Abstract
Introduction:
Beyond our population growing older and living longer, there is an increased risk of developing a cognitive disorder. Standardized screening during a routine visit in primary care may be ideal for early detection of mild cognitive impairment (MCI) and follow-up for cognitive changes.
Aim:
This quality improvement (QI) project aimed to determine the impact of implementing the Mini-Cog© quick screening for early dementia detection to identify and follow up on the cognitive impairment of older adults in a primary care clinic setting.
Methods:
Implementation started in February 2024 in a primary care clinic in Southern California. Data was collected for this project over a total of 16 weeks. This QI project implemented a routine cognitive screening using the Mini-Cog©. Cognitive impairment was identified, and if indicated by the Mini-Cog© scores, follow-up for a cognitive assessment and care plan services were initiated. Data were obtained from the project site’s electronic medical record on a total sample size of 471 participants (n = 382 in the pre-implementation group and n = 89 in the post-implementation group).
Results:
Pearson’s chi-square test indicated a statistically significant improvement in the identification rate of cognitive impairment, increasing from 11.8% (n = 45 out of 382) at pre-implementation to 34.8% (n = 31 out of 89) at post-implementation, and specifically, mild cognitive impairment increased from zero identified in pre-implementation to 12.4% (n = 11 out of 89) post-implementation. Lastly, follow-up rates improved from 91.1% (n = 41 out of 45) to 100% (n = 31 out of 31) in post-implementation, and clinical significance was evident based on the phi-coefficient (φ = 0.196), indicating a small effect size and a 100% follow-up rate.
Conclusions:
The findings of this project suggest older adults should receive cognitive screenings to help identify early cognitive impairment and increase follow-up for further evaluation, treatment, and advanced care planning.
Introduction
Millions of Americans suffer from cognitive impairment. 1 When loss of cognition is severe enough to interfere with daily life, it has been given the broad term dementia. 1 Dementia reflects physical changes in the brain that affect a person’s memory and intellectual ability, with Alzheimer’s disease being the most common form of dementia today. 1 As the older adult population (defined as 65 and older) in the United States (U.S.) continues to increase, so does the number of Americans being diagnosed with Alzheimer’s or other dementias. 2 The number of Americans being diagnosed with dementia is expected to grow from just over 50 million in 2020 to over 80 million in 2050. 2 Along with the increased risk of developing Alzheimer’s dementia is the increased burden this disease puts on our patients, our patient’s families, our communities, and our healthcare system. Healthcare costs accrued in 2020 by individuals with dementia were estimated to be $305 billion, an amount up to 20 times greater than older adults without dementia. 2 These are staggering costs to our healthcare system and our society.
Early stages of dementia are often unrecognized in our older adult population due to its difficulty in diagnosing. Many of our older adults are living with multiple chronic comorbidities and polypharmacy, and there is an underuse of cognitive diagnostic assessment tools available for its early detection. 3 Primary care providers (PCPs) may also feel uncomfortable making the diagnosis on observation alone. Furthermore, some older adults may not have the insight into their cognitive deficits and may become proficient at covering these up to their family members and providers; hence, early detection of mild cognitive impairment (MCI) can be easily missed. 2
MCI refers to individuals with cognitive impairment without obvious impairment of their functioning level; therefore, it can be considered an in-between state of typical signs of aging and dementia. 4 Older adults with MCI are at a ten times greater risk of developing dementia than those without MCI, and it represents an early stage of dementia in some older adults. 4 Standardized screening by PCPs during a routine visit may be ideal for early detection of MCI and follow-up for cognitive changes.
Problem Description
The U.S. Preventative Task Force recommends that providers assess cognitive functioning when a patient is suspected of having cognitive impairment. 3 However, there is a lack of attention by caregivers and providers to the issues faced by those suffering from cognitive impairment, with approximately 65% going undiagnosed in primary care, which negatively impacts these patients by not implementing advanced care planning before dementia progresses. 3 This lack of attention can cause severe long-term effects to those suffering as the disease progresses.
