Abstract
Background
Glioblastoma, a highly aggressive and complex brain tumor, poses significant challenges in diagnosis and treatment. This bibliometric analysis aims to explore the trends and key players in artificial intelligence (AI)-driven glioblastoma research over the past two decades using the Web of Science core database.
Methods
This bibliometric analysis utilized the Web of Science Core Collection to identify English-language articles (n = 1487) published between 2004 and 2024, focusing on AI applications in glioblastoma. Data extraction and preliminary statistics were performed using Microsoft Excel. Subsequently, CiteSpace and VOSviewer were employed for in-depth analysis and visualization of research trends, collaboration networks, keyword cooccurrence, and emerging hotspots.
Results
The results reveal a remarkable upward trend in research output, particularly after 2015, reflecting the growing interest in leveraging AI to address the complexities of glioblastoma. The United States and China were identified as the dominant contributors, with the University of Pennsylvania and Harvard Medical School emerging as key institutions. The analysis also identifies prominent authors such as Bakas Spyridon and highlights the pivotal role of journals like Neuro-Oncology and Cancers in disseminating cutting-edge research. Keyword analysis pinpointed “transfer learning” and “radiogenomics” as emerging frontiers, alongside established foci on machine/deep learning for imaging analysis and imaging biomarker development.
Conclusions
This comprehensive bibliometric study first delineates the evolving intellectual structure of AI in glioblastoma. It identifies the pivotal shift from methodological development to clinical translation and highlights radiogenomics as a key convergent frontier, providing a foundational map for future research prioritization.
Introduction
Glioblastoma, a common and aggressive primary brain tumor in adults, remains a significant challenge in neuro-oncology.1,2 Classified as a Grade IV glioma by the World Health Organization (WHO), it is distinguished by rapid growth, invasion of surrounding brain tissue, and resistance to conventional therapies.3,4 Histologically, glioblastoma is distinguished by necrosis, endothelial proliferation, and cellular pleomorphism. 5 These tumors often present with nonspecific symptoms such as headaches, seizures, and cognitive decline, which can result in delayed diagnosis. Despite advances in surgery, radiotherapy, and chemotherapy, the prognosis remains poor. The median survival rate is approximately 15–23 months postdiagnosis,6,7 with the 5-year survival rate being less than 6%.6,8 This poor prognosis emphasizes the urgent need for novel therapeutic approaches and diagnostic tools. Recent research has shown increasing numbers of studies exploring molecular mechanisms, targeted therapies, and immunotherapies. 9 However, the complex and heterogeneous nature of glioblastoma still hinders effective treatment development. Gaining new insights into tumor biology and microenvironment is crucial for improving patient outcomes.
In recent years, artificial intelligence (AI) has emerged as a transformative force in the clinical and research landscape of neurosurgery. 10 AI technologies, particularly machine learning and deep learning algorithms, have been extensively applied to analyze complex medical data, providing novel insights and strategies for diagnosis, treatment, and prognosis.11,12 In the diagnostic realm, AI has shown remarkable potential in medical imaging analysis. 13 For instance, convolutional neural networks (CNNs) have been utilized to analyze magnetic resonance imaging (MRI) scans, enabling the detection and classification of brain tumors with high accuracy. 14 Xia et al. developed a multiparametric CNN model that integrates different MRI sequences to distinguish primary central nervous system lymphoma from glioblastoma, achieving an accuracy of 0.899. 15 AI is also being employed to predict treatment responses and improve treatment planning. By integrating clinical data with molecular information, AI models can offer personalized treatment recommendations. 16 Reinforcement learning algorithms have been used to optimize treatment strategies based on individual patient characteristics and treatment responses. 17 In terms of prognosis, AI models can predict patient survival and tumor recurrence by analyzing various data types, such as imaging and genomic data. For example, Wang et al. developed a novel DNA methylation-driven gene-based molecular classification and predictive model for overall survival and immunotherapy response in glioblastoma patients. 18 These predictive models assist clinicians in making more informed decisions and enhancing patient management. The integration of AI into glioblastoma research and clinical practice is continuously evolving, offering innovative solutions to the challenges posed by this aggressive tumor. Machine learning has been used to identify predictors of glioma evolution from longitudinal sequencing. 19 AI-driven precision therapies are also emerging in the context of tumor drug resistance. 20 The core of AI lies in machine learning, which encompasses powerful algorithms such as deep learning, CNNs, and decision trees. 11 These advanced AI algorithms build models that enhance medical image and data analysis, enabling efficient analysis of multidimensional datasets and providing decision support systems for researchers and clinicians to navigate complex data and offer valuable insights and recommendations for precision medicine.21–23
Bibliometric analysis is a powerful tool that statistically evaluates scientific publications to identify research trends, influential authors and institutions, and emerging topics within a specific field. It provides an objective and comprehensive overview of the research landscape, allowing researchers to understand the historical development, current status, and future directions of a particular area of study.24,25 In the context of glioblastoma research, bibliometric analysis can reveal the most productive countries, journals, and authors, as well as the most frequently explored topics and research hotspots. This information is crucial for guiding future research efforts, optimizing resource allocation, and cultivating international collaboration. 26 However, a dedicated, comprehensive bibliometric analysis specifically focusing on the convergence of AI and glioblastoma is currently lacking. As this interdisciplinary field experiences explosive growth, the literature has become vast and fragmented, making it difficult to grasp the overall knowledge structure, key turning points, and the collaborative networks driving innovation. This gap is particularly critical to address now, as the field is at a tipping point. The past 5 years have witnessed an unprecedented surge in AI applications for glioblastoma, moving from proof-of-concept studies toward potential clinical translation. A systematic analysis is thus urgently needed to consolidate this rapidly expanding body of knowledge, distinguish sustained research streams from transient trends, and provide a strategic map of the intellectual landscape and collaborative networks. Such a map is essential for both new entrants and funding agencies to identify key players, potential partners, and underserved research areas, thereby guiding future investment and fostering targeted collaboration.
The current study intends to conduct a comprehensive bibliometric analysis of the application of AI in glioblastoma research over the past two decades. It aims to identify the main sources of publication, collaboration networks, primary research topics, and emerging trends in this area. We anticipate that this study will uncover significant growth in AI-related research in glioblastoma, with a particular focus on the application of machine learning and deep learning algorithms for diagnosis and treatment. The findings are expected to highlight the growing importance of AI in this field and provide a basis for further research.
