Abstract
To determine the effects of Tacrolimus (FK506) on Treg cells and subpopulations in SSc patients and assess the ability of FK506 to modify the immune imbalance of Treg/Th17 cells. We analyzed PBMC from five SSc patients and six healthy control by flow cytometry after cultured with 0, 0.1, 1, or 10 ng/ml FK506 in vitro. The number of Treg cells decreased in SSc patients treated with FK506. The number of FrI cells were decreased in SSc following FK506 treatment. The drug did increase the frequency of FrII/Treg cells, but not FrII cells. However, FK506 significantly decreased FrIII in both SSc patients and controls. FK506 clearly decreased the numbers of Th17 cells and FoxP3+IL-17+ cells. The proliferation capacity of cells was also inhibited by FK506, which had a greater effect on FoxP3− cells than FoxP3+ cells. FK506 did inhibit the proliferation of FrIII cells, but not FrI or FrII cells. Our study provides that FK506 reduced the number of FoxP3low CD45RA− T cells (FrIII) by inhibiting its proliferation. Therefore, FK506 modifies Treg cells and the immune imbalance between Tregs and Th17 cells in SSc patients.
Introduction
Systemic sclerosis (SSc), also called scleroderma, is a complex immune-mediated rheumatic disease that can be defined as a combination of microvascular damage, slow-developing fibrosis, and an abnormal immune system. 1 Effective treatments for SSc are currently not available, and standard of care options that target inflammation, immune system imbalance, and fibrosis are largely ineffective. 2
FK506 (Tacrolimus), which is a macrocyclic lactone, acts as a calcineurin inhibitor, which is over 100 times more potent than ciclosporin A (CsA) at inhibiting T cell proliferation, and its effectiveness as an immunosuppressant in transplant patients.3,4 FK506 down-regulates E-selectin, ICAM-1, and VCAM-1 expression.5,6 The endothelial cells (ECs) from donors survived the allograft while FK506 was effective.7,8 Transforming growth factorβ (TGFβ) plays a prominent role in the fibrosis of skin and internal organs in SSc patients.9–11 Importantly, FK506 significantly reduced the TGFbeta1-induced expression of type I procollagen protein, gained the anti-fibrotic effect by inhibiting TGFβ.12–14
In the past, FK506 was often used to treat localized scleroderma in the form of topical ointment,15–20 while recent studies have found that it is also effective for systemic sclerosis.4,21 FK506 has a prominent effect on the pathogenesis of SSc by inhibiting the immune response, protecting the vascular endothelium, and inhibiting the development of fibrosis. Thus, FK506 is a promising drug for the treatment of SSc.
Immunological activity, especially that of T lymphocytes, is considered to be a key stimulus in promoting the vascular abnormalities and fibrosis observed in SSc patients.22,23 The regulatory T cell (Treg) is indispensable for the maintenance of dominant self-tolerance and immune homeostasis. Most reports have shown that the ratio of Treg was elevated in the peripheral blood mononuclear cells (PBMCs) compartment in SSc, while others have reported normal or decreased Treg levels with or without dysfunction.24–29 Our previous work and recent report showed that the number of Treg cells is elevated in SSc patients together with dysfunction of the peripheral blood mononuclear cell (PBMC) and skin compartment.30,31 CD4+ T cells are also comprised of Th17 cells that secrete IL-17A and IL-17F, which can induce inflammation. 32 Interestingly, both Treg and Th17 levels are elevated in SSc patients. In fact, immune imbalance between Th17 and Treg cells is a well-documented characteristic in SSc patients.33–35 Miyara et al. 36 and Sakaguchi et al. 37 defined the subtypes of Treg cells based on the expression of FoxP3 and CD45RA, including subtypes such as CD4+CD25+FoxP3lowCD45RA+ (FrI), CD4+CD25highFoxP3highCD45RA− (FrII), and CD4+CD25+FoxP3lowCD45RA− (FrIII). The FrII subtype consists of activated Treg (aTreg) cells, which have a suppressive function. The FrI subtype consists of resting Treg (rTreg) cells, which can convert to aTreg cells, while the FrIII subtype consists of T cells that are not suppressors, can produce IL-17, and hence have Th17 potential. Therefore, FoxP3low CD45RA− (FrIII) T cells are not the real Treg cells.
