Abstract
Introduction and objectives:
Sepsis is a critical, life-threatening condition marked by organ dysfunction. Patients undergoing surgery for cancer are especially susceptible, facing a significantly increased risk compared to the general population. Prompt diagnosis is essential for optimal treatment outcomes. This study investigates the expression levels of IL-17 and CD4/CD25 in postoperative surgical oncology patients diagnosed with sepsis and examines their association with various clinicopathological parameters, including mortality.
Methods:
Peripheral blood samples were collected 48 h following admission to the surgical ICU. Flow cytometry was employed to measure the expression of IL-17 and CD4/CD25 in both septic patients and non-septic controls.
Results:
The study included 30 septic patients and an equal number of controls. The expression of IL-17 and CD4/CD25 was significantly higher in septic patients compared to non-septic patients, demonstrating high diagnostic performance. Additionally, elevated IL-17 expression was strongly associated with an increased risk of mortality.
Conclusion:
Our study provides insight into immune dysregulation in postoperative cancer patients with sepsis, highlighting the dual arms of active inflammation and immunoparalysis. Elevated IL-17 levels, coupled with Treg activation, were linked to poorer outcomes. Therapeutic strategies that target IL-17 signaling and modulate Treg activity may help restore immune balance and improve prognosis in immunocompromised individuals. Furthermore, developing predictive models using machine learning to classify sepsis severity based on immune markers could enhance diagnosis and prognosis.
Introduction
Sepsis is a life-threatening condition characterized by organ dysfunction resulting from a dysregulated host response to infection, with significant morbidity and mortality rates. 1 Every year, sepsis and septic shock affect thousands worldwide, posing a major healthcare challenge. With the rapidly growing elderly population and the associated immune senescence and frailty, sepsis-related mortality rates are projected to rise substantially over the next two decades.2,3
Cancer patients face a significantly higher risk of sepsis, up to tenfold greater than the general population, with incidence varying by cancer type. 4 Sepsis affects approximately 16.4 per 1000 cancer patients annually, and about 20% of all sepsis cases occur in this population. The prevalence of cancer diagnoses in ICUs is rising, with sepsis and acute respiratory failure being the most common critical illness syndromes, alongside other frequent complications.5–8
Although mortality rates among this population have decreased over time due to improvements in sepsis management, cancer therapies, and ICU protocols, the growing threat of antimicrobial resistance remains a major challenge, negatively affecting patient outcomes. 8 The interplay between sepsis and cancer presents a complex pathophysiological challenge, as both conditions involve immune dysregulation that compromises the host’s ability to respond to infections. This shared feature underscores the importance of understanding their mutual influence, as such insights could open new avenues for modulating immune dysfunction and controlling the hyperinflammatory state, potentially improving sepsis outcomes. 3
Surgical cancer patients are at elevated risk of postoperative sepsis due to immune suppression triggered by surgery, compounded by factors such as physiological stress, transfusions, anesthetic agents, and the complexity of oncologic procedures. 9 This risk is further intensified by the immunocompromised state associated with malignancy and cancer therapies. Early recognition of sepsis in this group is challenging but critical, and the use of biomarkers for timely detection can greatly improve patient outcomes by reducing morbidity and mortality. 9
Current biomarkers such as procalcitonin, lactate, and CRP do not consistently distinguish sepsis from noninfectious systemic inflammatory response syndrome (SIRS), highlighting the urgent need for effective screening tools and reliable biomarkers. 10 Moreover, these markers lack specificity and may also increase in non-infectious conditions.11,12
IL-17 plays a central role in infectious, inflammatory, autoimmune diseases, and cancer, offering valuable insight into immune dysregulation. Emerging evidence highlights its dual role as a biomarker for early detection and a potential therapeutic target for modulating immune responses in sepsis. 13 IL-17A contributes to the severity of tissue inflammation by stimulating the release of pro-inflammatory mediators, including IL-1, TNF, IL-6, IL-8, CCL20, and G-CSF, which collectively promote neutrophil recruitment. 14
In the context of immune dysregulation, CD4+CD25+Foxp3+ regulatory T cells (Tregs) serve as critical indicators of immunoparalysis. These cells play a vital role in modulating immune responses by exerting strong anti-inflammatory effects through contact-mediated inhibition of other immune cells and by producing high levels of soluble CD25, IL-4, and IL-10. Tregs contribute to an imbalance in the Th1/Th2 immune response (interferon-γ/IL-4) and induce the differentiation of CD4+ T cells into Th2 cells. Under septic conditions, the presence of CD4+CD25+Foxp3+ Tregs, considered natural Tregs, is indicative of immunoparalysis. 15
The dynamic interplay between IL-17A and Tregs underscores the complex immune landscape in sepsis and highlights potential avenues for therapeutic intervention. This study aims to assess the expression of IL-17 and CD4/CD25 in the peripheral blood of postoperative surgical oncology patients with sepsis using flow cytometry, and to investigate the correlation of these markers with various clinicopathological parameters, including mortality.
