Abstract
Background:
Alzheimer’s disease (AD) impacts an estimated 7.2 million people aged 65 and older in the United States and remains undiagnosed in numerous additional patients. Earlier detection requires awareness and established care pathways for timely screening, diagnosis, and referrals in the primary care setting.
Methods:
A consensus AD care pathway was developed by 8 multidisciplinary clinicians and piloted across 2 health systems representing 3 geographies, University of North Carolina at Chapel Hill (NC) and Midwestern University (AZ and IL). Twenty primary care practitioners (PCP) completed baseline surveys before receiving disease state and care pathway education. Subsequent 1- and 3-month surveys assessed understanding, confidence, and experience diagnosing, screening, referring, and implementing suggested AD practices.
Results:
At baseline, 15% of respondents felt sufficiently trained on AD referral guidance, and 10% felt trained on the use of noninvasive tests to inform referrals. After 3 months, 85% agreed or strongly agreed receiving sufficient training on appropriate referrals for patients with suspected AD, and 90% agreed or strongly agreed receiving sufficient training on utilizing noninvasive tests to inform referrals and detect AD.
Conclusion:
A standardized AD care pathway improved PCP disease awareness and confidence assessing patients suspected of having AD. Earlier AD detection in primary care may enable prompt intervention and resource accessibility.
Introduction
Alzheimer’s disease, a progressive neurodegenerative disorder that impairs memory, thinking, and behavior, is the most common cause of dementia, accounting for 60% to 80% of cases. 1 An estimated 7.2 million Americans over 65 are impacted by Alzheimer’s disease, making it one of the top 5 contributing causes of death in the United States. With an increasingly aging population, the prevalence of AD is expected to rise. 1
Early diagnosis and intervention of AD are critical for improving patient quality of life, clinical outcomes, and decreasing medical expenditures. 1 The Alzheimer’s Association recognizes the economic burden of AD on patients and the health care system with projected health and long-term care costs for AD-related expenses reaching $384B in 2025, and out-of-pocket costs reaching $97B. 1 In addition to the financial implications of AD, the nearly 12 million caregivers provided 19.2 billion hours of unpaid care for someone with dementia, an estimated value of $413B. 1 Earlier detection of cognitive impairment might allow for advanced planning for patients and their families/caregivers.1,2 Primary care practitioners play a pivotal role in the early detection of AD, often being the first point of contact for patients and their families, and providing continuity of care.1,3,4 However, estimates range that 29% to 76% of dementia is unrecognized in the primary care setting. 2 Despite their role in earlier AD detection, many PCPs report challenges in diagnosing and managing AD. 1 Four of every 10 PCPs reported never or only sometimes being comfortable making a diagnosis of AD or another dementia. In addition, once diagnosed, the patient is often unaware of the diagnosis.1,3 Reasons for nondisclosure of the diagnosis to the patient and their caregivers are multifactorial, such as the degree of the provider’s certainty of the diagnosis of dementia, the degree of insight of the patient, the severity of the dementia, time restraints, and fear of causing emotional distress.1,5
Various legislative and innovative efforts have begun to empower primary care to take on the diagnosis and management of Alzheimer’s disease and related dementias, especially as part of integrated health systems. In 2011, the Affordable Care Act enacted a policy meant to accelerate systemic transformation for those with dementia, by adding the Medicare Annual Wellness Visit (AWV). A requirement of the AWV is an assessment for cognitive impairment. 6 Additionally, in 2024, the Center for Medicare and Medicaid Innovation (CMMI) launched the Guiding an Improved Dementia Experience (GUIDE) Model within health systems to support patients diagnosed with Alzheimer’s disease and related dementias and their unpaid caregivers by requiring coordinated dementia care programs, including care navigation, caregiver education, and respite services for beneficiaries. 7 The GUIDE Model supports team-based, collaborative care through monthly payments to participating organizations. 7 By connecting families to community-based resources and providing caregivers with 24/7 access to a support line, the GUIDE model helps provide a bridge for primary care and health systems to connect people with dementia to critical services, allowing them to remain safely in their homes longer, reduce reliance on institutional care and improving quality of life for both patients and caregivers. 7 The Alzheimer’s Association, a leading dementia advocacy group, continues to drive policy and lead systemic community efforts through education, consensus building, and the development of solutions such as a toolkit with recommendations for cognitive assessment, complete with tests to identify cognitive decline and AD.6,8
Since primary care is most often the entry point for patients, an AD care pathway, combined with peer education, can support PCPs in performing assessments to identify patients with AD by raising awareness of the importance of early diagnosis and providing a timely stepwise guide to incorporate the pathway into clinical practice. This educational feasibility pilot seeks to evaluate PCP awareness of AD and associated care practices, understand the feasibility of implementing a consensus-driven AD care pathway across 2 different health systems, and understand factors impacting provider adoption of care pathways. Findings gleaned from this quality improvement initiative can be leveraged to enhance and scale future implementation of care pathways across diverse care settings.
