Abstract
Aim:
To explore Ocimum basilicum L. (sweet basil) and linalool for their antifibrotic activity in an arecoline-induced in vitro fibrotic model.
Methods:
Leaf extract of O. basilicum L. (LEOB) and linalool were used as experimental agents to test their antifibrogenic activity in vitro. Half-maximal inhibitory concentration (IC50) for arecoline, ethanolic LEOB, and linalool was determined using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. To evaluate the antifibrotic effect of ethanolic LEOB and linalool on pretreatment, that is, both the testing agents were added to the human buccal fibroblasts (HBFs) prior to induction with arecoline, and reverse transcriptase polymerase chain reaction (RT-PCR) was carried out to study the response of transforming growth factor beta (TGFβ), collagen 1 subtype A2 (COL1A2), and collagen 3 subtype A1 (COL3A1). To appreciate the morphological alterations in HBFs on treatment with arecoline, ethanolic LEOB, and linalool, Masson’s trichrome staining was performed.
Results:
Arecoline enhanced fibrotic activity by upregulating TGFβ1, COL1A2, and COL3A1 levels, whereas ethanolic LEOB and linalool on pretreatment significantly downregulated the increased levels of TGFβ1, COL1A2, and COL3A1 in primary HBF cell cultures.
Conclusion and implication to clinic:
Both ethanolic LEOB and linalool exhibited significant antifibrotic activity in an in vitro model. Further studies in an in vitro model can help attain a foundation for an herbal formulation in gel form that can be prescribed to patients diagnosed with oral submucous fibrosis for topical application. It can also be used synergistically with Western medicine.
Introduction
Oral submucous fibrosis (OSF) is defined as a potentially malignant disorder of the oral mucosa induced by areca nut chewing. 1 The disease is predominantly seen in Southeast Asian countries, namely India, Bangladesh, Pakistan, Sri Lanka, Taiwan, and China, with an incidence ranging up to 0.03–3.2% in the Indian rural population alone. According to epidemiological studies, OSF is reported to have malignant transformation rates of 2.3–7.6% and 3–19%. 2,3
Exposure of the oral mucosa to areca nut constituents generates reactive oxygen species (ROS) causing oxidative stress 4 and produces proinflammatory cytokines leading to the development of fibrosis. 5 The two distinct stages involved in OSF are fibroblast proliferation and the production of extracellular collagen matrix. 5 Varied groups of fibrogenic mediators and cytokines mediate collagen synthesis, via transforming growth factor-beta 1 (TGFβ1) being a predominant mediator of tissue fibrosis. Tissue fibrosis occurs through the TGFβ1/Smad signaling pathway; regulatory Smads are activated by TGFβ1 to mediate fibrosis via transactivation of type I and type III collagen.
Various treatment modalities (conservative physical therapy; medical therapy: steroids, interferon gamma injections, pentoxyfylline, and collagenase; and surgical therapy: extraoral and intraoral flaps, microvascular free flaps, and alloplasts) 6 have been tried time and again, but none have proven effective, as over a period of time the fibrotic bands recur. Pharmaceutical preparations are synthetic substances that cause intolerable short- and long-term side effects. Traditional medicines involving herbal drugs/preparations from natural compounds have been used as an accepted and cost effective treatment option with less side effects for many chronic ailments. Hence, investigating its use for OSF offers a new line of research and hope for treatment.
Ocimum basilicum Linn. (O. basilicum L.; sweet basil) is a perennial herb that belongs to the Lamiaceae family and linalool is the active secondary metabolite present. Plants of the Lamiaceae family and their active compounds are known to impart antifibrotic effect by inhibiting the TGFβ pathway and by promoting enzymatic digestion of the fibrillar deposits. 7 O. basilicum L. downregulates carbon tetrachloride–induced hydroxyproline levels in liver fibrosis. 8 With this background, our aim was to evaluate the antifibrotic effect of ethanolic leaf extract of O. basilicum L. (LEOB) and linalool (monoterpene alcohol) on arecoline-induced fibrosis in primary human buccal fibroblasts (HBFs).
On reviewing the literature, the authors concluded that it is the TGFβ/Smad signaling pathway that gets stimulated on induction with arecoline initiating the upregulation of downstream molecules like collagen 1 subtype A2 (COL1A2) and collagen 3 subtype A1 (COL3A1) that get deposited in the extracellular matrix (ECM) causing tissue fibrosis. Therefore, in the present study, the testing agents (ethanolic LEOB and linalool) were targeted against the three most crucial fibrotic molecules TGFβ1, COL1A2, and COL3A1.
Materials and methods
Preparation of herbal extract
The extraction of the herbal plant O. basilicum L. was carried out according to a previously published protocol. 9 Commercially available, standard linalool (Sigma Aldrich, Bangalore, Karnataka, India) was used.
