Abstract
Background
Artificial intelligence (AI) applications in medicine are rapidly expanding, revolutionizing the field of Chronic Kidney Disease (CKD) through diagnostics, prognosis prediction, and treatment decision-making. Despite significant progress, systematic analyses integrating AI with CKD remain limited.
Methods
Literature included in this study was sourced from the Web of Science Core Collection. Using tools such as CiteSpace, VOSviewer, and R-Bibliometrix, 888 relevant publications were analyzed. Research data encompassed dimensions including annual publication trends, author influence, institutional contributions, national output, keywords, and co-citation evolution.
Results
Research on AI and CKD has experienced exponential growth, particularly since 2019. China and the United States dominate paper publications, with leading institutions including the Sun Yat-sen University and University of California System. Core authors focus on AI-driven non-invasive biomarkers and CKD diagnosis via histopathological images. Global research trends shift from traditional machine learning to deep learning, emphasizing digital pathology and multimodal models to improve diagnostic and prognostic outcomes.
Conclusion
Between 2019 and 2025, the number of related publications grew rapidly, accelerating AI-driven advancements in CKD. Research emphasis has evolved from initial exploratory studies to clinically oriented applications centered on “deep learning models for image analysis, disease diagnosis, outcome prediction, and multimodal data integration.” Future efforts should prioritize integrating multi-omics technologies into multimodal models and developing fully automated hybrid models to advance AI from diagnostic support to clinical decision-making. These insights provide direction for future AI-driven innovations, promising enhanced precision in CKD management and improved patient outcomes.
1. Introduction
CKD represents a major global public health issue. 1 However, the absence of noticeable symptoms in the early stages often hinders early detection, leading to delayed intervention and increased mortality. 2 Regular screening of the general population and high-risk groups—such as individuals with diabetes or hypertension—facilitates early identification of CKD and enables timely intervention to prevent the progression to end-stage renal disease. 3 Despite advances in CKD diagnosis and treatment over recent decades, routine screening requires serum or urine samples, resulting in low compliance. Furthermore, the complexity of this chronic disease necessitates experienced physicians to develop personalized treatment strategies. Consequently, there is an urgent need for new technologies that facilitate easier diagnosis and access to professional treatment decisions.
As technology advances, AI is increasingly demonstrating its growing influence and potential. AI uses data processing, image recognition, and automation technologies to diagnose, provide treatment strategies, and predict disease prognosis. The emergence of large language models (LLMs), such as ChatGPT, has become a milestone in multiple fields, especially in medicine and healthcare. 4 AI’s application in medicine is spreading rapidly, including in various areas such as a general multimodal AI assistant for pathology, 5 imaging diagnostics for chronic kidney disease screening, 6 and the discovery and verification of CKD biomarkers. 7 In basic medicine and biology, AI excels in accurately predicting protein structures and biological molecule interactions. 8 The growing intersection of AI and medical research has sparked widespread interest, highlighting the need to analyze this field’s academic achievements and progress. 9
Although the potential of AI in the medical field has gained widespread recognition, with research literature on AI in CKD experiencing explosive growth, no recent study has comprehensively integrated the vast body of literature in this domain to systematically map the knowledge structure, academic collaboration networks, and emerging research frontiers of this dynamic discipline. Bibliometrics, an interdisciplinary field that quantitatively analyzes literature to reveal trends, research hotspots, collaboration networks, and influence within academic domains, not only quantifies scholarly dynamics but also provides researchers with evidence-based decision-making support to optimize academic resource allocation. Through bibliometric analysis, both quantitative and qualitative assessments of a field’s literature can be conducted to understand its current state and predict future directions. 10 To address these research gaps, this study conducts a comprehensive bibliometric analysis, systematically reviewing literature on AI applications in CKD. It quantitatively examines publication trends, core journals, and key contributors while employing keyword analysis, co-occurrence of references, and emergent detection techniques to identify and visualize research hotspots and thematic evolution patterns. The aim is to infer future research. This study seeks to provide nephrologists, clinical researchers, and patients with a panoramic reference for AI technology applications, thereby facilitating the clinical translation and widespread adoption of these emerging technologies.
2. Materials and methods
2.1. Data retrieval and collection
This study utilizes the Web of Science Core Collection (WoSCC) database—the largest and most internationally recognized comprehensive academic information repository—as our literature source. The search strategy employed is as follows:TS = ((“Chronic Renal Insufficiencies” OR “Chronic Kidney Insufficiency” OR “Chronic Kidney Insufficiencies” OR “Chronic Renal Insufficiency” OR “Chronic Kidney Diseases” OR “Chronic Kidney Disease” OR “Chronic Renal Diseases” OR “Chronic Renal Disease”) AND (“artificial intelligence” OR “AI” OR “machine learning” OR “Computer Reasoning” OR “Machine Intelligence” OR “Computational Intelligence” OR “deep learning” OR “neural network*” OR “convolutional neural network*” OR “random forest” OR “support vector machine” OR “natural language processing” OR “computer vision” OR “predictive model*”))
A total of 888 records were retrieved from the WoSCC database. After removing duplicates, two reviewers (W.Q.Y and S.Y.S) independently screened all titles and abstracts based on predefined inclusion criteria: (1) studies focused on artificial intelligence or machine learning applications in chronic kidney disease; (2) original articles or reviews; (3) published in English; (4) published between 2005 and 2025. Any disagreements between the two reviewers were resolved through discussion or, if necessary, adjudication by a third reviewer (D.J.Y). The full texts of potentially eligible studies were then retrieved and independently assessed by the same two reviewers using the same criteria. Search results were exported as plain text files and saved in. txt format. The screening and selection process is illustrated in Figure 1. The workflow of current study that illustrated research strategy and significance.
