, and accuracy were 1, indicating that the NLP algorithms were able to detect all people with dementia. Since the AD sample only contained positive cases for AD, we combined the AD and dementia data samples together (n = 104 + 125) for the AD cohort evaluation. Quality measure scores for the AD cohort ranged from 0.72 to 0.89, with the highest score for recall, meaning some false positives were detected by the algorithm.
The mean (SD) age of the dementia and AD cohort was 78.86 (0.19) and 76.13 (1.07), respectively (Table 1). Over half were female (dementia: 54.9%; AD: 61.9%), and most were living in urban settings (dementia: 87.3%; AD: 90.1%). The mean (standard deviation, SD) follow-up time in years was 1.24 (1.57) for the dementia cohort and 2.03 (1.94) for the AD cohort. The most common dementia comorbidity within 1 year of index was diabetes (30.2%) followed by anxiety (3.1%) and cardiovascular disease (2.6%). The most common AD comorbidity was diabetes (23.9%) followed by depression (18.3%).
Characteristics of the Dementia and AD Study Cohorts at Baseline.
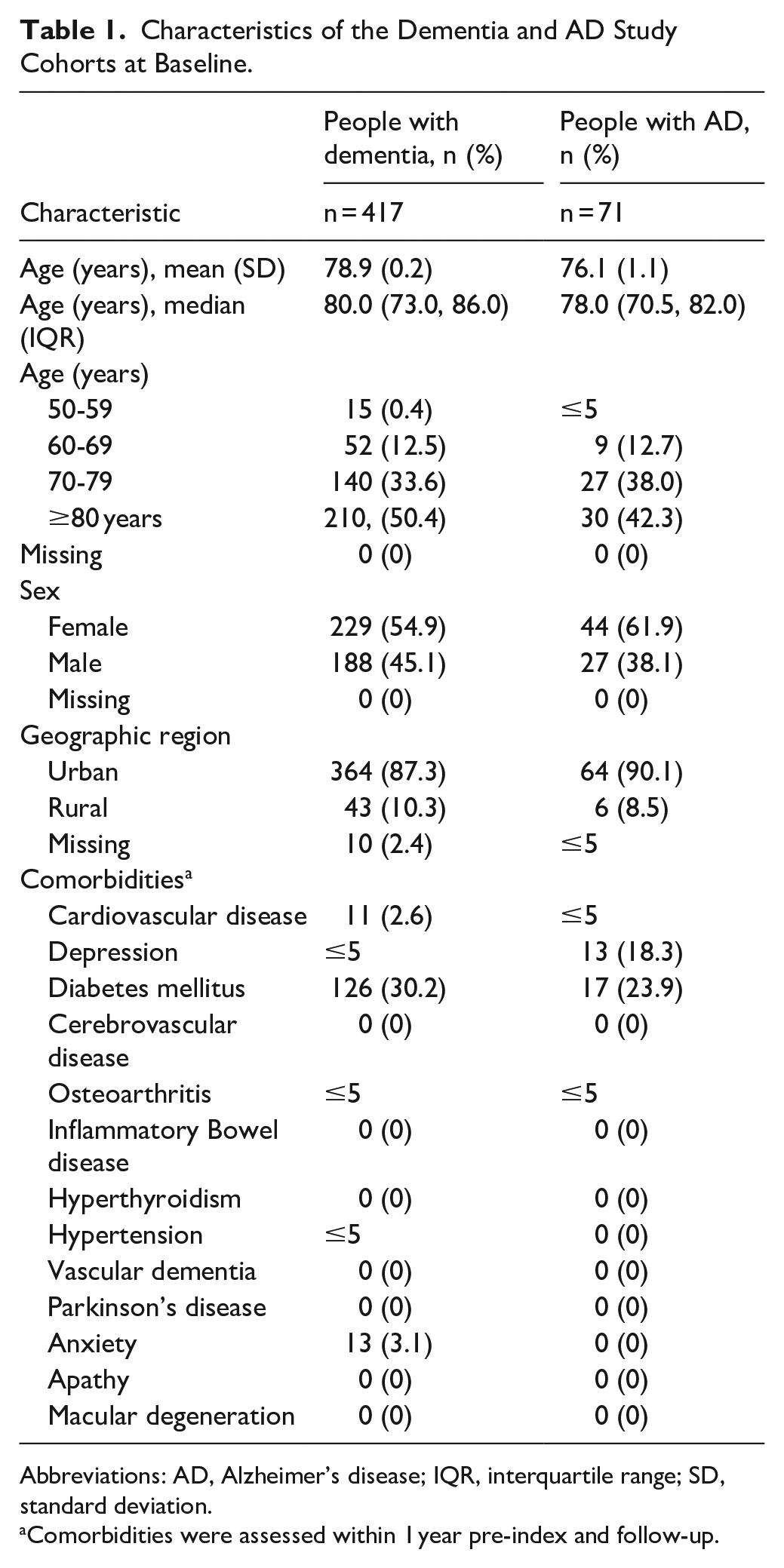
Abbreviations: AD, Alzheimer’s disease; IQR, interquartile range; SD, standard deviation.