Gas chromatography/mass spectrometry
Stocks of O. basilicum L. (200 mg/mL) and linalool (100 μg/mL) were prepared in ethanol. Samples were analyzed using gas chromatography/mass spectrometry (GC/MS), GC 7890 A (Agilent, China) gas chromatograph coupled to a mass spectrometer (MS) 5975 C (Agilent, Santa Clara, California, USA). Fast liquid injection mode was used to operate the GC fitted with a 10-μL syringe and a fused silica capillary column. Helium was used as the carrier gas with a flow rate of 1.5 mL/min. The initial oven temperature was at 70°C for 3 min and later increased to 300°C at 10 min for 9 min. The injector was maintained at 250°C and the transfer line at 240°C. A 1 μL portion of the sample volume was injected with a split ratio of 10:1. Fast injection was carried out with an inlet pressure of 14.187 psig. Seventy electron volt was recorded as the positive ion electron spectra. Retention time of linalool was determined and peak was optimized for the same. A comparison was established and the compound in the crude extract at the same retention time was identified and quantified using MS.
Primary cell culture
Primary HBFs were cultured for the present study. Six biopsy specimens of approximately 4 mm in dimension were derived from histologically normal mucosal sites of patients who underwent surgical third molar extraction after attaining informed consent. 2 mg/mL collagenase (HiMedia, Mumbai, Maharashtra, India) was used for digestion of the tissue samples. Of the six samples processed, three tissue samples yielded viable cells ranging from 105 to 106/mL that were further seeded with adjusted appropriate volumes of harvested cells at the rate of 103/mL. The growth medium, Dulbecco’s Modified Eagle’s Medium (DMEM) contained antibiotics (10,000 units penicillin, 10 mg streptomycin, 25 mg amphotericin B (HiMedia), and nystatin along with 10% Fetal Bovine Serum (FBS). The cells were allowed to grow in a monolayer. Cultures were maintained at 37°C in a humidified atmosphere of 5% CO2 and 95% air. Cells between the first and third passages were used in the present study. 10
Cytotoxicity assay
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) colorimetric assay was used to monitor cell viability and proliferation. MTT assay was measured by mitochondrial enzyme succinate dehydrogenase, as described by Mossman and with minor modification by Alley. 11,12 Then, 1 × 104 cells were seeded per well onto 96 well culture plates and incubated for 24 h. MTT solution was freshly prepared in Phosphate Buffered Saline (PBS) (5 mg/mL) just before use and filtered through 0.22-μm filter. Post 24 h, the supernatant was discarded and the monolayer was washed once with 1× PBS; 100 μL of different drug concentrations (arecoline, O. basilicum L., and linalool) were added to cells and incubated for 48 h; and 20 μL of MTT solution was added to each well. Plates were incubated for 4 h at 37°C in 5% CO2; 100 μL of DMSO was added to dissolve the metabolized product of MTT and incubated for 1 h. An absorbance of 590 nm was used to measure the proportion of viable cells. Triplicates were performed for the same.
Masson’s trichrome staining
The cells (1 × 104) suspended in growth media were seeded onto a coverslip placed in 12 well culture plates and were allowed to attain 40–50% confluency. On attaining the required confluency, the growth media was discarded and the cells on the coverslips were washed thrice in 1× PBS at room temperature. Cells were fixed in 3.7% formaldehyde and incubated for 10 min. Staining was carried out stepwise according to protocol standardized by the Lillehei Heart Institute (Histology and Microscopy Research Facility), University of Minnesota (Minneapolis, Minnesota, USA).
Reverse transcriptase polymerase chain reaction
TRIsoln (Merck, GeNei, Bangalore, Karnataka, India) was used to extract total RNA, according to the protocol stated by the manufacturers. RNA was spectrophotometrically measured at 260 nm by resuspension of RNA pellets in 20 μL molecular biological grade water, placed at 55°C for 5 min. A Thermo Scientific Verso cDNA synthesis kit (Abgene, Portsmouth, New Hampshire, USA) was used to synthesize complementary DNA (cDNA). The conversion and cycling program was performed as per the manufacturer’s instructions. The PCR contained a forward and reverse primer (Bioserve, Coimbatore, Tamil Nadu, India), Taq DNA polymerase (Ampliqon, Odense, Denmark), and 10 ng cDNA. β-actin was used as a housekeeping gene, to confirm the homogeneity of RNA loading. Detailed temperature and time for denaturation, annealing, and extension of the genes mentioned earlier are given in Table 1, which has been previously published. 13 Then, 2% agarose gel electrophoresis was used to analyze the PCR products; GelDoc (Biorad, Hercules, California, USA) was used to photograph the intensity of each band, post-normalization with β-actin messenger RNA (mRNA). The bands were further analyzed using ImageJ software (National Institutes of Health, Bethesda, Maryland, USA). List of nucleotide sequences, base pair (bp) product size, and annealing time and temperature for β-actin, TGFβ1, COL1A2, and COL3A1 was followed as previously published. 13
To study the antifibrogenic effects of ethanolic LEOB and linalool, HBFs were treated with the testing agents prior (pretreatment) to induction with arecoline as the experiment was carried out in an in vitro setting. The work plan for the present study is summarized in Figure 1.

