Abstract
Background:
Oral submucous fibrosis (OSF) is an oral potentially malignant disorder depicting extensive fibrosis of the submucosa affecting most parts of the oral cavity, including pharynx and upper third of the oesophagus. Despite its high potentially malignant nature, transformation of the epithelium towards malignancy in the background of fibrosis has not been studied extensively till date. Hypoxia Inducible Factor-1α (HIF-1α) is a master transcription factor that is induced by hypoxia and has found to be significantly associated with both fibrosis and malignancy.
Aim:
To test the hypothesis of differential expression of HIF-1α in malignant transformation of OSF.
Materials and methods:
Fifteen samples each of Normal Mucosa (NOM), OSF and Oral Squamous Cell Carcinoma (OSCC) making total of 45 tissues were studied by formalin fixed Haematoxylin and Eosin (H&E)-stained sections and frozen fixed for Reverse Transcription Polymerase Chain Reaction (RT-PCR). The relationship between differential HIF-1α messenger RNA expression level and oral epithelial dysplasia, inter incisal opening and grade of OSCC was studied.
Conclusions:
An upregulation of HIF-1α expression level is positively correlated with oral carcinogenesis and grade of OSCC, whereas downregulation is associated with fibrosis. Hence, it can be used as both diagnostic as well as a prognostic marker.
Keywords
Introduction
Mass ignorance and poverty along with deleterious oral habits among Indians have resulted in an alarming increase in the number of oral squamous cell carcinoma (OSCC) patients. Of 270,000 cases of oral cancers reported worldwide annually, 82,000 are diagnosed in India alone. 1 Oral submucous fibrosis (OSF), now globally accepted as an Indian disease, has one of the highest malignant transformation rate among oral potentially malignant disorder. The epidemiological data has indicated an alarming rise in OSF cases in India, from an estimated 250,000 cases in 1980 to 2 million cases in 1993. This could be due to an upsurge in the popularity of commercially prepared areca nut products (pan masala) in India and an increased uptake of this habit by young people (Gupta et al, 1998) due to easy access, affordable price and marketing.
OSF is characterized by changes in the connective tissue fibres of the lamina propria and this extensive fibrosis of the connective tissue also causes reduction of vascularity and subsequently hypoxia in oral mucosa. 2 Malignant transformation is the key mechanism in mucosal transformation to OSCC via potentially malignant mucosa. Adaptation to low oxygen tension, that is, hypoxia in cells and tissues leads to transcriptional induction of series of genes that participate in angiogenesis, iron metabolism, glucose metabolism and cell proliferation/survival. The primary factor mediating this response is Hypoxia Inducible Factor-1α (HIF-1α) Fig 3, an oxygen-sensitive transcriptional activator. HIF-1α is a master transcription factor that is stabilized in hypoxic conditions, after which it translocates into the nucleus to interact with hypoxia-responsive elements. 3 HIF-1α is a key regulator responsible for the induction of the genes that facilitate adaptation and survival of cells from normoxia to hypoxia. Recent literature shows that HIF-1α is associated with the upregulation of various growth factors associated with OSCC like vascular endothelial growth factor (VEGF), transforming growth factor-beta (TGF-β), fibroblast growth factor (FGF), platelet-derived growth factor (PDGF) and epidermal growth factor receptor (EGFR). Studies have also found association between hypoxia and fibrosis in renal fibroblasts 4 and lung fibroblasts. 5
Thus, we hypothesize that fibrosis in OSF and malignant transformation in the background of fibrosis mediates via HIF-1α. This study is an attempt in this regard.
Materials and methods
Ethical permission for the study was obtained from the ethical committee of the institution. The patients were considered on first come first serve basis and informed consent was subsequently obtained. Our study samples comprised of fresh frozen biopsies from a total of 45 patients. Fifteen samples each of NOM, OSF and OSCC were obtained to study the differential expression level of HIF-1α messenger RNA (mRNA) by reverse transcription-polymerase chain reaction (RT-PCR), also samples were subjected to routine formalin fixed Haematoxylin and Eosin (H&E) stained histopathological examination.