Comorbidities were assessed within 1 year pre-index and follow-up.
Scales were commonly used in the 1-year pre-index period (dementia: 65.2%; AD: 90.1%; Table 2). The most-used scales for both cohorts were the MoCA (dementia: 53.2%; AD: 84.5%) and MMSE (dementia: 33.8%; AD: 60.6%). Most people had diagnostic imaging (dementia: 91.9%; AD: 95.8%).
Diagnostic and Screening Tools Identified in EMR in the 1-year Pre-index Period (Dementia and AD Cohort).
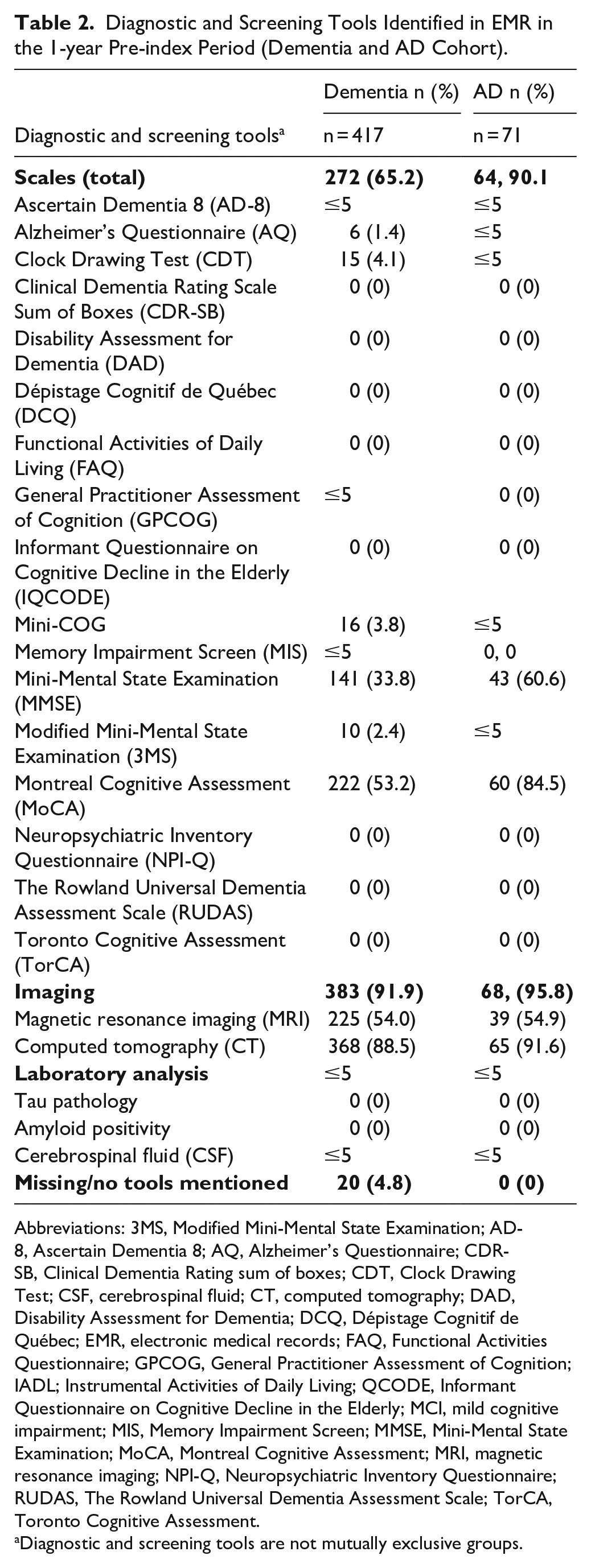
Abbreviations: 3MS, Modified Mini-Mental State Examination; AD-8, Ascertain Dementia 8; AQ, Alzheimer’s Questionnaire; CDR-SB, Clinical Dementia Rating sum of boxes; CDT, Clock Drawing Test; CSF, cerebrospinal fluid; CT, computed tomography; DAD, Disability Assessment for Dementia; DCQ, Dépistage Cognitif de Québec; EMR, electronic medical records; FAQ, Functional Activities Questionnaire; GPCOG, General Practitioner Assessment of Cognition; IADL; Instrumental Activities of Daily Living; QCODE, Informant Questionnaire on Cognitive Decline in the Elderly; MCI, mild cognitive impairment; MIS, Memory Impairment Screen; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; MRI, magnetic resonance imaging; NPI-Q, Neuropsychiatric Inventory Questionnaire; RUDAS, The Rowland Universal Dementia Assessment Scale; TorCA, Toronto Cognitive Assessment.
Diagnostic and screening tools are not mutually exclusive groups.
Compared to the 1-year pre-index period, the mean (SD) frequency of scale administration per patient-year was higher in the 1-year follow-up period (dementia: 0.07 [0.38] versus 0.10 [0.51], Supplement 6; AD: 0.21 [0.73] versus 0.23 [0.95], Supplement 7).