We have shown that decreased aTreg (FrII) levels and their functional deficiency together with an increase in FoxP3lowCD45RA− T cells are responsible for the increased levels of dysfunctional Tregs in SSc patients, which potentially cause the immune imbalance between Treg and Th17 cells. 31 In this study, we confirmed that FK506 can reduce the level of FoxP3low CD45RA− T cells and augment aTreg levels by inhibiting FoxP3low CD45RA− T cell proliferation, which can modify the abnormal Treg levels and immune imbalance between Treg and Th17 cells in patients with SSc.
Materials and methods
Study subjects
Five Chinese patients with SSc who fulfilled 2013 ACR/EULAR (American College of Rheumatology/European League) classification criteria 38 were chosen for this prospecive experimental study. All patients in this study had new-onset SSc and had not received any previous treatment (Table 1), and excluded potentially confounding comorbidities such psoriasis, spondyloarthritis, inflammatory bowel diseases, etc. The study consisted of 3 dcSSc and 2 lcSSc patients (two males and three females) with an average age of 36.60 ± 15.04 year and average disease duration of 1.80 ± 1.92 year. Six age- and sex-matched healthy volunteers were enrolled as controls. This study was approved by the Medical Ethics Committee of Peking Union Medical College Hospital and the Ethics Committee of EUSTAR. The methods used in this study were in accordance with the approved guidelines. All patients enrolled in this study provided written informed consent for the samples collected for the research study protocol.
Participant demographics and clinical characteristics.

F: Female; M: Male; dcSSc: diffuse cutaneous SSc; lcSSc: limited cutaneous SSc.
Antibodies
Anti-human mouse antibodies (conjugated with FITC, PE, PerCP, allophycocyanin (APC), Alexa Fluor 488, or PE-cyanin 7 (PE-Cy7)) used in this study were as follows: CD4-PerCP (OKT4, Biolegend), CD25-APC (BC96, eBioscience), FoxP3-Alexa Fluor 488, FoxP3-Alexa Fluor647, (PCH101, respectively, eBioscience), CD45RA-PE, CD45RA-PE-Cy7 (HI100, L48, respectively; BD Pharmingen), and IL-17-PE (eBio64DEC17, eBioscience). A control antibody of the respective IgG isotype was included in all experiments.
Flow cytometric cell phenotypic analysis
The peripheral blood from patients and healthy individuals was collected and PBMCs were prepared using Ficoll gradient centrifugation. Cells (5 × 105) were incubated with 0, 0.1, 1, or 10 ng/ml FK506 for 72 h and then washed with PBS containing 2% fetal calf serum. The cells were then incubated with anti-CD4, -CD25, and -CD45RA antibodies in the dark at 4°C for 30 min. Subsequently, intracellular FoxP3 staining was performed after fixation and permeabilization (eBioscience). For the detection of intracellular cytokine production, cells were stimulated with 20 ng/mL PMA and 1000 ng/mL ionomycin in the presence of Golgi-Stop (BD Biosciences) for 5 h and then incubated with anti-CD4, -CD25, -CD45RA, -FoxP3, and -IL-17 antibodies after fixation and permeabilization (eBioscience). Stained cell images were acquired using a FACSCalibur flow cytometer (Becton Dickinson) and analyzed using FlowJo v.7.6.4 software (Tree Star).