Materials and methods
Data source and search strategy
The Web of Science (WoS) core database from Clarivate Analytics was chosen for this bibliometric analysis due to its reputation as the most reliable source for such research. A comprehensive search was conducted on 15 February 2025 to identify all articles related to the application of AI in glioblastoma from 1 January 2004, to 31 December 2024, using the search query: (TS = (“glioblastoma*” OR “malignant brain tumor*” OR “glial cell tumor*” OR “astrocytoma*” OR “glioblastoma multiforme*” OR “high grade glioma*”)) AND (TS = (AI OR “Artificial intelligence*” OR “machine learning*” OR “deep learning*” OR “neural network*” OR “Robotics” OR “Autoregressive Model*” OR “Generative Adversarial Networks*” OR “Variational Autoencoder*” OR “Transformers*” OR “Convolutional Neural Network*” OR “GANs” OR “VAEs” OR “ CNN”)).The wildcard symbol (*), used in this search, served to increase the breadth and flexibility of the query, ensuring that variations of relevant literature were captured. Only English-language articles, review articles, and editorial materials were selected, resulting in a total number of 1487 of articles that met the inclusion criteria. The literature inclusion criteria were that the manuscript must focus on the role of AI in glioblastoma. The exclusion criteria were that articles must not be related to the role of AI in glioblastoma or could not be evaluated in the context of this research, and documents such as meeting abstracts, news articles, briefings, or other non-peer-reviewed content were excluded. The process of retrieval is exhibited in Figure 1.

Publications screening flowchart.
Data analysis
This study employed three principal tools: CiteSpace (version 6.3.R1), VOSviewer (version 1.6.18), and Microsoft Excel 2021, for the analysis and visualization of bibliometric data. A refined raw dataset, comprising crucial information such as titles, authors, keywords, institutions, countries, citations, journals, and publication dates, was the foundation of our analysis. This dataset was initially cleaned and organized using Excel and subsequently analyzed with CiteSpace and VOSviewer to reveal insights into research trends, author collaborations, institutional networks, and geographical distributions.
CiteSpace was primarily utilized to explore the evolution, trends, and identification of research hotspots. Its powerful analytical capabilities, including time slicing, betweenness centrality analysis, and burst detection, enabled us to visualize cooccurrence patterns, identify emerging trends, and highlight influential researchers, institutions, and countries. Burst detection allowed for the identification of topics experiencing a surge in research activity during specific periods, providing a deeper understanding of the dynamic changes within the field. Betweenness centrality analysis revealed key nodes and influential actors within the network, indicating which authors, institutions, or countries served as critical bridges, facilitating the flow of information and innovation.
VOSviewer, in contrast, was employed to construct detailed visual maps of keyword cooccurrence, author collaborations, and institutional relationships. These maps illuminated the connections between keywords and collaboration networks, providing insights into prominent research themes and patterns of cooperation. VOSviewer's visualization features, including node size and edge thickness, reflect the frequency of keyword occurrences and the strength of collaborative relationships, respectively, enabling the identification of core collaborative networks and influential research areas.
The integration of Excel's fundamental statistical functions with the advanced visualization capabilities of CiteSpace and VOSviewer provided a comprehensive understanding of the bibliometric landscape. Excel facilitated the tracking of publication trends, citation counts, and distributions across institutions and countries, establishing a robust statistical foundation for the analysis. CiteSpace and VOSviewer offered intricate visual maps and networks that highlighted influential research collaborations, emerging research topics, and geographical trends. The combined use of these tools enabled the identification of key patterns, hotspots, and collaborations, offering a more nuanced and dynamic perspective of the research field.
Results
Global publication and citation trend
Figure 2 presents the temporal distribution of publications and citations related to AI-based glioblastoma research from 2004 to 2024. Both indicators show a pronounced upward trajectory over time. Publication output remained low before 2015, followed by a rapid increase thereafter, reaching a peak in recent years. Citation activity mirrors this pattern, with a substantial rise after 2018, reflecting the growing academic impact of the field. The apparent decline in citations in the most recent year is attributable to incomplete citation accumulation. Overall, these findings demonstrate the rapid expansion and increasing influence of AI applications in glioblastoma research.

Annual trends in publications and citations related to artificial intelligence research in glioblastoma from 2004 to 2024. Bars represent the number of publications per year, while the line indicates the annual citation counts.
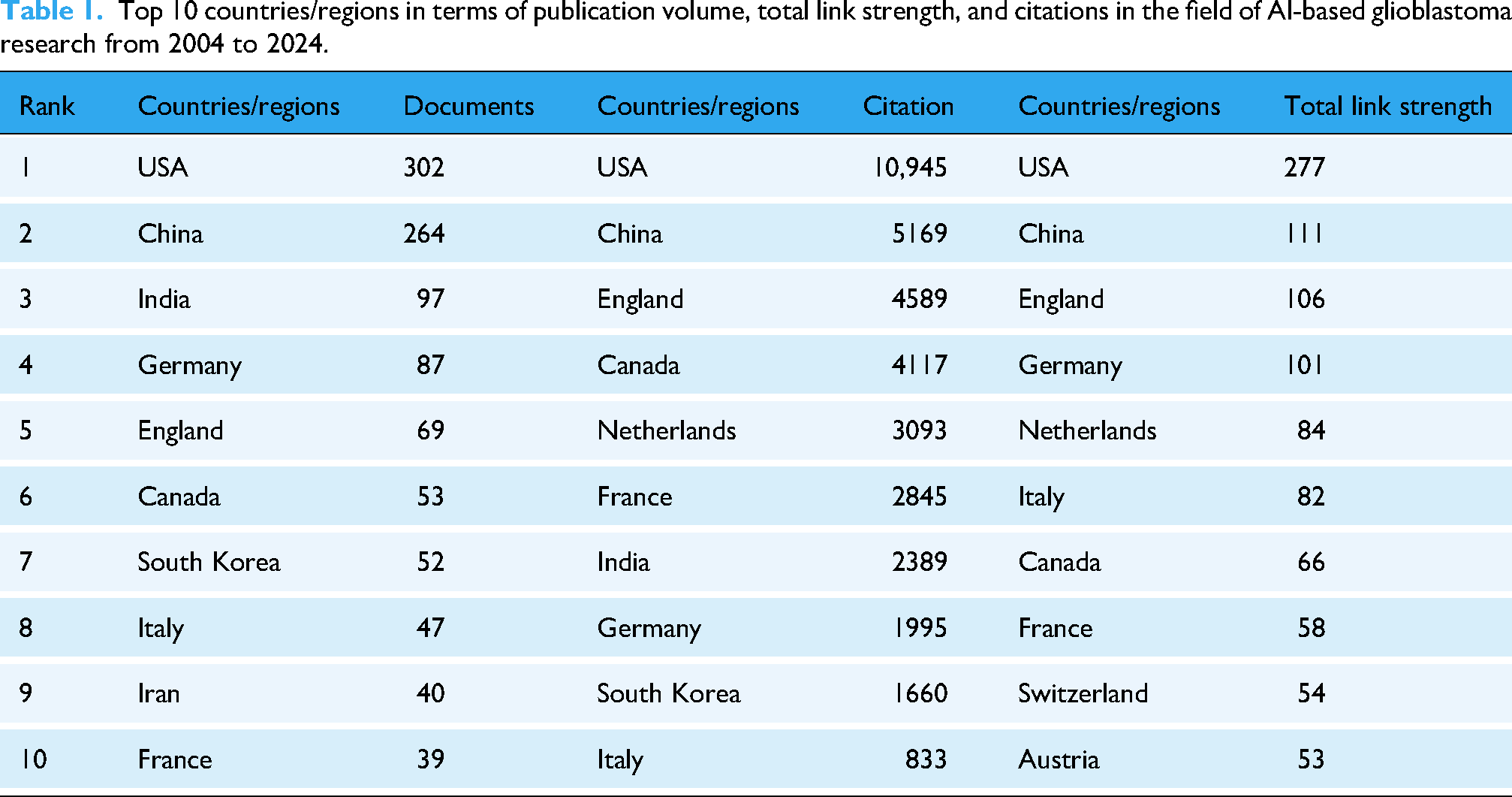
Contributions of countries/regions
The leading countries by publication volume and citation impact are quantified in Table 1. The collaboration network and geographical distribution in Figure 3(a) and (b) provide complementary spatial and relational insights. While both the USA and China are central hubs, the temporal analysis in Figure 3(a) reveals a critical distinction: the USA's collaborative network exhibits cooler colors, indicating earlier and sustained centrality, whereas China's hub displays warmer colors, signifying its more recent emergence as a dominant collaborative force. This pattern highlights an evolving global leadership structure.