Currently, there is a lack of screening and diagnosis of cognitive impairment, and this can partly be attributed to the growing shortage of geriatricians and clinicians who specialize in the complex care of the older adult population. 1 Thus, the burden of diagnosis and follow-up then falls on our PCPs, where there is currently no standard of care in place for cognitive screening, and therefore, it is rarely performed. 1 A recent survey reports that PCPs consider it essential to screen for cognitive impairment. 1 However, fewer than half are currently doing so, with the most common reasons being lack of time and absence of reported or observed symptoms. 1
Time constraints in busy primary care offices are often an issue when diagnosing and managing dementia. The time allotted for a typical office visit makes performing a full cognitive assessment challenging. 3 To assist with these perceived challenges, brief cognitive assessment tools have been designed and tested for early detection of cognitive changes and to decrease diagnostic uncertainty. 5 If the cognitive assessment tool indicates possible impairment, further evaluation by their PCP is warranted. 5 These diagnostic and management concerns highlight the need to optimize dementia care within our primary care system in the U.S.
Early detection, follow-up, and treatment, while not curative, may help to slow down the disease process of dementia, enabling the time required by caregivers, in collaboration with the PCPs, to develop a plan of care that can lighten the symptom burden and access to a broader range of support and treatment options; all while, the older adult still can be involved in the decision-making regarding their care. 6 This collaboration between patients, family members, and providers has long been a gold standard of the U.S. healthcare system. 1 Medicare recommends and supports a cognitive screening for all patients 65 and older; moreover, as of January 1, 2024, Medicare Part B reimburses a separate follow-up visit for a cognitive assessment and care planning services for beneficiaries who show cognitive impairment during a routine visit or as part of the Annual Wellness Visit (AWV). 7
Specific Aim
The project site, a primary care clinic in Southern California, did not have a systematic, routine cognitive screening for assessing cognitive changes in older adults. Before this quality improvement (QI) project, this site relied on clinical observation and the self-disclosure of cognitive changes from patients and their caregivers during regular office visits. Providers at this project site felt this was a diagnostic concern and sought a valid and reliable tool to help objectively identify cognitive impairment among older adult patients. Subsequently, if impairment was found, the hope was that the screening results would open that line of communication between the provider and the patient regarding possible follow-up and treatment for cognitive concerns. To address this gap in practice, a QI project was implemented to determine the impact of implementing the Mini-Cog© on identification rates and follow-up of cognitive impairment in adults aged 65 and older in a primary care office. The clinical question for this QI project was: Does implementing the Mini-Cog© quick screening for early dementia detection improve early identification and follow-up rates for cognitive impairment among adults aged 65 and older in a primary care office?
Ethical Considerations
Before conducting the project, Institutional Review Board (IRB) approval was obtained from the primary investigator’s university. The IRB determined this was a quality improvement project and did not meet the federal definition of human subject research. In this QI project, the intervention was offered to every patient over 65 who came into the project site to be seen within the 8-week implementation period and met the inclusion criteria. The Mini-Cog© quick screening for early dementia detection was implemented as part of routine care for this patient population. It involved no more than minimal risk to the participants involved in this project. Confidentiality of the subjects’ private information was protected according to the HIPPA Privacy Law, and no personal identifying information was collected or stored by the primary investigator. All data were kept on an encrypted external USB drive in a locked office at the project site. Lastly, all participation in this project was strictly voluntary. Thus, the appropriate steps were taken before this QI project began to protect the rights and welfare of the participants.
Methods
This QI project occurred in a primary care office in Southern California over 16 weeks. Before project implementation, 8 weeks of control data were collected, and then 8 weeks of intervention data were collected post-intervention. This primary care office serves a diverse community population in an underserved area of a large metropolitan city. This site is a primary care office whose patient population is an excellent, diverse representation of the city. This site serves an estimated population of 40% African American, 25% Hispanic/Latino, 10% Asian, and 25% Caucasian. Lastly, about 80% of the patient population at this site was over 65. Approximately 200 patients are seen at this site monthly, or nearly 2400 yearly.