Methods
Sample size calculation
The sample size was calculated using Minitab 17.1.0.0 for Windows (Minitab Inc., 2013, Pennsylvania, USA). Assuming a Type I error (α) of 0.05 and a Type II error (β) of 0.15 (corresponding to 85% power), the calculation was based on a meta-analysis evaluating the role of IL-17 in predicting mortality in septic patients, which reported a 5.96-fold increase in IL-17 levels among non-survivors compared to survivors. 16 Additionally, according to recent global data from the Intensive Care Over Nations (ICON) audit, 29.5% of ICU patients developed sepsis during their stay, with a reported variation ranging from 13.6% to 39.3%. 17
Based on these parameters, the minimum required sample size to achieve 85% power was 56 participants (28 septic patients and 28 controls).
Study design
A cross-sectional study was conducted at the National Cancer Institute (NCI), Cairo University. The study received approval from the Ethics Committee of the National Cancer Institute, Cairo University (approval no IRB000004025). Written informed consent was obtained from all recruited patients’ guardians.
Patient recruitment
Sixty cancer patients admitted to the surgical ICU after cancer-related operations were recruited for the study from November 2023 to January 2025. Half of the participants were diagnosed with sepsis, while the remaining matched individuals served as non-septic controls. Patients aged 18–65 years, of either sex, classified as American Society of Anesthesiologists (ASA) physical status II or III, with no preoperative signs or symptoms of infection and normal results for complete blood count, renal and hepatic function tests, and coagulation profiles were included in the study. Exclusion criteria were: refusal to participate by the patient’s guardian, abnormal preoperative kidney or liver function, or significant intraoperative blood loss requiring massive transfusion.
The primary outcome was the evaluation of IL-17A and CD4⁺CD25⁺ in patients with sepsis to assess their potential as early markers for sepsis. The secondary outcomes included correlating these markers with patient prognosis and other clinical and laboratory data.
Samples were collected 48 h following admission to the surgical ICU. Sepsis diagnosis was confirmed based on the International Guidelines for Management of Sepsis and Septic Shock 2021. 18 Patients were then classified into the sepsis group upon confirmation of a sepsis diagnosis, or into the control group if no evidence of sepsis was present at this point.
Clinical evaluation
All recruited patients underwent a comprehensive clinical assessment, including age, sex, smoking status, and the presence of comorbidities. Sepsis was evaluated using the Quick Sequential Organ Failure Assessment (qSOFA) score, which consists of three criteria: respiratory rate ⩾22/min, altered mental status, and systolic blood pressure ⩽100 mmHg. Each criterion is scored on a binary scale (0 for absence, 1 for presence), with a total score of ⩾2 indicating a high risk of organ dysfunction. 19
Additionally, data on the use of steroids, vasopressors, insulin, and respiratory support (oxygen face mask, nasal oxygen, Continuous Positive Airway Pressure (CPAP), or invasive mechanical ventilation, were collected. The length of hospital stays, and mortality rate were also recorded.
Flow cytometry
Data collection and measurement
Monoclonal antibodies with different specificities, along with their respective isotype controls, were utilized as outlined in Supplemental Table 1. These antibodies were obtained from BD Biosciences (Franklin Lakes, New Jersey, USA) and Beckman Coulter (Miami, Florida, USA). Flow cytometric analysis was performed using a six-color flow cytometer from Beckman Coulter, acquiring 10,000 events per sample. Data analysis was carried out using Kaluza software (Beckman Coulter).