Strategy to develop OSMF Cell Line model for screening anti-fibrotic compounds.
Statistical analysis
All data are expressed as mean ± standard error of mean. The differences in gene expression were evaluated by Kruskal–Wallis rank sum test. Post Tukey–Kramer (Nemenyi) test was used to evaluate statistically significant and effective concentrations of arecoline, O. basilicum L., and linalool.
Experimental results
Crude ethanolic LEOB was prepared using the maceration technique. The plant material, on removal of impurities, weighed 4.5 kg on purchase and yielded a final extract of 102 g. The final ethanolic extract was dark green in color, tasted minty bitter, and had an aromatic smell (Figure 2).

Crude ethanolic LEOB. Fresh leaves of O. basilicum weighed 4.5 kg on purchase. Dried leaves (shade dried) after removal of impurities appeared dark green in color. Final extract yield was 102 g. LEOB: leaf extract of Ocimum basilicum L.
GC/MS analysis of linalool in ethanolic LEOB
On evaluating the standard linalool as a reference, a retention time of 6.95 was observed (Figure 3). On analyzing the ethanolic LEOB, a similar peak with a retention time of 6.95 was observed, confirming the presence of linalool (Figure 3). On calculation, 0.2 g of linalool was present in 200 g of ethanolic LEOB.

GC/MS analysis of linalool. Linalool was detected at a retention time of 6.95 (red box) in the ethanolic LEOB; 0.2 g of linalool was quantified in 200 g of ethanolic LEOB. GC/MS: gas chromatography/mass spectrometry; LEOB: leaf extract of Ocimum basilicum L.
Cytotoxic effect of arecoline, ethanolic LEOB, and linalool on HBF
Safe dose (half-maximal inhibitory concentration, IC50) of arecoline, ethanolic LEOB, and linalool was determined on HBF in vitro using the MTT colorimetric assay. Arecoline induced cell death of 50% and 46% at 50 and 25 μg/mL, respectively (Figure 4). Ethanolic LEOB and linalool induced cytotoxicity to HBFs at a concentration above 125 µg/mL and 125 µM, respectively (*p < 0.05), during 48-h incubation. At concentrations of 250, 500, and 1000 µg/mL of ethanolic LEOB and 250, 500, and 1000 µM of linalool, fibroblast cell proliferation was inhibited up to 54.3%, 56.5%, and 60.15% for ethanolic LEOB and 54.4%, 55.21%, and 58.8% for linalool, respectively (Figure 4).
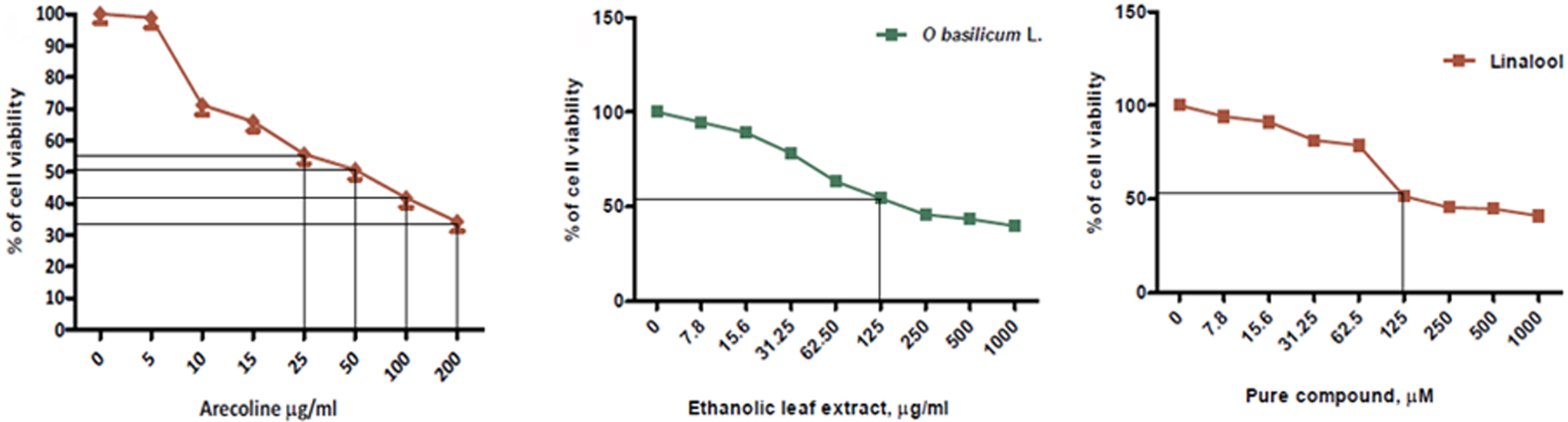
MTT assay to determine the viability of cells. Graphical representation of the percentage of cell viability on treatment of HBFs with different concentrations of arecoline, ethanolic LEOB, and linalool. Data presented are mean ± SD of results from three independent experiments. Half-maximal inhibitory concentration (IC50) for arecoline was determined as 50 μg/mL, for ethanolic LEOB as 125 μg/mL, and for linalool as 125 μM. MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; HBF: human buccal fibroblast; LEOB: leaf extract of Ocimum basilicum L.