Methods
Alzheimer’s Disease Care Pathway Development
In May 2023, a multidisciplinary team of 8 clinicians representing neurology, neuropsychology, geriatrics, primary care, and social work convened virtually to discuss existing peer-reviewed literature, pinpoint challenges and opportunities surrounding AD care delivery, and begin designing a framework for an AD care pathway (Figure 1 and Table 1). The expert panel included leaders representing health systems and organizations across the United States, including Advocate Health (Chicago, IL), Mount Sinai Health System (New York, NY), Ochsner Health (New Orleans, LA), Emory Health (Atlanta, GA), University of Southern California Keck School of Medicine (Los Angeles, CA), Atrium Health Wake Forest Baptist (Winston-Salem, NC), Bronson Healthcare (Kalamazoo, MI), and Primary Care Metabolic Group (Winnsboro, SC). Additional Brain Health Leaders Network members facilitated the discussion among the clinicians to support care pathway design. Following this meeting, the panel participants engaged in ongoing correspondence to provide additional feedback and reach consensus on the AD care pathway and evidenced-based screening and diagnostic tools to pilot across primary care settings to test feasibility.
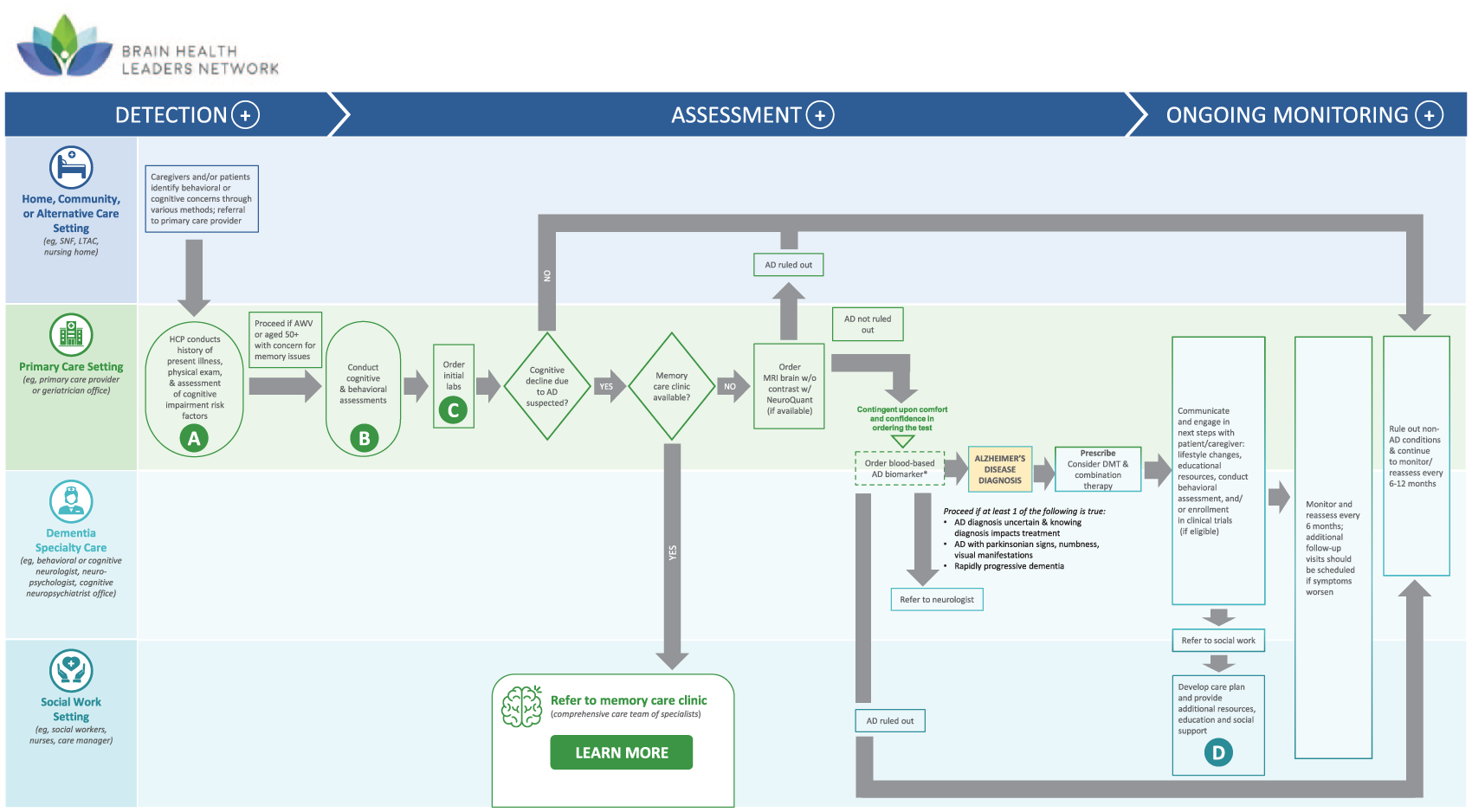
Alzheimer’s disease care pathway overview.
