Abstract
The study was aimed to explore the possible function of thermal ablation treatment on T helper 17 (Th17) cells and regulatory T (Treg) cells in transplantation of hepatocellular carcinoma in mice. In total, 60 male C57BL/6 mice were divided into control group, model group, and treat group. Flow cytometry was used to detect the frequency of Th17 and Treg cells in peripheral blood. The levels of interleukin (IL)-17, IL-23, IL-10, and transforming growth factor beta (TGF-β) in serum were detected by enzyme-linked immunosorbent assay (ELISA).The levels of IL-17, RORγt, Foxp3, and TGF-β mRNA in tumor tissues were detected by real-time fluorescence quantitative PCR (qRT-PCR). Compared with the model group, tumor size was significantly decreased after thermal ablation treatment. After treatment, the frequency of Th17 cells in peripheral blood was significantly decreased, while the frequency of Treg cells was profoundly increased (
Introduction
Thermal ablation therapy (TAT) of tumor is the direct application of extreme temperatures, either high or low in the focal solid tumors (single or multiple) with the aid of image-guided techniques using heating to induce irreversible cell injury and ultimately lead to tumor apoptosis and necrosis. 1 The thermal damage induced by heating is dependent on both temperature in the tissue and the heating duration. When the tumor target temperature is 50°C to 55°C, coagulation necrosis takes 4 to 6 min; when the temperature rises to 60°C to 100°C, protein denaturation of the target tissue occurs immediately, resulting in cytoplasm, destruction of mitochondrial enzymes, formation of nucleic acid histone complexes, and irreversible coagulative necrosis of cells in an extremely short period of time. 2
Hepatocellular carcinoma (HCC) is the most common primary liver malignancy, which is not only the fifth most common cancer but also the third most common cause for cancer death worldwide. 3 The incidence of HCC is expected to rise in the next two decades especially in the developing world. 4 Although cancer treatment technology continuously improved, the prognosis of liver cancer has not been significantly improved, and its 5-year survival rate has been ranging from 17% to 53%. 5 At present, radical treatment of liver cancer includes surgical resection, liver transplantation, and ablation therapy (microwave heating, radiofrequency or alcohol ablation, etc.).6–10 Unfortunately, 70% of patients were diagnosed as the middle or advanced stages of HCC, which makes patients lose the chance of surgical resection. Therefore, TAT has been rapidly developed due to its invasiveness and simple operation, and it has achieved good results in the treatment of small HCC. 11 It has become an important treatment method for early small HCC and postoperative recurrence of liver cancer.12,13
Since the rapid development of thermal ablation in 1990s, it has become the third major method of surgical resection and interventional therapy for liver cancer. Clinical trials have shown promising results in the treatment of a broad spectrum of cancers, including HCC, hepatic and lung metastases, renal, thyroid, and breast cancers. 14 Several thermal ablation strategies have been developed to treat the primary liver malignancies. 15 Thermal ablation has the advantages of low cost, lower morbidity, and mortality, and can also take advantage of real-time imaging guidance. Clinically, it has been observed that after the ablation treatment of liver cancer, enhanced immunity of the patient can even make distant metastases shrink or disappear.16,17 However, the specific mechanism of this anti-tumor immunity enhancement after ablation therapy remains unclear. Thus, this study uses mice tumor model to detect changes in regulatory T (Treg) cells after thermal ablation treatment of HCC to explore the mechanisms that involve in the immune-enhancing effect of thermal ablation on HCC patients.
Materials and methods
Animals
A total of 60 male C57BL/6 mice with body weight of 18 ± 2 g and aged 4 to 6 weeks were purchased from Jinan Pengyue Experimental Animal Breeding Company (Jinan, China) (animal production license number: SCXK (Lu) 20140007). Mice were maintained at a temperature of 25 ± 5°C and humidity of 55% ± 5%, light/dark cycle of 12 h, and free access to normal diet and water. The research protocol was approved by the Ethics Committee of the Affiliated Yantai Yuhuangding Hospital of Qingdao University (20180112) and performed in accordance with the “Guidelines for the care and use of experimental animals.”