Fibrotic effect of arecoline on HBFs: In vitro semiquantative assessment of fibrotic (TGFβ1, COL1A2, and COL3A1) markers post-arecoline treatment
Data presented in Figure 5 demonstrated that HBFs incubated with arecoline at increasing concentrations (0–50 µg/mL) for 48 h were normal showing uniform expression of actin (630 bp) levels. Fibrogenic mediators TGFβ1, COL1A2, and COL3A1 showed upregulation of expression with increasing arecoline treatment. TGFβ1 activity increased by 3- to 5-fold, COL1A2 by 8- to 10-fold, and COL3A1 by 20-fold at 25 µg/mL and 50 µg/mL, respectively, post-arecoline treatment.
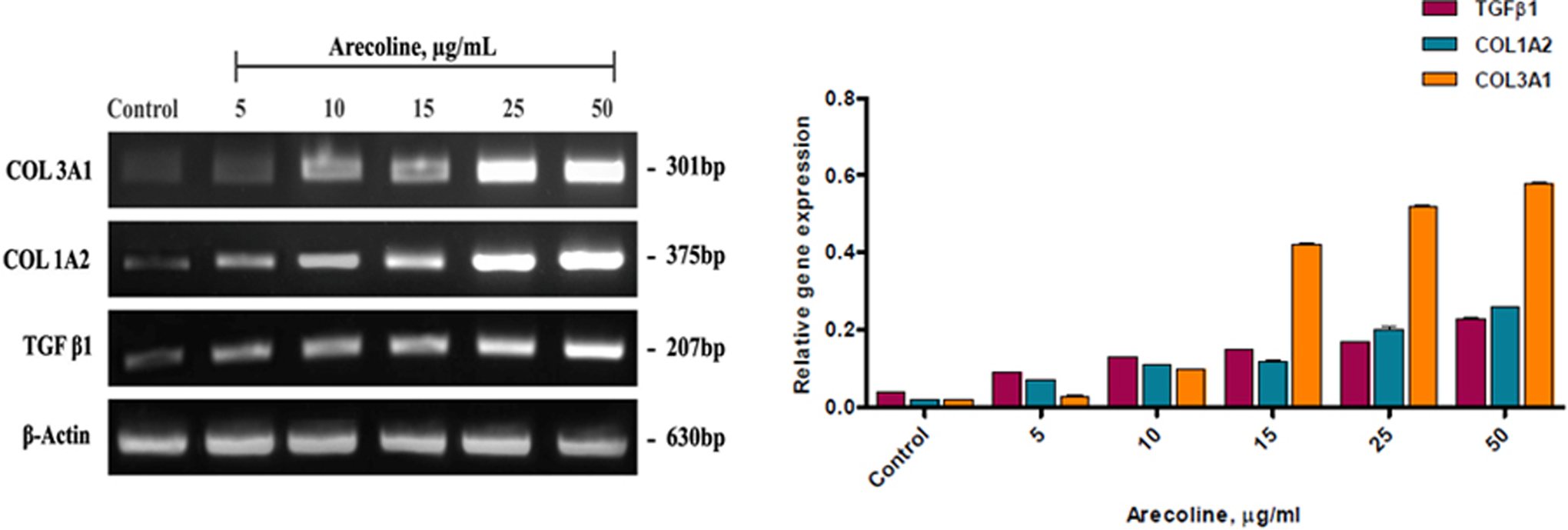
Expression of TGFβ1, COL1A2, and COL3A1 on arecoline-treated HBFs by RT-PCR assay. Cells were exposed to arecoline concentrations for 48 h containing as indicated. β-actin mRNA served as the loading control. Gel image showing a dose-dependent semiquantative increase in the mRNA expression levels of COL3A1, COL1A2, and TGFβ1 on arecoline treatment with nontoxic concentrations (0–50 μg/mL) reproduced with permission from reference no: 4314081379259; graphical representation showing a comparison between arecoline-treated samples and control (untreated). Quantification was done using ImageJ software and normalized using β-actin. Results were reported as mean ± standard error of means for at least three analyses for each sample. **p < 0.05 was considered statistically significant. Post Tukey–Kramer (Nemenyi) test was used to evaluate statistically significant and effective concentrations of arecoline. TGFβ1: transforming growth factor-beta 1; COL1A2: collagen 1 subtype A2; COL3A1: collagen 3 subtype A1; RT-PCR: reverse transcriptase polymerase chain reaction; mRNA: messenger RNA; HBF: human buccal fibroblast.