Similarly, the frequency of MoCA and MMSE administration increased for both cohorts. In follow-up, the mean (SD) rate of MoCA and MMSE administration was 2 times higher than pre-index for the AD cohort (MoCA: 0.29 [0.82] versus 0.67 [1.19]; MMSE: 0.13 [0.63] versus 0.24 [1.37]).
Among the scales examined, only 2 had severity scores reported: the MMSE (n ≤ 5) and MoCA (n = 22; Table 3). The mean (SD) score for the MoCA was 19.62 (4.28), consistent with MCI according to the scoring algorithm (range for MCI: 18-25). 25 The mean (SD) score for the MMSE was 14.00 (6.32), well below the “normal” (non-dementia) range of ≥24 out of 30. 16 The mean (SD) time to first severity assessment for the MMSE was 1.93 (4.03) months and 0.60 (1.19) for the MoCA.
Diagnostic and Screening Tools and Disease Severity Identified in EMR in People With AD at Index Or in the 1-year Period Following Index.

Abbreviations: 3MS, Modified Mini-Mental State Examination; AD, Alzheimer’s disease; AD-8, Ascertain Dementia 8; AQ, Alzheimer’s Questionnaire; CDR-SB, Clinical Dementia Rating sum of boxes; CDT, Clock Drawing Test; cerebrospinal fluid (CSF); Computed tomography (CT); DAD, Disability Assessment for Dementia; DCQ, Dépistage Cognitif de Québec; EMR, electronic medical record; FAQ, Functional Activities Questionnaire; GPCOG, General Practitioner Assessment of Cognition; IADL; Instrumental Activities of Daily Living; IQR, interquartile range; QCODE, Informant Questionnaire on Cognitive Decline in the Elderly; Magnetic resonance imaging (MRI); MCI, mild cognitive impairment; MIS, Memory Impairment Screen; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; N/A, not applicable; NPI-Q, Neuropsychiatric Inventory Questionnaire; RUDAS, The Rowland Universal Dementia Assessment Scale; SD, standard deviation; TorCA, Toronto Cognitive Assessment.