After receiving IRB approval and approval from the site’s healthcare providers and owners of the primary care clinic, the primary investigator initiated this QI project. Staff who helped to implement the project were front desk staff, who determined whether the client met the criteria based on the requirements laid out in this QI project to have a cognitive screening, licensed vocational nurses (LVNs) to administer the screening to the participants, and then PCPs to review the screening results and discuss follow-up and treatment if needed. Before implementation, the primary investigator held an educational luncheon with all healthcare providers and staff involved to discuss the implementation protocol and the proper usage of the Mini-Cog©. The education given to the staff on the Mini-Cog© can be located online at https://mini_cog.com/. Step-by-step instructions are available on the website, and a printout was given to each LVN to carry with them on their clipboard or pocket to refer to if they had questions about the screening tool or needed help with scoring.
At this implementation meeting, the team was introduced to the QI project protocol, the rationale for implementing the Mini-Cog©, and the process for collecting data. Convenience sampling was utilized. The inclusion criteria were: any patient coming in for a routine or follow-up visit who was aged 65 or older, did not already have a diagnosis of MCI, dementia, or other cognitive disorder, was able to read and understand English, and had not received a dementia screening within the last year. The exclusion criteria were: Adults under 65 years of age, already had a known diagnosis of MCI, dementia, or other cognitive disorder noted in their chart, were unable to read and understand English, and had already received a dementia screening within the last year.
Intervention
After confirming patient eligibility with the front desk staff, the LVNs conducted the Mini-Cog© screening with older adults in private patient rooms. The PCP then met with the patient following the screening to discuss the results and follow-up. All patients who scored a four and above were considered to have normal cognitive results, and the PCP recommended a repeat screening in 1 year. All patients who scored a three or below were considered to have abnormal results, and the PCP recommended the patient schedule a follow-up appointment for a further, more comprehensive assessment to determine a possible diagnosis and treatment, if any. (See Figure 1) According to the Centers for Medicare and Medicaid Services (CMS), at these follow-up visits, an independent historian must be present for evaluations, and the independent historian can be a family member or someone who can provide patient history when the patient cannot. 7 The PCPs were the ones to relay this vital information regarding their follow-up appointment.

Intervention implementation process.
For this more comprehensive follow-up appointment, this project site decided to follow the recommendations laid out on CMS.gov. This project site then cross-checked those recommendations with the recommendations also laid out by the Alzheimer’s Association website for what is to be included in the follow-up cognitive assessment and care planning appointment.7,8 According to CMS.gov and the Alzheimer’s Association, a complete examination by the PCP focuses on observing cognition and reviewing the patient’s history, reports, and records.7,8 CMS.gov suggests conducting a functional assessment of basic and instrumental activities of daily living, which should include decision-making capacity. 7 The next part of this follow-up appointment is to use a standardized instrument for staging dementia, like the Functional Assessment Staging Test (FAST) or the Clinical Dementia Rating (CDR).7,8 The expectation is for the PCP to then reconcile and review for any high-risk medications the older adult may be on as well as use any standardized screening instruments to evaluate for neuropsychiatric and behavioral symptoms,7,8 which this project site has been using the Geriatric Depression Scale-15 to screen all their patients 65 and older for depression. Lastly, CMS.gov also suggests a safety evaluation for the home and vehicle operation, identifying any social supports, including caregiver support the older adult has, and addressing any advance care planning and palliative care needs. 7
Study of the Intervention
This QI project aimed to determine whether the implementation of the Mini-Cog© would increase the rate of identification and follow-up of cognitive impairment among patients aged 65 and older in a primary care office. The Mini-Cog© quick screening tool is valid and reliable for detecting early cognitive impairment. Screening data were documented and collected through the patient’s electronic medical records (EMRs). First, a retrospective EMR review was completed on all patients aged 65 and older seen during the 8-week pre-implementation. The data collected on pre-implementation were patients with a progress note indicating cognitive impairment or symptoms of a cognitive disorder. This data provided comparative baseline information on older adults who were not screened for cognitive impairment but had a note of cognitive impairment documented in their medical records. Then, the data that was collected post-implementation included (1) the number of patients screened using the Mini-Cog© over the 8-week implementation period, (2) the number of patients who screened with a three or below, and (3) the number of patients who scheduled a follow-up appointment for further cognitive assessment and plan of care with their primary care provider (See Figure 2).