Global collaboration patterns in AI-based glioblastoma research (2004–2024). (a) Country collaboration network colored by average publication year, showing temporal evolution of international partnerships. (b) Geographical distribution of research output and collaboration intensity. AI: artificial intelligence.
Top 10 countries/regions in terms of publication volume, total link strength, and citations in the field of AI-based glioblastoma research from 2004 to 2024.
Analysis of authors and cocited authors
Table 2 lists the most prolific authors. Figure 4(a) to (c) together map the social and intellectual structure of the field. The coauthorship network (Figure 4(a)) reveals a clear thematic division between a clinical–translational research cluster (green) and a computational–imaging cluster (blue). This structural separation is reflected in the cocitation network (Figure 4(b)), while the collaboration intensity map (Figure 4(c)) identifies the most active nexus within these clusters. This tripartite analysis underscores a community organized around distinct yet interdependent intellectual foci.

Author networks in AI-based glioblastoma research. (a) Coauthorship network colored by average publication year, showing temporal development of research teams. (b) Author cocitation network revealing intellectual alliances. (c) Collaboration intensity map highlighting core-periphery structure. AI: artificial intelligence.
Ranking of the top 10 major authors in terms of documents and cocitations in the field of AI-based glioblastoma research from 2004 to 2024.

Contributions of institutions
The most productive institutions are listed in Table 3. Their collaborative relationships, shown in Figure 5(a), reinforce the national trends, with strong intraregional networks in North America and Asia. The institutional cocitation network (Figure 5(b)) further demonstrates that these leading institutions form a tightly knit intellectual core, jointly shaping the field's conceptual foundations through high mutual citation.
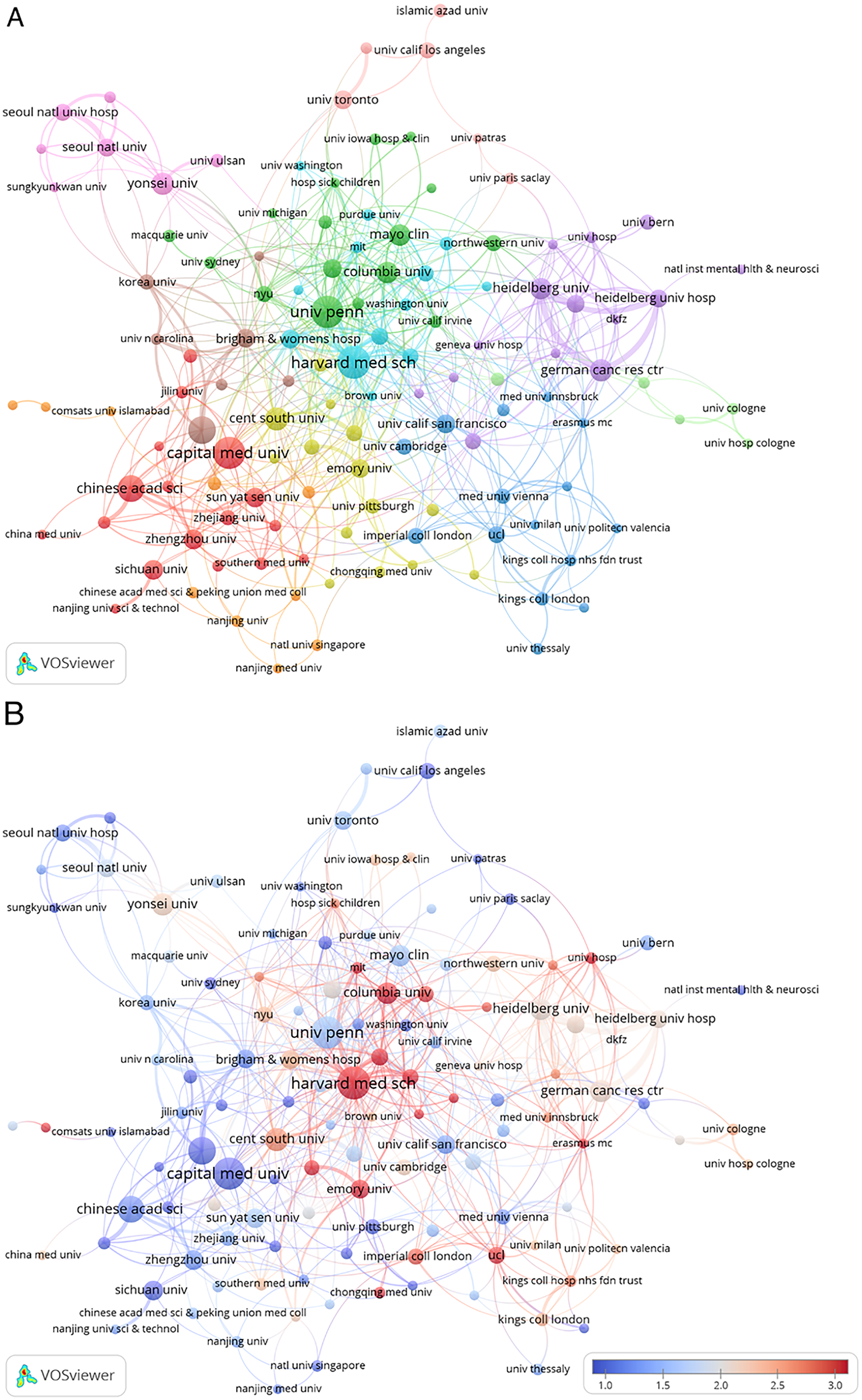
Institutional engagement in AI-based glioblastoma research. (a) Institutional collaboration network colored by average publication year, showing timing of research partnerships. (b) Institutional cocitation network revealing shared intellectual influence over time. AI: artificial intelligence.