Donepezil was the most frequently prescribed ChEI (dementia: 23.5%; AD: 47.9%). A substantial proportion of the dementia and AD cohorts received antidepressants (dementia: 49.6%; AD: 71.8%; Supplement 8).
Discussion
We aimed to understand real-world clinical practice patterns related to dementia and AD in primary care, to determine how future care needs will evolve as DMTs become available. Charts were a feasible data source, and most cases were confirmed through our validation study. However, our objective to describe the range of severity scores was not met since disease severity was often unspecified. Some results aligned with previous literature, such as people with AD tending to be older and female, 4 and the MoCA and MMSE being commonly used scales, 27 but inconsistencies and gaps remained. The fact that most individuals lived in urban settings and all attended the Queen’s Family Health Team (QFHT), where specialist care was more accessible, 28 could partly explain why diagnostic imaging and antidepressants and donepezil prescribing were more frequent.
The MoCA and MMSE are top choices for cognitive screening scales in clinical practice 27 despite numerous alternatives being available (Supplement 4 and others 29 ). The choice of MoCA/MMSE may have been influenced by levels of cognitive impairment. While the scales are correlated, 27 the MoCA is more sensitive for detecting MCI,29,30,31 and the 5th CCCDTD recommends the MoCA specifically for MCI detection. 12 Generally, we found MoCA and MMSE utilization to be more frequent during follow-up than pre-index, possibly because cognitive impairment was identified and subsequently monitored longitudinally.
While EMR-derived severity scores were limited, the available scores implied that some individuals who visited PCPs around the time of diagnosis already had early AD. MoCA mean severity scores were consistent with MCI, 26 suggesting clinicians may have administered this scale when individuals were still able to complete it. In contrast, the MMSE mean scores were lower, suggesting worse cognitive impairment, possibly when individuals were further along the AD continuum (e.g., an event triggered the PCP visit). These patterns align with the 5th CCCDTD recommendation to utilize the MoCA when MCI is suspected and utilize the MMSE for separating moderate dementia from normal cognition.11,12 We could not discern whether other cognitive scales or informant-reported measures were underutilized or underreported.
For people with AD, medications may have been underestimated as these individuals were likely also referred to specialists.32,33 Specialist referrals could explain the short mean follow-up times we observed since those with no PCP visit at the QFHT for 2 years were censored. Nevertheless, ChEI prescriptions gave insight into disease severity and reflected Canadian trends. In 2020, donepezil had the highest ChEI market share (75.8%) followed by galantamine (18.7%) and rivastigmine (5.5%), reflecting prescriber preferences and provincial coverage. 34 All are approved by Health Canada for managing symptoms of mild-to-moderate AD, with donepezil also indicated for moderate-to-advanced AD. 35 Therefore, PCPs were managing AD in its mild-moderate and possibly advanced clinical stages. The NMDA antagonist memantine, intended for moderate-to-advanced AD, 35 is not reimbursed in Ontario and only accessible to individuals paying cash or with third-party coverage.36,37
Antidepressants were prescribed in 77.8% of people with AD, a higher proportion than for depression as a comorbidity in the pre-index and follow-up year (18.3%) and higher than ChEIs (57.8%). This elevated proportion of antidepressants, found previously, 38 may reflect the high neuropsychiatric symptom burden in AD 39 and that antidepressants have demonstrated high-quality clinical trial evidence for treatment of agitation in AD.40,41 Alternatively, the high antidepressant rate may reflect EMRs painting an incomplete picture of AD care.
Our study raises awareness about the importance of the use of validated tools for early detection of dementia and may lead to earlier coordination of care, which could alleviate PCPs’ patient load while improving therapeutic access and quality of life for people living with dementia and AD and their care partners.