Cell culture
HCC cell line Hepa 1–6 cells (American Type Culture Collection (ATCC), CRL-1830, Biotechnology Co., Ltd. Shanghai enzyme research, Shanghai, China) were selected and cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) (GIBCO, Invitrogen, Grand Island, NY, USA) containing 10% heat-inactivated fetal bovine serum and 1% penicillin–streptomycin antibiotics at 37°C, 5% CO2 incubator (Thermo, Shanghai, China). Cells in logarithmic phase were used for subsequent experiments.
Establishment of animal models and groups
Liver cancer cells were digested and resuspended in phosphate buffer solution (PBS) with 2 × 107 cells/mL. Under aseptic conditions, 0.1 mL of liver cancer cell suspension was inoculated into the right axilla of the mice. During the inoculation, the skin at the inoculation site was slowly uplifted, and gradually became a rounded rind, and the needle was pulled out. If local bleeding or infiltration was observed, pinch the pinhole for a moment.
The mice were divided into control group (n = 12), model group (n = 24), and treat group (n = 24). After the tumor diameter grew to 6 to 10 mm, mice in the treat group were anesthetized with intraperitoneal injection of 0.6% sodium pentobarbital (40 mg/kg) and fixed in a supine position on the electrode plate. Radiofrequency ablation needle was inserted into the tumor center. The condition of ablation was 70°C, 5 min and power was 5 W. In the model group, radiofrequency needles were inserted into the tumor center without ablation.
Measurement of tumor volume and size
Tumor growth was monitored by measuring the perpendicular diameter with a caliper every 3 days, and the changes in the tumor volume within 15 days were observed in each group. The average tumor volume in each group was calculated: volume (mm3) = (length × width2)/2. After anesthetizing mice intraperitoneally with 0.6% sodium pentobarbital (40 mg/kg), six mice in each group were sacrificed by cervical dislocation and the tumor tissues were weighed. The inhibition rate (%) of the subcutaneous tumors = (VModel – VTreat)/VModel × 100%. Partial tumor tissue was collected and fixed with 4% paraformaldehyde buffer and then embedded in paraffin for immunohistochemical detection. The rest tumor tissue was quickly frozen in liquid nitrogen and transferred to a −80°C refrigerator for preservation by qRT-PCR.
Tumor re-challenge experiments
The same amount of tumor cells was inoculated into the left axilla of selected 12 mice that survived 30 days from the model and treat groups, and the tumorigenic rate of re-inoculated tumors was observed 15 days after inoculation.
Flow cytometry analysis
The peripheral venous blood was collected every 3 days. The peripheral blood cells were stimulated with 50 ng/mL propylene glycol monomethyl ether acetate (PMA), (BioVision, San Francisco, California, USA), 1 µg/mL ionomycin (Enzo Life Sciences, San Francisco, California, USA), and 500 ng/mL monensin (eBioscience, San Francisco, California, USA) for 4 h, and then cells were collected and stained with anti-mice CD4-fluorescein isothiocyanate (FITC) and anti-mice IL-17-PE antibodies (Bioss, Beijing, China). Briefly, cells were incubated with these antibodies at 4°C in the dark for 20 min and washed twice with FACS buffer, fixed, and permeabilized. For Treg cell staining, after permeabilization, cells were stained with anti-mice CD4-FITC and anti-mice CD25-APC (eBioscience, San Francisco, California, USA), and incubated at 4°C in the dark for 20 min. Cells were washed twice with fluorescence activating cell sorter (FACS) buffer, fixed, and permeabilized, and then cells were stained with anti-mice Foxp3-PE (eBioscience, San Francisco, California, USA). Flow cytometry analysis was performed by using a FACS Canto II flow cytometer (BD, San Francisco, California, USA).
Enzyme-linked immunosorbent assay
Serum inflammatory cytokines IL-17 (EK0431, Boster, Wuhan, Hubei, China, USA), IL-23 (EK0866, Boster, Wuhan, Hubei, China), IL-10 (EK0417, Boster), and TGF-
qRT-PCR analysis of related mRNA level in tumor tissue
The collected tissue samples were homogenized and centrifuged at 4°C (12,000 r/min, 15 min). Total RNA was extracted using TRIzol (Life Technologies, Carlsbad, CA, USA). OD260/OD280 demonstrated RNA purity between 1.8 and 2.0. RNA was reversely transcribed into complementary DNA (cDNA) by reverse transcription kit (Applied Biosystems, Waltham, MA, USA). Real-time fluorescence quantitative polymerase chain reaction (PCR) was performed by using Mastercycler nexus X2 (Eppendorf, Hamburg, Germany) with the condition: 95°C 1 min, 95°C 15 s, 61°C 15 s (40 cycles). Data were processed using the 2–ΔΔCt method, and relative expression levels were calculated using β-actin mRNA as an internal reference. The primer sequences are listed in Table 1.