RT-PCR
A maximum of 30 mg of tissues were homogenized in a homogenizer machine. mRNA was extracted directly from the tissue using the RNeasy-Mini-Kit (Qiagen, Germany) and extracted mRNA was evaluated for purity and concentration in Thermo Scientific Nanodrop 1000 (Holmes Chapel, Cheshire, UK). mRNA was reverse transcribed into complementary DNA using the One Step SYBR® Prime Script TM RT-PCR Kit (TaKaRa Biotechnology, Dalian Co Ltd, USA) and processed according to the manufacturers’ protocols. Predesigned PCR primers used for human HIF-1α and β-actin were selected by primer basic local alignment search tool (BLAST) (National Centre for Biotechnology Information/Primer-BLAST). Primer sequence for HIF-1α (based on GenBank accession no. NM_001530.2) was sense, 5-GATAGCAAGACTTTCCTCAGTCG-3 and antisense, TGGCTCATATCCCATCAATTC-3 (90-bp PCR product). Primers for the housekeeping gene, β-actin were sense 5′–TTCCAGCCTTCCTTCCTGG-3′ and anti-sense 5′–TTGCGCTCAGGAGGAGCAAT-3′ (141-bp PCR product).
Semi-quantitative PCR
Semi-quantitative PCR conditions were RT step: 48°C, 30 min, hold; enzyme activation: 95°C, 10 min, hold; denaturation: 95°C 15 s, 40 cycles and anneal 60°C, 1 min, 40 cycles in Multicolor Real Time PCR ( Bio Rad , Serial NO 584BR4114, USA) detection system to obtain the
Statistical analysis
The statistical analysis was performed using 2−▵▵Ct (Livak) method, Relative Quantification (RQ) = 2−▵▵Ct. To get the relative expression of target gene in the target group in comparison with the control group in terms of fold change. Log10 of RQ was calculated to know the upregulation or downregulation of the target gene.
Histopathological examination