Ranking of the top 10 major institutions of AI-based glioblastoma research from 2004 to 2024.

Analysis of journals and cocited journals
Core journals disseminating research are identified in Table 4. Figure 6(a) to (c) offer a layered analysis of their roles. The cocitation network (Figure 6(a)) shows strong interdisciplinary linkages. Temporal analysis (Figure 6(b)) indicates the rising contribution of multidisciplinary open-access journals in recent years. Most significantly, the dual-map overlay (Figure 6(c)) visually traces the primary knowledge flow, revealing that the field is fundamentally driven by the application of research from “Systems, Computing, Mathematics” and “Molecular Biology, Genetics” to clinical domains (“Medicine, Medical, Clinical”).

Journal citation networks in AI-based glioblastoma research. (a) Cocitation network showing journal relationships. (b) Temporal evolution of journal influence, colored by publication recency. (c) Dual-map overlay of citation flows between disciplines. AI: artificial intelligence.
Ranking of the top 10 major journals of AI-based glioblastoma research from 2004 to 2024.

N/A: not available.
Keyword analysis of research hotspots
Analysis of keyword trends (Table 5, Figure 7) reveals the conceptual structure and evolution of AI-glioblastoma research. Core techniques (“machine learning,” “deep learning”) and clinical targets (“glioblastoma,” “MRI,” “prognosis”) dominate the landscape, while emerging methods like “transfer learning” show strong citation bursts, indicating a shift toward data-efficient modeling.

Visualization of keyword cooccurrence and citation trends in AI-based glioblastoma research (2004–2024). (a) Detailed keyword cooccurrence network; (b) Keyword density heatmap; (c) Analysis of keyword citation trends; (d) Timeline of leading keywords with notable citation bursts. AI: artificial intelligence.
Ranking of the top 20 major keywords of AI-based glioblastoma research from 2004 to 2024.

The cooccurrence network (Figure 7(a)) demonstrates tight integration between computational methods and clinical applications, with distinct clusters highlighting key research themes. The density visualization (Figure 7(b)) confirms the central importance of machine learning and MRI analysis. Temporal analysis of citation bursts (Figure 7(c)) reveals the field's evolution from foundational concepts (“neural networks”) to contemporary focuses on “survival” prediction and “transfer learning.”
The timeline visualization of research clusters (Figure 7(d)) further charts this progression, from early work on tumor characterization to recent clusters focused on learning-based radiomic analysis (#7) and IDH mutation profiling (#4). This evolution reflects a consistent movement toward precision medicine, where AI increasingly bridges imaging features, molecular markers, and clinical outcomes to enable more personalized glioblastoma management.
Analysis of top-cited references and cocitation networks
Analysis of highly cited references and cocitation networks reveals the intellectual foundation of AI-based glioblastoma research (Table 6, Figure 8). The most influential works span methodological advances in deep learning architectures for tumor segmentation, standardization of radiomic feature extraction, and integration of molecular diagnostics. Table 6 presents the 15 most frequently cited references that collectively illustrate the breadth and progression of AI-based glioblastoma research, spanning innovative deep learning architectures, advanced imaging techniques, and translational clinical applications. Among these, the first reference by Kamnitsas et al. 27 introduces an efficient multiscale 3D CNN framework combined with fully connected conditional random field (CRF), substantially improving brain lesion segmentation across various MRI modalities. The second reference by Havaei et al. 28 details a deep neural network approach tailored to glioblastoma segmentation, showcasing how local and global contextual features can be captured simultaneously to enhance accuracy and speed. The third reference by Bi et al. 29 provides a comprehensive overview of AI in cancer imaging, including its application to brain tumors, and highlights clinical challenges such as data standardization, model validation, and the integration of AI-driven tools into existing diagnostic workflows. Taken together, these top studies underscore the field's ongoing shift toward more robust, scalable, and clinically impactful AI methodologies for glioblastoma diagnosis, treatment planning, and outcome prediction.

Highly cited references and cocitation network in AI-based glioblastoma research (2004–2024). (a) Clustered Network (CiteSpace): Each color-coded cluster represents a distinct research focus, with node size proportional to citation frequency and node color indicating publication time; (b) Reference Network Map: Displays individual references by cocitation strength; larger nodes indicate higher impact, and links denote interrelated studies; (c) Timeline of References with Notable Citation Bursts: Highlights the periods when references experienced significant surges in scholarly attention, often marking pivotal developments or methodological advances. AI: artificial intelligence.
Ranking of the top 15 major highly cited references of AI-based glioblastoma research from 2004 to 2024.