Primer sequences.

Statistical analysis
SPSS19.0 statistical software was used to analyze all the results, and quantitative data are expressed as mean ± SD (standard deviation). The t-test was used for the data analysis between the two groups and one-way ANOVA (analysis of variance) was used for the comparison of multiple groups followed by least significant difference (LSD) test for subsequent analysis. The difference was statistically significant at
Results
Thermal ablation treatment inhibits tumor growth in mice tumor model
After subcutaneous inoculation of Hepa 1–6 cells, the tumor formation rate was 100% in model group and the tumor growed rapidly. From the second day of inoculation, mice developed subcutaneous nodules which were visible to the naked eye. On the eighth day, the average diameter of the tumor was 6 mm. Tumor tissue was collected after 15 days. General observation of the tumor showed nodular expansive growth, hard texture, central tissue necrosis, and no adhesion to the surrounding muscle tissue. Three days after thermal ablation treatment, the tumor size in treat group was significantly reduced as compared to model group (240.5 versus 172.6 mm3,
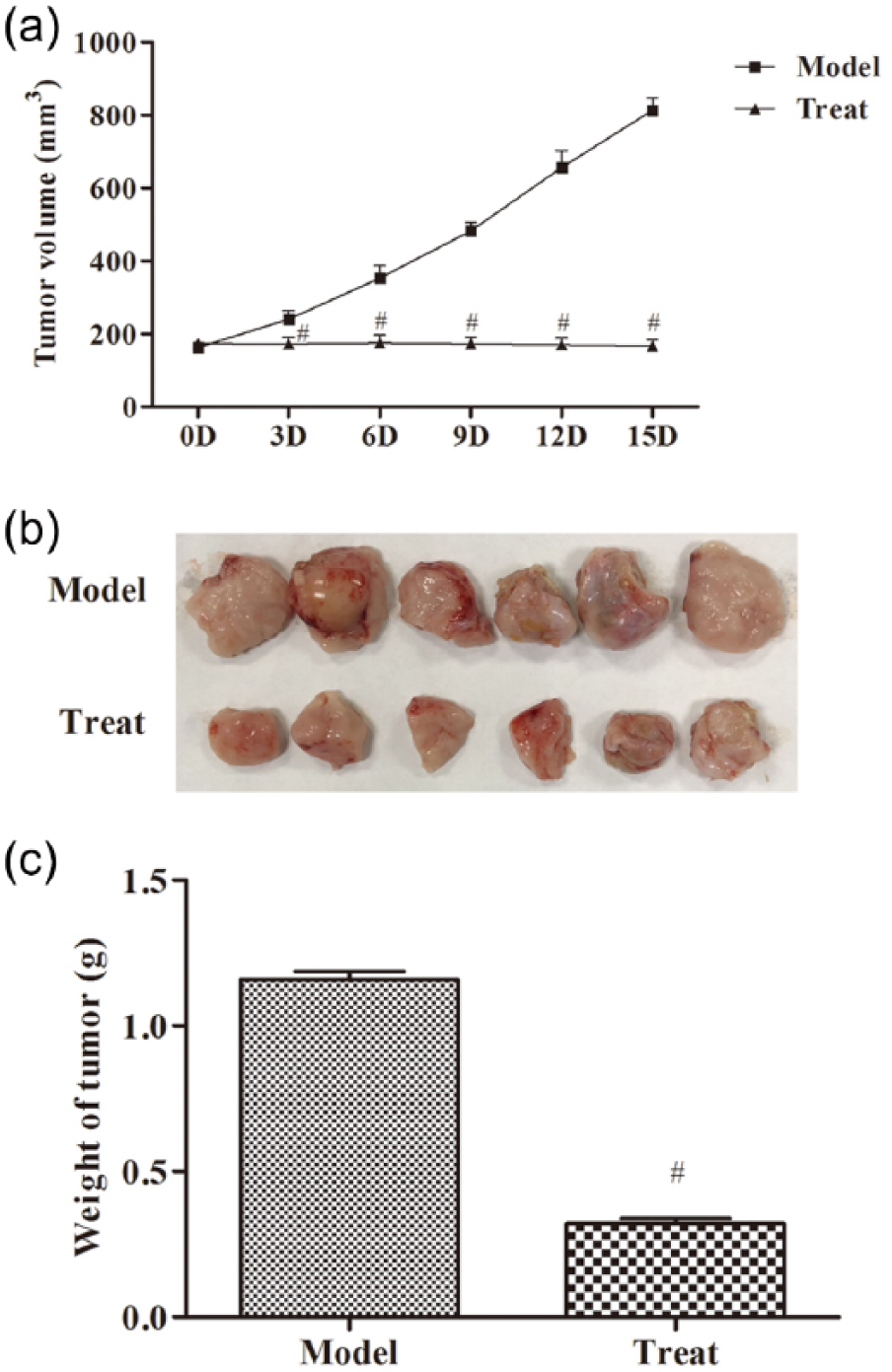
Thermal ablation treatment inhibits tumor growth: (a) the tumor size of mice in the model and treat groups, (b) the images of transplanted tumors in mice, and (c) the body weight in the model and treat groups. The data were expressed as the mean ± SD. Compared with model group, #
Thermal ablation affects the Th17/Treg cell imbalance in peripheral blood