Antifibrotic effect of ethanolic LEOB: In vitro semiquantative assessment of fibrotic (TGFβ1, COL1A2, and COL3A1) markers on pretreatment with ethanolic LEOB on arecoline-induced fibrosis in HBF
On observation, arecoline (25 µg/mL) induced fibrosis by upregulating TGFβ1 expression by 103-fold, COL1A2 by 200-fold, and COL3A1 by 104-fold when compared to baseline control (0 µg/mL; Figure 6). On pretreatment with ethanolic LEOB, reduction in TGFβ1 level was observed, with maximum downregulation of 18-fold at 62.5 μg/mL and 52-fold at 125 μg/mL (see Figure 6; *p < 0.05). COL1A2 level decreased by 9-fold at 62.5 μg/mL and by 14-fold at 125 μg/mL (*p < 0.05), COL3A1 levels decreased by 4-fold and 6-fold at 62.5 and 125 μg/mL (*p < 0.05), respectively. On an overall observation, ethanolic LEOB exhibited maximum antifibrotic activity at higher concentrations, although it demonstrated its antifibrotic efficiency even at lower concentrations.

Effect of ethanolic crude LEOB on the expression of TGFβ1, COL1A2, and COL3A1 on arecoline-treated HBFs by RT-PCR assay. Induced control group was treated with 25 μg/mL arecoline for 48 h and the baseline control group was untreated. Gel image showing semiquantitative mRNA expression levels of TGFβ1, COL1A2, and COL3A1 after LEOB extract induction at different concentrations (7.8, 15.6, 31.25, 62.5, and 125 μg/mL) for 48 h. β-actin was used for normalization and verification; graphical representation showing a comparison between the ethanolic LEOB–treated samples and control (induced). Quantification was done using ImageJ software. Data presented are the mean ± standard error of means of results from three independent experiments (**p < 0.05 and *p < 0.001 vs. control). Post Tukey–Kramer (Nemenyi) test was used to evaluate statistically significant and effective concentrations of ethanolic LEOB. LEOB: leaf extract of Ocimum basilicum L.; TGFβ1: transforming growth factor-beta 1; COL1A2: collagen 1 subtype A2; COL3A1: collagen 3 subtype A1; HBF: human buccal fibroblast; RT-PCR: reverse transcriptase polymerase chain reaction; mRNA: messenger RNA.
Antifibrotic effect of linalool: In vitro semiquantative assessment of fibrotic (TGFβ1, COL1A2, and COL3A1) markers on pretreatment with linalool on arecoline-induced fibrosis in HBF
On observation, arecoline (25 µg/mL) induced fibrosis by upregulating TGFβ1 expression by 37-fold, COL1A2 by 55-fold, and COL3A1 by 52-fold when compared to baseline control (Figure 7). On pretreatment with linalool (Figure 7), effective reduction in TGFβ1 level by almost 12-fold was observed at lower concentrations of 7.8 and 15.6 µM (*p < 0.05) and by 11-fold at 31.25 µM. COL1A2 levels decreased by 2-fold at concentrations of 7.8–31.25 µM (*p < 0.05) of linalool, whereas COL3A1 levels decreased significantly by 17-fold at 7.8 µM, 13-fold at 15.6 µM, and 15-fold at 31.25 µM (*p < 0.05), respectively. Overall, reduction in expression levels of TGFβ1, COL1A2, and COL3A1 was observed at lower concentrations predominantly; increasing concentrations up to 125 µM showed lesser fold downregulation of fibrotic markers.