Cells sorting and proliferation assay
PBMCs were stained with anti-CD4, -CD25, and -CD45RA antibodies in the dark at 4°C for 30 min. and then sorted using a FACS AriaII flow cytometer (Becton Dickinson). RPMI 1640 medium supplemented with 10% fetal bovine serum, 100 IU/mL penicillin, and 100 mg/mL streptomycin was used for cell culture. Ten thousand sorted cells were labeled with 1 μM CFSE (Invitrogen) from patients and control donors. One hundred thousand PBMCs from the same healthy donor were incubated with 25 μg/mL mitomycinC (MMC) at 37°C for 30 min and then added to the co-cultures as feeder cells. Co-cultures were stimulated with 0.5 μg/mL anti-CD3 (OKT3, BD Pharmingen) and 1 μg/mL anti-CD28 (CD28.2, BD Pharmingen) antibodies in 96-well round-bottom plates supplemented with RPMI medium, and then incubated with 0, 0.1, 1, and 10 ng/ml FK506 for 72 h, respectively. Proliferation of CFSE-labeled cells was then assessed by flow cytometry.
Measurement of cytokine levels in serum and culture supernatants
The human Th1/Th2/Th9/Th17/Th22 13plex FlowCytomix Multiplex Kit (Bender MedSystems, eBioscience thermo) was used for high-throughput immunoanalysis of cytokines in the sera and supernatants by using flow cytometry. All assays were performed according to protocols specified by the manufacturers and with the respective reagents (diluents, calibrators, blocking reagents, and detecting-antibody mixtures) included with their kits.
Statistical analysis
Data were analyzed with the SPSS v.17.0 statistics software (IBM, USA). Variables were summarized as counts and percentages or as medians and ranges. The independent samples
Results
FK506 decreases the relative number of Treg cells and FoxP3low CD45RA− T cells and increases the number of aTreg cells
The number of CD4+CD25+FoxP3+ Treg cells in PBMC samples obtained from SSc patients decreased after treatment with 1 or 10 ng/ml FK506 compared to treatment with 0.1 ng/ml (

The effect of FK506 on Treg cell and its subtypes on the SSc patients: (a) a representative flow cytometry analysis of CD25+ FoxP3+ cells of CD4+ gated dot-plot of the indicated conditions in healthy controls (
The number of FrI/CD4+ cells within the samples obtained from SSc patients was reduced after treatment with 10 ng/ml FK506 compared to those treated with 0.1 ng/ml FK506 (
FK506 did not induce significant effects on the FrII/CD4+ cell population obtained from SSc patients compared to healthy controls (
In SSc patients, the number of FrIII/CD4+ cells decreased after treatment with 10 ng/ml FK506 compared to untreated controls (