Peripheral blood mononuclear cell (PBMC) isolation
Peripheral blood samples were obtained from patients and processed within 2 h of collection. Samples were allowed to equilibrate to room temperature (~30 min) prior to processing. The whole blood was diluted 1:1 with sterile phosphate-buffered saline (PBS). One milliliter of room temperature Polymorph density gradient medium (Ficoll) was gently layered into an 8 mL round-bottom polystyrene tube, followed by the careful addition of 3 mL of the diluted blood on top, avoiding mixing of the layers.
Samples were centrifuged at 400–500 × g for 45 min at room temperature. Following centrifugation, the peripheral blood mononuclear cells (PBMCs) become separated from other blood components and form a distinct, cloudy layer beneath the plasma. This PBMC layer typically appears as the first opaque band from the top. The upper plasma layer was carefully removed, and the PBMC layer was transferred to a 15 mL or 50 mL conical tube using a micropipette.
PBMCs were washed twice with PBS, centrifuging at 500 × g for 5 min each time, and the supernatant was discarded after each wash. The isolated cells were counted and diluted to a concentration of 1 × 10⁶ cells/mL for staining procedures.
Surface and intracellular staining
For surface staining, 10 µL of fluorochrome-conjugated MoAb was added to 100 µL of the diluted lymphocyte suspension, incubated in the dark for 20 min, followed by a PBS wash. The cells were then resuspended in 500 µL of PBS and analyzed by flow cytometry.
For intracellular staining, 100 µL of diluted lymphocytes were resuspended in 1 mL of PBS, then fixed with a mixture containing 500 µL of 4% paraformaldehyde, 500 µL of PBS, and 5 µL of Tween 20. The suspension was incubated for 10 min at room temperature. After washing, 10 µL of the relevant MoAb was added, and the cells were incubated for 30 min at 4°C in the dark. Following a final wash, cells were resuspended in 500 µL of PBS and analyzed. The protocol was adapted from Menon et al. 20
Analysis strategy
A three-antibody panel was utilized to evaluate immune markers in cancer surgical patients with sepsis: surface CD4-FITC, intracellular IL-17-PE, and surface CD25-PE-Cy5.5. Initially, doublets were excluded based on forward scatter (FSC) and side scatter (SSC) characteristics by gating on singlet events. Lymphocytes were subsequently identified and gated using an FSC versus SSC plot. After acquiring 10,000 events per sample, lymphocytes were selected for further analysis.
CD4 expression was plotted against CD25 to quantify the percentage of CD4⁺ T-helper cells, CD25⁺ cells, and the co-expression of CD4⁺CD25⁺ regulatory T cells within the gated lymphocyte population (Supplemental Figure 1).
For the analysis of interleukin-17a, CD IL-17a expression was plotted against cell count and quantified as mean fluorescence intensity using the mean channel fluorescence in the FL2 channel. Isotype controls were used to account for autofluorescence and nonspecific binding. Fluorescence intensity was presented as a histogram plotted against cell counts and recorded for further analysis. The mean fluorescence intensity of CD IL-17a in patient samples was then superimposed on that of the isotype control (Supplemental Figure 2).
Statistical analysis
Data were entered into a Microsoft Excel spreadsheet and analyzed using SigmaPlot for Windows (version 12.5.0.38) and Minitab for Windows (version 17.1.0.0, Minitab Inc., 2013, Pennsylvania, USA). The Shapiro-Wilk test was applied to evaluate the normality of data distribution.
For numerical variables, results were expressed as mean ± standard deviation for normally distributed data and as median with interquartile range for data not following a normal distribution. Categorical variables were summarized as frequencies and percentages.
Comparisons between two numerical groups were made using the independent t-test for normally distributed data and the Mann-Whitney U test for non-normally distributed data. The chi-square test was employed to compare categorical variables.