2.2. Data analysis
In this study, all metadata retrieved from WoSCC were imported into multiple tools—CiteSpace (version 6.3. R3), VOSviewer (version 1.6.20), R-Bibliometrix (version 4.3.0), and Pajek (version 6.01)—to generate visualizations for quantitative and qualitative analysis.
CiteSpace (Version 6.3. R3) analyzed the most influential authors, keywords, and references, generating keyword networks, timelines, and time zone maps, while also performing reference clustering. VOSviewer (Version 1.6.20) visualized co-occurrence networks for countries, institutions, authors, keywords, and references. Each node in these co-occurrence networks represents a country, institution, author, keyword, or reference, grouped by type. R-Bibliometrix (Version 5.0) is used for metadata acquisition and descriptive statistical analysis. Pajek (Version 6.01): Optimizes network visualization effects.
3. Results
3.1. Basic information and annual publications of the included literature
General characteristics of the analyzed publications.


The publication trend of AI in CKD research from Jan 1, 2005, to Dec 31, 2025. Annual publications and citation counts of literature related to AI applications in the field of CKD from 2005 to 2025.
3.2. Country and Region Analysis
The top 10 countries in terms of the number of published papers in the AI and CKD field are primarily from East Asia, North America, South Asia, and Europe. The respective numbers are as follows: China (N=1525), United States (N=637), South Korea (N=198), India (N=183), Germany (N=144), Japan (N=133), France (N=131), Italy (N=118), United Kingdom (N=118) and Spain (N=111). Extensive international collaboration exists among these nations, with China and the United States demonstrating the closest cooperative ties (Figure 3(a)). Country/region analysis of AI applications in CKD. (a) country/region collaboration network diagram. (b) Distribution of emerging countries/regions. (c) Top 20 countries by publication volume. SCP: single country publications, referring to literature where all authors originate from a single country, indicating research without transnational academic collaboration; MCP: multiple country publications, referring to literature where authors come from two or more countries, indicating transnational collaboration. A higher number of MCPs signifies greater participation in international collaborative projects, broader cooperation scope, and higher academic collaboration activity for that country.
In recent years, countries such as Cambodia, Argentina, Tunisia, and North Macedonia have also begun prioritizing research in this field, emerging as new research nations (Figure 3(b)). Their focus primarily centers on efficient diagnostic and therapeutic needs under resource-constrained conditions, reflecting developing countries’ technological catch-up and localized innovation in this domain. Furthermore, using “corresponding author contributions” as an indicator, Figure 3(c) lists the top 20 countries: China, the United States, India, South Korea, and Japan are the core contributors. Among these, China has the highest number of “multinational collaborative papers (MCPs)” (N=37). Among countries publishing over ten papers, Germany and Egypt exhibit significantly higher “proportions of multinational collaborative papers,” at 70.0% and 66.7% respectively.
3.3. Institutions analysis
Statistical results indicate that a total of 1,379 institutions participated in the study. To optimize resource utilization and enhance the accuracy and practicality of analytical outcomes, we selected 59 institutions with publication counts greater than or equal to 10 and constructed an interactive network diagram (Figure 4(a)). Two prominent color chains emerged, indicating exceptionally strong connections between institutions at either end. One chain featured Duke-NUS Medical School, The Singapore National Eye Centre, and National University of Singapore—all located in Singapore—forming the widest link chain among these institutions, signifying their closest ties. The other chain connected China’s Sun Yat-sen University with The Hong Kong Polytechnic University, exhibiting a connection density second only to that of the Singaporean institutions. Institutional analysis of AI applications in CKD. (a) Institutional collaboration network diagram. (b) Top 10 institutions by total publication volume: Funnel chart. (c) Institutional paper output: Line chart.
Based on contributions in AI and CKD research, we list the top ten institutions by publication volume. The United States had four leading institutions on the list, making it the country with the most represented institutions. These included renowned institutions such as the University of California System (N=44), Harvard University (N=31), the University of Michigan (N=31), and the University of Pennsylvania (N=31) (Figure 4(b)). Among all institutions, China’s Sun Yat-sen University had the highest number of publications (N=61). Additionally, we analyzed the publication timeline and volume of these institutions. Findings indicate that the Sun Yat-sen University began publishing relevant papers as early as 2013, making it the earliest institution to initiate research in this field. Starting in 2018, other institutions, represented by Egyptian Knowledge Bank, also progressively engaged in related research (Figure 4(c)).