H&E-stained sections were prepared from all the cases. The presence of oral epithelial dysplasia (OED) was evaluated in the epithelium of OSF tissue. OSCC was graded into grade I, II and III according to Bryne’s et al. (1992) deep invasive cell grading system. 6 (Figs 1 & 2; Tables 1 & 2)

Relative quantification of HIF-1α for different paired comparisons. HIF-1α: hypoxia inducible factor-1α.
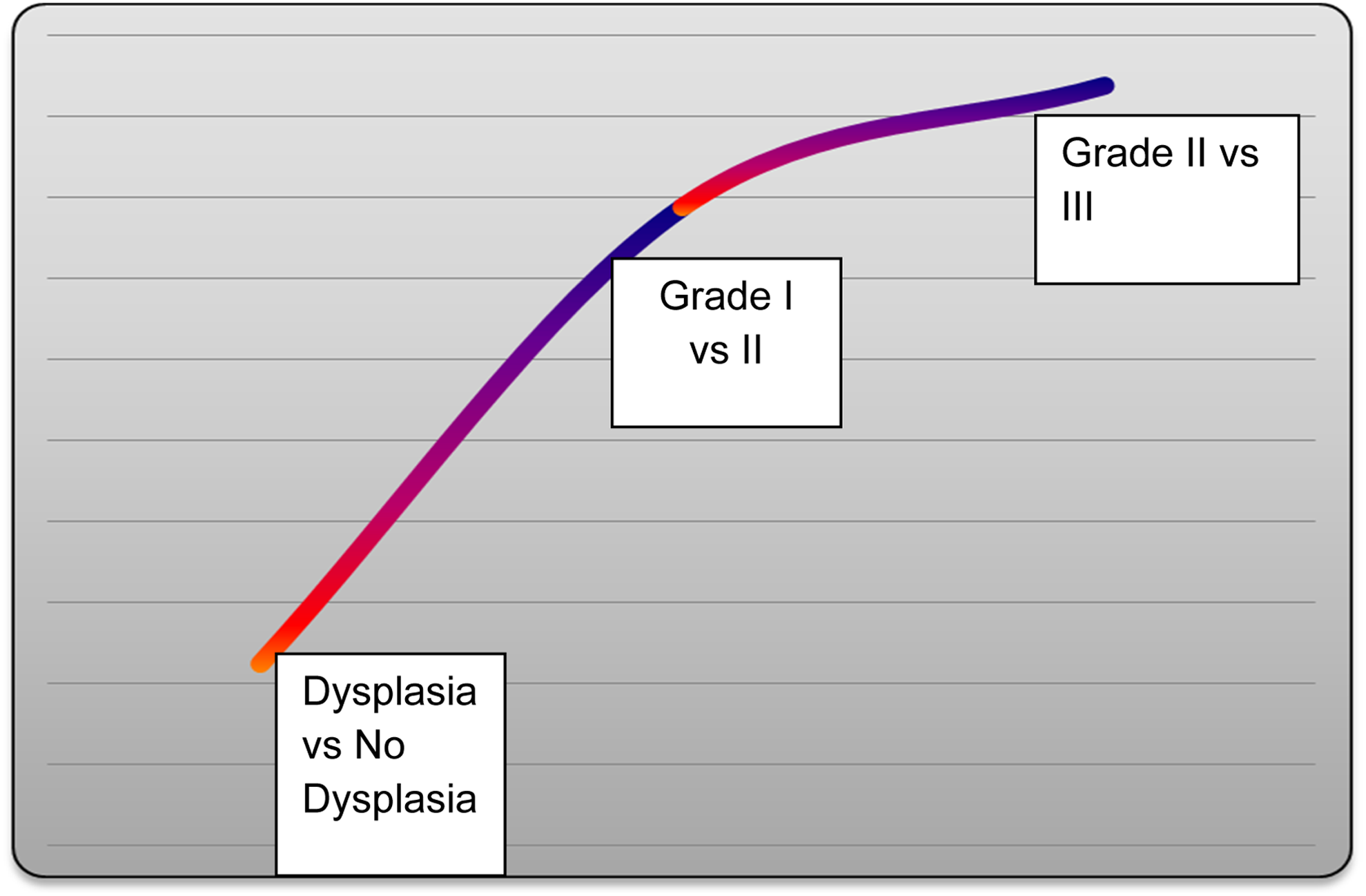
A gradual upregulation of HIF-1α mRNA expression from OSF without dysplasia to OSF with dysplasia to Grade I , II & III OSCC.

Schematic representation of the HIF-1 pathway. (a) undernormoxic conditions, HIF-1α is rapidly degraded because of hydroxylation of HIF-1α by prolyl hydroxylase and subsequent binding of the VHL. (b) physiologic stress (hypoxia and reactive oxygen and nitrogen species), mutations in the VHL and P13K/AKT/mTOR activity contribute to HIF-1α stabilization and accumulation. Upon stabilization of HIF-1α, it binds to HIF-1β. This HIF-1 complex binds to the hypoxia-responsive element of the DNA, thereby initiating the transcription of its target genes. The results in a glycolytic tumour metabolism among others. The glycolytic end products pyruvate and lactate induce HIF-1α accumulation, which in turn increases HIF-1 activity. HIF-1α: hypoxia inducible factor-1α; HRE: hypoxia-responsive element; AKT: protein kinase B; EGFR: epidermal growth factor receptor; mTOR: mammalian target of rapamycin; P13K: phosphatidylinositol-3-kinase; VHL: von-Hippel–Lindau protein.
Dysplasia, IIO in OSF and grade of OSCC in patients.

OSF: Oral Submucous Fibrosis; OSCC: Oral Squamous Cell Carcinoma; IIO: Inter Incisal Opening; NOM: Normal Mucosa.