To evaluate the diagnostic performance of the markers, a receiver operating characteristic (ROC) curve was generated, with an area under the curve (AUC) >0.6 considered acceptable. To assess the robustness of the diagnostic performance and address potential overfitting, internal validation was performed using both bootstrapping and leave-one-out cross-validation (LOOCV). For bootstrapping, 1000 resamples with replacement were used to estimate the confidence interval of the AUC for each marker. LOOCV was also conducted, where each patient was left out once as a test case while the model was trained on the remaining 59 patients, and the AUC values were averaged across all iterations.
To address the potential influence of confounding factors, survival analysis was performed using Kaplan-Meier survival curves, cumulative hazard analysis, and Cox regression modeling (proportional hazards model) to identify factors influencing survival probability in septic patients. Variables with p < 0.1 in univariate analysis or clinical relevance were included in the multivariate model. All tests were two-sided, with a p-value <0.05 considered statistically significant.
This manuscript was prepared in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies.
Results
The study included 30 patients with sepsis and an equal number of non-sepsis patients. Supplemental Table 2 presents the clinical and demographic characteristics of the study groups, which were matched for sex, smoking habits, and prevalence of comorbidities. The septic group was significantly older than the non-septic group (mean age 53.9 ± 14.1 vs 45.2 ± 19.3 years; p = 0.05). No significant differences were observed in sex distribution, smoking status, or comorbidity prevalence. Regarding arterial blood gases, septic patients had significantly lower pH (7.26 ± 0.12 vs 7.34 ± 0.11; p = 0.02) and HCO₃⁻ levels (17.19 ± 6.78 vs 21.63 ± 5.01 mmol/L; p = 0.01), while other ABG parameters were comparable. Treatment differences were notable: steroid and vasopressor use were significantly higher in septic patients (p = 0.001 each), as was the use of invasive mechanical ventilation (IMV; 43.3% vs 10%; p = 0.004). Nasal prongs were used more often in non-septic patients (26.7% vs 3.3%; p = 0.01).
Figure 1 displays the expression levels of inflammatory markers in septic versus non-septic patients, demonstrating statistically significant elevations across all five markers in the septic group. Specifically, CD4% was significantly higher in septic patients (median 43.32 (IQR: 30–56)) compared to non-septic patients (29.63 (IQR: 12–37); p = 0.02). Similarly, CD25% (6.14 (5–9) vs 2.22 (0.6–3.3); p = 0.001), CD4⁺CD25⁺ (4.38 (4–7) vs 0.53 (0.12–1.1); p = 0.001), and mean fluorescence intensity (MFI) IL-17/ISO (4.05 (3–5) vs 1.58 (1–2); p = 0.001), all showed highly significant increases in septic patients.

Expression of inflammatory markers in septic and non-septic groups.
Additionally, Figure 2 illustrates the ROC curves of each inflammatory marker for diagnosing sepsis. IL-17% achieved an AUC of 1.00 (95% CI: 1.00–1.00), followed by CD4+CD25+ (AUC = 0.99, 95% CI: 0.95–1.00), CD25% (AUC = 0.99, 95% CI: 0.95–1.00), and CD4% (AUC = 0.74, 95% CI: 0.62–0.86). These results demonstrate very good to excellent discriminatory power. To assess the robustness of these findings, a bootstrapping analysis with 1000 resamples was performed, confirming the stability of the results. The bootstrapped mean AUC for IL-17% remained at 1.00 with a narrow 95% confidence interval, indicating consistent and strong discriminatory performance across resamples (Supplemental Table 3).