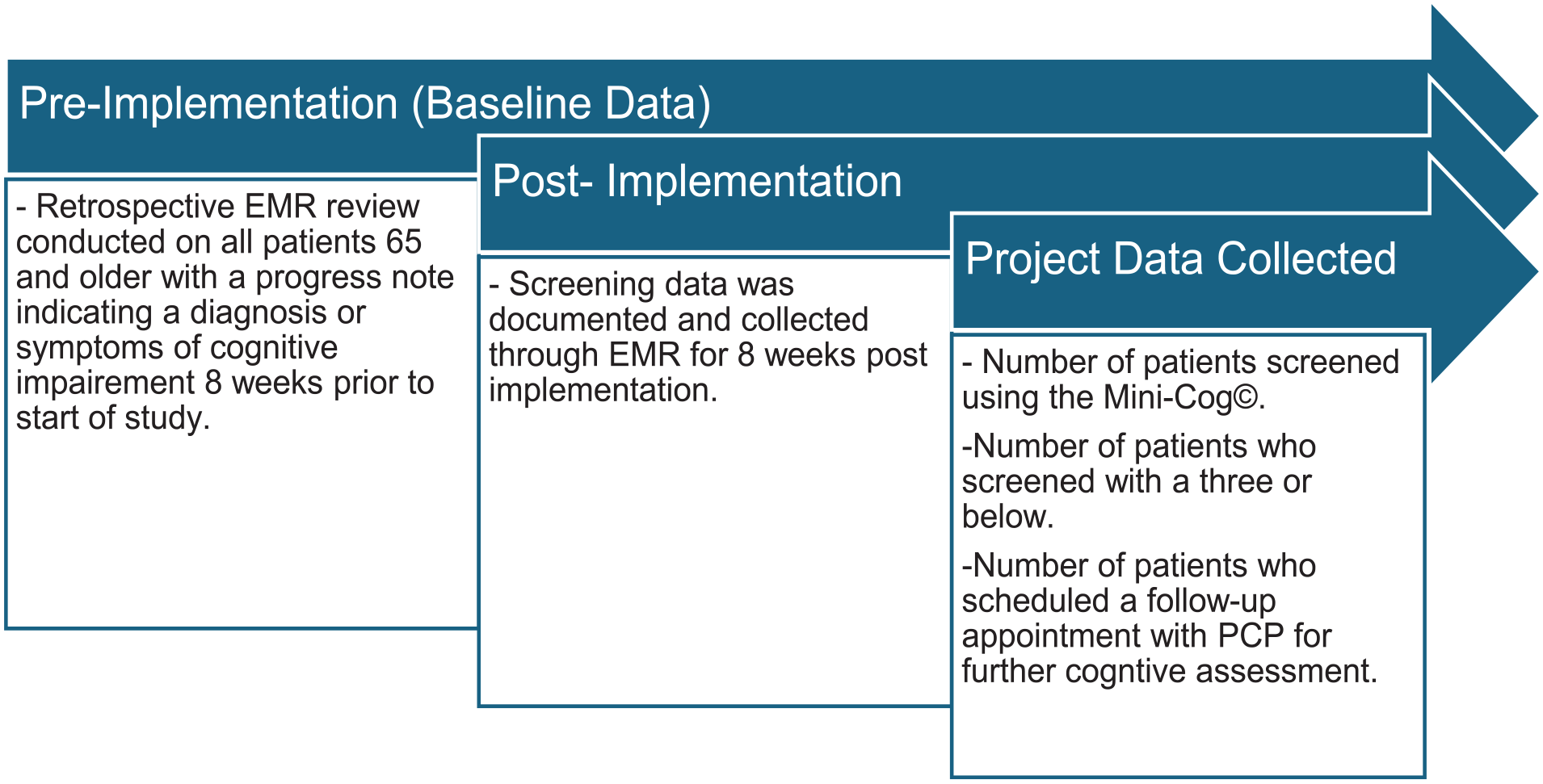
Data collection process.