The number of Th17 and FoxP3+IL-17+ T cells decreases after treatment with FK506
The number of CD+IL-17+ cells in samples obtained from healthy controls decreased after treatment with 10 ng/ml FK506 compared to untreated controls (
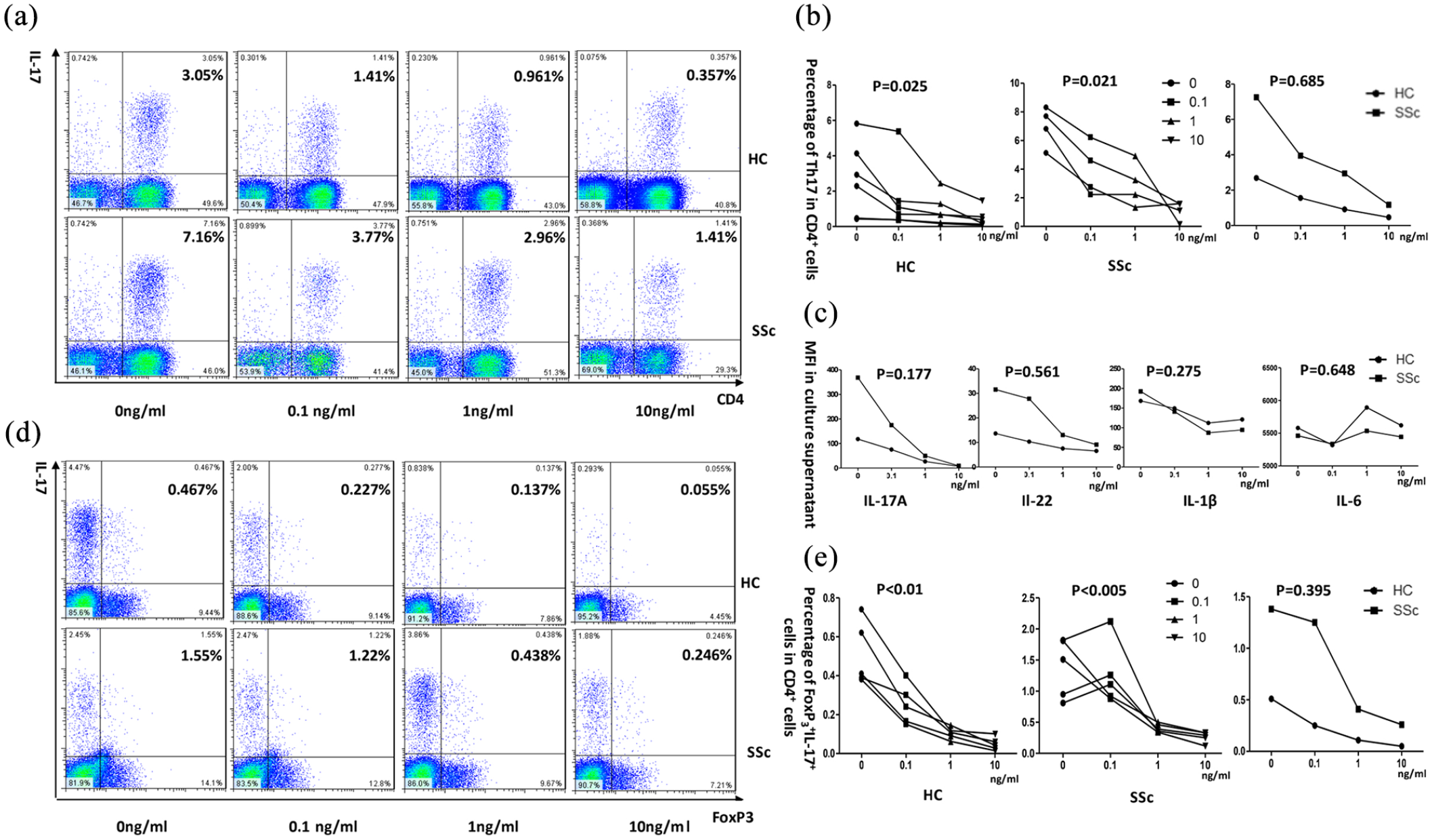
Influence of FK506 on Th17 cells and FoxP3+IL-17+ T cells on the SSc patients: (a) a representative flow cytometry analysis of CD4+ IL-17+ cells dot-plot of the indicated conditions in healthy controls (
The mean fluorescence intensity (MFI) of IL-17A, IL-22, IL-6, and IL-1β were higher in the serum of SSc patients than controls (
In healthy control samples, the number of FoxP3+IL-17+ cells decreased after treatment with FK506 at all doses tested compared to untreated controls (
FK506 inhibits the proliferation of FoxP3low CD45RA− T cells
We next assessed the proliferation capacity of T cells and its subpopulations from healthy controls and SSc patients after treatment with FK506. Proliferation of CD4+ cells was reduced in samples from SSc patients after treatment with 1 and 10 ng/ml FK506 compared to 0 ng/ml FK506 (
In healthy controls, the proliferation of FoxP3− cells treated with 1 and 10 ng/ml FK506 was decreased compared to untreated cells (

The influence of FK506 on the proliferation of CD4+ T cells and its subsets on the patients of SSc: (a) a representative flow cytometry analysis of CD4+ cells, FoxP3+ and FoxP3− cells proliferation of the indicated conditions in healthy controls (
FK506 had no effect on the proliferation of FrI cells from SSc patients. In addition, there was no difference between SSc patients and healthy controls (
However, the proliferation of FrIII cells from SSc patients decreased after treatment with 10 ng/ml FK506 compared to cells treated with 0.1 ng/ml FK506 or untreated (

The influence of FK506 on the proliferation of Treg subtypes on the patients of SSc: (a) a representative flow cytometry analysis of FrI, FrII, and FrIII cells proliferation of the indicated conditions in healthy controls (
Discussion
Systemic sclerosis (SSc) is characterized by the presence of autoantibodies and progressive tissue fibrosis, 39 causing excessive extracellular matrix (ECM) deposition and vasculopathy. The production of ECM responses to Th17 cells. 40 And Th17 cell specifically secrete angiogenic factor, contribute to angiogenesis. 41 Treg and Th17 cells were served as hallmark of SSc, act as pro-fibrotic T cells. 42 And the imbalance of Th17 and Treg responses contributes to the pathogenesis of SSc. 43 Depletion or restoration of Treg cells may become a viable approach in controlling fibrosis.