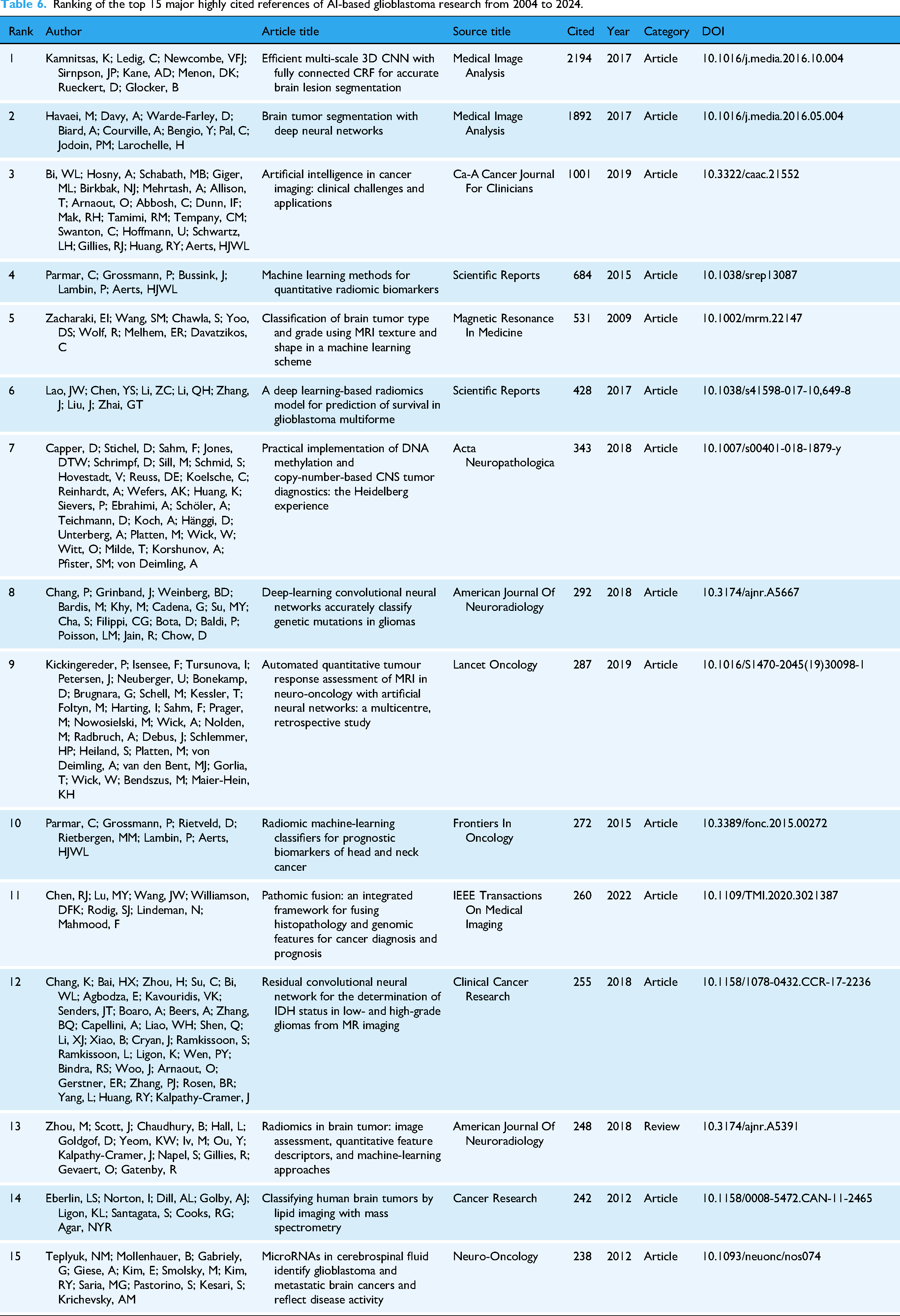
Figure 8(a) visualizes a cocitation network of key references in AI-based glioblastoma research, where each node represents a publication and the node size corresponds to its cocitation frequency. Warmer-colored nodes generally indicate more recent references, while cooler colors represent older or foundational works. The dense clustering at the center suggests that these references are frequently cited together, reflecting a convergence of seminal studies on topics such as deep learning architectures, advanced imaging modalities, and radiomic analyses. In contrast, nodes on the periphery may indicate emerging or specialized research areas that have not yet been integrated into the central knowledge base. In the cocitation map, the 2016 WHO Classification of Tumors of the Central Nervous System by Louis DN et al. 3 occupies a prominent position, reflecting its pivotal role in unifying histological and molecular perspectives within glioblastoma research. The reference is frequently cited alongside studies on IDH1 mutations and advanced imaging or AI-based diagnostic approaches, underscoring its influence on both foundational tumor classification and cutting-edge computational methods. By formalizing the integration of molecular markers into diagnostic criteria, this work established a conceptual framework that resonates throughout the network, as evidenced by its large, centrally located node and numerous links to other high-impact publications. Consequently, it has become a cornerstone for subsequent investigations aiming to refine prognostic models, enhance personalized treatment strategies, and harmonize pathological classification with AI-driven clinical insights. Additionally, the 2020 Radiology article stands out as a pivotal node linking radiomics-based methodologies with AI-driven glioblastoma research. 30 Its presence among frequently cited works reflects the field's growing emphasis on reproducibility and comparability in imaging-derived biomarkers, as the study provides a standardized set of radiomic features that can be reliably implemented across diverse software platforms. By defining reference values and validating them through iterative testing phases, the publication has become an anchor for researchers seeking to ensure consistency in quantitative image-based phenotyping. Consequently, it is often cocited alongside seminal works on AI-based segmentation and molecular characterization, underscoring the importance of robust feature standardization for advancing multicenter collaborations and improving the clinical utility of radiomics in neuro-oncology.
Figure 8(b) provides a denser map of individual publications, illustrating how frequently they are cocited, where each node represents a scientific publication and each cluster (labeled with a “#” followed by a topic, such as “#0 brain tumor segmentation” or “#6 classification”) denotes a set of closely related references. Nodes are colored and sized according to factors like publication year and citation frequency, with larger nodes typically indicating higher impact. Lines between nodes represent cocitation links, showing how often these publications are cited together. The dense connections and overlapping clusters illustrate the interdisciplinary nature of AI-based glioblastoma research, reflecting various thematic focuses such as molecular markers, advanced imaging, and clinical applications. This visualization offers insight into the field's key research directions, the relationships between different topics, and how these areas have evolved over time. For example, Menze BH appears as a central node within the “brain tumor segmentation” cluster, reflecting its foundational influence on AI-driven segmentation studies in neuro-oncology. 31 This publication introduced the Multimodal Brain Tumor Image Segmentation Benchmark (BRATS), which evaluated 20 cutting-edge algorithms on a series of 65 real and 65 simulated multicontrast MR scans from glioma patients. The study's findings highlighted both the complexity of accurately delineating tumor subregions, underscored by substantial variation among human annotators, and the potential for algorithmic fusion methods to exceed individual performance levels. By providing publicly available datasets and an ongoing online evaluation platform, Menze BH et al. set a widely recognized standard for reproducible, comparative research in automated glioma segmentation, a legacy clearly evidenced by its prominent position and extensive interconnections in the figure.
Figure 8(c) pinpoints the temporal spikes in citations for influential studies. Red spikes along the timeline indicate periods of sudden scholarly attention to specific references, often corresponding to methodological breakthroughs or the introduction of clinically relevant findings. Shifts in research focus are evident: early bursts (e.g., around 2012–2014) typically relate to pioneering work in radiomics and segmentation techniques, whereas more recent bursts (2018–2022) center on integrated analyses of molecular markers, machine learning-based outcome prediction, and the refinement of deep learning architectures. These patterns reflect the evolving priorities of the field, from basic feasibility demonstrations of AI techniques to more sophisticated, translational applications that inform patient management. Article published in 2021 by Weller et al. (Nat Rev Clin Oncol) presents the European Association of Neuro-Oncology guidelines for the diagnosis and management of diffuse gliomas in adults. 32 By incorporating the 2016 WHO Classification of Tumors of the Central Nervous System and subsequent cIMPACT-NOW recommendations, this comprehensive guideline addresses the latest diagnostic algorithms, therapeutic modalities (surgery, radiotherapy, and pharmacotherapy), and follow-up strategies, aiming to balance clinical efficacy with the avoidance of unnecessary interventions. Its strong citation burst underscores the immediate relevance and broad adoption of these evidence-based recommendations in clinical practice. Another representative study published in 2014 by Aerts et al. (Nat Commun) introduces a pioneering radiomics framework that quantitatively characterizes tumor phenotypes based on hundreds of image-derived features. 33 Conducted across a large cohort of lung and head-and-neck cancer patients, this study demonstrates how noninvasive imaging can capture intratumor heterogeneity and link it to underlying gene expression profiles. The significant citation burst highlights the paper's foundational role in establishing radiomics as a powerful tool for bridging imaging and molecular oncology, paving the way for subsequent advances in AI-driven precision medicine.