The frequency of T helper 17 (Th17) cells and Treg cells in mice peripheral blood was analyzed by flow cytometry (Figures 2 and 3). Compared with the control group, the frequency of Th17 was increased with time in model group, while the frequency of Th17 was increased with time in model group (
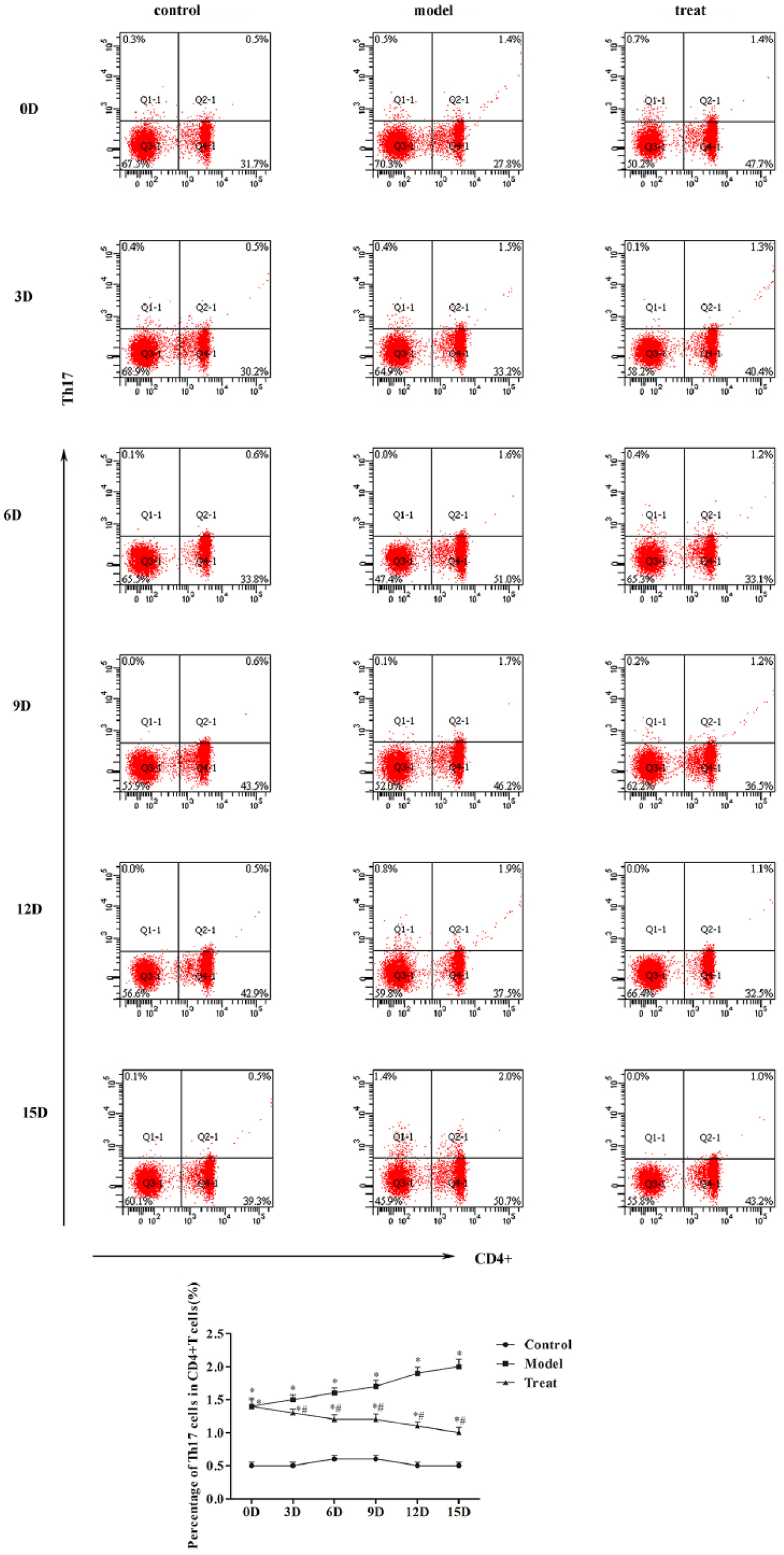
Thermal ablation affects the Th17 cells in peripheral blood: (a) the cell frequency of Th17 cells analyzed by flow cytometry and (b) the percentage of Th17 cells in CD4+ T cells were quantified. Compared with control group, *
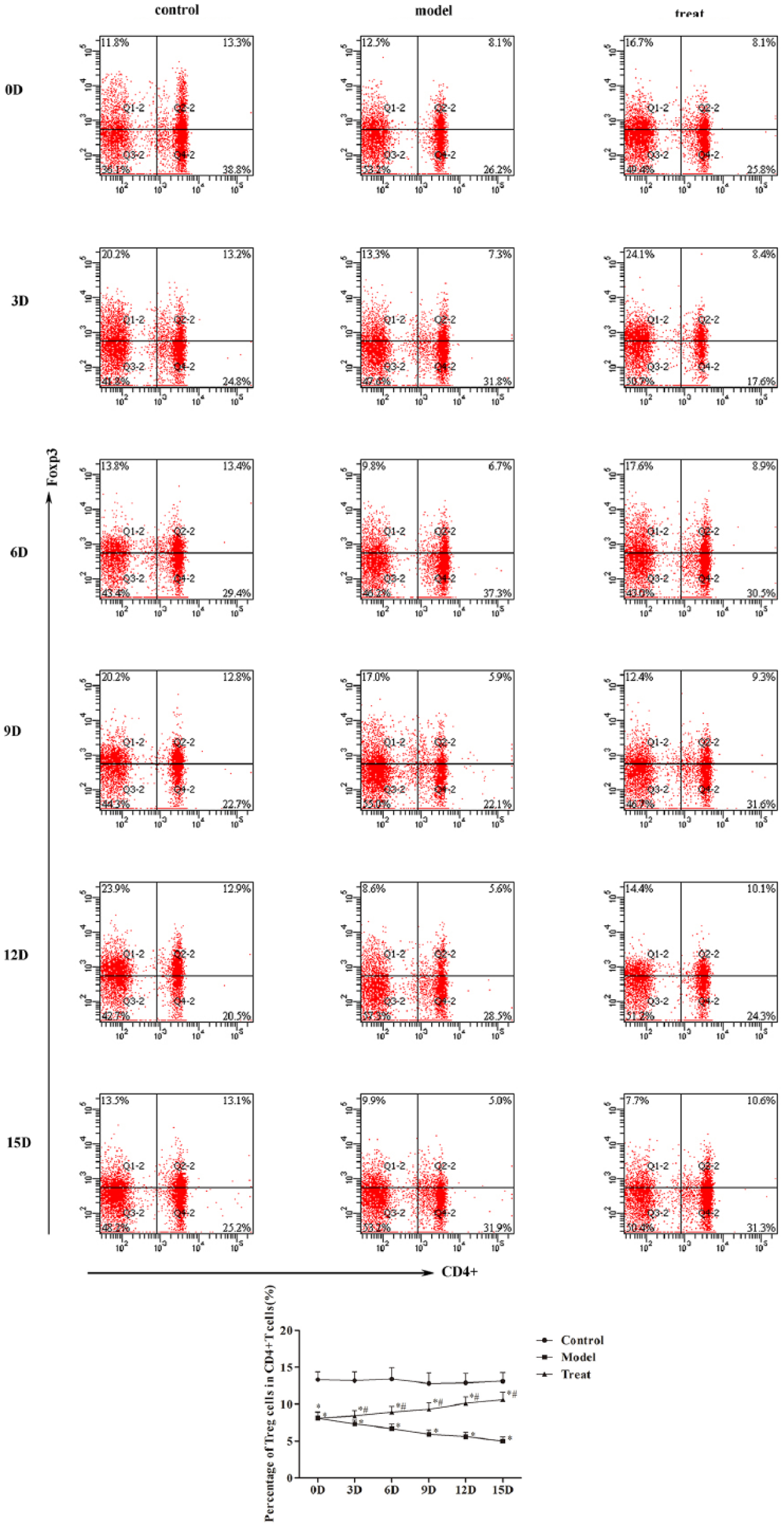
Thermal ablation affects the Treg cells in peripheral blood: (a) the cell frequency of Treg cells analyzed by flow cytometry and (b) the percentage of Treg cells in CD4+ T cells were quantified. Compared with control group, *