Tacrolimus (FK506) is widely used as an immunosuppressive drug for preventing the rejection of transplants. The effect of FK506 on Treg cells is controversial. Several reports have found that FK506 has no effect on Treg cells, 44 others suggest that FK506 may increase the number of Treg cells in organ transplant recipients,45–47 while some others have proved that FK506 can reduce the frequency of Treg cells in MuSK-antibody positive myasthenia gravis and systemic lupus erythematosus patients,48,49 a finding that is consistent with transplantation studies. 50 Therefore, the effect of FK506 on Treg cells is worthy of in-depth study. In this study, we divided Treg cells into three subgroups and made our findings. We found that FK506 had no effect on the CD4+CD25+FoxP3+ T cells from healthy donors, which was in agreement with previous findings. 44 Furthermore, we found that FK506 could decrease the number of CD4+CD25+FoxP3+ T cells from SSc patients. These results suggest that FK506 may adjust abnormal Treg cells in SSc patients.
Our study had confirmed that Treg levels are elevated in patients with SSc; interestingly, these Tregs lack normal immune suppression capacities. Instead, the main Treg compartment was found to be composed of non-suppressive FoxP3lowCD45RA− (FrIII) cells with no suppressive function, which indicates that the FoxP3+IL17+ cells can secrete IL-17 to promote differentiation. However, aTreg levels (FrII) are decreased and have reduced function. 31 In this study, we further verified that FK506 could decrease the level of non-suppressive FoxP3lowCD45RA− cells, but had no effect on aTregs, which suggests that the relative level of aTregs was increased.
We also assessed the effects of FK506 on the Th17 and FoxP3+IL-17+ cell populations. The levels of Th17 and FoxP3+IL-17+ cells from SSc patients and healthy controls decreased after treatment with FK506. In addition, the key cytokines secreted by Th17 cells, IL-17A, and IL-22, were increased in the serum from SSc patients. The levels of IL-17A and IL-22 on culture supernatants were also decreased after treatment with FK506, which is consistent with the effects of FK506 on the Th17, FoxP3+IL-17+, and FoxP3lowCD45RA− (FrIII) cells populations.
The relationship between Th17 and Treg cells is complex, and while they have opposing functions, they are related to each other in the process of differentiation. 51 IL-6 has key effects on T cell differentiation and can work in conjunction with TGF-β produced by thymus-derived natural regulatory T cells (nTregs) to convert them into Th17 cells.52,53 IL-6 also controls Th17 immunity by inhibiting the conversion of conventional T cells into Foxp3+ regulatory T cells. 54 We have also confirmed that the FoxP3lowCD45RA− cell compartment may represent a stage in the development of CD4+FoxP3+ plasticity. The numbers of Treg cells and IL17+ cells are both elevated in SSc patients, and the increased number of FoxP3low CD45RA− T cells was the reason for the imbalance between Treg and Th17 cells. This finding also suggests that Th17 cells preferentially increase in the imbalance between Treg and Th17 cells. Many cytokines can affect and control the differentiation of Th17 cells, such as IL-6, TGF, IL-1β, IL-27, and IL-32.55–57 IL-1β production is a critical step for Th17 differentiation and generation of human memory Th17 cells,58–60 and IL-17 production was shown to be initiated by IL-23 and amplified by IL-1β in Th17 cells. 61 We found that the levels of IL-6 and IL-1β were increased, which favored Th17 cells in the imbalance between Treg and Th17 cells and maintained IL-17 secretion during the progress of differentiation. FK506 decreased the level of IL-1β in the culture supernatant and helped to reduce the stability of Th17 cells and the production of IL-17. A recent report also showed that tacrolimus significantly inhibited IL-17-producing cells and the levels of IL-1β and IL-6. 62 A previous report showed that FK506 could decrease IL-6 levels in rat adjuvant-induced arthritis, 63 but this study did not obtain similar results in SSc patients.