Collectively, these references trace a trajectory from technical validation to clinical integration, emphasizing automated segmentation as a foundational step, radiomic–molecular correlation for prognostic refinement, and guideline development for real-world implementation. The cocitation patterns underscore the field's maturation toward clinically validated, standardized AI applications in neuro-oncology.
Discussion
This study provides a detailed bibliometric analysis of AI applications in glioblastoma research over the past two decades. Our findings reveal a significant upward trend in publications, reflecting the growing interest and investment in this field. Through our analysis, we have identified key contributors, including prominent authors such as Bakas Spyridon and leading institutions like the University of Pennsylvania. The study also underscores substantial international collaboration and knowledge exchange, as evidenced by extensive collaborating networks that connect the USA, China, Germany, and other countries. AI's influence on glioblastoma research is further highlighted by its diverse applications, ranging from enhancing diagnostic accuracy through advanced imaging techniques to improving treatment outcomes via machine learning algorithms. The emergence of research hotspots, such as transfer learning and radiogenomics, indicates a shift toward more precise and personalized treatment approaches. Overall, these findings demonstrate the potential of AI to transform glioblastoma research and clinical practice, offering promising new avenues for improved patient outcomes.
The current research landscape in glioblastoma AI applications has shown a clear upward trajectory. Our analysis indicates a significant increase in publications over the past two decades, with a notable acceleration in research output after 2015. Research activity remained relatively limited until 2015, after which a noticeable acceleration occurred. This period coincided with several major technological and clinical milestones that reshaped the landscape of AI-driven neuro-oncology research. The emergence of deep learning models such as AlexNet and ResNet,34,35 the release of the U-Net architecture in 2015 which transformed medical image segmentation, and the establishment of public imaging datasets through initiatives such as the MICCAI BraTS challenges31,36 collectively enhanced data accessibility and algorithmic innovation. Furthermore, the 2016 revision of the WHO classification of central nervous system tumors, which incorporated molecular markers into diagnostic criteria, provided new opportunities for AI-assisted classification and prognostic modeling. 3 Although direct causation cannot be established, the timing of these advancements parallels the sharp rise in publication output observed after 2015. This growth mirrors the broader trends in AI-assisted medical research and reflects the increasing recognition of AI's potential in tackling complex medical challenges like glioblastoma. 37 For example, the USA and China are at the forefront of this research, with the USA exhibiting the highest citation count, indicative of its strong academic influence. Meanwhile, China leads in the overall number of publications, reflecting its rapid growth in research output. Additionally, several European countries, such as Germany and England, have demonstrated high citation/publication ratios, highlighting the impact of their research despite fewer total publications. These findings are well-supported by the data from our bibliometric analysis and align with observations in AI-assisted drug and biomarker discovery for neuro-oncology,38–42 where similar growth patterns and research hotspots have been identified.43,44
The analysis of authors in AI-driven glioblastoma research reveals a dynamic and collaborative research community. Bakas Spyridon, Louis DN, and Davatzikos Christos stand out as prominent figures with significant contributions. Bakas Spyridon has a substantial number of publications and a broad collaborative network, while Louis DN has made substantial contributions to glioblastoma classification, and Davatzikos Christos has been influential in developing imaging biomarkers. Their work has been pivotal in advancing the field, particularly in these areas. The coauthorship network illustrates a global research community, with authors from North America, Europe, and Asia collectively driving innovation. This aligns with the findings of Bakas et al., 45 which emphasized the importance of international collaboration in accelerating progress in oncology research. The cocitation network further underscores the collective impact of these key authors. For example, Louis DN and Stupp R are part of a green cluster that emphasizes clinical and pathological aspects, including standard-of-care treatments, clinical trials, and translational research leveraging AI-based tools. Bakas Spyridon and Merna BH are in a blue cluster focusing on advanced imaging techniques and the development of machine learning or deep learning algorithms for tumor segmentation, prognostic modeling, and large-scale data sharing frameworks. This multidisciplinary approach integrates different fields to enhance diagnostic accuracy and treatment personalization.
The collaboration between these authors facilitates knowledge exchange and fosters innovation, as seen in the integration of clinical insights with computational methods. Their collective efforts not only advance the understanding of glioblastoma but also pave the way for more effective AI-based clinical applications. In addition, the institutional analysis shows that leading universities and research centers, such as the University of Pennsylvania, Harvard Medical School, and Capital Medical University, dominate AI-focused glioblastoma research. These institutions act as hubs in the global research network, promoting knowledge exchange and collaborative projects. The institutional collaboration network shows a dense network of partnerships, with North American and Chinese institutions occupying central positions. This mirrors the observations of Kickingereder et al., 44 which indicated that leading institutions in AI and medical research often form the backbone of international scientific cooperation. While our network analysis identifies key hubs such as the University of Pennsylvania and authors like Bakas Spyridon, the underlying drivers of this topology warrant further discussion. The high betweenness centrality of these nodes likely reflects a combination of genuine intellectual leadership—pioneering public datasets like the BraTS challenge and foundational segmentation algorithms—and the structural advantages conferred by preferential attachment mechanisms common in scientific collaboration networks. 46 In such systems, established and highly visible research groups naturally attract more collaboration opportunities. While this concentrates expertise and can accelerate standardization, future research should combine bibliometric analysis with qualitative methods to better understand the relationship between network position and scientific innovation in this field. Furthermore, the concentration of influential research within a limited set of actors presents a dual narrative: one of focused expertise driving progress, and another underscoring the need for intentional efforts to broaden participation and mitigate potential redundancy, thereby ensuring the robustness and generalizability of translational AI research.