Limitations
EMRs are not intended for research, 42 possibly explaining inconsistent reporting and missing values. The dementia case definition used in the study does not allow for reliable differentiation between dementia and non-AD dementias. Furthermore, screening scales were limited to those documented at the QFHT, although they were likely treated in multiple settings. 28 Due to the evolving nature of the use of diagnostic tools/tests due to copyright enforcement there is a need for ongoing research in this space to understand how this is impacting real world diagnostic processes. While cognitive symptoms were the focus of the study, neuropsychiatric symptoms are prevalent across the AD cognitive continuum and almost ubiquitous in AD dementia 43 these symptoms are associated with AD biomarkers and represent sequelae of neurodegenerative disease.41,44,45 The high use of antidepressants and antipsychotics may have reflected this behavioral symptom burden, but without rating scales used to measure or monitor. Future studies should incorporate neuropsychiatric symptoms into protocols as assessment of these symptoms is recommended by CCCDTD5. 14 We could not confirm whether scales or neuroimaging studies, noted for >90% of individuals, were ordered by PCPs since our NLP algorithm could not distinguish this. Future research using enhanced NLP or chart reviews may clarify. Additionally, the case algorithms may have overlooked some people with AD because eligibility criteria were partly contingent on factors under-recorded in EMRs. For example, some PCPs may have been uncomfortable applying AD diagnostic codes and recorded AD <5 times in 3 years. Our findings may not generalize to all primary care practices, especially rural settings. The QFHT, representing approximately 18,000 people, is a single academic family health practice where trainees assess a large proportion of patients and specialist referrals were feasible. Thus, selection bias is a possibility.
Conclusion
PCPs play a role in early and late detection of dementia, facilitating access to therapies. Our study and methods are novel in Canadian literature, but data linkages between primary care and specialty clinics are still needed to understand the patient journey and identify early intervention opportunities for PCPs. With innovations for early AD coming to market, PCPs might feel unprepared for their new, evolving roles. 19 The study raises awareness regarding the need for integrated care approaches for the early identification of dementia and AD patients.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251363156 – Supplemental material for Diagnosis and Management of Alzheimer’s Disease in Primary Care: A Real-World Study in Ontario, Canada
Supplemental material, sj-docx-1-jpc-10.1177_21501319251363156 for Diagnosis and Management of Alzheimer’s Disease in Primary Care: A Real-World Study in Ontario, Canada by Zahinoor Ismail, Melanie Wilson, Hany Khalifa, Daniela Belovich, Eileen Shaw, Tram Pham, Suzanne McMullen, Yuhao Chen, Nafiz Sadman, Jackson Cai, Farhana Zulkernine and David Barber in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
This study was supported by Hoffmann-La Roche Ltd. This study used data from Eastern Ontario Network and Queen’s Family Medicine Health Team. We would like to thank Heather Neilson, who is an employee of Medlior Health Outcomes Research Ltd., for her support in medical writing and editorial assistance.
Ethical Considerations
The study was approved by the Queen’s University Health Sciences and Affiliated Teaching Hospitals Research Ethics Board
Consent to participate
A waiver of consent was sought from the Health Sciences and Affiliated Teaching Hospitals Research Ethics Board (HSREB) at Queen’s University.
Consent for Publication
Not applicable
Author Contributions
ZI, MW, HK, DBe, ES, TP, and SM made substantial contributions to the design of the study and were involved in the interpretation of data and drafting or revising the manuscript for content. FZ, YC, NS, JC, and DBa were involved in the acquisition of data, analysis, interpretation of data, and revising the manuscript for content.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Hoffmann-La Roche Ltd.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ZI is funded by Alzheimer’s Drug Development Foundation, Brain Canada, CCNA, CIHR, NIA, and the Weston Foundation. He has been an investigator on clinical trials of monoclonal antibodies developed by Biogen and Roche and has served on Advisory Boards and Consultancies for Acadia, Biogen, Lundbeck/Otsuka, and Roche. MW, HK, and DBe are employed by Hoffmann-La Roche, who funded this study. MW and DBe hold Hoffmann-La Roche stock. ES, TP, and SM are employed by Medlior Health Outcomes Research Ltd., which received funding for the study from Hoffmann-La Roche. FZ, YC, NS, JC, and DBa are employed by Queen’s University, which also received funding for the study from Hoffmann-La Roche.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