The expression of IL-17, IL-23, IL-10, and TGF-β in serum
As shown in Figure 4, the levels of IL-17 and IL-23 in the model group were significantly higher than those in the control group (
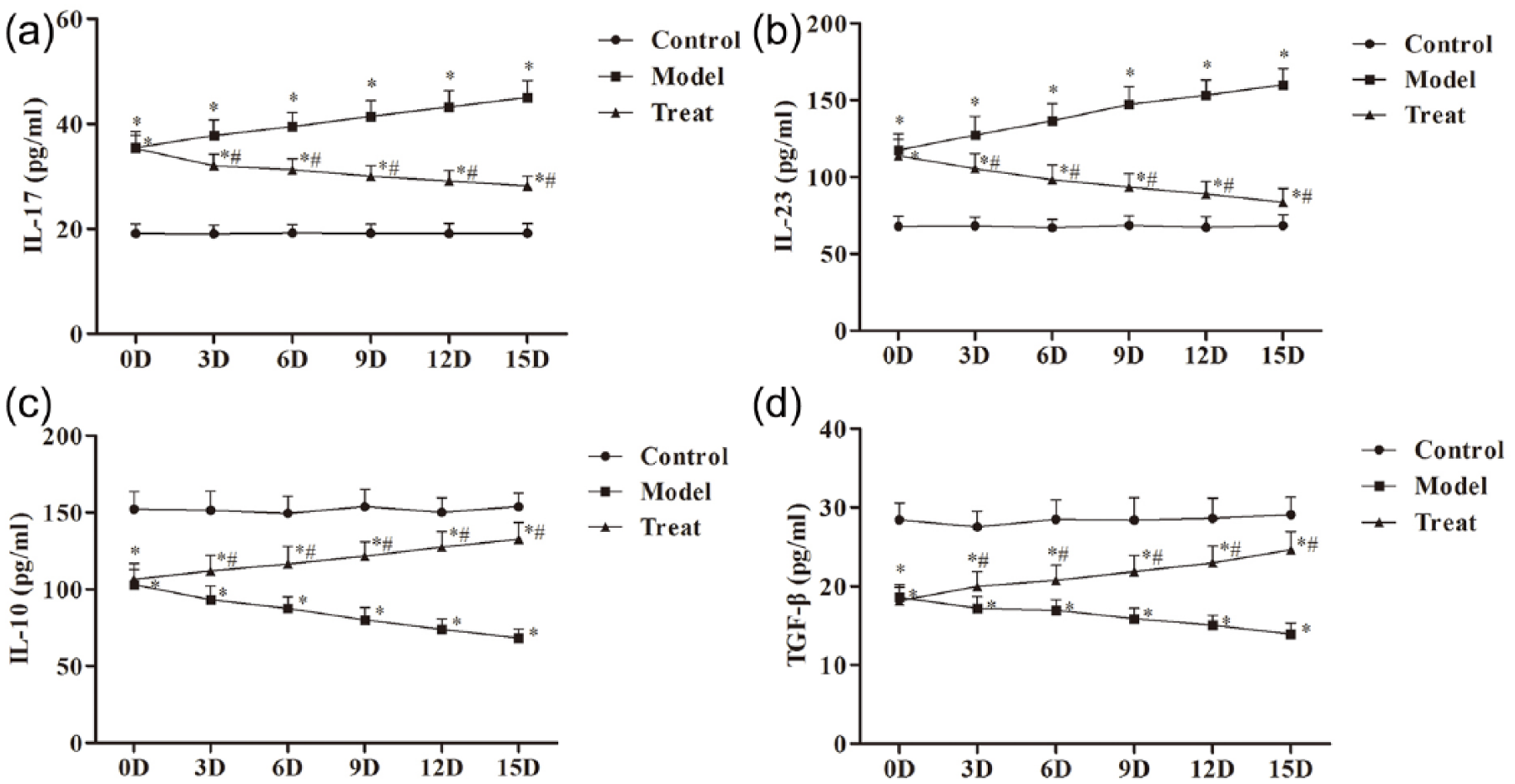
The expression of (a) IL-17, (b) IL-23, (c) IL-10, and (d) TGF-β in serum measured by ELISA. The data were expressed as the mean ± SD. Compared with control group, *
The mRNA expression level of IL-17, RORγt, Foxp3, and TGF-β in tumor tissues
As shown in Figure 5, compared with the model group, the levels of IL-17 and RORγt mRNA were notably reduced; at the same time, the levels of Foxp3 and TGF-β mRNA were increased obviously (
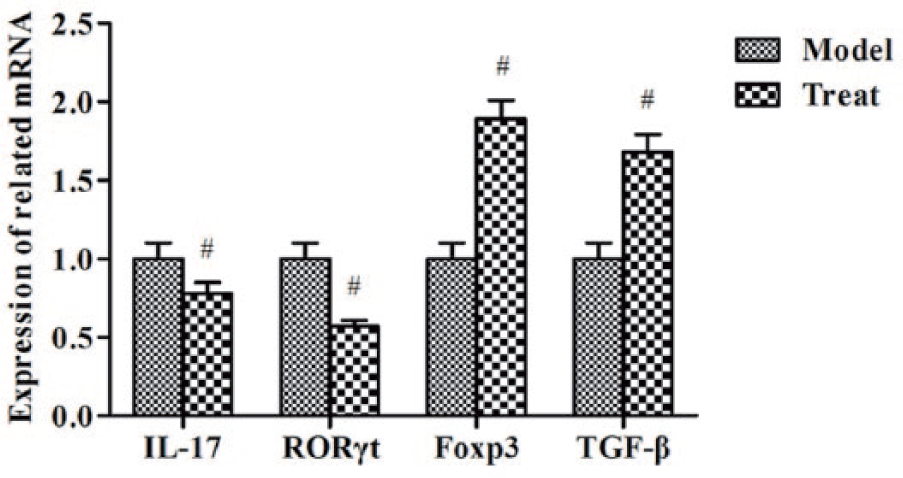
The mRNA expression level of IL-17, RORγt, Foxp3, and TGF-β in tumor tissues measured by qRT-PCR. Compared with model group, #