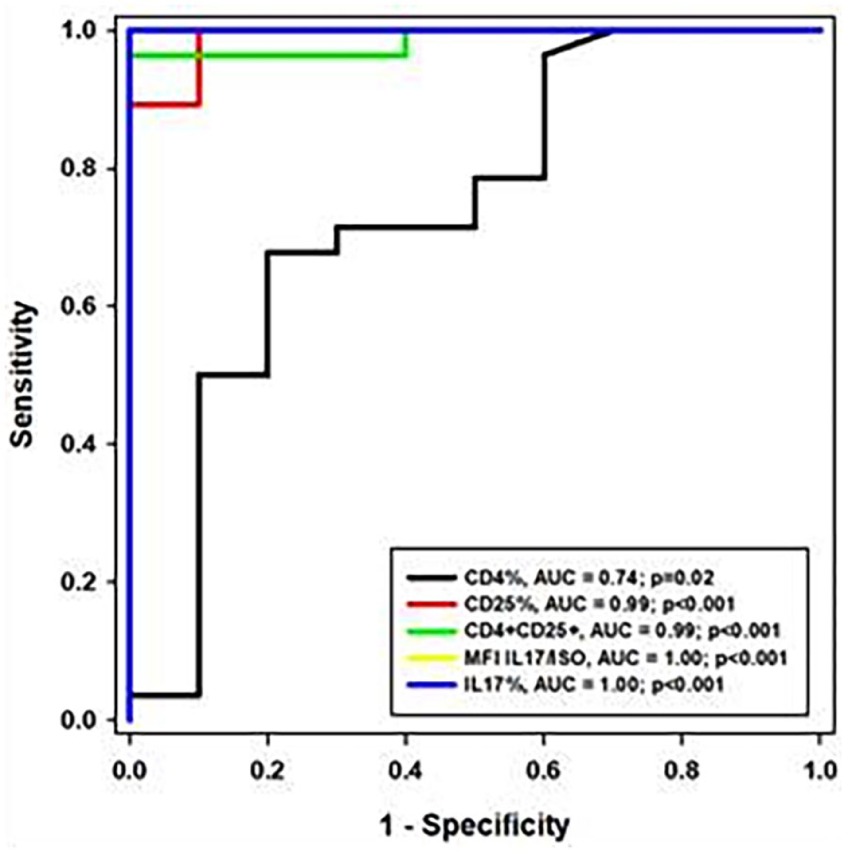
Receiver Operating Characteristic (ROC) curve illustrating the diagnostic performance of immune markers in distinguishing septic from non-septic cancer surgical patients. The ROC analysis demonstrates the sensitivity and specificity of individual markers: CD4%: AUC = 0.74, p = 0.02, CD25%: AUC = 0.99, p < 0.001, CD4⁺CD25⁺ T cells: AUC = 0.99, p < 0.001, IL-17 MFI/ISO: AUC = 1.00, p < 0.001, and IL-17%: AUC = 1.00, p < 0.001.
Moreover, Table 1 shows the diagnostic performance of these inflammatory markers; IL-17%, MFI IL17/ISO, and CD4+CD25+ demonstrated perfect diagnostic accuracy (100% sensitivity and specificity, PPV and NPV = 100%). CD25% showed 100% sensitivity and 90% specificity, while CD4% had a more modest performance (sensitivity = 71%, specificity = 70%).
Diagnostic performance of immune markers for identifying sepsis in postoperative surgical oncology patients.

MFI: mean fluorescence intensity; IL: Interleukin; ISO: isotype control; CI: Confidence interval; PPV: positive predictive value; NPP: negative predictive value.
Septic patients had a median qSOFA score of 2 (IQR: 2–3). All patients had positive blood cultures; 90% of isolates were multidrug-resistant (MDR), and 10% were pan-drug-resistant (PDR). The most frequently isolated organisms were Klebsiella (50%), Acinetobacter (40%), and Pseudomonas (30%; Supplemental Table 4).
Septic patients had a significantly longer median hospital length of stay (33 days (IQR: 11–72) vs 4 days (IQR: 2–17), p = 0.001) and markedly higher in-hospital mortality (56.7% vs 0%, p = 0.001), confirming the severe clinical burden of sepsis in this cohort (Supplemental Table 5).
Table 2 presents the univariate prognostic role of different marker expressions and the qSOFA score. While none of the markers showed significant prognostic value, a higher qSOFA score was significantly associated with mortality (median 3 vs 2; p = 0.03).
Univariate analysis of prognostic markers in relation to mortality among septic surgical oncology patients.