3.4. analysis of authors
This study’s statistics indicate that 5,323 authors participated in AI research within the CKD field. The author collaboration network reveals a tightly knit core research community within this domain (Figure 5). Among them, 113 core authors—each publishing two or more papers and connected to other authors—formed eight collaborative clusters with a total link strength of 1,634. Authors such as Kretzler, Matthias, Bitzer, Markus, and Hodgin, Jeffrey B. exhibit the strongest collaboration, forming key collaborative hubs. Additionally, Liu, Fei and Zhang, Li are the most frequently collaborating researchers among emerging scholars. Author collaboration and emerging scholar clustering network diagram for AI applications in CKD research.
Top 10 authors by publication output in AI in CKD research.

3.5. Journals
888 publications appeared in 366 journals. Using Bradford’s Law of Scatter, 20 core journals were identified. Their publication counts, geographic distribution, impact factors (IF), and JCR category information are shown in Figure 6(a): The UK journal Scientific Reports ranked first with 48 publications; followed by BMC Nephrology (UK) with 24 publications. Among these journals, 60% (12 journals) were classified as Q1, while 7 and 1 belonged to Q2 and Q3, respectively. In terms of geographical distribution, Switzerland contributed the most with 9 journals, followed by the United Kingdom with 5 journals, the United States with 4 journals, and Italy and the Netherlands each with 1 journal. Clustering analysis of the journals yields the top five cluster labels: Chronic kidney disease classification, Non-alcoholic fatty liver disease, Nutrition diagnosis expert system, Diabetes mellitus patient, and Deep learning (Figure 6(b)). Regarding H-index and G-index (Figure 6(c) and (d)), IEEE Access and Scientific Reports consistently ranked at the top. Citation analysis (Figure 6(e)) revealed that Nature Machine Intelligence had the highest total citations (N=5046), followed by Jama-Journal Of The American Medical Association (N=1014) and IEEE Access (N=600). Figure 6(f) shows that starting in 2019, IEEE Access became the journal with the highest publication volume in this field, while other core journals also exhibited an upward trend in publication volume during the same period. This timeframe coincided with the accelerated development of AI applications in the CKD field. Disciplinary and journal analysis of AI in CKD research. (a) Twenty core journals screened based on Bradford’s dispersion law; (b) journal clustering diagram; (c) number of publications in top journals based on H-index; (d) number of publications in top journals based on G-index; (e) number of publications in top journals based on total citations; (f) annual publication trends in leading journals. IF: impact factor; JCR: journal citation reports.
3.6. Keywords