Care Pathway Process Suggestions.

Abbreviations: ADL, activities of daily living; CBC, complete blood count; CMP, comprehensive metabolic panel; HIV, human immunodeficiency virus; IADLs, instrumental activities of daily living; MMSE, mini-mental state examination; MoCA, Montreal cognitive assessment; PHQ-9, patient health questionnaire-9; PHQ-2, patient health questionnaire-2; RPR, rapid plasma reagin; RUDAS, Rowland Universal Dementia Assessment Scale; TSH, thyroid-stimulating hormone.
Consensus Care Pathway Development
The expert panel designed the Alzheimer’s disease consensus care pathway (Figure 1), which includes recommendations for various steps within the pathway (Table 1). The panel outlined 4 key steps labeled A-D and can be triggered either when a patient or caregiver brings a cognitive concern to the PCP or during the Medicare Annual Wellness Visit. During the patient’s visit, the provider assesses cognitive impairment through a thorough history, including medication review for potentially cognitive-impairing medications, and subsequently conducts a cognitive assessment using a primary screening or risk-stratification tool, followed by relevant clinical assessments and laboratory tests to rule out reversible conditions of cognitive impairment. Thereafter, if cognitive impairment is still suspected with no known reversible causes, the panel recommended the PCP either refers the patient to a memory care clinic if resources permit, or orders neuroimaging such as magnetic resonance imaging (MRI). Once AD diagnosis is strongly suspected, a specialist referral can be made; however, if the case is uncomplicated, the patient can continue ongoing management in the primary care setting. Recognizing primary care clinics have unique approaches to managing AD clinical pathways, all steps within the care pathway are suggestions and are to be used based on clinician discretion and can be tailored to an organization’s unique workflows.
Participants
A total of 20 PCPs across the 3 locations participated in the feasibility pilot, including 4 participants from UNC, 3 participants from Midwestern University in Illinois, and 13 participants from Midwestern University in Arizona. For this pilot, PCP is defined as primary care practitioners including physicians (MDs or DOs), physician assistants, and nurse practitioners, with 15%, 20%, and 65% of participants representing nurse practitioners, physician assistants, and physicians, respectively. The participants represented a wide range of practice experience; 35% had up to 5 years of experience, 20% had 6 to 10 years of experience, 15% had 11 to 15 years of experience, 20% had 16 to 20 years of experience, and 10% had greater than 20 years of experience. Participants were identified and enrolled in the pilot through voluntary sampling, and each site was supported by a principal investigator (PI) and/or co-investigator who held clinical and decision-making roles (eg, medical director or division chief).
Feasibility Pilot Go-Live Preparation
Prior to pilot go-live, a study protocol was developed along with the PI and co-investigator, physicians with subject matter expertise, to ensure alignment with institutional workflows; the protocol was reviewed by each site’s research office and received an exemption from the local institutional review boards. The pilot’s educational intervention included each site’s PI or co-investigator delivering a minimum of 1, 1-h educational session at pilot kick-off to the site’s respective participants.