Relative quantification of HIF-1α for different paired comparisons.

HIF-1α: Hypoxia Inducible Factor-1α; NOM: Normal Mucosa; OSF: Oral Submucous Fibrosis; OSCC: Oral Squamous Cell Carcinoma; IIO: Inter Incisal Opening; ddCT:delta-delta-Ct; RQ: Relative Quantification.
a
Results
(a) Epithelial dysplasia: all 6 OSF cases with dysplasia had higher expression level of HIF-1α than OSF cases without dysplasia. (b) Inter Incisal Opening: 7 OSF cases with IIO ≤ 27mm expressed lower level oh HIF-1α than 8 cases of IIO > 27mm. (c) Different grades of OSCC: Grade I, II & III OSCC showed gradual upregulation in the expression level of HIF-1α. (d) HIF-1α expression between different groups: progressive upregulation in HIF-1α expression level was found in OSCC than normal mucosa than OSF cases.
OSF cases with dysplasia and IIO > 27mm showed higher expression than cases withot dysplaia and IIO < 27mm. Grade I, II & III showed gradual upregulation in the expression level.
Discussion
The change in global trends in epidemiology of OSCC and indulgence of habits in teenagers at an early age is resulting in higher incidence among the youth (<30 years), hence the need for early detection of malignant transformation. It is proposed that cancer may arise de novo or directly as a transition from potentially malignant lesions which allows carcinomatous transformation of normal progenitor keratinocytes brought about by well-recognized DNA aneuploidy and loss of heterozygosity.
Studies in Indian population have reported malignant transformation rate of 2.3–7.6% during 10–17 years of follow-up. 7,8 Wide array of studies have focused on various molecular markers which could be helpful in early diagnosis and expecting therapeutic implications for OSF and oral carcinogenesis. 9 HIF-1α could be a key gene-regulating carcinogenesis in the background of fibrosis and partly being responsible for fibrosis as suggested in recent research studies. Measuring gene expression is an important part of many life sciences – particularly the ability to quantify the level at which a particular gene is expressed within a cell, tissue or organism can give a huge amount of information. However, it is often easier to detect one of the precursors, typically mRNA, and infer gene expression level quantitatively by RT-PCR, which is a very sensitive, semi-quantitative, simple, convenient, non-radioactive and rapid technique, suitable for a routine clinical laboratory. Also, immunohistochemistry studies do not recommend any uniform cut-off point of HIF-1α immunostaining. Hence, this study was planned to measure the differential mRNA expression level of HIF-1α gene as the target gene while keeping β-actin (housekeeping) gene as the reference gene by RT-PCR method.
In this study, in OSCC group, HIF-1α m-RNA expression was found to be upregulated by 2.056 fold in respect to OSF group, similarly 1.248-fold upregulation in comparison to normal group. This finding is in accordance with, an in vitro study by Mohamed et al. 10 where hypoxia was found to up regulate HIF-1α mRNA expression on both normal and oral cancer cell lines by Northern blot analysis. Hence, suggesting that hypoxia regulates HIF-1α expression in head and neck carcinoma cell lines.
Within different grades of OSCC, progressive upregulation was found from grade I to grade II to grade III OSCC. Upregulation by a factor of 2.948 in grade III in respect to grade I, 2.749 fold in grade II in respect to grade I and 1.189 fold in grade III in respect to grade II OSCC was found, which suggests its prognostic significance. Literature reveals significant association between upregulation at both protein and mRNA level with oral carcinogenesis along and different grades of cancer; metastasis, disease-free period and determination of prognosis.
A recent meta-analysis on the prognostic significance of HIF-1α in oral cavity cancer has shown HIF-1α upregulation to be associated with poorer overall survival. 11 Dunkel et al. 11 found that the CD44 low HIF-1α high signature was associated with poorer disease-free survival. Uehara et al. 12 found expression of HIF-1α in OSCC is likely to be of great value in treatment planning and to predict the prognosis of OSCC. Lee et al. 13 found that HIF-1α expression is significantly upregulated in areca quid chewing-associated OSCC. Zheng et al. 14 in their RT-PCR study measured mRNA levels of HIF-1α and found HIF-1α mRNA levels were significantly increased in carcinoma of the tongue and a positive correlation was observed with pathological differentiation grade.