The keyword co-occurrence network visually depicts major research themes and trends. “Chronic kidney disease” occupies a central position, surrounded by artificial intelligence terms such as “machine learning” and “artificial intelligence”, alongside clinical keywords like “mortality”, “risk” and “biomarkers” (Figure 7(a)). Salience analysis identified the top 16 keywords with significantly increased frequency, arranged in order of their emergence (Figure 7(b)). The timeline view (Figure 7(c)) illustrates the historical evolution of research, while the time-zone view (Figure 7(d)) highlights the co-occurrence of high-frequency keywords in specific years, revealing concentrated academic focus on particular clinical issues: early keywords such as “chronic kidney disease” and “glomerular filtration rate” represent disease definitions and core physiological indicators for CKD. Beginning in 2013, terms like “artificial neural network” and “prediction” signaled the initial penetration of artificial intelligence into CKD research. From 2019 onwards, keywords such as “deep learning,” “random forest,” and “diabetic nephropathy” indicate a recent shift toward diagnostic approaches driven by increased model complexity and accuracy, alongside growing emphasis on analyzing comorbidities. Keyword co-occurrence analysis of AI in CKD research. (a) Co-occurring keyword network. (b) Top 16 keywords with highest emergence intensity. (c) Timeline view of keyword co-occurrence and clustering. (d) Time-zone view of keyword co-occurrence displayed by year.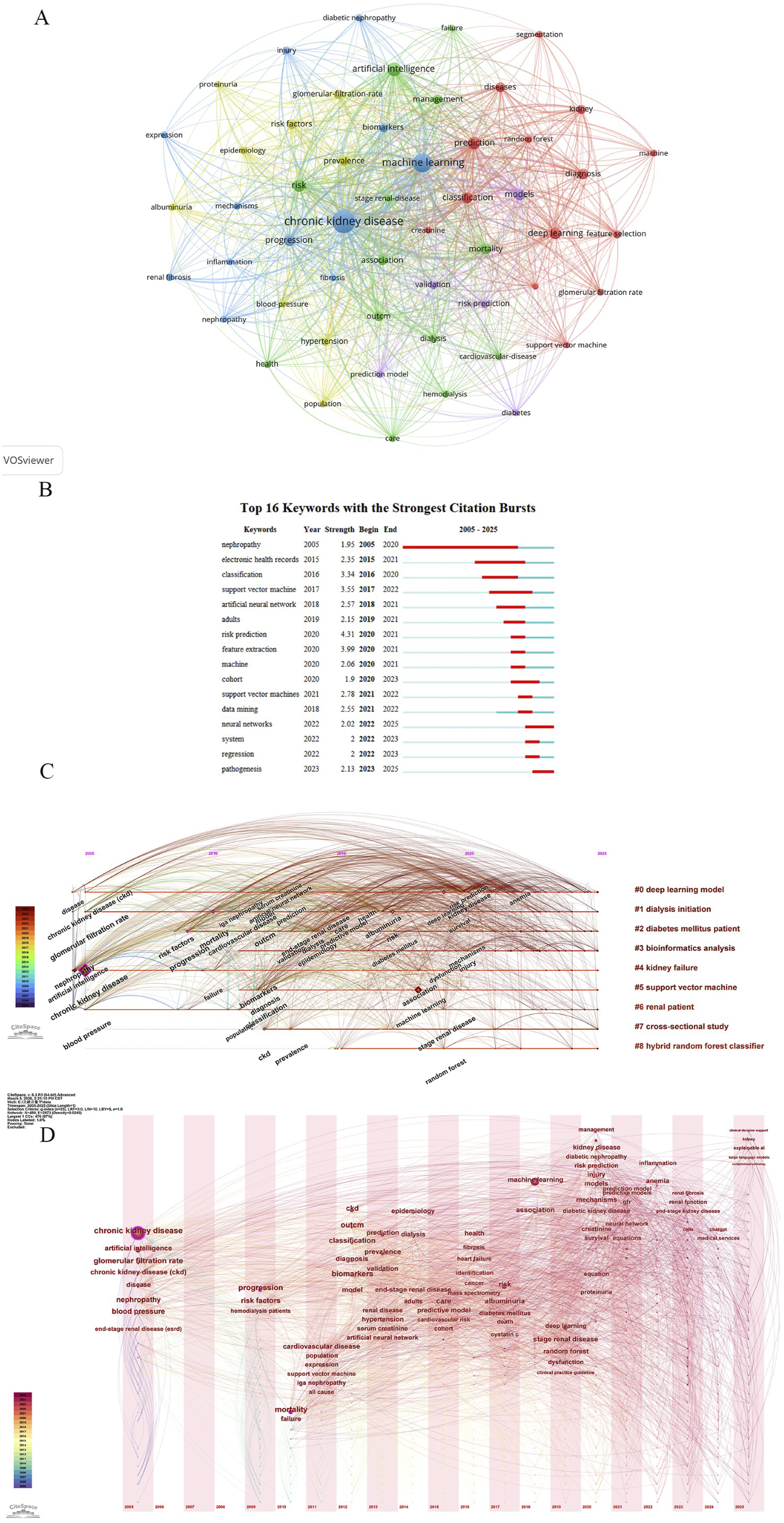
3.7. Cited references and co-cited references
The top 10 cited publications.

Furthermore, using CiteSpace’s Log-Likelihood Ratio (LLR) algorithm, we performed a clustering analysis on the 691 references, generating a peak map (Figure 8(a)). Through 20 clusters, we identified the research focus and development trends in this field (Figure 8(b)), which can be broadly divided into three evolutionary stages. 1) 2010–2017: Individual clusters emerged successively, but no significant interconnections existed between them across annual time slices in CiteSpace. 2) 2017–2020: Intercluster connections emerged within time slices, forming pairwise links. In 2017, Clusters 1 and 12 connected via Chen ZW’s paper; Clusters 0 and 10 in 2018 were linked through Esteva A’s paper. This indicates the initial establishment of early predictive models. 3) 2020–2025: Multiple clusters became interconnected, marking the transition toward refined, high-quality development in AI-assisted renal pathology research. For example, 2025 represents tighter connections between clusters 0, 1, 9, and 15, which encompass large language models and biomarkers. Co-citation analysis of artificial intelligence in CKD research. (a) Co-citation peak map. (b) Cocitation cluster time-slice map. (c) Co-citation cluster map. (d) Co-citation emergence map.
To explore the strength of connections among co-cited references, this study adjusted the citation frequency threshold to ≥15 in VOSviewer, selecting 68 highly cited references to construct a reference co-citation network (Figure 8(c)). Analysis of total link strength revealed that three publications—Xiao J, 2019, J TRANSL MED 11 ; Bikbov B, 2020, lancet 12 ; and Almansour NA, 2019, comput biol med 13 —occupied a prominent core position within the network, with total link strengths of 268, 264, and 240, respectively. This indicates that their research conclusions or methodologies have been extensively referenced by subsequent highly cited publications, playing a significant guiding role in shaping research directions.