Thermal ablation treatment inhibits subcutaneously tumor revaccination
Tumors were successfully inoculated subcutaneously in mice in both model and treat groups. In the model group, the tumor formation rate of revaccination in mice was 100%, and the tumor formation rate of the re-inoculated mice in the treat group was only 50% (
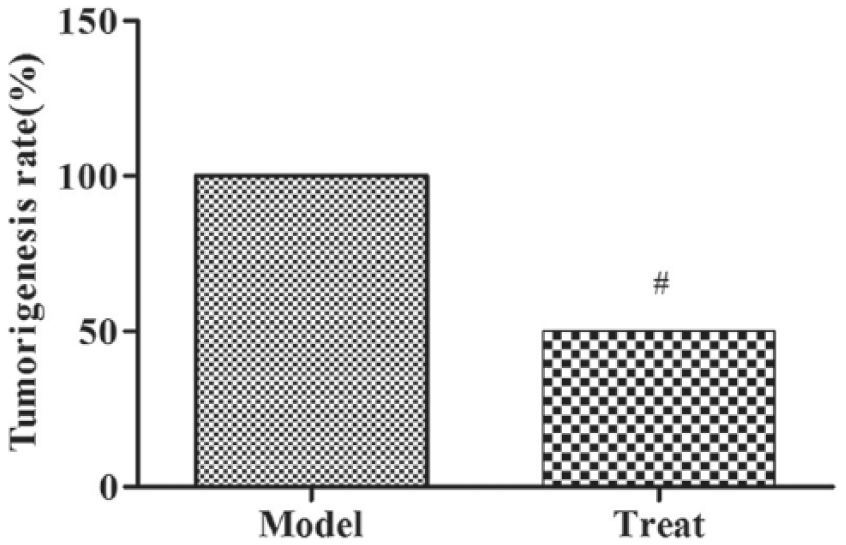
Thermal ablation treatment inhibits subcutaneously tumor re-inoculation. Compared with model group, #
Discussion
In this study, we found the significant effect of thermal ablation in transplantation of HCC in mice. Tumor growth was significantly inhibited after thermal ablation treatment. More importantly, re-inoculation experiments indicated that the thermal ablation treatment has a positive effect on the mice with HCC.