In preparation of the initial education session, a baseline survey was completed by all PCP participants, followed by the education session which included use of an educational slide deck that each site’s PI or co-investigator was able to modify depending on site-specific needs, workflows, and preferences. One site provided 2 additional case study sessions with the participants during the pilot time frame, and participants were encouraged to reach out to their respective investigators throughout the pilot duration with any questions. Information within the standard educational slide deck included an overview of the feasibility pilot purpose and expectations, the consensus AD care pathway, insights into the clinical profile of AD (eg, preclinical AD, mild cognitive impairment due to AD, mild dementia due to AD, moderate dementia due to AD, and severe dementia due to AD). Additionally, insights surrounding instruments to assess for cognitive impairment were included at the institution’s discretion and PI guidance, including insights into the Montreal Cognitive Assessment (MoCA), Functional Assessment Staging Tool (FAST), and Mini-Cog as appropriate. Utilization of any suggested tools or assessment practices were taken based on clinician judgment. Educational sessions were conducted virtually or in-person, based on PI or co-investigator choice. After completing the baseline survey and participating in the initial educational session, participants were asked to use the education and guidance provided in practice when appropriate (eg, during AWV or if patient presents with memory concern). The feasibility pilot went live and piloted across all sites for 3 months from May to November 2024.
Data Collection and Analysis
Surveys were developed in collaboration with the investigators and included a variety of question types to measure both qualitative and quantitative data, including Likert scale, multiple choice, and open-ended responses. Surveys were distributed to participants using Survey Monkey, an electronic survey platform. The questions assessed awareness and understanding of AD and clinical stages, confidence with assessing and diagnosing AD, and screening and referral practices across 3 time points, including baseline (prior to educational intervention), One-month post-intervention, and 3-months post-intervention (Tables 2 and 3). The survey also included open-ended questions during the 1-month and 3-month follow-up periods surrounding barriers and opportunities to adopt the AD care pathway. All participants completed the baseline survey, 1-month, and 3-month follow-up surveys. All data collected were de-identified to ensure participant confidentiality and were subsequently analyzed to understand trends or changes in perception and confidence throughout the duration of the pilot. Each Likert survey question was analyzed for statistical significance with the Kruskal-Wallis H non-parametric test to compare the ordinal dependent variable across the 3 time points. To assess the differences baseline and follow-up timepoints, Wilcoxon rank sum tests were performed with a P-value of .05 (Table 4).
Agreement Assessment Across Baseline, 1-Month, and 3-Month Survey Responses.

Confidence Assessment Across Baseline, 1-Month, and 3-Month Survey Responses.

CT=computed tomography; MMSE=mini mental state examination; MoCA = Montreal Cognitive Assessment; MRI=magnetic resonance imaging; PET=positron emission tomography.
Baseline, 1-month, and 3-month Survey Response Analysis.

CT, computed tomography; MMSE, mini mental state examination; MoCA, Montreal Cognitive Assessment; MRI, magnetic resonance imaging; PET, positron emission tomography.
Bolded text indicates statistically significant data.
Results
Baseline Results: Initial State of AD Care Delivery, Training, and Institution Protocol
The baseline survey distributed to the PCP participants assessed their perceptions on AD care delivery prior to receiving an educational session on AD and the associated care pathway. The initial results revealed varying levels of confidence, agreement, and experience among participants regarding their understanding of disease-specific knowledge, perceptions of prior training, and actions taken when managing patients suspected of having AD. Overall, the baseline findings indicate that while participants consistently conduct cognitive exams and demonstrate some awareness of AD-specific resources, most lack confidence in their ability and training to differentiate between stages of AD, confirm a diagnosis, and have limited understanding of the appropriate protocols for referring patients to specialty care or ordering additional diagnostic tests to support an AD diagnosis. Despite these gaps in confidence and variability in reported practices, the sweeping majority expressed positive responses toward managing patients with AD within the primary care setting and recognized the potential for improved patient outcomes through the adoption of the standardized care pathway.
One-Month Post-Educational Intervention: Initial Impact of Education on AD Awareness and Care Delivery Practices
One month after the educational session, participants demonstrated improvements in their understanding and actions related to AD knowledge, perceptions of their training, and ability to implement their institution’s AD care protocol, relative to the baseline survey results. Participants indicated an 80% increase in agreement with understanding of risk factors for developing AD, with 90% agreeing or strongly agreeing 1-month post-education (P = .022; Tables 2 and 4). Participants’ understanding of the differences across the stages of AD also improved. Findings from the 1-month assessment show that 65% of participants agreed or strongly agreed they understood the stages of AD, compared to 10% at baseline (P = .001).