In bibliometric analysis, references exhibiting citation burst characteristics provide a direct reflection of core research outcomes that garnered high attention and concentrated citations from scholars during a specific period. This study identified the top 25 references with the highest citation burst intensity in this field using CiteSpace (Figure 8(d)). The red bars in the figure correspond to the citation burst periods for each reference. Results indicate that citation surges in this field were particularly pronounced at two distinct time points: 2020 and 2023. The study “Polat H, 2017, J MED SYST” titled “Diagnosis of Chronic Kidney Disease Based on Support Vector Machine by Feature Selection Methods” exhibited the highest citation burst intensity (8.28) during 2017-2022. This indicates that Support Vector Machine-based CKD diagnostic techniques became a core research direction in this phase, with its methodologies and conclusions providing crucial references for subsequent studies. 14 The 2019 paper “Xiao J, 2019, J TRANSL MED” has maintained a high-citation emergence state from 2019 to present, highlighting the application value of AI-driven machine learning and neural network approaches in CKD and establishing them as an emerging research focus. 11
4. Discussion
AI has not only transformed people’s daily lives but also revolutionized medical research and practice, offering innovative diagnostic, prognostic, and therapeutic solutions. Our bibliometric analysis spanning 2005 to 2025 captures the evolution and impact of AI in the field of CKD, synthesizes relevant research findings, and highlights patterns of research growth, shifts in research themes, and academic milestones.
4.1. What is the current state of research in this field?
Over the past two decades, AI research in CKD has expanded significantly: the number of papers increased from 3 in 2005 to 251 in 2025. While citation counts peaked in 2020 and subsequently declined, the volume of research continues to grow steadily. This trend reflects growing interest in applying artificial intelligence to CKD research. This heightened focus on AI-driven solutions has spurred a series of emerging technological advancements, particularly in the capabilities of deep learning and neural networks for early detection, diagnosis, and personalized treatment strategies for CKD.15–17
From a geographical perspective, several influential countries stand out in the field of CKD artificial intelligence research. China has consistently led in the volume of publications, an advantage stemming from its national AI strategy. This strategy has funded multiple significant AI initiatives in nephrology and driven the development of the world’s largest kidney disease database (3.63 billion records). Coupled with the healthcare system’s urgent need for AI tools to support primary care, these factors collectively fuel the country’s prolific research output. However, China’s share of MCPs lags behind that of the United States. Moving forward, greater collaboration with other nations can leverage multinational technological strengths to make more contributions to AI in the CKD field and enrich the global research landscape. Institutions are key drivers in advancing AI within CKD research. China’s Sun Yat-sen University leads globally in publication output, followed closely by the University of California System in the United States and Seoul National University (SNU) in South Korea. Network analysis reveals strong collaborative ties among top institutions—particularly those in the United States and China—validating both nations’ academic standing and global influence.
Among the most frequently cited articles globally, the study by Ogunleye A et al. published in IEEE/ACM Transactions on Computational Biology and Bioinformatics stands out particularly. 18 This paper examined several representative and recent artificial intelligence algorithms within the context of CKD, selecting Extreme Gradient Boosting (XGBoost) as its high-performance base model. After optimization, the optimal full model trained on all features reported accuracy, sensitivity, and specificity of 1.000 on the test set, demonstrating the model’s potential efficacy for CKD diagnosis within the specific dataset. It should be noted that this study did not involve external validation on independent external datasets; its reported high performance was achieved under a specific data distribution, and caution is warranted when generalizing to different populations and clinical settings. The true generalizability of these results remains to be further validated through external testing across multicenter settings and diverse populations to mitigate potential overfitting risks. Nevertheless, this study provides valuable exploration into the application of XGBoost for CKD diagnosis and establishes an algorithmic foundation for subsequent research. Another study by Singaporean researchers published in the Journal of Clinical Epidemiology found that logistic regression achieved the highest area under the receiver operating characteristic curve (0.905 [0.88, 0.93]) in CKD prediction. 19 These studies represent highly influential and advanced research outcomes in this field.
4.2. Which journals and individuals lead in publications?
Among the top 20 publishing journals, publications related to computer science and nephrology stand out, alongside public health and general medicine journals. This indicates that artificial intelligence is primarily applied to direct clinical scenarios such as CKD staging, prognosis assessment, dialysis/transplant management, and precision medication.16,20–22 Nature Machine Intelligence received the highest number of citations, reaching 5,046, indicating greater academic influence compared to other computer science and nephrology journals such as IEEE Access and Kidney International Reports.
Within the authors’ co-author network, key figures such as Kretzler, Matthias, Bitzer, Markus, and Hodgin, Jeffrey B. exhibit high connectivity and “tipping point” effects, bridging academic communities and fostering interdisciplinary convergence. For instance, these three authors have collaborated on four papers in the AI and CKD field, including work on precision medicine for CKD using non-invasive biomarkers 23 and spatial analysis based on unsupervised machine learning algorithms to predict CKD using histopathology images. 24 Their academic focus on digital pathology imaging for CKD represents an emerging research perspective. Other influential authors like BARBIERI C have examined anemia management in dialysis patients. They developed an anemia control model using machine learning to improve anemia outcomes in hemodialysis patients, minimizing the use of erythropoiesis-stimulating agents. 25 Subsequently, they enhanced hemoglobin concentration prediction for large hemodialysis cohorts using deep recurrent neural networks. 26 Their latest findings reveal that the use of the Anemia Control Model is associated with reduced hospitalization risk in hemodialysis patients. 27 These studies have garnered widespread attention and made outstanding contributions to the advancement of this field.