In order to determine the role of thermal ablation on Th17 and Treg cells in peripheral blood, we analyzed the changes of frequency of Th17 and Treg. Th17 and Treg are two lymphocyte subsets with opposite functions. They all derived from the same original T cells. Th17 cells increase the immune response through release of inflammatory cytokines, while Treg cells secrete immunosuppressive cytokines to suppress the immune response. Previous studies demonstrated that the levels of Treg and Th17 cells are increased in patients with chronic hepatitis B infection, 18 but the changes of these two cell subsets in HCC in mice are still unclear. We next checked the frequency of Th17 and Treg cells in each group to explore the role of these cells. Our data suggest that Th17/Treg cell imbalance was affected with the treatment, with a reduction of Th17 cells in the peripheral blood and an increase of Treg cells in the peripheral blood. Few cytokines related to the regulation of Th17/Treg cell imbalance were also discussed in this study. In this study, the levels of IL-17, RORγt and IL-23 were decreased in treat group. As known, IL-17 is secreted by Th17 cells, and the orphan nuclear receptor RORγt is defined as the key transcription factor involved in Th17 cell differentiation, and IL-23 promotes the survival and expansion of Th17 cell.19,20 The levels of IL-10 and TGF-β were increased in treat group. IL-10 might play a negative role for the IL-17 production and TGF-β was reported to induce the differentiation of Th17 cells.19,20 Thus, the Th17/Treg cell imbalance might play a positive role to affect the immune function from the treatment of thermal ablation. The other T cells, such as Th1, Th2, and Th9, may also play a positive role in the immune function. We will continue to research these factors in the future.
Thermal ablation has long been developed and used as a non-surgical alternative treatment of all kinds of tumors, which include liver, kidney, lung, bone, adrenal, and breast malignancies, with the advantage of minimal invasiveness, safety profile, and especially a good choice for patient with late stage of HCC, who lost the surgical chance to remove the tumor. 21 Thermal ablation treatment can directly destroy solid tumors by induceing apoptosis and necrosis of tumor cells.22,23 Although this treatment method has been used both by research and clinical treatment, the mechanism involved in this treatment is still unclear.24,25 Exploring the immune regulation of this treatment is crucial for further development of more efficient thermal ablation method. 26 In this study, we proved that thermal ablation treatment plays a significant anti-tumor role through regulation of Th17/Treg cell imbalance.
HCC is the fifth most common cancer and its incidence is increasing worldwide because of the hepatitis B and C virus infection. At present, several treatment methods were introduced to treat liver cancer, which includes surgical resection, liver transplantation, and ablation therapy (microwave heating, radiofrequency or alcohol ablation, etc.). Because of the high recurrence rate after surgical resection, and more than half of the patients diagnosed as the middle or advanced stage of HCC which makes patients lose the chance of surgical resection, TAT has been rapidly developed due to its invasiveness and simple operation.
Conclusion
In this study, thermal ablation treatment was introduced to treat HCC of mice to explore the mechanism involved in this therapy. Current data suggested that Th17/Treg cell imbalance change by thermal ablation might provide a new sight to explore the mechanism of this treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