Statistically significant changes were noted in participants’ perceptions of their training related to AD care delivery. After 1 month, 85% of participants agreed or strongly agreed to having sufficient training on when to refer a patient with suspected AD to a specialist (P < .001), while 75% agreed or strongly agreed that their training was sufficient regarding the use of non-invasive tests to detect AD and inform referral decisions (P < .001). Additionally, participants’ confidence in diagnosing mild cognitive impairment due to AD pathology increased after 1 month, where 45% of respondents reported feeling fairly or completely confident (P = .008).
Although 55% of participants did not report barriers using the care pathway, the remaining 45% identified challenges. Barriers outlined included unfamiliarity with the care pathway protocol, time constraints, and insufficient support staffing. To mitigate these issues, participants proposed future opportunities including ongoing education and EHR integration of the care pathway protocol. Additional solutions focused on expanding cross-collaborative care with the inclusion of social workers, AD specialists to provide consultation within the primary care clinic, ancillary staff involvement in workflows, and support services for patients and caregivers. These findings reinforce the positive impact of the educational session on participants’ awareness and confidence regarding AD screening, identification, and referral practices, while also indicating opportunities to better address barriers.
Three-Months Post-Educational Intervention: Improved Confidence and Reformed Perspectives
While participants demonstrated a sustained increase in confidence in conducting and interpreting cognitive screening exams for AD throughout the duration of the pilot, statistically significant improvements were observed exclusively 3 months post-education in their ability to counsel patients with AD and perceptions of training received for conducting cognitive exams.
Agreement in ability to implement institutional protocol related to AD screening and identification saw a significant rise after 3 months. At the 3-month assessment, 50% agreed or strongly agreed on their ability to use the institution’s protocol (P = .003). Participant responses regarding their ability to counsel a patient about AD showed an 87.5% increase or 75% (P < .001) agreeing or strongly agreeing after 3 months post-education on their ability to counsel patients. Respondents also reported an increase in agreement regarding the sufficiency of the training received on how to conduct a cognitive assessment for cognitive decline, where 90% either agreed or strongly agreed after 3 months (P = .01). In addition to participants’ perceptions of their training and ability to counsel patients, they reported a significant increase in their confidence in conducting and interpreting cognitive screening exams, with 65% of respondents reporting feeling completely confident (P < .001).
Participants also acknowledged a statistically significant reduction over time in facing barriers to implementing their organization’s protocol for screening, assessing, and identifying patients with AD. A decrease of 55% was recorded from the 1-month follow-up where 45% reported these barriers, to the 3-month follow-up, where 20% reported facing these barriers (P < .004). Barriers recognized in the 3-month follow-up reflected those noted in the 1-month follow-up, including time constraints, limited access to support staff and caregiver support, and limited access to imaging services. Reported opportunities to overcome barriers included integration of clinical decision support (CDS) tools into the EHR, such as EHR-embedded cognitive screeners, dot phrases, flow sheets, best practice alerts, and order sets designed to alleviate time constraints, guide care, and prompt PCPs to follow the AD protocol. In addition to EHR tools, participants emphasized the need for ongoing training and support from a multidisciplinary care team, which could allow for specialist consultation, inclusion of ancillary staff, and patient resources within the primary care setting.
Consistency Across Pilot Duration: Care Delivery Behaviors and Perceptions
The impact of the AD care pathway demonstrated meaningful improvement in awareness and perceptions on feasibility, illuminating enhanced ability to use care pathways for screening and identification of patients suspected of having AD in the primary care setting. Although the pilot indicates the positive overall improvement of PCPs confidence, awareness, and knowledge related to the screening and identification of patients suspected of AD, some practices remained consistent over time. While perceptions on confidence surrounding how to conduct cognitive assessments improved over time, self-reported practices for conducting a cognitive assessment remained steady throughout the pilot duration. At both baseline and 1-month post-education, 75% of participants indicated having conducted a cognitive exam within the past month which slightly increased to 80% at the 3-month follow-up. Additionally, self-reported referrals to dementia specialists remained consistent throughout the pilot. The 1-month results showed that 50% of respondents reported having referred a patient to specialists in the last month, whereas 55% of respondents had indicated making a referral in the 3-month follow-up survey. Throughout the pilot duration, participants overwhelmingly agreed (90% at baseline and 1-month post-education; 95% 3-months post-education) that certain patients suspected of AD can continue to be managed within primary care, especially those with mild cases that have access to appropriate resources.