Measures
The Mini-Cog© is a cognitive screening tool initially developed in 2000 by Borson et al 9 The screening tool combines a three-word recall with a clock drawing test. 9 It was designed as a brief test to identify cognitively impaired individuals from non-cognitively impaired individuals in a community sample of culturally, linguistically, and educationally diverse older adults. 9 In 2003, Borson et al conducted a study that compared the Mini-Cog© with the Mini-Mental Status Exam (MMSE) and the Cognitive Abilities Screening Instrument and found that in a population of almost 250 older adults, the Mini-Cog© had the highest sensitivity (99%) and correctly classified the highest percentage of participants. 10 Furthermore, the time to administer the Mini-Cog© was 3 min versus upwards of 7 min for the MMSE. 10 After conducting this study, Borson et al concluded that the Mini-Cog© was ready to be incorporated into primary care or general practice settings as a routine cognitive vital sign measure. Many studies have since been conducted using the Mini-Cog© since Borson et al ’s first study and found the Mini-Cog© sensitivity to be between 76% to 99% and specificity to be between 89% to 96%.4,9 -14
The Mini-Cog© can have a total score of five, with three points associated with the recall task and two points related to the clock drawing task. Borson et al determined that a score of 0, 1, or 2 suggested a high likelihood of cognitive impairment, and a score of 3, 4, or 5 indicated a low probability of cognitive impairment, with a score of 3 being suggestive of a possible mild cognitive impairment or early stages of cognitive impairment.9,10 Because of this finding, and for the purpose of this QI project to also identify mild cognitive impairment, the chosen cut-off between normal and abnormal results was a score of 3.
Ultimately, the Mini-Cog© was chosen for this project over more widely used tools like the MMSE or the Montreal Cognitive Assessment (MoCA). Although the MMSE has a brief administration time of only 5 to 10 min, it is not sensitive to MCI. 4 The MoCA is known to have a higher sensitivity to MCI but takes much longer to administer (10-15 min) and is unsuitable for an already busy outpatient primary care setting. 4 The Mini-Cog© only takes an average of 3 min to administer and is readily accepted by patients and providers, as it has a higher sensitivity and specificity of identifying MCI in patients than the MMSE; therefore, it makes an excellent, valid, and reliable tool for identifying cognitive impairment in outpatient settings like primary care. 4
Data Procedures and Analysis
The patients’ demographic data on sex, age, race/ethnicity, and Mini-Cog© screening scores were collected from the EMR and de-identified by the project site office manager. Data were also collected on any scheduled follow-up appointments for further neurocognitive testing and plan of care if indicated and de-identified by the site’s office manager. All data were input into a Microsoft Excel spreadsheet, and the IBM Statistical Package for the Social Sciences (SPSS) version 29 software was utilized to analyze the data. Before starting this study, the primary investigator ran a sample size test using G Power 3.1.9.7 with the parameters set of 95% power, a moderate effect size (w = 0.30), and an alpha set to 0.05 for a chi-square test. The result was a total minimum sample size of N = 145. Pearson chi-square tests were conducted to answer the clinical question for this QI project: Does implementing the Mini-Cog© quick screening for early dementia detection improve early identification and follow-up rates for cognitive impairment among adults aged 65 and older in a primary care office? Statistical significance was set to 0.05.
Data Analysis Results
Analysis of the data on cognitive disorder identification using Pearson chi-square tests showed that the post-implementation identification rate increased significantly, from 11.8% (n = 45 out of 382) at pre-implementation to 34.8% (n = 31 out of 89) at post-implementation. Statistical significance was shown, X2 (1, N = 471) =28.34, P < .001. Follow-up appointment rates also improved, from 91.1% (n = 41 out of 45) at pre-implementation to 100% (n = 31 out of 31) at post-implementation, although it was not statistically significant, X2 (1, N = 76) =2.91, P = .090. (See Figure 3) However, clinical significance was evident, based on the phi-coefficient (φ = 0.196), indicating a small effect size and a 100% follow-up rate for patients in the implementation group.