The analysis of journals in AI-driven glioblastoma research reveals a group of prominent publications that play a crucial role in advancing the field. Neuro-Oncology stands out as a leading journal, with a high volume of articles and a robust impact factor, reflecting its central role in integrating neurosurgical, oncological, and computational research. Other high-impact journals such as Cancers and Scientific Reports have also made significant contributions by publishing numerous studies focused on AI-driven diagnostics, prognostic modeling, and treatment strategies for glioblastoma. Frontiers in Oncology and Radiology further highlight the growing interest in leveraging imaging-based AI methods to enhance tumor characterization and therapy planning. These journals not only disseminate cutting-edge research but also foster interdisciplinary collaborations essential for advancing AI-based strategies in glioblastoma diagnosis and treatment. For example, Cancers and Frontiers in Oncology have shown a notable increase in publications and citations in recent years, mirroring the expanding role of AI in precision oncology and radiomics. This aligns with the findings by CA: A Cancer Journal for Clinicians, which emphasized the importance of interdisciplinary journals in advancing AI applications in cancer imaging. 29 Additionally, the dual-map visualization of journal interactions underscores how various academic disciplines interconnect in AI-based glioblastoma research, highlighting the pivotal role of journals like Neuro-Oncology and Radiology in fostering cross-disciplinary partnerships. These efforts are essential for personalized therapy, immunotherapy, and novel drug development, reflecting the extensive interdisciplinary integration required for AI-based glioblastoma research. From molecular markers and genomics to medical imaging and clinical assessment, these journals facilitate the development and validation of computational algorithms, underscoring the pivotal role of AI in fostering collaborative progress against this complex disease. This high concentration of literature in a core set of journals aligns with known bibliometric distributions. While it signifies efficient knowledge consolidation, it also necessitates awareness that such centralization can inherently shape the field's agenda from a consolidated perspective, highlighting the importance of diverse publication avenues for methodological and clinical innovation.
The keyword analysis of research hotspots in AI-based glioblastoma research reveals several key themes that have emerged as central to the field. Machine learning and deep learning, particularly in imaging-based analyses, are prominent research areas.39,40 These advanced computational techniques are being applied to improve diagnostic accuracy and treatment planning for glioblastoma. Molecular markers such as IDH1 have also gained significant attention, underscoring their importance in glioblastoma classification and prognosis. 47 The emergence of transfer learning as a research hotspot indicates a shift toward leveraging pretrained models to overcome data limitations, particularly in resource-limited settings where large datasets are not readily available. This approach has shown promise in improving diagnostic accuracy and treatment personalization. The integration of these technologies reflects a movement toward more precise and personalized medicine. The citation trends analysis shows that early research focused on foundational machine learning applications and tumor characterization, while recent years have seen a surge in studies exploring the integration of molecular markers and advanced computational methods. This evolution reflects the field's increasing focus on precision diagnostics and tailored treatment strategies. The dual-map visualization of keyword citation trends provides additional insights into the interdisciplinary nature of AI-based glioblastoma research, illustrating how knowledge flows between different fields, with prominent connections between computer science, molecular biology, and clinical medicine. 1 This underscores the importance of cross-disciplinary collaboration in advancing the field and highlights the potential of AI to transform glioblastoma research and clinical practice. 48
The analysis of top-cited references and cocitation networks in AI-based glioblastoma research offers valuable insights into the field's evolution and key research directions. Kamnitsas et al. 27 developed a multiscale 3D CNN framework integrated with a fully connected CRF, significantly advancing brain lesion segmentation across various MRI modalities. This work has become a cornerstone for automated tumor segmentation, which is crucial for accurate diagnosis and treatment planning. Havaei et al. 28 also made significant contributions by detailing a deep neural network approach tailored for glioblastoma segmentation, demonstrating how local and global contextual features can be simultaneously captured to enhance accuracy and speed. These studies highlight the growing sophistication of AI methodologies in glioblastoma research, moving from basic feasibility to clinically impactful applications. Bi et al. 29 provided a comprehensive overview of AI in cancer imaging, underscoring clinical challenges such as data standardization and model validation. This work has been pivotal in addressing the practical hurdles of integrating AI-driven tools into diagnostic workflows. The cocitation network analysis reveals that foundational studies on deep learning architectures, advanced imaging modalities, and radiomic analyses are frequently cited together, reflecting their collective influence on the field. The 2016 WHO Classification of Tumors of the Central Nervous System by Louis DN et al. 3 occupies a central position in the cocitation map, emphasizing its role in unifying histological and molecular perspectives within glioblastoma research. This classification has become a cornerstone for studies aiming to refine prognostic models and enhance personalized treatment strategies. A qualitative sampling of the most-cited references contextualizes the bibliometric trends. These seminal works are primarily foundational, establishing methodological benchmarks (e.g., for segmentation) or demonstrating predictive potential in retrospective studies. Notably, few employ multicenter validation designs, and prospective trials or reports of clinical deployment are absent from this high-impact cohort. This suggests that the field's scholarly influence is currently driven by proving technical capability. The next translational leap will require generating similarly influential evidence from rigorous external validation and prospective clinical utility studies. Collectively, these references and cocitation networks illustrate a research trajectory driven by automated tumor segmentation, which serves as a foundational step for reproducible tumor volume quantification, essential for accurate diagnosis, treatment planning, and response monitoring.
The evolution of AI techniques in glioblastoma research demonstrates a clear trajectory from traditional machine learning reliant on hand-crafted radiomic features to the current dominance of deep learning architectures. CNNs, particularly 3D U-Net-based architectures, have become the cornerstone for automated and precise tumor subregion segmentation from multimodal MRI, providing a critical tool for quantitative assessment. 49 Furthermore, the application of CNNs has expanded beyond anatomical delineation to predicting molecular markers, such as the noninvasive classification of MGMT promoter methylation status from multiparametric MRI, showcasing their power in linking imaging phenotypes to genomic characteristics.50,51 While the field continues to explore more advanced architectures like state space models (e.g., Mamba) for their efficiency in modeling long-range dependencies in volumetric MRI, 52 a critical parallel focus must be on the clinical translatability of these technologies. Important methodological studies highlight that deep learning models, including those for computational pathology tasks like glioma IDH1 mutation prediction, can exhibit significant performance disparities across different racial patient cohorts, revealing demographic bias as a substantial challenge for real-world implementation. 53 Therefore, future research must not only pursue algorithmic advancements but also prioritize robustness, generalizability, and rigorous bias mitigation to ensure AI models can be equitably and reliably integrated into clinical neuro-oncology workflows.