Discussion
At baseline, the data demonstrated large gaps in understanding and awareness of disease risk factors or stages, and a lack of confidence with conducting AD screening and identification steps. Follow-up surveys indicated that after receiving education, participants expressed improved disease awareness, increased confidence conducting and interpreting screening exams, as well as enhanced confidence diagnosing mild cognitive impairment due to AD. Although participants reported improved confidence and knowledge, a portion of participants indicated they continued to experience barriers to incorporating AD screening and identification practices, including:
o Time limitations to conduct cognitive or functional tests
o Limitations interpreting screening tests over time
o Specialist availability for e-consultations
o Limited support staff
o Availability of resources for patients and caregivers
o Access to non-invasive tests such as imaging (eg, MRI, PET, CT)
Some of the barriers identified in this pilot are similar to challenges found in other studies such as limited specialized AD training and support for PCPs and minimal awareness of resources for patients and caregivers. However, participants did not bring forward concerns for barriers commonly cited in other similar studies such as healthcare system limitations including inadequate reimbursement for assessments or optimal cognitive tests for primary care settings.3,9
Participants also noted opportunities to encourage adoption of an AD care pathway in primary care, including:
o Distribution of information to access regional resources, including insights into local memory care clinics and patient or caregiver resources (eg, nursing coverage, caregiver respite)
o Continued clinical education and training for both PCPs and support staff (eg, training support staff on administration of cognitive assessment, e-consultations)
o Sharing educational information for patient and caregiver distribution (eg, coping with an AD diagnosis, treatment, disease progression, and home care)
o Integration of AD care pathway within EHR systems leveraging CDS tools (eg, dot phrases, flow sheets, best practice alerts, order sets, etc.)
Recognizing the rapidly evolving AD diagnostic and therapeutic landscape, participants’ feedback presents opportunities for optimizing future care pathways, including incorporation of clinical decision support tools to streamline processes, a broader understanding of non-invasive tests such as emerging blood-based biomarkers, and tools to facilitate interpretation of test results over time. The development of EHR-enabled clinical-decision support tools can further streamline AD care pathways and reduce the time burden currently placed on PCPs while also improving the quality of referrals to specialists that have lengthy wait times. To scale care pathways more broadly, future consideration could be given to training ancillary team members in administration of cognitive assessments to help address the time constraints many PCPs face in their daily practice. In addition to resources for PCPs, alignment with existing value-based programs or models such as the Medicare AWV or GUIDE may enable interventions that support early detection practices. While this feasibility study spanned 3 months, future research opportunities include measuring long-term outcomes, including but not limited to consistent use of cognitive assessment tools, completion of appropriate tests to rule out reversible causes of cognitive impairment, number of specialty referrals, and number of patients diagnosed with Alzheimer’s disease.
The pilot study had limitations of small participant number (n = 20) and a relatively short duration of follow-up (3 months). With this small sample size, there is potential bias introduced through convenience sampling, as some respondents who chose to participate may have a special interest in AD and related dementias. To understand broader long-term adherence to the care pathway and whether participants exhibited sustained knowledge, additional study participants and intervention duration (within health systems or community-based settings) is needed to study the feasibility of long-term sustainability.
The results of the AD care pathway pilot study reinforce the ability PCPs play as an integral role in the detection of AD across health systems and community settings. Although an overwhelming majority of participants reported that an AD care pathway could facilitate earlier detection of the disease and improve patient outcomes, additional evidence generation is needed to illustrate the impact of the care pathway on provider behaviors and clinical outcomes.
Footnotes
Author Contributions
All authors meet the criteria for authorship stated in the Uniform Requirements for Manuscripts Submitted to Biomedical Journals. ICMJE criteria and provided final approval to submit the manuscript for publication. The authors all contributed to this work, including concept and design, drafting, revision of the manuscript, and final review.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding for this initiative was provided by Novo Nordisk, Inc. Members of the Novo Nordisk, Inc.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The funding for this initiative was provided by Novo Nordisk, Inc. Members of the Novo Nordisk, Inc. Medical Affairs team were silent attendees to consensus discussions and did not contribute to the initiative design, conduct, or writing of the manuscript. In the last 24 months, Dr. Bozoki has received funding as a Site Principal Investigator from: CervoMed (formerly EIP Pharma), Cognition Therapeutics, Alector, and Transposon. She has also received consulting support from Cognition Therapeutics and Creative Biopeptides. In the last 12 months, Dr. Bozoki, Dr. Huntington-Alfano, and Dr. Reeder have received funding from Petauri Kinect for their work on this project.
Data Availability Statement
The datasets used and/or analyzed during this study are available from the corresponding author on reasonable request.