FoxP3− cells, including Th1, Th2, and Th17 cells, play a role in the induction of autoimmune tissue injury and are linked to active disease of SSc. We found that FK506 inhibited the proliferation of both FoxP3+ and FoxP3− cells, but the impact on FoxP3− cells was more significant than that on FoxP3+ cells. This result is consistent with previous studies.45,62 Ren et al. 64 also found that tacrolimus could inhibit the proliferation of CD4+CD25− T cells by modifying tolerogenic dendritic cells (tDCs) in collagen-induced arthritic mice. FoxP3lowCD45RA− cells can secrete IL-17 with no suppressive function, and our results indicate that the proliferation capacity of FoxP3lowCD45RA− cells is significantly decreased after incubation with FK506, but not rTregs and aTregs. These results are consistent with the effects of FK506 on FoxP3+ and FoxP3− cells. Kogina et al. 45 also reported that FK506 could not suppress the proliferation of CD4+FoxP3high cells, but rather promoted cell proliferation.
A prior study in SSc patients showed that FK506 with an average trough concentration of 4.7 ng/ml (range 1.75–9.5 ng/ml) could achieve favorable clinical outcomes. 65 Our study supports that medium and high concentrations, especially high concentrations of FK506(10ng/ml), have profound inhibitory effects on the proliferation capacity of FoxP3lowCD45RA− cells and the levels of CD4+CD25+FoxP3+ T cells, Th17 cells in SSc patients. This suggests that similar concentration target could be used to guide theapy in future studies of SSc patients.
There were limitations of this study. We focus on the effect of different concentration of FK506 by the present small sample size. In recently, SSc were classified into four subsets based on the expression profiles of disease-associated genes in the involved skin of SSc patients, such as fibroproliferative subset, inflammatory subset, normal-like subset and limited subset. In general, inflammatory subset of SSc patients is highly responsive to immunosuppressants. 66 In the future, it is essential to expand the sample size of each subtype of SSc, to identify the response of Treg/Th17 cells to FK506 among the four sutsets of SSc.
Conclusions
In conclusion, we found that FK506 could inhibit the proliferation of the non-suppressive subtype of Treg cells and consequently preserved the level of suppressive cells. An increase in FoxP3lowCD45RA− T cells secreting IL-17 with no suppressive function was the reason for the observed elevation in the number and dysfunction of Tregs in SSc patient samples, which may explain the Treg/Th17 imbalance observed in SSc patients. FK506 decreased the level of CD4+CD25+FoxP3+Treg cells by inhibiting the proliferation of FoxP3lowCD45RA− T cells and improving the imbalance of Treg/Th17 cells in SSc patients.
Footnotes
Acknowledgements
We are indebted to Dr. Zhuang Tian, who screened and selected the patients for the study according to the study design and approved protocol. We also thank the healthy donors and SSc patients who participated in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by EULAR Scleroderma Trial & Research group (EUSTAR), National Natural Science Foundation of China [Grant No.81571597, 81300294].
Ethical approval
Ethical approval for this study was obtained from the Medical Ethics Committee of Peking Union Medical College Hospital and the Ethics Committee of EUSTAR.
Informed consent
All patients enrolled in this study provided written informed consent for the samples collected for the research study protocol.