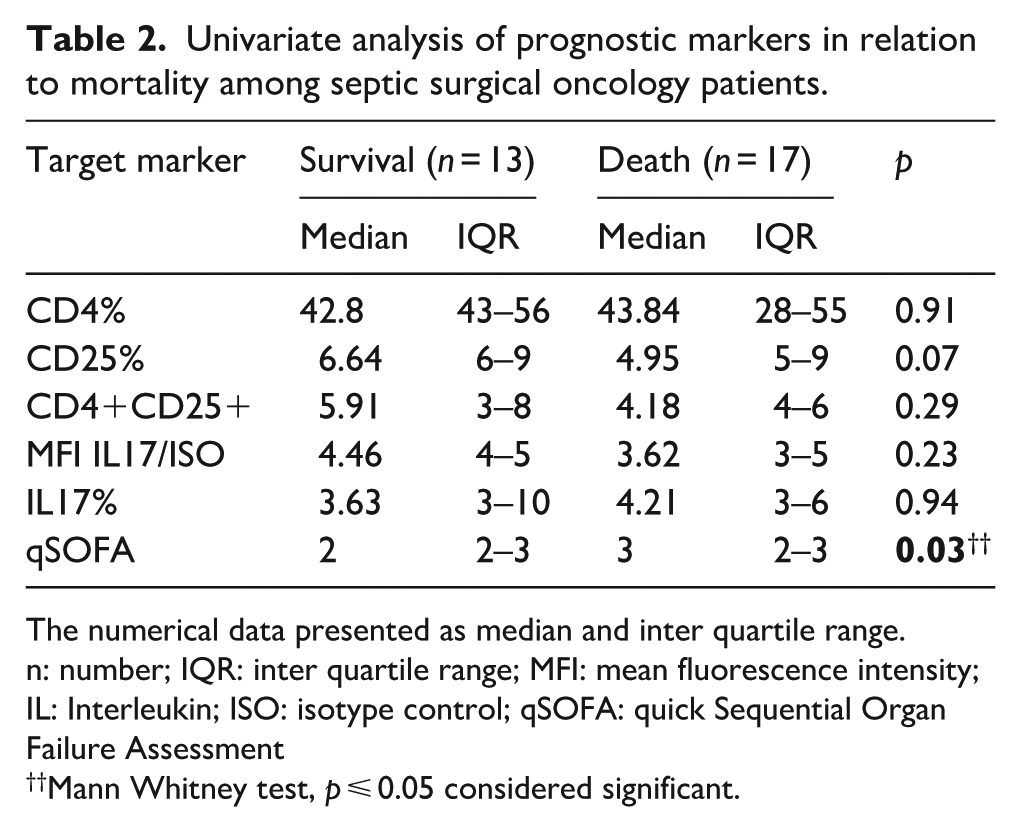
The numerical data presented as median and inter quartile range.
n: number; IQR: inter quartile range; MFI: mean fluorescence intensity; IL: Interleukin; ISO: isotype control; qSOFA: quick Sequential Organ Failure Assessment
Mann Whitney test, p ⩽ 0.05 considered significant.
Multivariate Cox regression (Table 3) revealed that higher CD4% was protective (HR: 0.88, 95% CI: 0.78–0.98; p = 0.02), as was lower MFI IL-17/ISO (HR: 0.44, 95% CI: 0.21–0.91; p = 0.03). Additionally, diabetes (HR: 0.01, 95% CI: 0.00–0.62; p = 0.03) and Pseudomonas infection (HR: 0.01, 95% CI: 0.00–0.47; p = 0.02) were protective, while ischemic heart disease (HR: 8.67, 95% CI: 1.08–69.66; p = 0.04) and absence of Klebsiella (HR: 463.8, 95% CI: 7.3–29390.5; p = 0.001) were associated with increased mortality risk.
Determinants of survival outcomes in patients with sepsis.

The test of significant: Cox regression analysis with proportional hazard model, p ⩽ 0.05 considered significant.
MFI: mean fluorescence intensity; IL: Interleukin; ISO: isotype control; qSOFA: quick Sequential Organ Failure Assessment; DM: Diabetes Mellitus; HTN: Hypertension; IHD: Ischemic Heart Disease; CE: Coefficient; HR: hazard ratio; CI: confidence interval.
Furthermore, Supplemental Figure 3 presents the Kaplan–Meier survival analysis for septic patients. The curve demonstrates survival probability over time in septic patients. The cumulative hazard function reflects increasing mortality risk, with a steeper decline in survival observed among patients with ischemic heart disease and those without Klebsiella infection, as identified in the multivariate Cox regression model.