Effect of linalool on the expression of TGFβ1, COL1A2, and COL3A1 on arecoline-treated HBFs by RT-PCR assay. Induced control group was treated with 25 μg/mL arecoline for 48 h and the baseline control group was untreated. Gel image showing semiquantitative mRNA expression levels of TGFβ1, COL1A2, and COL3A1 after linalool induction at different concentrations (7.8, 15.6, 31.25, 62.5, and 125 M) for 48 h. β-actin was used for normalization and verification; graphical representation showing a comparison between the linalool-treated samples and control (induced). Quantification was done using ImageJ software. Data presented are the mean ± standard error of means of results from three independent experiments (**p < 0.05 and *p < 0.001 vs. control). Post Tukey–Kramer (Nemenyi) test was used to evaluate statistically significant and effective concentrations of linalool. TGFβ1: transforming growth factor-beta 1; COL1A2: collagen 1 subtype A2; COL3A1: collagen 3 subtype A1; HBF: human buccal fibroblast; RT-PCR: reverse transcriptase polymerase chain reaction; mRNA: messenger RNA.
Morphological assessment of HBFs and qualitative assessment of collagen, post-arecoline treatment and on pretreatment with linalool
Masson’s trichrome staining (MTS) was performed to observe fibroblast morphology and to qualitatively assess collagen deposition in the ECM, post-arecoline treatment, and compare it with linalool pretreated cells. From the data shown in Figure 8, effective concentrations of linalool that downregulated the expression of fibrotic markers were 7.8 and 15.6 µM. Hence, to assess morphological restoration of arecoline-induced fibrosis, HBFs were pretreated with 15.6 µM linalool for 48 h. Fibroblasts exhibited spindle morphology and scanty ECM deposition was observed to be present in the untreated (control) group (Figure 8). In contrast, it was observed that HBFs treated with 25 μg/mL of arecoline appeared distorted, rounded, and stained strongly positive for collagen demonstrating excessive deposition of ECM (Figure 8). In the linalool treated group, fibroblasts retained their spindle morphology and showed very faint collagen staining, thereby decreasing ECM deposition. The group treated with linalool appeared similar to the control group (Figure 8). In the present study, no attempt was made to identify the presence or absence of myofibroblasts.

Masson’s trichrome staining for comparing collagen production qualitatively between control and treated group of HBFs. Control group (untreated); HBFs treated with 25 μg/mL arecoline for 48 h reproduced with permission from reference no: 4314081379259; HBF pretreated with 15.6 μM linalool for 48 h. HBF: human buccal fibroblast.
Discussion
OSF is a chronic, insidious disease of the oral mucosa that is associated with significant functional morbidity and an augmented risk of malignancy. 14 The etiopathogenesis of OSF was earlier believed to be multifactorial; a number of factors trigger the disease process by causing juxta-epithelial inflammatory reaction followed by epithelial atrophy. The prime factor recognized responsible for OSF supported by epidemiological case control, animal experiments, and tissue culture studies is chewing of betel quid, containing betel leaf, areca nut, tobacco, slaked lime, and other spices.
Areca nut is largely composed of carbohydrates, fats, proteins, crude fiber, polyphenols, alkaloids, and mineral matter. Among the chemical constituents, alkaloids like arecoline (being the most abundant), arecaidine, guvacine, isoguvacine, and arecolidine are most toxic. 15 Arecoline undergoes nitrosation and has been tested in in vitro experiments to cause lung adenocarcinoma and esophageal papilloma in mice models. Locally, areca nut chewing is known to cause trauma and injury to the oral mucosa due to its abrasive nature, and continuous contact between the quid (combination of betel leaf, areca nut, and slaked lime) and the oral mucosa leads to absorption and metabolism of alkaloids; this continuous irritation leads to injury-related oxidative stress, chronic inflammation, and constant stimulation of fibroblasts, leading to upregulation of varied groups of cytokines such as TGFβ1, connective tissue growth factor, platelet-derived growth factor (PDGF), and basic fibroblast growth factor (bFGF). 16 TGFβ1, a potent fibrogenic cytokine, stimulates collagen production via the TGFβ/Smad signaling pathway; on regular external stimulation, it loses its autocrine activity. It upregulates tissue inhibitor matrix metalloprotienases (TIMPs) and downregulates matrix metalloprotienases (MMPs) producing an imbalance between collagen production and degradation, which leads to excessive accumulation of type I and type III collagen, hyalinization, and occlusion of blood vessels in the submucosa. 17
Fibrosis is a condition that lacks a universal effective therapeutic regimen. Treating patients diagnosed with OSF is an enigma, and the prime objective is to provide relief of symptoms (burning sensation and intolerance to spicy food), to improve mouth opening, and to restore functional normalcy. Studies have recommended discontinuation of the habit (areca nut chewing), physiotherapy, combination therapies (steroids and hyaluronidase injections), collagenase, and placental extracts, but none have been effective. Ayurvedic medicines have proven to exhibit potent antioxidant, antimicrobial, analgesic, anti-inflammatory, antiulcerogenic, hepatoprotective, and antifibrotic activities in in vitro and in vivo models. These medicinal properties can be attributed to the presence of principal constituents such as phenols, flavanoids, terpenoids, alkaloids, and aglycones.