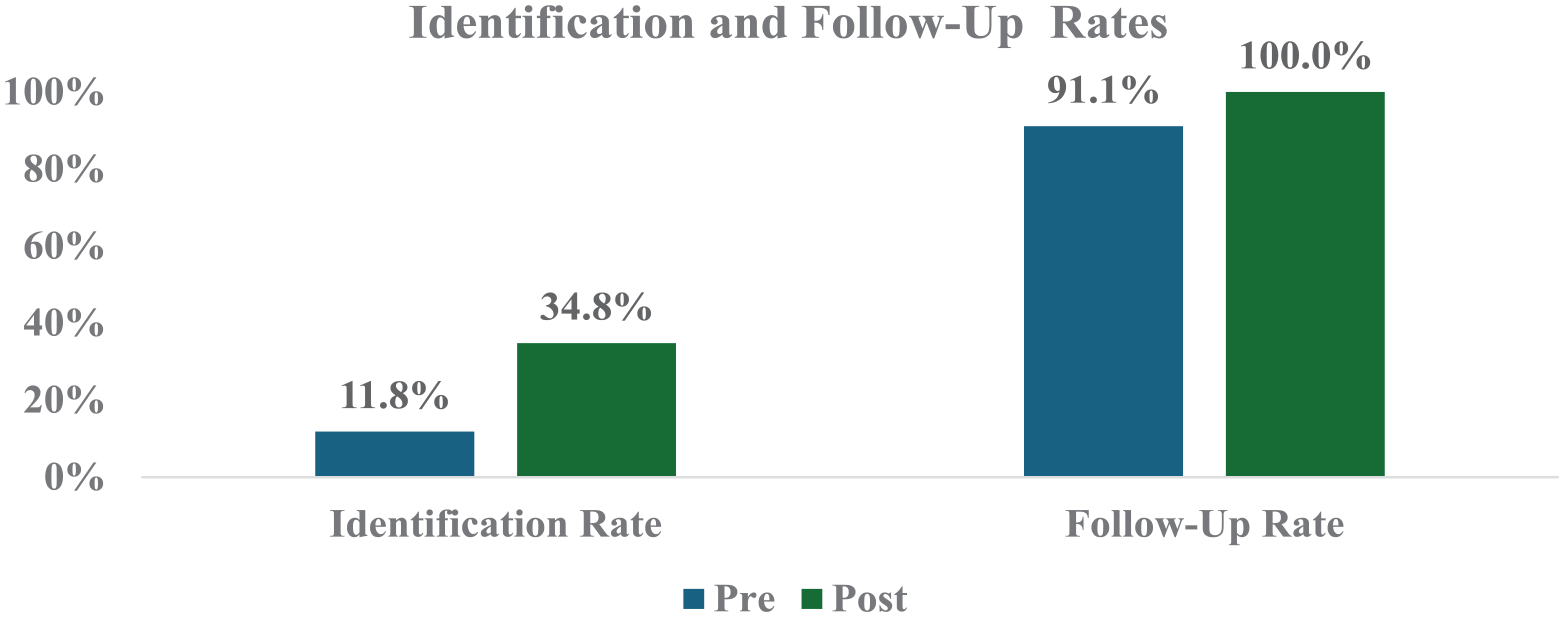
Identification and follow-up rates.
When evaluating according to the degree of cognitive disorder, zero patients with mild cognitive impairment were identified during pre-implementation. This increased to 12.4% (n = 11 out of 89) post-implementation. Thus, the implementation was able to significantly improve the identification of mild cognitive disorder at a substantially higher rate post-implementation (12.4%), compared to pre-implementation (0%), X2 (1, N = 471) =48.34, P < .001. Patients with mild cognitive disorders were not identified or followed up during pre-implementation. In contrast, during post-implementation, there were 11 patients identified with mild cognitive disorder, and all (100%) had a scheduled follow-up in primary care. Moderate/severe cognitive disorder was identified for 11.8% (n = 45 out of 382) at pre-implementation and increased to 22.5% (n = 20 out of 89) at post-implementation. (See Figure 4)

Identification and follow-up rates according to degree of cognitive disorder.
The Mini-Cog© scores, according to cutoff scores of 2 or lower, 3, and 4 or higher, were computed for demographics. Table 1 displays the Mini-Cog© scores by demographics (age, gender, and race). The intervention group consisted of 89 patients. Most were female (67.4%, n = 60); the most common age range was 65 to 74 (n = 41, 46.1%). Race was diverse, with 46.1% (n = 41) of Black/African American patients, 30 (33.7%) Caucasian patients, and 11 (12.4%) Hispanic/Latino patients.