4.3. What research topics do these articles primarily focus on?
Keyword co-occurrence analysis identified two major research clusters: CKD disease itself and the evolution of artificial intelligence algorithms. Early studies concentrated on traditional machine learning for clinical diagnosis, such as CKD core physiological indicators including glomerular filtration rate, creatinine, and blood pressure. 28 Later research shifted toward multi-omics approaches based on deep learning for screening, diagnosis, and prognostic analysis. Multimodal models integrate clinical data across different scales and modalities, multi-omics data (proteomics, metabolomics), and pathological imaging to predict CKD progression. 29 The following is introduced and elaborated upon from four aspects:
In the field of imaging, diagnosing nephropathy using fundus images, 30 predicting advanced CKD via retinal fundus imaging, 15 and employing renal ultrasound as a non-invasive diagnostic method, 31 multifrequency magnetic resonance elastography combined with automated segmentation for assessing renal function in CKD patients, 32 classification and segmentation of renal magnetic resonance imaging for CKD detection, 33 and nuclear medicine imaging—particularly planar scintigraphy and single-photon emission computed tomography (SPECT)—all offer non-invasive renal function assessment, though further research is needed. 34 Additionally, Opt-Forest technology utilizes electrocardiogram data to enhance the accuracy of early chronic kidney disease diagnosis. 35
In pathology, artificial intelligence enables precise image recognition of complex histopathological slides, possesses robust computational capabilities for processing whole-slide imaging, and leverages advanced deep learning technologies trained on large datasets. 36 For instance, “virtual staining technology” integrated with AI extends imaging capabilities to infer missing data, addressing the common challenge of data gaps in large-scale computational pathology studies—a transformative advancement for pathology. 37 Research confirms that AI in digital pathology demonstrates high accuracy, specificity, and predictive performance in CKD cases, successfully handling complex, high-dimensional scenarios. 38 Nevertheless, areas for improvement remain, such as balancing sensitivity and specificity, and the need for further validation across diverse populations to mitigate effects of heterogeneity and lack of standardized predictive criteria. Additionally, certain clinically significant domains are underrepresented in the literature, including AI for providing interpretable decision support, longitudinal monitoring of treatment response, and AI-assisted humanistic care and doctor-patient communication. Addressing these gaps would help align research priorities more effectively with unmet clinical needs.
In nursing care, national and international nephrology organizations have issued statements regarding the application of artificial intelligence in nursing. For instance, the American Society of Nephrology states that AI can predict common complications during dialysis and alleviate nursing staff workload by monitoring treatment status in real time through audio signals generated by dialysis machines. 39 Concurrently, the European Renal Association asserts that wearable and implantable bioelectronic systems play a vital role in renal care by enabling non-invasive, continuous monitoring of physiological and biochemical parameters. 40 Examples include the Kardio-Net deep learning model for detecting severe hyperkalemia and systems measuring creatinine and uric acid in saliva to monitor hydration status and electrolyte imbalances in chronic kidney disease. These innovations provide critical care support for nephrology specialists and patients in remote areas with limited laboratory resources.40–42
In other areas, researchers have leveraged breathomics by analyzing volatile organic compounds in exhaled breath. Combining artificial intelligence with sensitive electrochemical sensing has demonstrated significant potential in diagnosing and monitoring CKD, chronic obstructive pulmonary disease, and lung cancer. 43 Another study employed machine learning algorithms—including LR, SVM, and ANN—to screen human breath for three distinct volatile organic compounds (VOCs) and carbon monoxide. 44 This approach yielded a machine learning-based chronic kidney disease classifier capable of distinguishing healthy from unhealthy populations. In genetics, screening for disease-causing genes aids in diagnosing CKD and other susceptible comorbidities. 45 These approaches offer intriguing insights that can inspire clinical practice in nephrology.
Interestingly, keywords outside the nephrology field, such as “cardiovascular disease,” began appearing in the keyword timeline starting in 2020. Its co-occurrence with chronic kidney disease often reflects the cross-disease application of artificial intelligence in diagnostic imaging and pathological analysis. 42 Cardiovascular disease and CKD share common pathophysiological features, and studies indicate that adults with primary glomerular disease and nephrotic syndrome have a relatively higher risk of cardiovascular events.43,44 Recent mechanistic studies reveal that circulating extracellular vesicles derived from CKD carry unique nephrogenic miRNAs, which mediate molecular crosstalk and contribute to heart failure pathogenesis in CKD patients. 45 This shared biological signature fosters interdisciplinary research and may enable the transfer of validated AI methods from CKD to cardiovascular disease, potentially accelerating clinical translation.46–49
Comparing the findings of this study with recent bibliometric research on AI applications in other subspecialties of nephrology facilitates a more comprehensive understanding of AI utilization patterns within nephrology. A bibliometric analysis of machine learning applications in nephrology from 2013 to 2024 similarly revealed exponential growth in this field, with research hotspots centered on early prediction models and clinical management optimization. 50 This aligns closely with the explosive growth post-2019 and the prognostic prediction theme identified in this study. The study further notes that while most predictive models report high accuracy, research on treatment optimization and complication prevention remains relatively scarce. This finding corroborates our conclusion that research objectives have extended from “disease diagnosis” to “prognostic prediction,” yet clinical decision support still requires deepening.