Discussion
The pathogenesis of sepsis involves a dysregulated host response to infection, marked by an imbalance between pro- and anti-inflammatory pathways. This immune dysfunction can result in both excessive inflammation and immunosuppression, leading to tissue damage and varied clinical outcomes. 21 In this context, our study shows that immunological markers assessed by flow cytometry have diagnostic and prognostic value in sepsis, aiding in the prediction of mortality among cancer patients following surgery.
IL-17 is derived from T cells and produced by various immune cells, including macrophages, dendritic cells, mast cells, and natural killer cells. 22 As a pro-inflammatory cytokine, IL-17 binds to specific receptors, promoting inflammatory cell infiltration, particularly neutrophils, contributing to tissue damage.23,24
Interestingly, interleukin-17 (IL-17) plays a dual role in host defense against infections and the development of inflammatory disorders. 25 It exerts both protective and detrimental effects by activating neutrophils and macrophages at sites of infection, thereby enhancing antimicrobial responses and contributing significantly to the body’s defense mechanisms. 26
Our study demonstrates that IL17 expression was significantly associated with the development of sepsis. Furthermore, its expression exhibited perfect predictive accuracy for the condition. A previous study also showed that serum IL-17 levels have been shown to predict sepsis development and mortality in polytrauma patients. 27 In another study, serum analysis for IL-10, IL-17, and procalcitonin (PCT) have demonstrated high diagnostic value in sepsis patients. The combination of these tests yields better diagnostic performance and more substantial clinical advantages than each test alone. 25 A recently published systematic review and meta-analysis, which included four studies, revealed that serum interleukin-17 levels were significantly higher in non-survivor sepsis patients compared to survivors. 16
Beyond its diagnostic and prognostic significance, IL-17 can also be a therapeutic target for neonatal sepsis. A study by Wynn et al. demonstrated that elevated IL-17A levels were associated with the detrimental effects of IL-18. The findings suggested that blocking IL-18, IL-1 receptor 1 (IL-1R1), or IL-17 signaling could serve as innovative therapeutic strategies to enhance outcomes for neonates with sepsis. 28
In our cohort, CD4+/CD25+ cells, alongside IL-17, demonstrated strong diagnostic value for sepsis, although they did not show a significant association with patient prognosis. Consistent with our findings, a study by Leng et al. 15 demonstrated that the proportion of CD4+CD25+Foxp3+ T cells was significantly higher in patients with early-stage septic shock compared to those with SIRS, which can be helpful for assessment of immune status and guiding appropriate immunoregulatory therapy. This finding also aligns with the study by Saito et al., which reported that septic patients exhibited significantly higher percentages of CD25(+) cells among CD4(+) T cells compared to the control group. The percentages of Treg cells were also notably higher in septic patients than in those with SIRS. Additionally, plasma levels of soluble CD25 were significantly elevated in septic patients compared to both SIRS patients and healthy volunteers. 29
Sepsis-induced apoptosis contributes to lymphopenia in patients with septic shock, affecting various T cell subsets, including CD4⁺, CD8⁺, and natural killer (NK) cells, while sparing regulatory T cells (Tregs). 30 Although Tregs constitute a small fraction of the T-lymphocyte population, they play a critical role in regulating immune activation, making them key players in suppressing excessive immune responses during sepsis. This regulatory role, however, can be a double-edged sword, acting as both angels and demons: while septic shock patients may benefit from the mitigation of hyperinflammatory responses, they simultaneously face an increased risk of secondary infections. 31 Interestingly, it has been reported that both CD4⁺ T cell subsets (CD25⁺ and CD25⁻) decrease immediately following the onset of sepsis. However, while regulatory T cells (CD4⁺CD25⁺) rapidly return to levels observed in healthy individuals, the CD4⁺CD25⁻ T lymphocyte population remains significantly depleted. 32
The persistent elevation of CD4⁺CD25⁺ regulatory T cells contributes to profound immunoparalysis and is linked to unfavorable outcomes. Consequently, sequential monitoring of these cells in the peripheral blood of septic patients may offer valuable insight into disease progression and assist in assessing prognosis. 33
Conclusions
Our study provides a snapshot of immune alterations in patients with sepsis following cancer surgery, emphasizing the coexistence of active inflammation and immunoparalysis. These opposing immune responses reflect dysregulated immune cell function, resulting in failure to restore homeostasis. The observed increase in pro-inflammatory markers, particularly IL-17 expression, alongside elevated Treg marker expression, sheds light on the early immunological events and consequences of sepsis. Notably, this heightened pro-inflammatory response is associated with poorer prognosis.