Mini-Cog© Scores According to Gender, Age, and Race.

For gender, the male and female distribution was similar among the scoring categories, X2 (1, N = 89) =1.01, P = .603. For age, the patients aged 85 and older had a higher proportion of patients than those younger age categories for scores of 3 or lower, whereas they had the lowest proportion of patients with a score of 4 or 5. The other age groups had similar distributions among the score categories, and no significant differences were identified between any of the age groups, X2 (1, N = 89) =8.48, P = .075. There were no Hispanic/Latino patients with a score of 2 or lower for race. Most Hispanic/Latino and Asian/Pacific Islanders scored 4 or 5 (at least 80%), whereas Caucasian and Black/African American patients showed only 57% and 63%, respectively, with scores of 4 or 5 on the Mini-Cog©. No significant differences were identified, X2 (1, N = 89) =6.32, P = .389.
Discussion
Pearson chi-square tests showed statistical significance in increasing identification rates of older adults with cognitive impairment, both in mild and moderate/severe cognitive impairment rates. Before implementing the Mini-Cog©, this project site did not identify any mild cognitive impairment in older adults, as seen in the pre-implementation data. Then, once the Mini-Cog© was in place, early changes in cognition were identified, along with an increase in some moderate to severe cases that were not identified on observation or self-disclosure prior. Next, clinically significant, all agreed to a follow-up visit with their primary care provider to complete a full cognitive assessment and care planning services. The demographic assessment of Mini-Cog© scores showed that patients aged 85 and older were more likely to demonstrate cognitive disorder, and Caucasians and Black/African Americans had the highest proportion of patients with scores of 3 or lower on the Mini-Cog©. Gender differences were not identified across Mini-Cog© scoring categories. However, no statistically significant differences in cognitive disorder scores were found for categories of gender, age, or race. These findings were a novelty to this study and added to the literature in that cognitive disorders can be seen across all races, ages, and genders.
The results of this project support the use of the Mini-Cog© to allow for early identification of cognitive disorders and prompt intervention for a more comprehensive follow-up appointment to reduce the worsening of symptoms, along with early implementation of advanced care planning. The results suggest that the answer to the clinical question in this QI project was affirmative: screening for cognitive impairment in adults 65 and older in a primary care office did result in early identification and follow-up of cognitive impairment. The results of this QI project align with published literature and recommendations by the USPSTF and Medicare on the benefits of routinely screening for cognitive impairment in the older adult population in primary care settings.1,3,5 -7
Similar studies found in the literature by Brooks et al and Fernandes et al support this QI project’s findings to screen for cognitive impairment in primary care.1,3 Limitations mentioned in the Brooks et al 1 study included provider buy-in, stating that although all providers initially agreed to participate, two of their providers completed little to no screenings. 1 Another limitation mentioned in the Brooks et al 1 study was that most participants screened were White; therefore, the generalizability of their results to other populations was limited. 1 In this QI project, nurses were the ones who were instructed, educated, and utilized to conduct the Mini-Cog© screening, allowing nurses to work at the top of their scope of practice and allowing PCPs to focus on the results and follow-up. This QI project was also conducted on a very diverse population, as shown in Table 1, allowing for more generalizability to other populations.
From a financial and treatment perspective, the Centers for Medicare and Medicaid Services encourages a separate visit to assess a patient’s cognitive function more thoroughly and develop a care plan. 7 This visit can be billed once every 180 days per Medicare Part B recipient. 7 Then, during this more comprehensive follow-up visit, possible treatments, such as anti-amyloid therapies, can also be discussed. Anti-amyloid therapies have been FDA-approved for the treatment of Alzheimer’s, a dementia-related disease.15,16 Anti-amyloid therapies are the breakthrough agents that have been shown to slow down the inevitable progression of dementia.15,16 Therefore, early identification, diagnosis, and follow-up with treatment is critical.