A separate bibliometric analysis of 30 years (1994–2024) of AI research in kidney transplantation revealed distinct application patterns: among 890 publications, the United States contributed most significantly (31.6% of papers), with Mayo Clinic and Harvard University leading institutions. Research themes centered on deep learning applications for donor-recipient matching optimization, postoperative monitoring, and personalized immunosuppression therapy. 51 Compared to the CKD domain examined in this study, AI research in kidney transplantation emphasizes decision support for “matching and allocation”—spanning preoperative donor kidney quality assessment (e.g., risk prediction based on KDPI scores), intraoperative augmented reality navigation, and postoperative rejection prediction and graft survival analysis. AI applications in CKD, however, emphasize “end-to-end management”—spanning early screening, risk stratification, and optimizing intervention timing for complications like renal anemia and CKD- Mineral and Bone Disorder. This divergence profoundly reflects distinct clinical needs: kidney transplantation addresses precision matching under organ scarcity, while CKD management tackles the challenges of managing a chronic, highly heterogeneous disease over extended periods.
Overall, AI in nephrology subfields exhibits a pattern of “common trends and distinct differences”. Common trends include deep learning-driven technologies, multimodal data integration, and prognostic prediction as shared focal points. Distinct differences manifest in kidney transplantation focusing on decision-making and matching, while CKD emphasizes comprehensive management throughout the disease course. Future research may draw inspiration from matching algorithms in kidney transplantation and dynamic prediction models in CKD to advance cross-subspecialty integration. Examples include applying donor-recipient matching principles from transplantation to individualized matching between CKD patients and intervention strategies, or integrating CKD disease progression prediction models into long-term postoperative follow-up management.
4.4. What are the research trends in this field?
Thematic evolution analysis further clarifies the development trajectory of the field: From 2005 to 2015, research focused on the clinical diagnosis of abnormal renal physiological indicators, closely tied to the widespread adoption of electronic health record systems and the maturation of traditional machine learning algorithms. During this period, algorithms such as support vector machines and random forests began to be applied to predictive analysis of routine indicators like serum creatinine and urine protein, with research emphasizing the validation of AI technology’s ability to mine existing clinical data. From 2016 to 2020, the research focus shifted toward establishing standardized diagnostics and screening biomarkers. This transition was driven by two major factors: First, the State Council of China issued the “New Generation Artificial Intelligence Development Plan” in 2017, establishing intelligent healthcare as a national strategic priority. Second, the rapid advancement of omics technologies enabled multidimensional biomarker research. In 2019, Zhang Luxia’s team at Peking University systematically outlined the application prospects of big data and AI in nephrology, providing a theoretical framework for subsequent research. From 2021 to 2025, driven by deep learning technologies (such as image segmentation, neural networks, and feature recognition) and multimodal integration, the field entered an “elevation phase.” Its core drivers stemmed from three aspects: First, the maturation of deep learning technologies, with convolutional neural networks achieving breakthroughs in medical image recognition, rapidly transferring these techniques to renal pathology image analysis.52–54 Second, the establishment of national-level data platforms—such as large-scale population cohort databases like the U.S. NHANES and China’s CHARLS, alongside open-access clinical databases including MIMIC and SEER—has significantly expanded the breadth and diversity of data sources. These repositories encompass multidimensional information including biochemical indicators, imaging data, and long-term follow-up outcomes, enabling researchers to develop CKD risk prediction models with enhanced generalization capabilities. Third, the “Digital Kidney” grand science initiative launched in 2025 by Peking University First Hospital in collaboration with the National Center for Biomedical Imaging explicitly proposes integrating multimodal, cross-scale imaging technologies with AI algorithms to construct the world’s first comprehensive digital kidney atlas. This marks the field’s transition from “technological exploration” to a “systemic engineering endeavor driven by national strategy.” This paradigm shift—from “traditional image analysis” to “AI-driven integrated diagnostic and therapeutic systems”—is an inevitable outcome of the convergence of technological, data, and policy dividends. Focusing on these key evolutionary directions can provide researchers with guidance for advancing the translational application of AI in CKD clinical practice.
Monitoring developmental trajectories aids in identifying evolving research hotspots. Year-by-year analysis reveals that the co-citation network converges into 10 major labeled clusters, which can be grouped into three themes: “disease prediction”, “pathology”, and “artificial intelligence models”. These themes experienced strong citation surges between 2019 and 2023, reflecting heightened focus on the efficiency of radiology and pathology workflows.6,55 These themes indicate tangible shifts in CKD management: AI-enhanced digital imaging and pathology enable automated, more precise classification, predicting disease onset and renal function progression. A critical question arises: given the limitations of laboratory tests and biopsies in diagnosing the etiology of different primary causes of CKD, can advanced imaging replace biopsy? 56 Therefore, developing an economical, non-invasive, and accurate diagnostic method represents a promising direction for AI applications in CKD, particularly considering constraints such as patient cognitive levels, coagulation function, and renal structure.