In the present study, our objective was to downregulate the potent profibrogenic mediator TGFβ1 and its downstream molecules COL1A2 and COL3A1. We considered a medicinal plant, O. basilicum L. and its active monoterpene linalool for attenuating arecoline-induced fibrosis in buccal mucosal fibroblasts.
Arecoline, a known fibrosis inducer, was used in the present study to establish a cultured human cell line in vitro model. To establish OSF as a collagen metabolic disorder, the fibrotic effect of arecoline was evaluated. On exposing HBFs with the determined nontoxic concentrations (0–50 μg/mL) of arecoline for 48 h (Figure 4), a significant elevation was observed in the mRNA expression levels of TGFβ1 by 3- to 5-fold with an annealing temperature of 56°C and duration of 40 s. COL1A2 levels upregulated by 8- to 10-fold with an annealing temperature of 54°C and duration of 40 s. COL3A1 levels increased by 20-fold at 25 and 50 μg/mL (*p < 0.05) with an annealing temperature of 54°C and duration of 30 s (Figure 5). Recent studies have demonstrated enhanced expression of TGFβ target genes, namely αSMA and collagen isoforms (COL1A1, COL1A2, and COL3A1) on exposing human gingival fibroblasts to areca nut. 18 Similarly, upregulation of downstream molecules of TGFβ pathway, namely COL1A2, COL3A1, LOX, and TIMPS, has been observed in OSF tissues. 19 It has been explained earlier that prolonged exposure of areca nut constituents to the oral mucosa causes the release of fibrogenic growth factors like TGFβ, PDGF, and bFGF, thereby stimulating excessive production of type I and type III collagen. 20
To evaluate the antifibrotic effect of O. basilicum L., freshly dried leaves of O. basilicum L. were subjected to maceration. A final yield of 102 g of ethanolic LEOB was obtained (Figure 2). On observation, arecoline (25 μg/mL) induced fibrosis by upregulating TGFβ1 expression by 103-fold, COL1A2 by 200-fold, and COL3A1 by 104-fold when compared to baseline (0 μg/mL, Figure 6). Data presented in Figure 6 indicated that pretreatment of HBFs with nontoxic concentrations (7.8–125 µg/mL) of ethanolic LEOB for 48 h (Figure 4) significantly downregulated the mRNA expression levels of the elevated fibrotic markers. TGFβ1 showed maximum reduction of 18-fold at 62.5 µg/mL and 52-fold at 125 µg/mL. COL1A2 levels decreased by 9-fold at 62.5 µg/mL and 14-fold at 125 µg/mL and COL3A1 level decreased by 6-fold at 62.5 and 125 µg/mL. The antifibrotic effect of ethanolic LEOB observed in our study model was concurrent to previously documented studies, wherein it was observed that O. basilicum L. of the Lamiaceae family efficiently retarded fibroblast turnover by a flow cytometry analysis. It also decreased TGFβ secretion and impaired collagen synthesis by upregulating matrix degradation enzymes MMP1 and MMP9, overall downregulating hydroxyproline content, an indirect measure to quantify collagen in cell culture supernatant. 7 It has also been reported that 200 mg/kg b.w of O. basilicum L. extract downregulated CCl4-induced liver fibrosis in a rat model, by increasing liver function enzyme activity and downregulating the serum marker enzymes such as alkaline phosphatase. It retained tissue architecture and reduced hydroxyproline deposition in hepatocytes, thereby preventing hepatic fibrosis. 8 Earlier studies have attributed the antifibrotic activity of O. basilicum L. to the presence of essential oils such as eugenol, linalool, thymol, and α-caryophylene and the presence of diterpenes and triterpenes. 21 In our study, leaves of O. basilicum L. were used for extract preparation and experimentation, as the leaves (young and mature) are known to contain the maximum amount of essential oils.