Combined strategies targeting both IL-17 signaling and Treg modulation could help balance inflammation and immune regulation, potentially improving outcomes in immunocompromised patients. Additionally, multiple sampling to monitor changes in these markers throughout the course of sepsis may provide valuable insights into the dynamic shifts in immune status.
Flow cytometry offers a rapid and effective method for analyzing immune cell populations and their functional states. This approach enhances the understanding of immune dysfunction in sepsis and supports the identification of potential novel biomarkers. Developing predictive models that integrate large panels of flow cytometry data with genomic profiling can improve patient stratification and classification into distinct subgroups. Additionally, incorporating machine learning techniques with various clinical and laboratory parameters could significantly improve the accuracy of diagnosing sepsis severity and predicting patient outcomes.
Strengths and limitations
We consider flow cytometry to be superior to serum measurement of IL-17, as it enables the evaluation of IL-17 expression in conjunction with CD4 and CD25 markers. This approach provides important insights into the balance between pro-inflammatory and regulatory immune responses, which is particularly critical in sepsis and cancer patients. Unlike serum IL-17 levels, which may be elevated for various reasons, flow cytometry offers cell-specific functional data that allow for a more precise assessment of immune dysregulation.
Our study has some limitations that should be acknowledged. First, we collected only a single sample from each patient, limiting our ability to capture dynamic changes in immune markers during the progression of sepsis. Another limitation is the relatively small sample size, which, although sufficient to meet power requirements, may limit the generalizability of our findings and introduces the potential for overfitting. While internal validation techniques such as bootstrapping and leave-one-out cross-validation were employed to mitigate this risk, the absence of an independent external validation cohort remains a limitation. To confirm and extend these results to broader ICU populations, larger, multicenter studies are warranted.
A limitation of the data analysis step was the absence of formal correction for multiple comparisons (e.g. Bonferroni adjustment), which was not applied due to the limited number of predictors and the exploratory nature of the analysis. Additionally, we did not assess the functional activities of IL-17-producing cells or Tregs or conduct genetic or molecular analyses to better understand the underlying mechanisms of immune dysregulation. The absence of a broader marker panel, such as TNF-α or PD-1, may have restricted our insights. Confounding factors, including variability in cancer types, treatments, and patient health status, could also influence the results.
Supplemental Material
sj-docx-1-iji-10.1177_03946320251375162 – Supplemental material for Flow cytometry-based evaluation of CD4/CD25 and IL-17 expression in sepsis of cancer surgical patients
Supplemental material, sj-docx-1-iji-10.1177_03946320251375162 for Flow cytometry-based evaluation of CD4/CD25 and IL-17 expression in sepsis of cancer surgical patients by Ahmed Samir Abdelhafiz, Asmaa Ali, Walaa Y Elsabeeny, Ahmed Fahmy, Merhan A Fouda, Mahmoud Ali Ayoub and Marwa Hanafi in International Journal of Immunopathology and Pharmacology
Footnotes
Author contributions
A.S.A. designed the experiment and wrote the manuscript. A.A. performed data analysis. W.Y.E. collected samples and data. A.F. collected samples and data. M.A.F. contributed to data collection. M.A.A. contributed to data collection. M.H. conducted the experiment and performed analysis. All authors reviewed and approved the final manuscript.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Human ethics and consent to participate
All procedures performed in this study involving human participants were in accordance with the ethics standards of the institutional research committee of the investigating hospital (Institutional Review Board (IRB), National Cancer Institute, Cairo University) and the 1964 Helsinki Declaration and its later amendments or comparable ethics standards.
Written informed consent was obtained from all guardians prior to study. Ethical approval for this study was obtained from the National Cancer Institute, Cairo University ethics committee (approval no IRB000004025)
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