It is worth noting that the observed disparity between the volume of academic research and the pace of clinical integration stems from fundamental differences in the primary objectives and evaluation paradigms of consecutive research phases. The exponential growth in publications largely captures the technology exploration and proof-of-concept phase, where innovation is driven by methodological novelty and performance on benchmark datasets. 54 Success in this phase, often measured by algorithmic accuracy on retrospective data, does not automatically translate to readiness for clinical use. The subsequent clinical translation and implementation phase demands evidence that aligns with clinical practice: robust performance across diverse, real-world patient populations and care settings, proven utility in improving workflows or decision-making, and compatibility with regulatory and reimbursement pathways.55,56 The current bibliometric landscape revealed in our study indicates a field that is exceptionally productive in the first phase. However, a commensurate volume of high-level evidence—such as prospective randomized trials or robust multicenter external validations—that directly addresses the criteria of the second phase is still emerging. 57 Therefore, the gap is not necessarily an indicator of failure but reflects the challenging, resource-intensive, and necessary progression of the field from demonstrating technical potential to proving clinical value. A critical implication of our findings, which we wish to explicitly underscore, is the fundamental distinction between bibliometric influence and clinical utility. Our study maps the propagation of ideas and scholarly attention within the academic community; it measures influence, not ground truth. A highly cited paper may introduce a novel AI architecture or demonstrate proof-of-concept on a single dataset. However, as noted by studies on AI in healthcare, 56 this citation velocity does not equate to proven model robustness, successful external validation across diverse clinical settings and patient populations, or feasibility for deployment in real-world clinical workflows. The ultimate test for the trends identified here will be their translation into measurable improvements in patient outcomes through rigorous prospective trials. While our analysis maps the rapid expansion of AI in glioblastoma research, bridging the gap between algorithmic development and clinical deployment requires addressing specific methodological hurdles. Accordingly, we propose three prioritized future directions: First, the field must shift from a primary focus on accuracy to demonstrable robustness and fairness. This entails rigorous external validation on multi-institutional datasets with explicit testing for demographic and scanner-related biases to ensure generalizability.58,59 Second, enhancing clinical interpretability through intuitive explainable AI is nonnegotiable for building trust; clinicians require clear rationales for model predictions to effectively integrate them into decision-making pathways.60,61 Finally, research should progress toward integrated, multimodal diagnostic systems that fuse imaging, histopathology, and genomic data within frameworks that are scalable and compliant with clinical data privacy and workflow constraints.62,63 Addressing these practical challenges of robustness, interpretability, and clinical integration is paramount for translating promising AI tools into tangible improvements in patient care.
Our bibliometric analysis of AI in glioblastoma research has provided a comprehensive overview of the field's development and key trends over the past two decades. However, this study has some limitations. First, our reliance on the WoS Core Collection, while providing high-quality, standardized data ideal for citation network analysis, inherently shapes the scope of our findings. This database offers extensive coverage of biomedical literature but has more limited indexing of conference proceedings compared to Scopus or IEEE Xplore. Given that our study is fundamentally oriented toward mapping AI applications within the clinical and translational research domain of glioblastoma—rather than surveying the entirety of AI methodological development—WoS was selected as the most appropriate source. We acknowledge that this choice may result in our bibliometric maps emphasizing networks and trends within the biomedical community, potentially offering a less complete representation of contributions from core computer science venues. Therefore, the trends identified herein are best interpreted as reflective of the medically integrated AI research landscape. Second, the use of English as the sole language for inclusion may have excluded valuable research published in other languages, potentially distorting the representation of international contributions. Future research could expand the scope by including more diverse data sources and employing multilingual search strategies to provide a more comprehensive and balanced view of global efforts in this field. Third, and most critically, our quantitative approach captures scholarly communication metrics but cannot assess research quality or translational maturity. We did not evaluate validation rigor (e.g., retrospective vs. prospective design), effect sizes, reproducibility, or any evidence of clinical deployment and patient-level outcomes. While we incorporated a qualitative assessment of top-cited papers to address this, a full-scale systematic review of validation rigor across the corpus was beyond our scope. Our sampling highlights the need for future bibliometric studies to systematically integrate such qualitative dimensions to better assess translational maturity. Finally, while our analysis maps the concentration of research output and collaboration, a pattern consistent with bibliometric laws such as those described by Bradford and Lotka, the quantitative data alone cannot arbitrate between the potentially positive interpretation of genuine specialization and the possible risk of incremental redundancy. Future research combining bibliometrics with content analysis could help differentiate these dynamics. Looking ahead, the future of AI in glioblastoma research appears promising. Machine learning and deep learning technologies are expected to play increasingly important roles in enhancing diagnostic accuracy and personalizing treatment plans.13,64,65 For instance, the integration of advanced imaging techniques with AI algorithms holds the potential to improve early detection and prognosis prediction. The continued development and refinement of these technologies, along with the establishment of robust validation frameworks and standardized data protocols, will be crucial for their successful translation into clinical practice, and realizing measurable improvements in patient survival and quality of life-core goals of digital health innovation. Furthermore, fostering greater interdisciplinary collaboration and ensuring equitable access to AI-driven innovations across diverse healthcare settings will be key to maximizing the benefits for patients worldwide and advancing the collective fight against this aggressive form of brain cancer.
Conclusion
In conclusion, our bibliometric analysis has provided a comprehensive overview of the research landscape, key players, and emerging trends in AI-driven glioblastoma research over the past two decades. The significant upward trend in publications and citations reflects the growing interest and investment in this field. Key authors and institutions have been identified, highlighting the importance of individual leadership and collaborative efforts in advancing AI applications for glioblastoma. The analysis of journals and cocitation networks highlights the pivotal role of high-impact, multidisciplinary journals in shaping the research agenda and fostering interdisciplinary collaborations. The identification of research hotspots, such as the application of machine learning and deep learning in imaging-based analyses and the emergence of transfer learning, indicates a shift toward more precise and personalized treatment strategies. Overall, this study highlights the transformative potential of AI in glioblastoma research and clinical practice, offering promising new avenues for improved patient outcomes. This focus on accessible, impactful digital health solutions is essential for driving meaningful progress in neuro-oncology.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076261428356 - Supplemental material for Mapping knowledge landscapes and emerging trends in artificial intelligence in glioblastoma: A bibliometric analysis
Supplemental material, sj-docx-1-dhj-10.1177_20552076261428356 for Mapping knowledge landscapes and emerging trends in artificial intelligence in glioblastoma: A bibliometric analysis by Pei Wang, Wen Liu and Cong Zhou in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076261428356 - Supplemental material for Mapping knowledge landscapes and emerging trends in artificial intelligence in glioblastoma: A bibliometric analysis
Supplemental material, sj-docx-2-dhj-10.1177_20552076261428356 for Mapping knowledge landscapes and emerging trends in artificial intelligence in glioblastoma: A bibliometric analysis by Pei Wang, Wen Liu and Cong Zhou in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors would like to thank those individuals who made the VOSviewer and the R package “bibliometricx” opensource software available.
Contributorship
PW contributed to study conception and design, writing–original draft, and writing–editing and review. WL contributed to study conception, data curation, investigation, writing–editing and review, and supervision. CZ had the idea of the article and its design, collected the data, contributed to study conception and design, and conducted the data analysis, investigation, writing–original draft, and writing–editing and review.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
No patients were involved in this research, so the ethical approval is not required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Graduate Education and Teaching Reform Research Project of Shandong Province (SDYJSJGC2024066), the Medical and Health Science and Technology Development Plan of Shandong Province (202304011343), the Cultivation Plan of High-level Scientific Research Projects of Jining Medical University (JYGC2021KJ006), the Practical Teaching Education Research Plan Project (JYSJ2024B14, JYSJ2024C23).
Data availability
The data sets analyzed during this study are available in the Web of Science core collections.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