Lastly, this QI project suggests that changes in cognition, especially early changes, can be challenging to identify without a routine cognitive screening, using a valid and reliable tool like the Mini-Cog© used in this project. However, once it is brought to the patient’s and provider’s attention, both were willing to follow up with a more thorough assessment and possible diagnosis. By implementing this screening and identifying the cognitive concerns early, interventions such as treatment and advanced care planning, including an advanced directive and creating a physician order for life-sustaining treatment, or POLST, can all be put into place. Having these interventions implemented and documented in the patient’s chart can ensure the healthcare treatment older adults receive in their later years is consistent with their wishes and preferences. 5 All to increase older adults’ quality of life, both mentally and physically, during their golden years.
Limitations
This QI project had five significant limitations. First, the sample was obtained from a single metropolitan primary care clinic site. This single site and small sample size, which excluded all non-English speaking patients and all older adults who already had a known cognitive disorder, significantly affect the generalizability of these findings. This leads to the second limitation, which was that this project was completed on an underserved population, which could have exaggerated or understated the intervention outcomes of this study as opposed to it being completed on a well-served population. The third limitation was the timeframe in which this QI project was completed. The 16-week period (8 weeks to collect pre-implementation data and 8 weeks for post-implementation) did not allow enough time to measure the completion of follow-up appointments and advanced care planning.
The fourth limitation was the design of this QI project. The comparison and implementation groups were sampled by convenience, not by random assignment, and were not of equal size; therefore, there may have been other unexplored differences between the groups. 17 Because of this limitation, this project only shows an association link between implementing the Mini-Cog© and the increase in identifying cognitive impairment in older adults. The fifth limitation of this project was the lack of patient-level data collected. Instead of patient-level data, aggregated data was collected, so a regression analysis could not be performed.
However, what can be reported is that only 11.8% of 382 adults 65 and older seen during the 8 weeks before the intervention had medical documentation of a cognitive disorder or cognitive impairment. Based on the results of this QI intervention, there is a significant under-reporting of cognitive impairment or cognitive symptoms when older adults are not screened. With this finding, this project still adds insight to the literature on increasing the identification rates for cognitive disorders in the older adult population within the primary care community.
Conclusion
Despite the limitations mentioned above, the results of this QI project are still significant. At the project site, increasing the identification and follow-up of older adults with cognitive impairment was achieved and sustained. Moreover, increasing identification and follow-up rates leads to a more extensive cognitive assessment with diagnosis, treatment, and advanced care planning to improve patient outcomes and overall quality of life. This QI project suggests recommendations for clinical practice and future projects.
Clinical Practice Implications
This project’s first and most crucial implication is that all adults 65 and older should routinely receive a cognitive screening using a valid and reliable tool, like the Mini-Cog©. Implementing this cognitive screening as part of a standard intake or annual wellness visit would be a proactive approach to cognitive decline. At the very least, this screening should be completed annually to detect cognitive changes so that interventions can be initiated early. Per Medicare recommendations, a second implication is that primary care providers can and should provide the follow-up cognitive assessment and care plan services appointment. 7 Then, the primary care provider can refer the patient to a specialist if found to be warranted during their follow-up cognitive assessment.
Recommendations for Future Quality Improvement Projects
First and foremost, a longitudinal project that includes a comprehensive cognitive assessment and care planning follow-up appointments should be attempted. Medicare is launching a relatively new initiative to recommend a separate cognitive assessment and care planning appointment in primary care, so it would be of great interest to see how many older adults complete this assessment and then complete the advanced care planning with their providers and family members. It would be interesting to see how completing this assessment affects the care they receive in their later years.
Furthermore, a study on a well-served population should be conducted to see if the intervention outcomes of that study are comparable to the outcomes of this study, which was done on an underserved population, as mentioned in the limitation section. Another suggestion would be to collect patient-level data so that regression analysis can be performed to more reliably assess the predictive value of demographics and clinical characteristics regarding risk, meaning low scores on the Mini-Cog©. In general, a more comprehensive project on a larger population at multiple primary care sites would address more variables, like treatments and referrals to specialists, and how implementing this screening and cognitive assessment affects overall medical costs and the quality of life of older adults. This follow-up over a more extended period to measure the patient outcomes of older adults would be valuable in guiding primary care practice for the future.
Footnotes
Acknowledgements
Keirstin E. Uomoto would like to thank Dr. David Kattan and Dr. Kirk Uomoto’s primary care office, where the data was collected, and their support.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