Based on the above bibliometric findings—particularly the explosive growth of keywords such as “imaging” and “pathology” over the past five years, along with the evolving research focus from “disease diagnosis” to “prognostic prediction”—we propose the following two future research directions: 1) Development of multi-modal deep fusion models based on multi-source data. Current clustering analysis indicates that “imaging” and “pathology” have emerged as distinct research clusters, yet their integration with laboratory data and genetic information remains rudimentary. Future research should overcome single-modality limitations by developing Transformer-based multimodal fusion models. These models should systematically evaluate the incremental value of different data combinations for CKD staging, pathological classification, and prognosis prediction, with external validation conducted on public datasets like NHANES, CHARLS, and UK Biobank. 2) Dynamic prediction models and intervention timing optimization for clinical decision-making. Keyword time-zone maps reveal that “prediction models” and “machine learning” frequently appeared between 2018 and 2021, yet existing research predominantly focuses on static diagnosis with insufficient attention to dynamic disease course management. Future research should develop fully automated hybrid models integrating time-series data (e.g., incorporating Long Short-Term Memory) for precise CKD diagnosis and staging. Furthermore, dynamic intervention timing optimization models should be constructed for specific complications (e.g., renal anemia, CKD-Mineral and Bone Disorder), advancing AI from “auxiliary diagnosis” to “clinical decision support systems” and ultimately achieving a closed-loop transition from “prediction” to “intervention.”
This study has limitations. First, we did not validate the results using other bibliometric tools (such as Gephi), which may affect the objectivity of the research. Due to space constraints, detailed discussions of some representative studies could not be included. Second, beyond these methodological limitations, potential biases should be noted: publication bias, where positive results or novel findings are more likely to be published and cited; language bias, as the study was confined to English-language literature, potentially obscuring research trends and contributions from non-English-speaking regions and limiting global representativeness; and exclusion of non-journal outputs (e.g., case reports, Editorials, Letters, Conference abstracts, gray literature), which may overlook emerging research in this rapidly evolving field. Additionally, the predominance of publications from countries such as China and the United States may be substantially influenced by national research policies and funding opportunities—for instance, China’s “New Generation Artificial Intelligence Development Plan” (2017) and the NIH’s “Advancing American Kidney Health” initiative. Our study did not quantitatively analyze the causal relationship between these macro-level policy factors and publication output, and readers should consider this contextual background when interpreting country-specific trends. These factors should be considered when interpreting the scope and generalizability of our conclusions. Finally, while this study focuses on AI applications precisely aligned with clinical needs at the core pathological stage of CKD, the application scenarios and effectiveness of AI may vary across different etiologies and disease stages. Future research could conduct in-depth analyses targeting specific etiologies and stages to further explore the academic impact and clinical value of AI in this field.
5. Conclusion
In summary, this bibliometric study indicates that AI research in the field of CKD is experiencing rapid growth, with a significant increase in the number of publications between 2019 and 2025. China and the United States are the primary contributing countries, and the research focus has undergone a notable evolution—transitioning from initial exploratory studies to clinically oriented research centered on “implementing image analysis, disease diagnosis, outcome prediction, and multimodal data integration through deep learning models.” Future research should prioritize: integrating multi-omics technologies into multimodal models to reduce operator dependency and enhance diagnostic accuracy; developing fully automated hybrid models to drive AI’s transition from diagnostic support to clinical decision-making. Findings suggest that sustained advancements in these areas hold promise for reshaping clinical practice patterns in CKD.
Supplemental material
Supplemental material - Artificial intelligence in chronic kidney disease: Bibliometric and visual analysis of trends and future directions
Supplemental material for Artificial intelligence in chronic kidney disease: Bibliometric and visual analysis of trends and future directions by Wenqian Yu, Siyuan Sun, Haohan Wang, Yurong Cheng, Dajun Yu in Digital Health.
Footnotes
Acknowledgements
We extend our gratitude to all researchers who have contributed to the fields of AI and CKD. The authors declare that no artificial intelligence (AI) tools or AI-assisted technologies were used in the preparation of this manuscript. All content, analysis, and writing were performed solely by the authors.
Ethical considerations
This study is solely based on public database and does not require ethical approval.
Author contributions
Wenqian Yu and Siyuan Sun were responsible for literature retrieval, data analysis, and manuscript drafting. Haohan Wang and Yurong Cheng made significant contributions to the design of retrieval strategies, data acquisition, analysis, and interpretation. Dajun Yu oversaw manuscript review and topic selection, and approved the final version for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Capital Health Development Research Project (No.: Shoufa 2022-2-4173), World Federation of Chinese Medicine Societies Science and Technology Project (No.: WFCMS2024038) and Construction Project for the Integrated Platform of Traditional Chinese Medicine Clinical Research at Xiyuan Hospital of China Academy of Chinese Medical Sciences (No.: XYZX0405-33).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Writing assistance and third-party submissions
This article does not involve any third-party writing or submission assistance.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