On the contrary, Santos et al.
15
showed a significant relationship between strong HIF-1α protein expression and lower local disease relapse (
In OSF patients with OED, upregulation by a factor of 1.295 was found in respect to no dysplasia patient’s, showing a gradual increase from non-dysplastic to dysplastic to squamous cell carcinoma. This is in agreement with studies done by Tilakratne et al. 16 who concluded that hypoxia is an early event in oral carcinogenesis. The reason for increase in expression of HIF-1α with increase in grades of OED and OSCC is HIF-1α gene regulating around 1% of genome, especially those factors involved in regulating carcinogenesis such as VEGF, TGF-B, FGF, PDGF and EGFR, even in the background of fibrosis. 16 Semenza (2000) suggested that HIF-1α is an essential component in changing the transcriptional response of tumours under hypoxia, targeting transcription of over 60 genes involved in many aspects of cancer biology including cell survival, glucose metabolism, cell invasion and angiogenesis. 17
But a 0.604-fold expression downregulation was found in OSF group as compared to normal group This is not in consensus by the study results demonstrated by Tilakratne et al. 16 who found upregulation in expression of HIF-1α when studied semi-quantitatively by PCR. The review of literature also has revealed excellent studies signifying the role of HIF-1α in fibrosis of kidney, lungs and oral mucosa. This downregulation could be either because of variation in the ethnicity of patients in both the studies or because of relative higher expression of HIF-1α in normal oral mucosal tissue as observed by Mohamed et al., ubiquitous presence of inflammation and its upregulating effect on HIF-1α should also be considered. If this finding is considered good enough, then a downregulation will be very much indicative of fibrosis progression. This is supported by the 0.608-fold downregulation in OSF patients with lesser mouth opening, that is, inter-incisal opening (IIO) ≤27 mm than patients with mouth opening of >27 mm.
In our study, HIF-1α was found to be upregulated in OSCC in comparison to normal and OSF group. This is in accordance with the literature. A sequential upregulation is observed from OSF without dysplasia to OSF with dysplasia to OSF with dysplasia to Grade I to Grade II to Grade III OSCC squamous cell carcinoma suggesting a possible role of HIF-1α in oral carcinogenesis, a marker for early detection of malignant transformation with prognostic significance.
On the contrary, a downregulation proportionally related with the degree of fibrosis suggest a positive correlation with extent of fibrosis. In other words, a downregulation suggests fibrosis, whereas an upregulation is suggestive of oral carcinogenesis in the form of dysplasia and poorer grade of OSCC.
Smaller sample size and relatively higher expression level of HIF-1α mRNA in NOM seem to be the limitation of our study. So, multicentric studies with larger sample size are required to further validate the study.
To conclude, to the best of our knowledge, till date no study has been reported comparing HIF-1α mRNA expression objectively in (1) normal, OSF and OSCC group simultaneously and (2) correlating it with maximum IIO, dysplasia in OSF and within different grades of OSCC, that too in Indian population. It is evident from this study that HIF-1α mRNA can be a promising marker for malignant transformation. It can also be targeted as a therapeutic agent very effectively to prevent the malignant conversion of OSF into OSCC.
Footnotes
Acknowledgement
The authors are thankful to Director ICMR, New Delhi India, Dr P K Pandilwar, Associate Professor Department of Oral and Maxillofacial Surgery, Government Dental College & Hospital Nagpur and to Mr. Dhananjay Raje, statistician.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study had received financial assistance from Indian Council of Medical Research, New Delhi India vide their correspondence number 3/2/2012 /PG Thesis HRD -10 dated 20/08/2013.