To evaluate the antifibrotic activity of the essential oil linalool, GC/MS was performed to standardize and quantify the amount of linalool in the ethanolic LEOB. The data presented in GC/MS (Figure 3) showed the presence of 0.2% linalool in 200 g of ethanolic LEOB with a retention time of 6.95. The presence of low quantities of linalool can be explained due to its volatile nature and low molecular weight, which makes the quantification of the compound arduous. It also largely depends upon the type of solvent, extraction method used, and geographical and seasonal variations. 22 Highest linalool concentration of 69.3% on GC/MS analysis from leaves of O. basilicum L. harvested at 16:00 h and dried at 40°C has been reported earlier. Drying leaves at higher temperatures of 50°C and 60°C decreases linalool content. 23 On the contrary, low essential oil content (linalool, eugenol, rosmarinic acid) of 0.33–0.47% on GC/MS analysis in fresh leaves of O. basilicum L. from Sudan has also been reported. 24 Recently, a study reported 95% of total essential oil obtained by hydrodistillation of fresh leaves of O. basilicum L. (Nigeria), and 24 compounds were identified by GC/MS analysis of which linalool was 10.8%, methylchavicol 60.3%, and methylcinnamate 6.3%. 25
On observation, arecoline (25 μg/mL) induced fibrosis by upregulating TGFβ1 expression by 37-fold, COL1A2 by 55-fold, and COL3A1 by 52-fold when compared to baseline (0 μg/mL, Figure 7). On evaluating the antifibrotic activity of linalool, data presented in Figure 7 showed that pretreatment to HBFs with nontoxic concentrations (7.8–125 µM) of linalool for 48 h (Figure 4) significantly downregulated the mRNA expression levels of COL3A1, COL1A2, and TGFβ1 at a minimum concentration of 7.8 µM. TGFβ1 decreased by almost 12-fold at 7.8 and 15.6 µM and 11-fold at 31.25 µM (*p < 0.05). COL1A2 levels also decreased by almost 2-fold at lower concentrations of 7.8, 15.6, and 31.25 µM. COL3A1 level decreased by 17-fold at 7.8 µM, 13-fold at 15.6 µM, and 15-fold at 31.25 µM, whereas a minimal reduction by only 1-fold was observed at higher concentrations of 62.5 and 125 µM. The antifibrotic activity exhibited by linalool at lower concentrations can be attributed to its purity. The antifibrotic effect of linalool observed in our study model was in concurrence with studies which reported that linalool treatment for 72 h in diabetes-induced male Wistar rats significantly decreased the levels of TNFα, IL-6, TGFβ1, and NF-κB, thereby prevented ROS formation and inhibited TGFβ1-mediated production of collagen type I and fibronectin. 26 The antifibrotic activity of linalool has been attributed not only to its radical scavenging potential (antioxidant activity) but also to its interactions with signaling cascades involving cytokines and regulatory transcription factors. 27
MTS showed that fibroblasts exhibited spindle morphology and deposition of ECM in the untreated (control) group (Figure 8); in contrast, HBFs treated with arecoline (25 μg/mL) appeared distorted, rounded, and stained strongly positive for collagen (Figure 8), demonstrating excessive ECM deposition. Haphazard arrangement of type I collagen in tissue sections of stage I OSF and parallel arrangement in stages II and III on using MTS technique have been observed previously. 28 A similar study demonstrated amorphous areas, an indication of early hyalinization in subepithelial buccal mucosal tissues of mice treated with aqueous areca nut extract for 300–600 days using MTS. On pretreating HBFs with linalool (Figure 8), fibroblasts retained their spindle morphology with a faint positivity for collagen staining, decreasing ECM deposition. 29 Our study is the first of its kind to evaluate the fibrotic activity post-arecoline treatment and antifibrotic activity on pretreatment with minimum concentration (15.6 µM) of linalool on oral buccal fibroblasts to study cell morphology and collagen deposition in the ECM qualitatively.
Inspite of the low yield, the process of crude extract formulation was economical when compared to the commercially purchased pure linalool essential oil. In the present study, both ethanolic LEOB and linalool demonstrated significant antifibrotic potential. On comparison, linalool exhibited effective antifibrogenic effect at lower concentrations, thus proving its effectiveness.
Conclusion
The above experiments cumulatively contribute in understanding the mechanisms of action and therapeutic values of ethanobotanical plants and their respective pure compounds. The present study was invaluable in the identification of the lead compound linalool and plant species O. basilicum L. with promising and efficient antifibrotic activity in an in vitro model of oral submucosal fibrosis. The study also highlights the importance of alternative medicine as an approach to mitigate the progression of fibrosis. To our knowledge, till date no in vitro study comprising HBFs has been reported in literature to evaluate the efficacy of O. basilicum L. and linalool in the management modality for OSF. It is evident from the study that both O. basilicum L. and linalool assure as promising antifibrotic agents in the treatment of OSF in future.
Footnotes
Acknowledgements
The authors wish to thank Dr C. Ravindran and the entire Department of Oral and Maxillofacial Surgery for their extended help in providing patient tissue samples. The authors also thank Dr. Rajasekaran and Dr.Dinesh for providing their guidance in performing Masson trichrome staining.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research work, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: One of the authors received funding under the ICMR - TALENT SEARCH SCHEME from the Indian Council of Medical Research, New Delhi, India.
